Abstract
Despite the increasing importance of cane rat (
Keywords
The cane rat (
Between February and August 1996, three suspected cases of tumors were detected during routine handling. These animals were treated and managed in the same way as the rest of the stock until they showed signs of disease that threatened their survival. Full necropsies were performed by trained technical staff from the farm, and material from the tumors was taken for laboratory investigations. In addition to the affected cane rats reported here, two other animals with suspected tumors died but samples were accidentally damaged and could not be processed.
Samples were fixed in nonbuffered formal saline and kept for several weeks until they could be processed for laboratory investigation. Calcified and bonelike material was sectioned, washed, and decalcified until soft. Samples were then embedded in paraffin, sectioned, and stained with hematoxylin and eosin.
A 36-month-old healthy looking female cane rat (No. 1) from West Africa presented after 2.5 years on the farm with a 4-cm-diameter circular swelling on the right side of the neck involving the jugular vein and vena cava. The mass was calcified and well encapsulated (Fig. 1). Four months later, following 2 days of anorexia, the female was found dead. No other lesions were reported at necropsy.
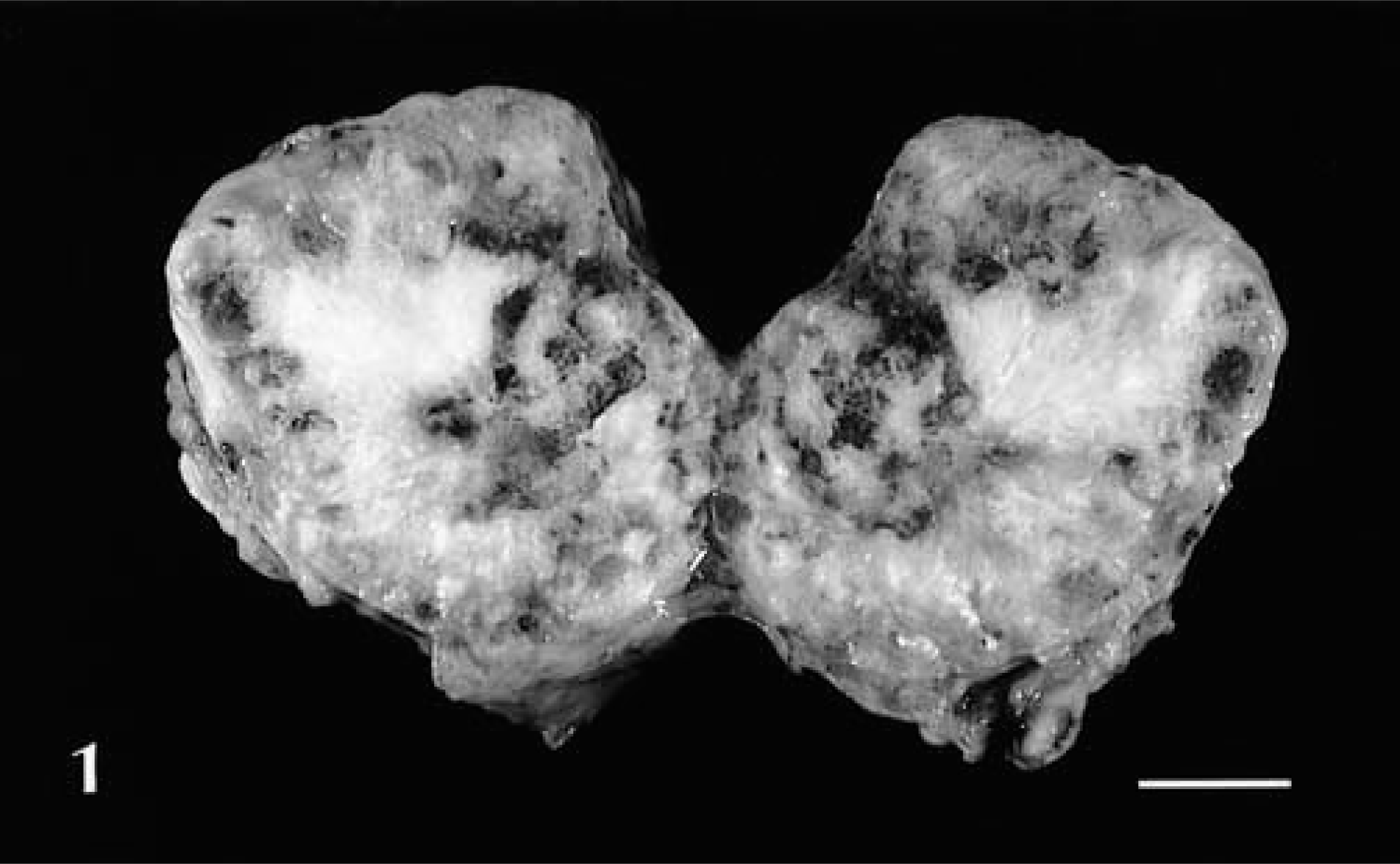
Chondroma; cane rat No. 1. Sagittal section. Note the well-defined encapsulation of the neoplastic mass and the bonelike appearance of the tissue growing from the center. Bar = 1.3 cm.
Histologically, there were areas of cartilaginous tissue growing from a thin layer of perichondrium-like cells. Some of these areas contained very large cells with large nuclei, but in other areas these cells grew in an orderly pattern, resembling normal cartilage. Calcification of the cartilaginous matrix was widespread. There were areas where the mineralized cartilaginous tissue had become necrotic. The growth was classified as a chondroma.
A 6-month-old male cane rat (No. 2) born on the farm was found with a 2.5-cm-diameter swelling on the left side of its rump. The mass, which was well encapsulated and of soft musclelike consistency, protruded 3.5 cm from the body, and the surface of the skin in this area had become hairless and ulcerated. The animal died a few weeks later from unknown causes, and the tumor was collected for histopathologic analysis. No lesions were observed in other organs.
The neoplastic tissue was composed of large fusiform and rounded, elliptic cells exhibiting diffuse growth, with <5% stroma. The margin of the lesion was irregular and poorly defined. The mitotic index was low. Many neoplastic cells showed karyorrhectic nuclei with margination and fragmentation of the chromatin. Multinucleated cells were present in small numbers. The tissue was well vascularized throughout, with many distended capillaries filled with erythrocytes and sometimes thrombosed. The neoplastic tissue was infiltrated diffusely by degenerating neutrophils. A zone showed bacterial colonization, possibly due to secondary infection of the ulcerated area. The lesion was classified as an hemangiosarcoma on the basis of the proliferation and abundance of blood vessels and endothelial cell polymorphism (Fig. 2).
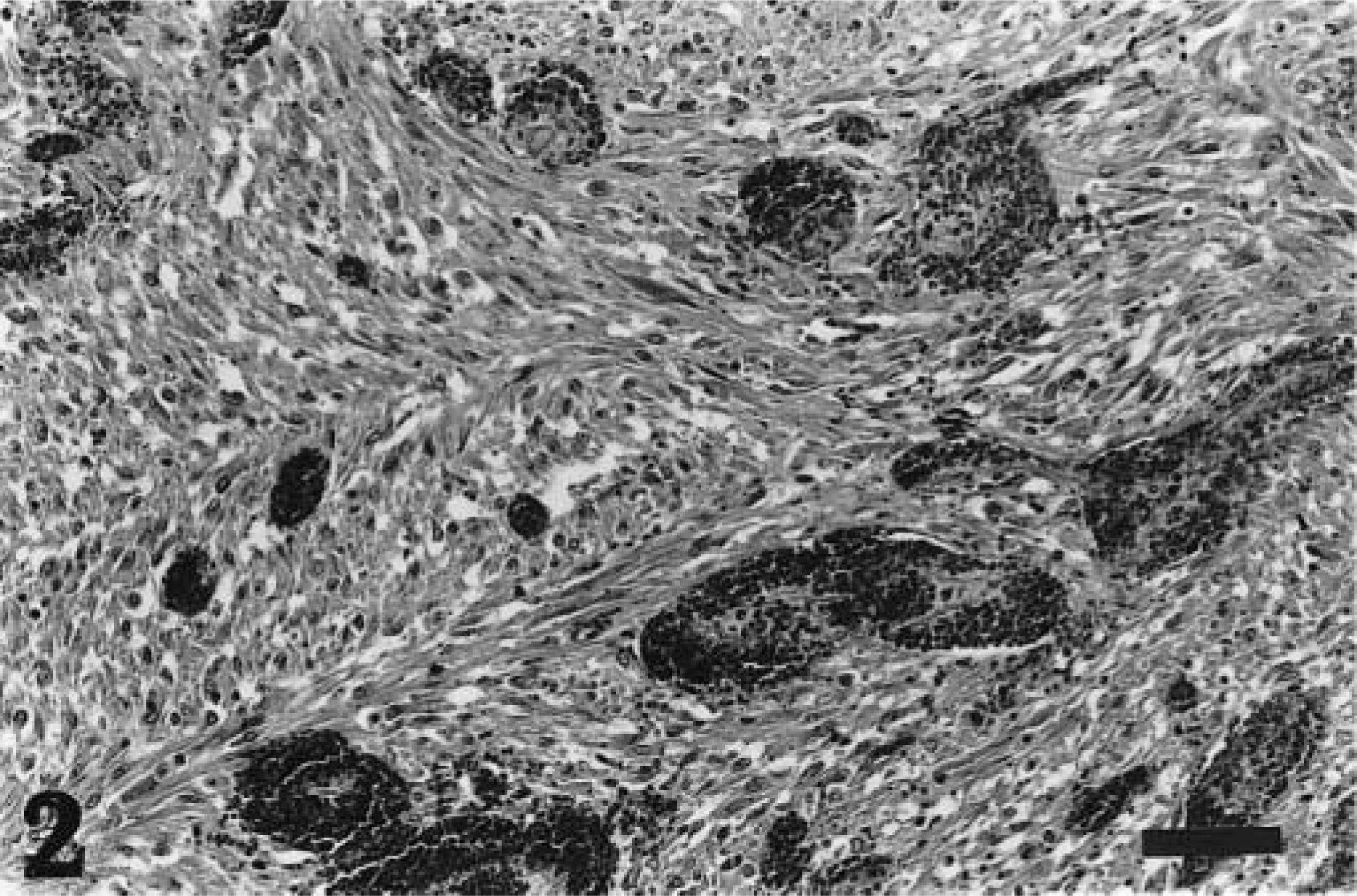
Hemangiosarcoma; cane rat No. 2. Neoplastic tissue composed of large fusiform and rounded cells in a stroma that is well vascularized, giving a cavernous appearance. HE. Bar = 20 µm.
After 2 years on the farm, a 29-month-old female cane rat (No. 3) imported from West Africa was found with an invasive, multilobular, calcified mass affecting its left hind leg. The swelling was hard and invaded the whole leg from the femur to the interphalangeal area. However, the animal was in good condition, ate well, and did not show other signs of disease except impaired locomotion. The hind limb was surgically excised under general anesthesia, but the animal died 2 days later from postoperative complications.
Margins of the lesion were sharp. Samples contained normal bone tissue and an adjacent neoplastic proliferation of chondroid tissue (Fig. 3), which arose multifocally from a thin layer of perichondrium-like cells. Chondroid cells were pleomorphic, with aberrant contours and large nuclei. Mitoses were not visible. There was extensive mineralization of the chondroid matrix, and near these areas groups of large spindle cells formed nonmineralized bone matrix. In one area, neoplastic chondroid tissue was present near the normal long bone. Many areas of the chondroid tissue showed necrosis. The growth was classified as a chondrogenic osteosarcoma.
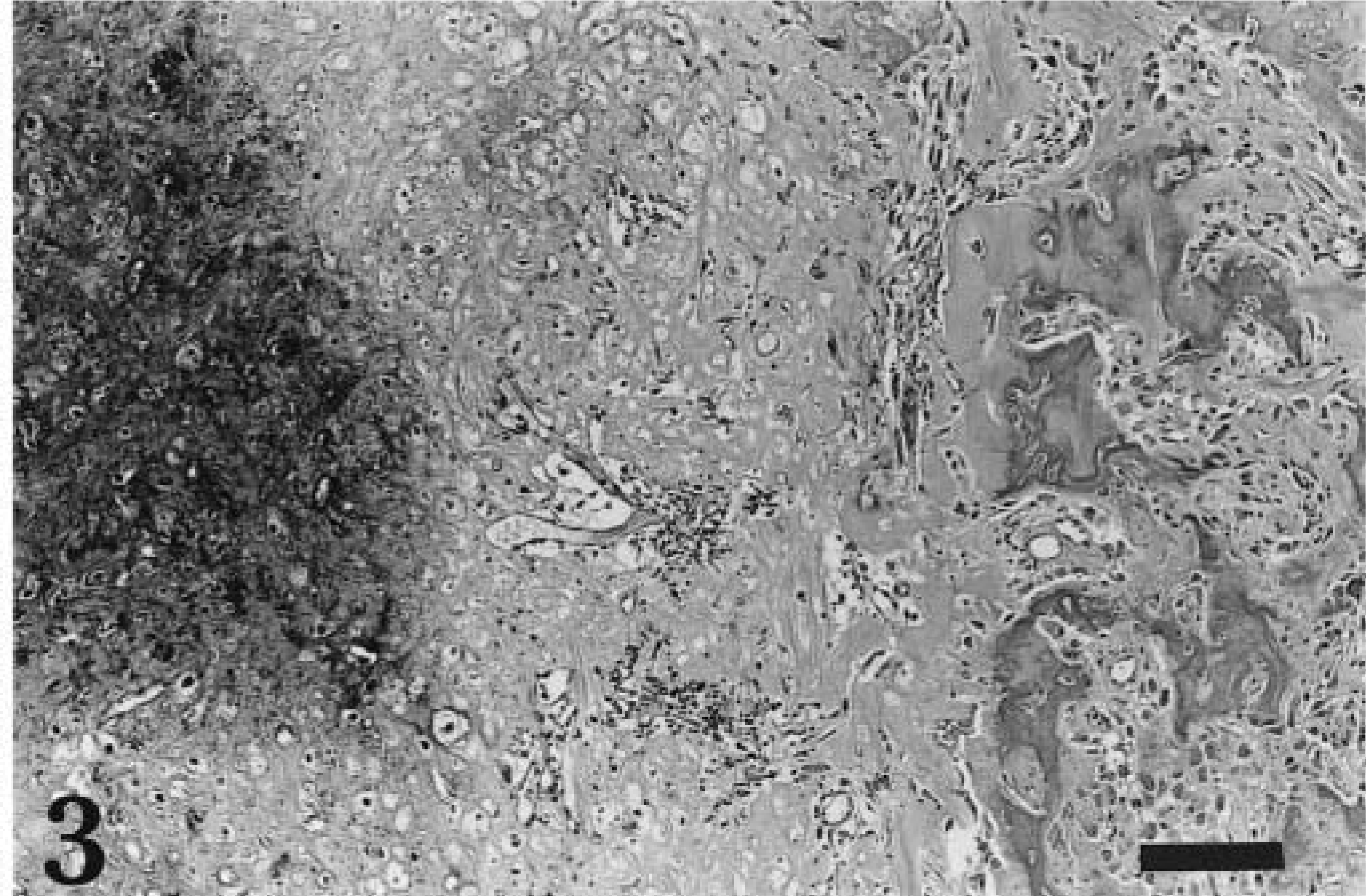
Chondrogenic osteosarcoma; cane rat No. 3. Gradual transition of osteoid tissue, some of it mineralized (left), into chondroid tissue with chondroblasts (right). HE. Bar = 50 µm.
Most information about tumors in rodents concerns laboratory and pet animals, but the captive breeding of wild rodents allows investigation of diseases of species for which very little information has been published to date. 1,4 The present report is the first record of tumors and is one of the few reports of pathologic changes in cane rats in captivity. 4 Two of the tumors described here involved chondrogenic tissue. This finding is uncommon in pet and laboratory animals; 6,9 bone tumors are occasionally reported in wild mice and gerbils. 7,8 Many lesions diagnosed as chondromas are actually low-grade chondrosarcomas, 6 and both types of tumor can be equivocal. In mice, spontaneous tumors with an osseous appearance appear to be potentially malignant. 10 Osteosarcomas have been occasionally reported in wild 1 and laboratory rodents; most are found in aged females and are located in the hind legs. 8,10 No metastatases were observed, and the histologic appearance of the tumor in cane rat No. 1 was such that it seemed unequivocally benign, and confusion with an osteosarcoma appears unlikely.
Hemangiosarcomas are more common in pet and laboratory rodents, being reported in gerbils, mice, and rats. 9 These tumors are highly malignant and metastasize easily because they have easy access to vascular channels. Unfortunately, the existence of possible metastases could not be determined in the present study.
After 3 years of monitoring an average of 400 cane rats from several farms in Gabon, no other cases of neoplasms were detected and none were reported in the literature. The tumors described here appeared over a period of 8 months, and at least three suspected cases presented simultaneously within a 3-month period. Therefore, the incidence of 3/235 (1.3%) in the colony seems abnormally high. The causes of these tumors in this colony remain unclear. However, because of the size and nature of the lesions described, it is doubtful that detection ability was influenced by closer attention.
In some rodent species, frequency of neoplasms increases with age. 7,9 However, ageing was not considered a predisposing factor in this colony. Cane rats can live for up to 4 years in captivity, 3 and only cane rat No. 1 might be considered old. Cane rat No. 2 was only 6 months old, and another affected rat not reported here was <1 year old. A high inbreeding coefficient has been suggested as a possible cause for high tumor prevalence in hamster colonies 2 but seems an unlikely factor in the present cases because the founder colony came from at least eight separate farms and the cane rats reported here included founders and descendants from the same colony. The sudden appearance of tumors in a short period and in animals of different ages and sexes may indicate an oncogenic virus on the farm. DNA polyomaviruses are enzootic in wild and laboratory mice, although tumor production seems to be a rare manifestation of the infection. Bone tumors and hemangioendotheliomas of laboratory rodents can be induced in experimentally infected newborns. 5 Several types of RNA C-type oncornaviruses induce bone tumors in mouse colonies; these neoplasms develop in the cranium, vertebral column, and ribs. 8 Chemical agent intoxication through contaminated food can induce cancer in different soft tissues in laboratory rodents. Green fodder for the cane rats was recovered from peri-urban industrial areas, so the possibility of contamination cannot be excluded. Further toxicologic analysis is needed to identify possible carcinogenic products in the cane rat diet.
The etiology of the tumors reported here remains unknown. However, evaluation of additional tumor cases should be encouraged to elucidate the etiology and prevalence of tumors in farmed cane rats.
Footnotes
Acknowledgements
We are very grateful to Dr. Mariano Domingo, Dr. Neus Prats, Pere Losada (Spain), and Blanca Pérez from the Veterinary School of Barcelona, and to Greendale Laboratories (UK) for their help in preparing histologic material. We also thank the staff of the VSF project, Coopération Française, and the Gabonese government for their contribution. This study formed part of the Pathology Programme at the Jersey Zoo funded by the Rufford Foundation.
