Abstract
Thirty-nine epithelial bladder tumor samples from 37 animals affected with bovine enzootic hematuria (BEH) were selected for immunohistochemical studies. The expression of structural proteins such as uroplakin III (UPIII) and cytokeratin 7 (CK7) and the cell cycle–related proteins cyclin D1 and p53 were evaluated in urothelial papillomas and carcinomas. Loss of UPIII and CK7 expression was seen in both high-grade and high-stage urothelial carcinomas (P < .001 and P < .02). Cyclin D1 expression showed no statistically significant association with grade or stage. In contrast, p53 immunoreactivity was positive in high-grade and high-stage carcinomas (P < .05 and P < .01), confirming its association with the highest malignant behavior of the bladder tumors in BEH.
Keywords
Bovine enzootic hematuria (BEH) is a disease characterized by the occurrence of hematuria and urinary bladder tumors, resulting at least partially from the chronic ingestion of bracken fern (Pteridium spp), the only plant known to cause cancer naturally. 17 This disease is very common in São Miguel Island, Azores, Portugal, 19 where bracken grows endemically, although it has been reported in many other countries in the world. As with many other tumors, the expression of biomarkers in BEH bladder lesions has been studied to estimate the degree of molecular changes occurring in the course of the disease. The studies on BEH have shown that up to 13% of the bladder lesions are nonneoplastic, and only half of the neoplastic lesions are malignant, 4 with urothelial carcinomas being the most frequent tumor type. 4,18
Cytokeratins are the most common markers used to identify epithelial cells, being expressed both in normal and malignant cells. 14 The urothelium is known to express the simple epithelium-type cytokeratin 7 (CK7), frequently used to identify tumors of urothelial origin. 14 The superficial urothelial cells also show a specialized plasma membrane, called an asymmetrical unit membrane (AUM), whose major components are the uroplakins (Ups), highly conserved transmembrane proteins found in a broad range of species. 25 Uroplakin III (UPIII) was reported to be lost in some high-grade urothelial tumors of both humans and domestic animals. 1,16,20
The expression of cyclin D1, a nuclear protein specifically involved in the G1/S phase transition, is frequently overexpressed in urothelial cancer in humans, 22 having been expressed in 100% of the hemangiosarcomas tested in bovine bladder tumors. 3 The alterations of the p53 pathway were identified as one of the most important events in the development of aggressive behavior in tumors. 10 Mutations of p53 are known to occur in 50% of human cancers, 24 including urothelial carcinomas. 23
Materials and Methods
Sample Collection
Thirty-nine bovine urinary bladder tumors of epithelial origin, each one corresponding to isolated, nonconfluent lesions, were collected from 37 Friesian dairy cows slaughtered in the abattoir of São Miguel Island, Azores, Portugal. Up to 90% of the animals had hematuria before or at slaughter, and all were known to have grazed on bracken-infested pasture. Their ages ranged from 3 to 12 years (mean, 6 years). In 2 cows, 2 independent tumor samples were retrieved from the bladder.
All tissues were fixed in 10% buffered formalin and processed for routine histopathology with embedding in paraffin. Three-micron sections were stained with hematoxylin and eosin.
Histological Typing and Immunohistochemistry
Histological typing was based on World Health Organization established criteria and on recent publications on BEH-associated bladder lesions. 4,13,18 Tumors were diagnosed by 2 authors (T.C. and M.C.P.) and a consensus achieved whenever disagreement in the classification occurred.
To determine whether immunoreactivity was associated with the stage, we classified tumors according to the degree of invasion as follows: Ta, noninvasive papillary tumor; T1, suburothelial tumor; and T2, muscle invasive. Two groups were considered: the noninvasive, including Ta, and the invasive, including T1 and T2. Tumors were also classified according to malignancy grade as follows: G1, well differentiated; G2, moderately differentiated; G3, poorly differentiated; and G4, undifferentiated. 7 Papillomas were included in the Ta noninvasive group and classified as G0 to allow intergroup comparisons. Regarding tumor grade, 2 groups were considered: low grade, including G0, G1, and G2, and high grade, including G3 and G4.
The primary antibodies, antigen retrieval methods, and detection systems used in immunohistochemistry are shown in Table 1. Due to tissue loss during antigen retrieval, it was not possible to perform staining in 3 samples for UPIII, in 2 for CK7, in 4 for cyclin D1, and in 13 for p53.
Antibodies Used and Technical Details.
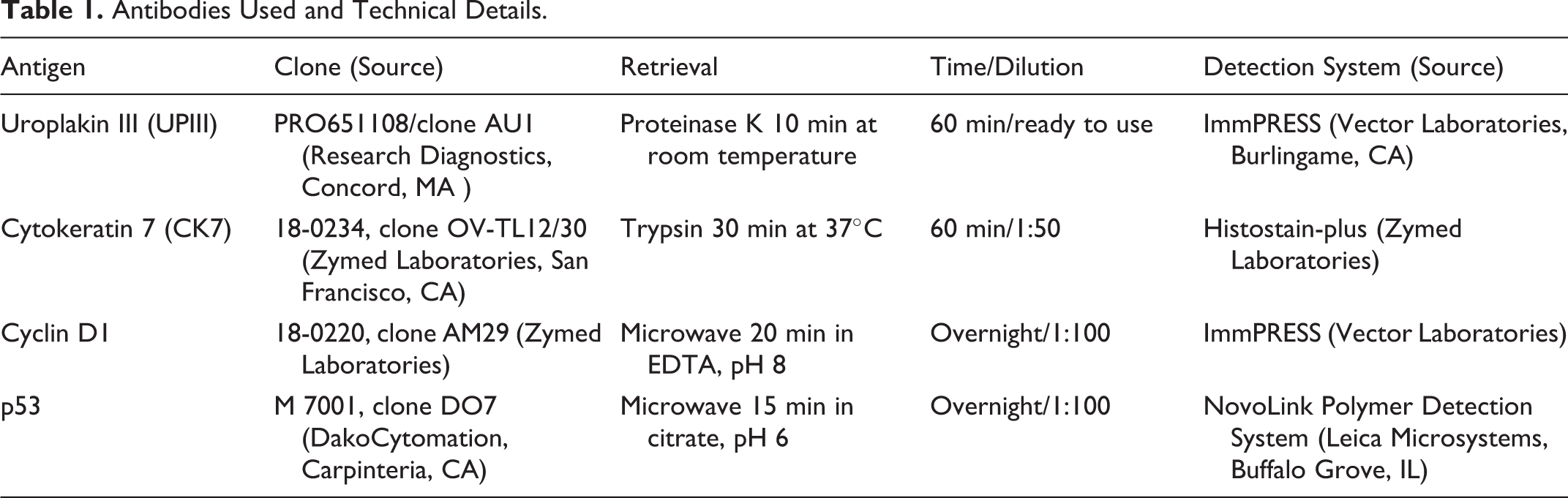
Antisera Validation
Normal urothelium served as an internal positive control for UPIII and CK7. Human mantle cell lymphoma was included as a positive control for human cyclin D1 antibody. Bovine ocular squamous cell carcinoma was used as a positive control for p53 immunodetection. A 5% cutoff value was accepted as positive staining with antibodies against UPIII and CK7. A 20% cutoff value was accepted as positive staining for cyclin D1 and p53. Negative controls for immunohistochemistry were processed identically to test slides, but the primary antibody was omitted.
Statistical Analysis
Association between the presence/absence of immunoreactivity for the antibodies tested and malignancy grade (G0–G4) vs stage (Ta–T4) was evaluated using a 2-sided Fisher’s exact test. Statistical analysis was performed using R-Stat version 2.12.1 (R Foundation for Statistical Computing, Vienna, Austria). The associations were considered significant when P values were less than .05.
Results
The selected urothelial tumors were classified as follows: 7 papillomas and 32 carcinomas, with representation of the various stages and grades (Tables 2 and 3).
Distribution of Grades and Stages of Bovine Epithelial Urinary Bladder Tumors Studied.
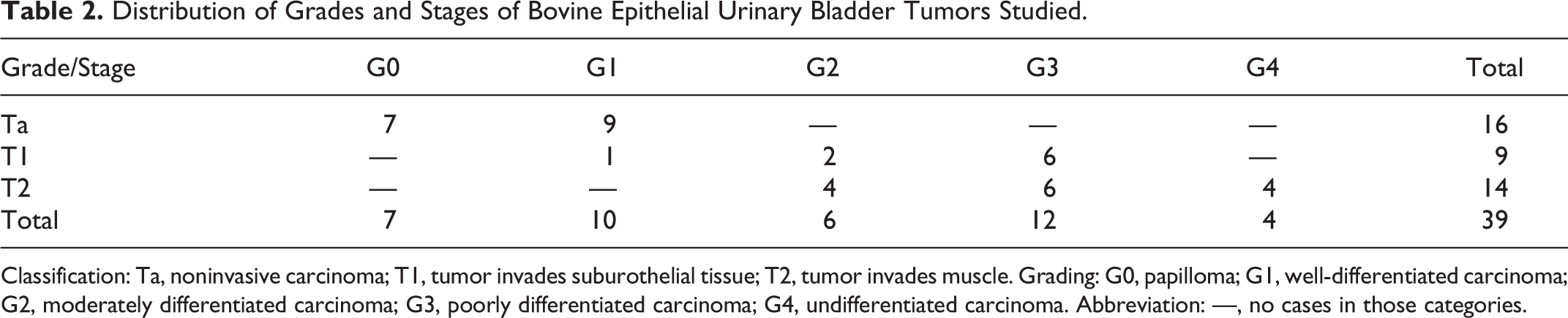
Classification: Ta, noninvasive carcinoma; T1, tumor invades suburothelial tissue; T2, tumor invades muscle. Grading: G0, papilloma; G1, well-differentiated carcinoma; G2, moderately differentiated carcinoma; G3, poorly differentiated carcinoma; G4, undifferentiated carcinoma. Abbreviation: —, no cases in those categories.
Immunohistochemical Results Obtained in Bovine Epithelial Urinary Bladder Tumors.
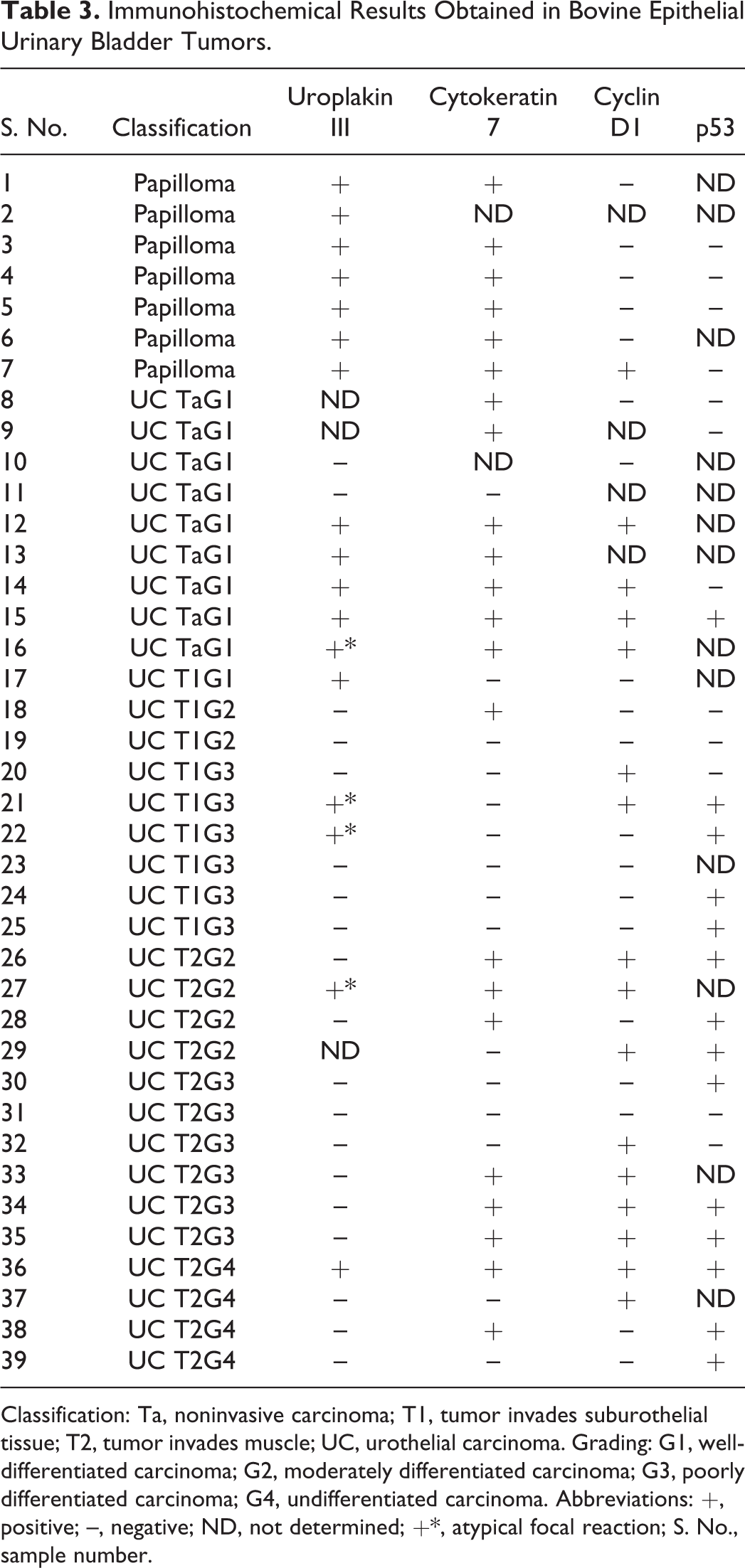
Classification: Ta, noninvasive carcinoma; T1, tumor invades suburothelial tissue; T2, tumor invades muscle; UC, urothelial carcinoma. Grading: G1, well-differentiated carcinoma; G2, moderately differentiated carcinoma; G3, poorly differentiated carcinoma; G4, undifferentiated carcinoma. Abbreviations: +, positive; –, negative; ND, not determined; +*, atypical focal reaction; S. No., sample number.
Uroplakin III and Cytokeratin 7
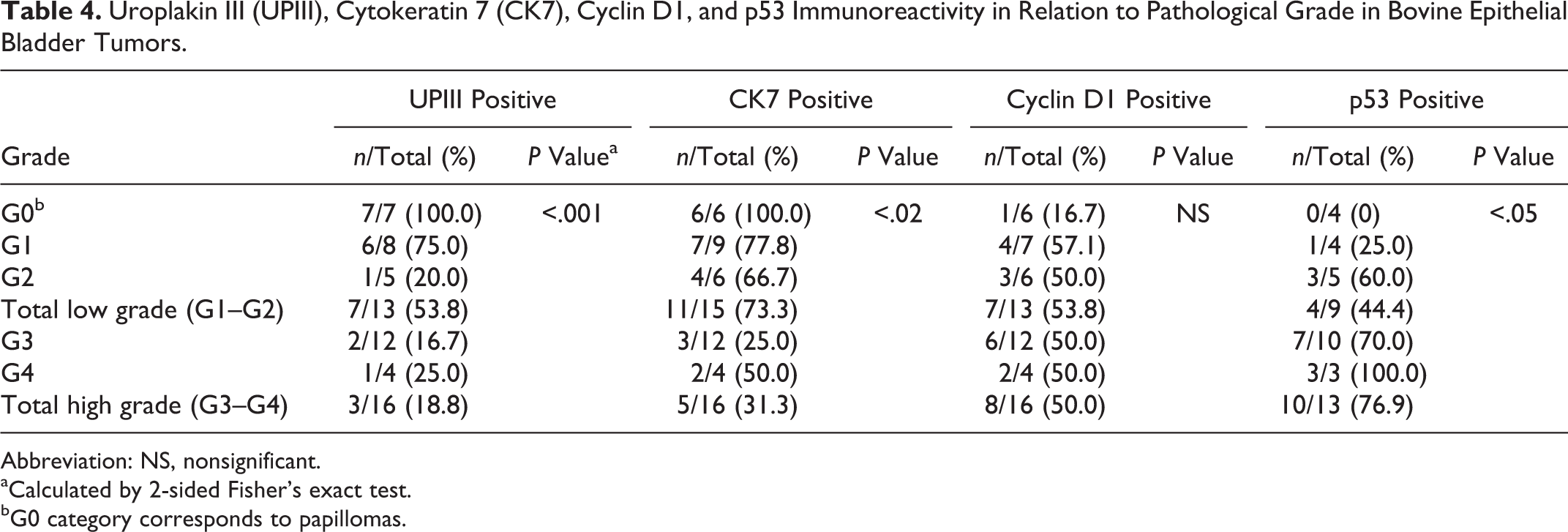
The UPIII staining pattern was very intense on the apical portion and cytoplasm of the normal urothelium. The same pattern was seen in all papillomas (Fig. 1) but was gradually lost with increasing tumor grade/stage. Ten of 29 (34.5%) urothelial carcinomas tested were positive to UPIII. Urothelial carcinoma cells exhibited plasma membrane and cytoplasmic staining lacking distinct polarity (Fig. 2). Most urothelial carcinomas showed scattered UPIII distribution, with areas of strongly positive cells adjacent to nonreactive areas. Most G4 urothelial tumors were negative. Details regarding the results in each stage/grade groups are shown in Tables 4 and 5. The loss of expression of UPIII was statistically significantly associated with increasing grade (P < .001) and stage (P < .001).
Urinary bladder; bovine. Urothelial papilloma displaying typical uroplakin III (UPIII) reaction, restricted to the apical cytoplasmic border of the most superficial cells. ImmPRESS-peroxidase and Mayer’s hematoxylin counterstain.
Uroplakin III (UPIII), Cytokeratin 7 (CK7), Cyclin D1, and p53 Immunoreactivity in Relation to Pathological Grade in Bovine Epithelial Bladder Tumors.
Abbreviation: NS, nonsignificant.
aCalculated by 2-sided Fisher’s exact test.
bG0 category corresponds to papillomas.
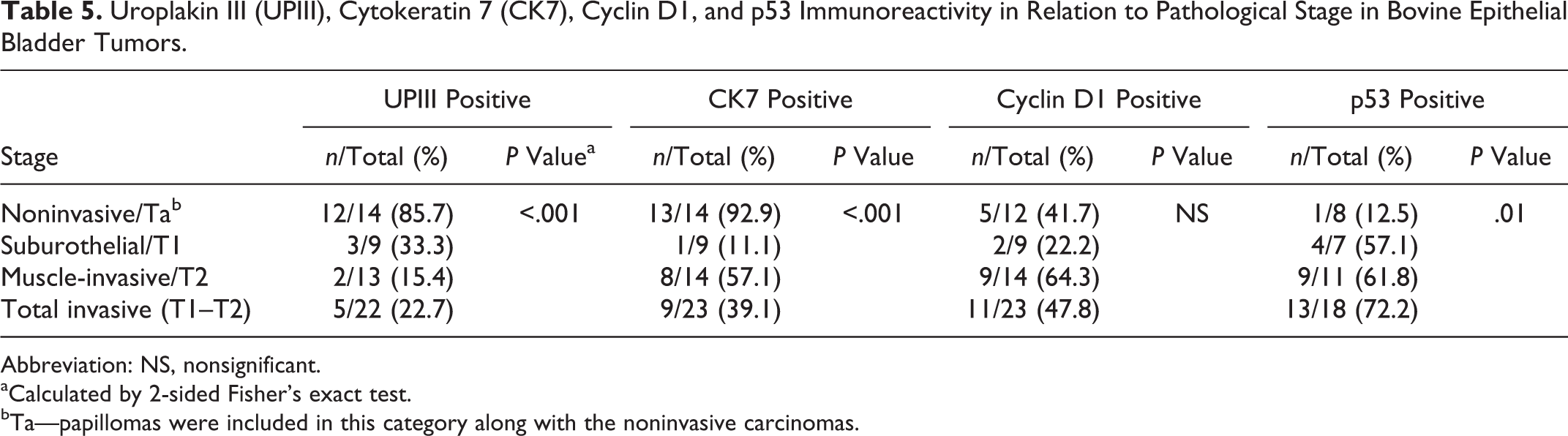
Uroplakin III (UPIII), Cytokeratin 7 (CK7), Cyclin D1, and p53 Immunoreactivity in Relation to Pathological Stage in Bovine Epithelial Bladder Tumors.
Abbreviation: NS, nonsignificant.
aCalculated by 2-sided Fisher’s exact test.
bTa—papillomas were included in this category along with the noninvasive carcinomas.
CK7 expression in urothelial papillomas was similar to that found in the normal urothelium, with positive cells just above the basal layer up to the more apical cells, with diffuse cytoplasmic staining (Fig. 3). CK7 expression was more frequent in high-grade urothelial malignancies than UPIII, with 16 of 31 (51.6%) carcinomas positive to this marker. The expression pattern in urothelial carcinomas was cytoplasmic, usually with heterogeneous distribution within the tumor (Fig. 4). Intensity was comparatively stronger than that observed with UPIII. Details regarding the results for CK7 in each stage/grade groups are shown in Tables 4 and 5. Compared with UPIII, loss of CK7 expression was statistically significantly associated with increasing grade (P < .02) and stage (P < .001).
In 9 of 16 (56.3%) high-grade urothelial carcinomas, expression of both UPIII and CK7 was considered negative, being positive for the 2 markers in only 1 case (6.3%). Comparing the overall results for both structural proteins, statistically significant differences were found only regarding tumor invasiveness (P < .05).
Cyclin D1 and p53
In total, 16 of 35 (45.7%) tumors tested showed overexpression of cyclin D1. The staining was nuclear and frequently strong (Fig. 5). Details regarding the results in each stage/grade groups are shown in Tables 4 and 5. The differences in overexpression of cyclin D1 in the various tumors analyzed were not statistically significantly associated with grade or stage.
No p53 immunoreactivity was detected in papillomas. In carcinomas, p53 labeling was nuclear and markedly strong, occasionally identified in 80% of the tumor cells (Fig. 6). Details regarding overexpression of p53 in each stage/grade groups are shown in Tables 4 and 5. The differences between grades were statistically significant (P < .05), with p53 overexpression being also significantly associated with tissue invasiveness, Ta vs T1–T2 (P = .01).
Discussion
Positive UPIII reactivity was found in 34.5% of urothelial carcinomas, with the loss of UPIII reactivity being significantly associated with an aggressive/invasive phenotype. Previous studies have revealed that uroplakins are redistributed during neoplastic transformation in cows. 1,21 The heterogeneity of UPIII staining was also reported in canine urothelial tumors. 20 Similar results were found in humans, where loss of UPIII expression was considered a powerful prognostic indicator. 12,16 In a rodent model of chemically induced urinary bladder carcinogenesis, uroplakin expression has been negatively correlated with tumor grade. 15
CK7 labeling was positive and heterogeneous in some cases of urothelial carcinoma. In addition, it was shown that the loss of expression was correlated with high grade (P < .02) and stage (P < .001). A heterogeneous staining pattern for CK7 also has been obtained in canine urothelial carcinomas. 20 In human urothelial carcinomas, CK7 expression seems to be more preserved than in bovine urothelial carcinomas, as it was detected in 82% to 100% tumors. 2,6 The preservation of the CK7 expression is also high in cats (75%) 9 and dogs (98.1%). 20
In summary, the expression of both structural proteins shows little variation within each group of low- and high-grade tumors, becoming quite relevant when referred to the stage, with the expression of CK7 being better preserved, particularly in the muscle-invasive group of tumors, representing the more aggressive phenotype.
In the present study, cyclin D1 overexpression was seen in nearly half of the cases of urothelial carcinomas studied (45.7%), but no statistically significant association was found between stage and grade. A lower percentage was reported previously in endothelial tumors in BEH. 3 The percentage of positive cases was very similar within the various stage/grade groups. Identically, in some studies in humans, it was not possible to correlate cyclin D1 overexpression with any particular pathological features of bladder tumors. 22 However, different authors reported that cyclin D1 overexpression was confined to noninvasive or T1 urothelial tumors in humans. 11
In contrast, in this study, it was shown that p53 overexpression in bovine urothelial carcinomas is significantly associated with both increasing tumor grade (P < .05) and stage (P = .01). The results obtained for p53 in muscle-invasive carcinomas are higher than what has been reported for the muscle-invasive stage hemangiosarcomas in BEH. 3 Mutation and deletion of the p53 tumor suppressor gene are considered the most frequent genetic alterations of bladder cancer in humans and are associated with advanced disease 8 and lower survival rates. 5
In conclusion, the present study showed that the expression of UPIII is lost in high-grade muscle-invasive bladder carcinomas of cows affected by BEH, while other structural proteins such as CK7 seem to be better retained in the process. In addition, the cell cycle regulatory pathways involving cyclin D1 may be compromised in all types of epithelial tumors, including benign ones, with no clear distinction regarding prognostic factors such as muscle invasion. The present work also shows that the more aggressive urothelial tumors are possibly associated with p53 mutations, similar to many other malignancies in humans and animals. It also shows that the more aggressive urothelial tumors are possibly associated with p53 mutations, as in many other malignancies in humans and animals. However, it should be borne in mind that the monoclonal antibodies recognize limited types of mutations. Screening for mutations in tumors negative for p53 could be interesting to validate the immunohistochemistry results.
Footnotes
Acknowledgements
We thank Sandra Carvalho and Maria Rosário Luis for expert technical assistance and Dr Telmo Nunes for his contribution to statistical analysis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research was supported by the Portuguese Foundation for Science and Technology and by the Interdisciplinary Centre of Research in Animal Health (CIISA/FMV).
