Abstract
Porcine circovirus (PCV)-2, a newly described single-stranded circular DNA virus pathogen of swine is the cause of postweaning multisystemic wasting syndrome (PMWS). In gnotobiotic piglets, PCV-2 infection alone produces asymptomatic infection without evidence of overt PMWS. Gnotobiotic piglets infected with PCV-2 were injected with keyhole limpet hemocyanin in incomplete Freund's adjuvant (KLH/ICFA), and the effects on virus production and development of PMWS were determined. In the first experiment, piglets were injected subcutaneously on the left hip and shoulder, and viral burden was assessed in regional lymph nodes draining the injection sites and in contralateral lymph nodes 13–14 days after infection. Immune activation increased the number of virus antigen-positive cells in draining lymph nodes and increased the amount of infectious virus recovered by 1–4 log10. In a second experiment, the effects of injections of KLH/ICFA with or without concurrent stimulation of peritoneal macrophages by intraperitoneal injections of thioglycollate broth on induction of PMWS was assessed. All immunized piglets developed moderate to severe PMWS, whereas none of the piglets infected with PCV-2 alone developed PMWS. In PMWS-affected piglets, extensive replication of PCV-2 was documented by both immunocytochemistry and quantitative viral titrations. Thus, immune activation is a key component of the pathogenesis of PCV-2-associated PMWS in swine.
It is now established that porcine postweaning multisystemic wasting syndrome (PMWS) is causally associated with infection by porcine circovirus-2 (PCV-2), a newly described circovirus 22 pathogen of swine. 1 Progressive wasting disease was first identified in several “high health” herds from western Canada 9 15 as a distinct entity with a high mortality rate and a spectrum of clinical signs, including progressive weight loss, jaundice, and respiratory disease. Gross lesions characteristic of PMWS include generalized lymphadenopathy, hepatitis with icterus and edema, nephritis, and pneumonia. Histologically, the disease features systemic angiocentric lymphoplasmacytic and histiocytic to granulomatous inflammation, syncytial giant cells, and the variable presence of prominent, intracytoplasmic basophilic viral inclusions, chiefly within macrophages. Since that first description, PMWS has been reported in swine throughout the world. 5 7 23 26 The prevalence of subclinical infection without overt clinical expression appears to be high. 1 Increasingly, clinical syndromes not overtly expressed as wasting disease are being associated with PCV-2 infection.
Little is known about the pathogenesis of this disease, critical pathogenic events in pigs, virulence determinants, and method of spread within a herd. Attempts to elucidate the natural history of the disease under field conditions are frustrated by the episodic nature of the wasting complex, the inconsistency of signs and lesions of PMWS within an infected herd or between infected herds, and the high incidence of asymptomatic infection with PCV-2 in PMWS-negative herds.
Reproduction of the clinical and pathologic features of PMWS in both conventional and gnotobiotic swine by inoculation of materials containing PCV-2 has now been accomplished. 2 6 14 20 However, unlike the infection experiments performed in conventional piglets, 2 experimental infection of gnotobiotes with PCV-2 alone produces mild, asymptomatic infection, modest lymphadenopathy, but no other manifestations of infection or inflammation in parenchymal organs. Wasting disease does not occur in infected gnotobiotes, 20 viral antigen is sparsely distributed, and lymphoid tissues show rare PCV-2–positive cells restricted to dendritic cells of germinal centers or medullary regions of lymph nodes. When gnotobiotes are coinfected with porcine parvovirus (PPV) and PCV-2, moderate to severe wasting disease is seen with deaths occurring between 25 and 35 days of age. 20 In these dually infected piglets, PCV-2 infection is widespread throughout all lymphoid tissues and within granulomatous lesions in parenchymal organs; PPV is not. Aside from the well-known effects of PPV on developing fetuses, postnatal infection with PPV is not associated with significant clinical disease. 10 20 Thus, in gnotobiotes, neither PCV-2 nor PPV, as single infections, are pathogenic. Both agents are nonenveloped DNA viruses highly resistent to disinfectants and chloroform and both have a strong cellular tropism for histiocytic cell lineages. 1 4 16 24 25 Replication of circoviral DNA is dependent upon host cell enzymes expressed during the S-phase of the cell cycle, and replication of viral DNA is initiated only after the cells have experienced mitosis. 1
The mechanism(s) whereby PPV potentiates PCV-2 and results in PMWS in dually infected gnotobiotes are not known. Although some field cases of PMWS are coinfected with PPV, 13 most are not, a finding that suggests that PPV is not obligatory for expression of PCV-2–related PMWS. Other pathogens such as the porcine reproductive and respiratory syndrome (PRRS) virus have been found to potentiate PCV-2 disease. 3 17 There are two general but not mutually exclusive mechanisms whereby PPV might promote PMWS in gnotobiotes. Parvoviruses are lympho- and monocytotropic 16 25 and have been associated with transient immunosuppressive effects (lymphopenia, reduced blastogenesis, etc.) in swine. 24 25 PPV-associated immunosuppression may account for PMWS in dually infected animals. Alternatively, since both are DNA viruses and depend upon cellular DNA synthesis for optimal virion production, stimulation of host DNA synthesis by PPV could inadvertently promote PCV-2 replication. Since the chief cellular target of both viruses appears to be macrophages and histiocytes, local and systemic immunostimulation with a noninfectious irrelevant immunogen incorporated into a strong adjuvant should potentiate PMWS in gnotobiotic piglets infected with PCV-2 alone. In the data reported here, we tested the hypothesis that PCV-2 virion production in vivo is upregulated as a result of immune stimulation, and this effect is mediated by activation of macrophages/histiocytes contained within lymphoid tissues. As a consequence of local upregulation, virion production is enhanced or prolonged and PCV-2 is disseminated to all tissues, resulting in PMWS in immunostimulated gnotobiotic piglets.
Materials and Methods
Piglets
Three date-mated pregnant sows were purchased from a closed-confinement swine operation, and a total of 27 piglets were derived by Caesarian section 18 into sterile pentub isolation units, 4–6 piglets per unit. To maintain biosecurity, uninfected control piglets were housed in separate self-contained isolation units.
Virus
A tissue culture–propagated strain of PCV-2 was used as the starting source of viral inoculum. 5 12 Virus was propagated in PCV-free PK15 cells 11 and confirmed to be PCV-2 by nucleotide sequence analysis 20 and reactivity with PCV-2–specific monoclonal antibody as described. 20 For challenge, a viral pool was prepared from a 10% (w/v) lymphoid tissue homogenate collected from gnotobiotic piglets infected with the 24th in vitro passage of this virus. 20 The inoculum contained 4.3 × 106 infectious viruses ml when titrated on PCV-free 11 porcine kidney cell monolayers.
Experimental design
The piglets were divided into short- and long-term experiments. For the short-term experiment, 11 piglets were used. Seven piglets were oronasally inoculated with 0.5 ml of PCV-2 inoculum/nare at 1 day of age; four received saline alone. At 3 days of age, four infected piglets were injected with a 2.0-ml volume of 1.0 mg keyhole limpet hemocyanin (KLH) emulsified in incomplete Freund's adjuvant (ICFA) in two sites: the left axilla and left hip (1.0 ml/site). The left-side injections were repeated at 7 days of age. The three remaining PCV-2–infected piglets were injected with KLH in saline. Uninfected controls were injected with KLH in saline (n = 2) or KLH/ICFA (n = 2). The piglets were terminated at 13–14 days of age for determination of the titer of infectious virus in lymphoid tissues, presence of histologic lesions, and distribution of viral protein by immunohistochemistry. Two hours prior to termination, replicating DNA as an index of cellular proliferation was labeled in situ with 200 µg bromo-desoxiuridine (BrDU) as described. 19
In the second (long-term) experiment, 16 piglets from two litters were used. Ten piglets were inoculated with the same PCV-2 pool as before at 1 day of age. At 3 days of age, piglets were injected in four sites (both axillas and both hips) with KLH/ICFA as above (n = 3) or were injected in four sites with KLH/ICFA and also given an intraperitoneal (IP) injection of 10.0 ml of thioglycollate broth (glycan) to stimulate peritoneal macrophage exudation into the peritoneal cavity (n = 4). The parenteral injections with KLH/ICFA were repeated at 7 days of age, and piglets received additional IP injections of glycan at 10 and 20 days of age. Three PCV-2–infected piglets were not immunized. Controls consisted of uninfected piglets injected with KLH/ICFA and IP glycan (n = 3) on the same schedule, and uninfected piglets that were not injected with adjuvant (n = 3). These piglets were terminated when moribund or on postinfection day (PID) 35 after in situ labeling with BrDU. 19
Tissue collection, histopathology, and immunocytochemistry
Samples of prescapular or axillary/prescapular and superficial inguinal lymph nodes draining injection sites; contralateral axillary/prescapular and inguinal lymph nodes; bronchial, mesenteric, and mandibular lymph nodes; spleen; and thymus, as well as liver, kidney, and lungs were aseptically collected from each piglet. Tissue samples were divided into fragments for virus reisolation and histopathologic examination. For the former, samples were immediately frozen in liquid nitrogen and stored at −70 C for quantitative virus titrations. For the latter, tissues were collected into labeled cassettes and fixed by immersion in 10% (v/v) phosphate-buffered formalin and processed by routine methods. Six-micron section replicates were stained with hematoxylin and eosin and Masson's trichrome stain for morphologic evaluation and for PCV-2 antigen using monoclonal antibody specific for PCV-2. 14 Section replicates were stained for incorporated BrDU by an indirect immunoperoxidase method. 19 Briefly, deparaffinized sections were quenched for endogenous peroxidase with 3% H2O2 in PBS, reacted with purified monoclonal antibody or monoclonal antibody–containing cell culture supernatants, blocked with 4% (v/v) heat-inactivated horse serum, reacted with biotinylated horse anti-mouse IgG, reacted with avidin–peroxidase, and visualized with DAB (Vectastain®).
Virology
Quantitative virus titrations were performed in microtiter plates on subconfluent monolayers of PK15 cells treated with glucosamine. 1 5 12 In the short-term experiment, viral titrations were performed on individual tissue samples from each piglet. In the long-term experiment, lymphoid tissues from each piglet were pooled and titrated as a set; homogenates of liver from each piglet were also separately assayed for infectious virus. A 10% (w/v) suspension of each freshly thawed tissue was made in Hank's Balanced Salt Solution (HBSS) with antibiotics and 2.5% HEPES buffer and homogenized with sterile Tenbroeck grinders or an ultraspeed tissue homogenizer. Homogenates were clarified by low-speed centrifugation and tenfold dilutions of each were made into medium. Four replicates of each dilution (50 µl) were inoculated into microtiter wells and 50 µl of medium with glucosamine was added. After 24 hours, monolayers were washed, and 200-µl tissue culture medium who added to each well. Five days later, supernatants were discarded and monolayers were fixed in acetone. Viral antigen was visualized by indirect immunoperoxidase method using first monoclonal antibody to PCV-2, biotinylated horse antimurine IgG, and streptavidin-conjugated peroxidase. Bound monoclonal antibody was visualized with DAB. Titers were determined by the 50% end-point method of Reed and Muench. 21
Results
Experiment 1 (local immunostimulation)
Gross lesions
No overt clinical evidence of PMWS was observed in either PCV-2–infected or control piglets. At necropsy on PID 13/14, lymph nodes draining the injection sites were three- to fourfold enlarged and edematous. Contralateral corresponding lymph nodes from these piglets were small and inactive. In uninfected KLH/ICFA-injected piglets, lymph nodes draining the injection sites were also enlarged, reddened, and edematous, whereas contralateral corresponding lymph nodes were equivalent to controls. Lymph nodes in piglets infected with PCV-2 alone were small, inactive, and indistinguishable in size from uninfected controls injected with KLH in saline. All piglets that received KLH/ICFA, but not KLH in saline, developed subcutaneous pyogranulomatous inflammatory lesions at the injection sights.
Histopathology and BrDU incorporation
Lymph nodes draining KLH/ICFA injection sites were histologically similar, regardless of PCV-2 infection status. The most prominent feature was the appearance of numerous vacuoles, presumed to be lipid contained within the ICFA, entrapped within medullary sinuses. Accompanying these lipid vacuoles was a mixed, reactive, predominantly granulomatous inflammatory response. Reactive lymphoid hyperplasia and germinal center formation were present. In addition, eosinophil and neutrophil infiltrates with formation of microabscesses was seen in some immunized nodal tissues. In contrast, contralateral lymph nodes from KLH/ICFA-injected piglets, lymph nodes from piglets infected with PCV-2 alone, and uninfected control piglets injected with KLH in saline were inactive. Lipid vacuoles were not observed, mixed granulomatous inflammation was not present, germinal centers were rare and underdeveloped, and associated lymphoid hyperplasia was absent. A mild multifocal histiocytic interstitial inflammatory cell infiltrate was seen in the liver of one PCV-2–infected KLH/ICFA-injected piglet. Except for mild vacuolar degeneration and glycogen accumulation in hepatocytes, microscopic lesions were not seen in any other tissue from piglets in any other infection treatment group.
Section replicates stained for incorporated BrDU into newly synthesized cellular DNA identified incorporated intranuclear BrDU in numerous cells contained within lymph nodes draining KLH/ICFA injection sites. BrDU was incorporated into macrophage/histiocytes within granulomas and regions of reactive lymphohistiocytic hyperplasia and was also free within the medullary sinuses (Fig. 1A). In addition, lymphocytes chiefly associated with germinal centers stained positively for BrDU. In contrast, only a few labeled cells, chiefly histiocytes, were seen in contralateral unstimulated lymph nodes (Fig. 1B). This BrDU staining pattern in unstimulated draining lymph nodes was indistinguishable from that seen in distal lymphoid tissues, and a similar pattern of scattered positive-staining cells was seen in uninfected piglets injected with KLH in saline. In piglets infected with PCV-2 alone, occasional histiocytes incorporated BrDU; in general, the pattern, intensity, and frequency of this staining was similar to that seen in uninfected control piglets.
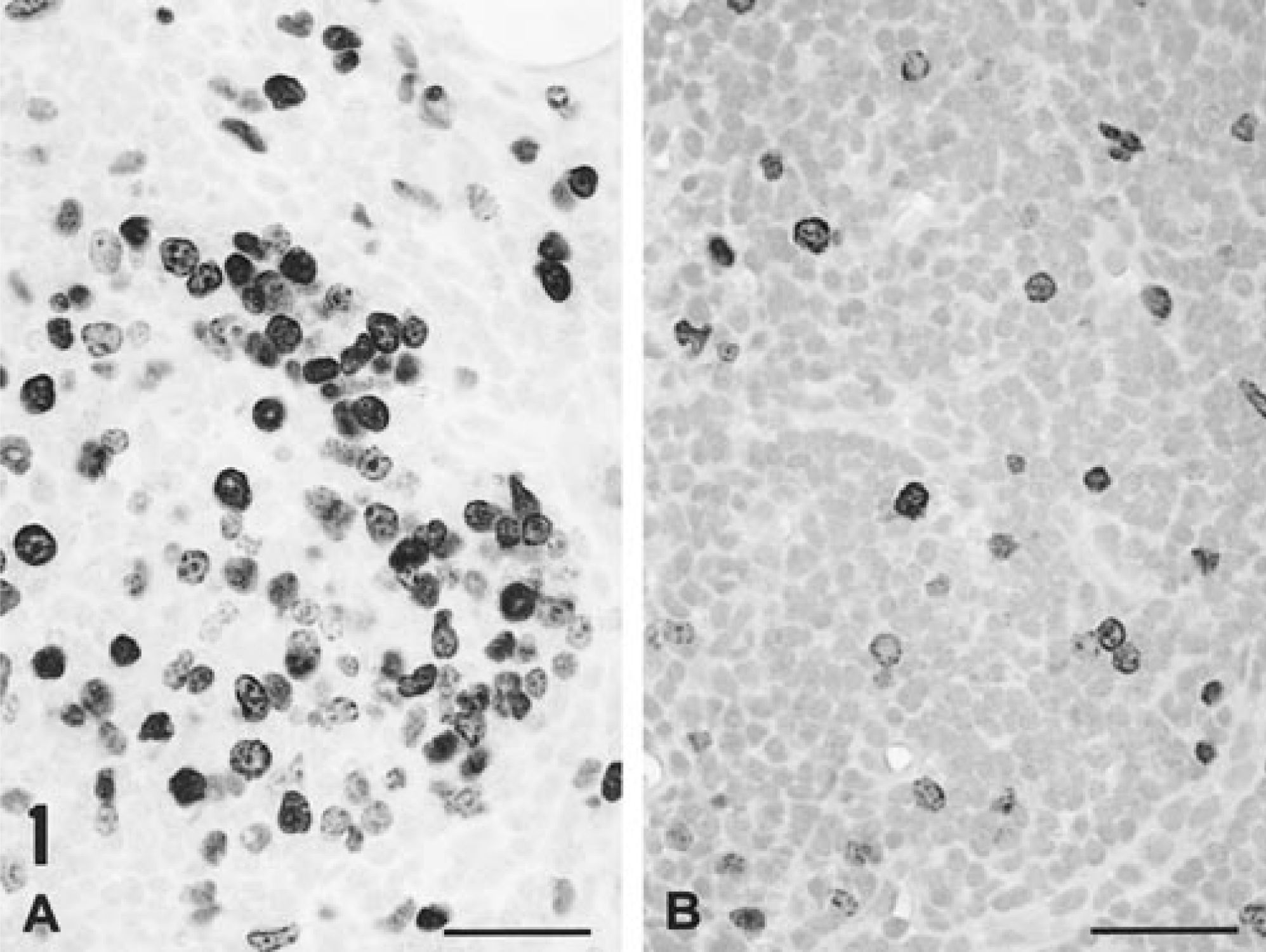
In situ BrDU incorporation into nuclear DNA in an axillary lymph node draining an immunization site 14 days after infection with PCV-2 (
Virology
Section replicates were stained for PCV-2 antigen with monoclonal antibody specific for PCV-2. Unequivocal accumulation of staining product was restricted to lymphoid tissues and was only seen in PCV-2–infected piglets injected subcutaneously with KLH/ICFA. In these piglets, the relative amounts of viral antigen in lymphoid tissues varied by anatomic site. Single or widely scattered virus-positive cells, primarily macrophages, were seen in some or all lymphoid tissues examined, except thymuses. In contrast, lymph nodes draining injection sites contained multiple foci of infected macrophages both within organizing granulomas and associated with the lipid adjuvant deposits. Parenchymal cells in parenchymal tissues (liver, kidney, lungs, myocardium) were virus antigen-negative. Viral antigen was not detected in the three piglets infected with PCV-2 alone or in the uninfected control piglets.
Table 1 summarizes the results of quantitative viral titrations performed on lymphoid tissues from the piglets of short-term experiment 1. PCV-2 was recovered only from piglets infected with PCV-2; tissues from uninfected controls were uniformly negative for infectious virus. The amount of recoverable virus varied not only by anatomic sight within infected piglets but also by the type of immunostimulation regimen used. The highest amount of infectious virus (103 to >106 infectious units/g tissue) was recovered from lymph nodes draining sites of injection with KLH/ICFA. In every instance, contralateral lymph nodes from these same piglets contained lower amounts of virus (≤102 infectious units/g tissue). PCV-2 was also recovered from distal lymphoid tissues. Bronchial lymph node homogenates contained lower levels of infectious virus than did distal nodes draining injection sites. The thymus was the only lymphoid tissue that did not contain recoverable PCV-2. Piglets infected with PCV-2 and injected with KLH in saline contained sparsely distributed, recoverable PCV-2. Bronchial node homogenates from one of three infected piglets was devoid of recoverable virus; low levels of virus were recovered from the KLH in saline-immunized inguinal lymph node of one piglet in this group.
Titratable infectious PCV-2 in tissues from piglets infected with PCV-2, injected with KLH/ICFA in the left axilla and hip, and terminated on postinfection day (PID) 13/14.
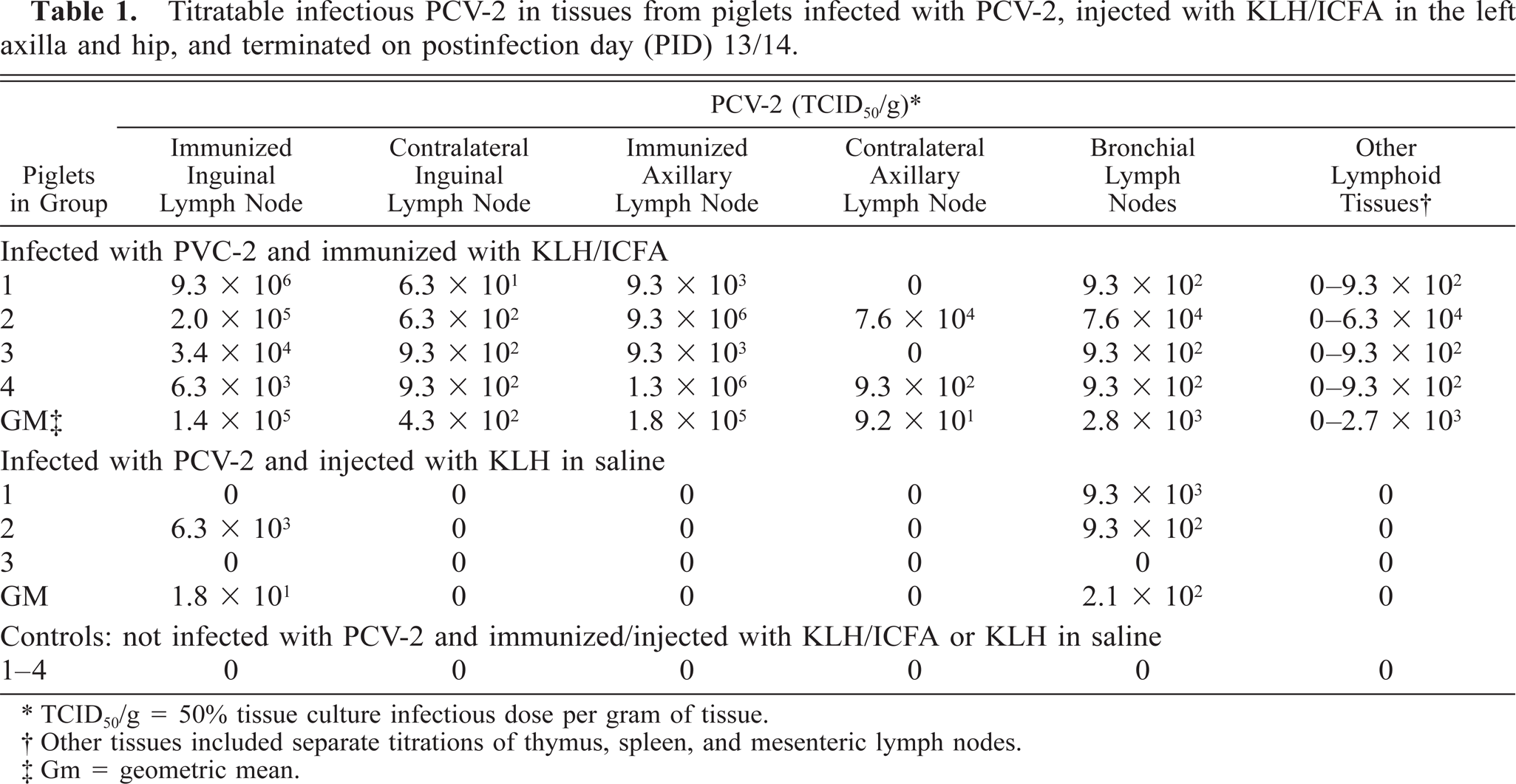
TCID50/g = 50% tissue culture infectious dose per gram of tissue.
Other tissues included separate titrations of thymus, spleen, and mesenteric lymph nodes.
Gm = geometric mean.
Experiment 2 (systemic immunostimulation)
Gross findings
Clinical findings and salient gross features in piglets injected subcutaneously with KLH/ICFA and with or without IP glycan are summarized in Table 2. Unlike experiment 1, all PCV-2–infected piglets injected with adjuvant exhibited signs and lesions of PMWS. One piglet died suddenly on PID 29; the remaining infected piglets were terminated with severe clinical signs of PMWS (PIDs 28, 29, 32) or with signs of moderate to severe PMWS on PID 35. A consistent finding was anasarca, which varied in severity between infected piglets. The majority (5/7) exhibited mild to severe icterus. Piglets infected with PCV-2 but not immunostimulated with adjuvant were clinically normal at termination on PID 35, as were piglets from both uninfected control groups.
A summary of gross lesions and clinical findings in gnotobiotic piglets infected with PCV-2 and injected with KLH/ICFA with or without thioglycollate (glycan) intraperitoneally (IP).
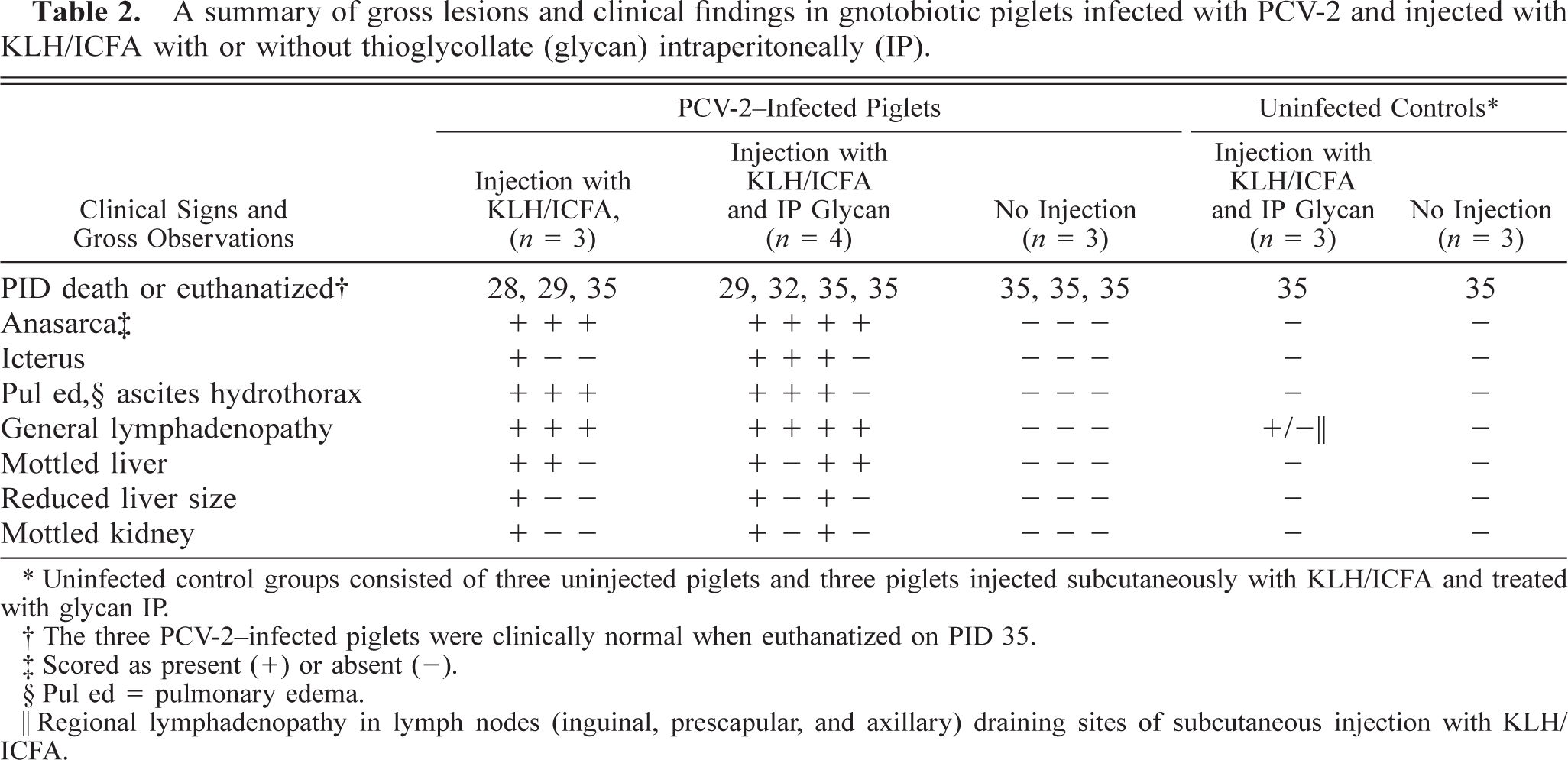
Uninfected control groups consisted of three uninjected piglets and three piglets injected subcutaneously with KLH/ICFA and treated with glycan IP.
The three PCV-2-infected piglets were clinically normal when euthanatized on PID 35.
Scored as present (+) or absent (-).
Pul ed = pulmonary edema.
Regional lymphadenopathy in lymph nodes (inguinal, prescapular, and axillary) draining sites of subcutaneous injection with KLH/ICFA.
Gross lesions in PCV-2 infected KLH/ICFA-injected piglets were compatible with PMWS. Icterus was confirmed, and all piglets had lesions of generalized edema (anasarca, ascites, pulmonary edema, hydrothorax, perirenal edema, and edema of the mesentery, especially prominent around the pancreas). Generalized lymphadenopathy was seen in all infected and immunostimulated piglets, although within individual piglets, the severity of nodal enlargement varied somewhat by anatomic sight. The least frequently involved nodes were those of the mesenteric chain; the most commonly involved were draining nodes from injection sites and bronchial nodes. Gross hepatic lesions of mottling, paleness (fatty degeneration and necrosis) were prominent in five of seven piglets. In addition, there was a striking reduction in liver parenchyma in three of seven animals. In these piglets, total liver volume was reduced to approximately one-third of normal. On cut section, stromal connective tissue elements were prominent within the reduced hepatic parenchyma. In three piglets, kidneys were mottled and enlarged and contained multiple petechial/ecchymotic hemorrhages. A deep gastroesophageal ulcer with associated melena was seen in one piglet. In the piglet that died suddenly on PID 29, peritoneal exudation and mesenteric fat necrosis associated with moderate to severe pancreatic necrosis was observed. Peritonitis anticipated to be present and associated with the IP injections of thioglycollate was not detected in the other animals of this group.
Gnotobiotic piglets inoculated with PCV-2 and not injected with KLH/ICFA were clinically asymptomatic at termination on PID 35. At necropsy, the only evidence of PCV-2 infection was modest lymphadenopathy of bronchial lymph nodes and inconsistent lymphadenopathy elsewhere. Except for regional lymphadenopathy of lymph nodes draining adjuvant injection sites, uninfected control piglets were clinically normal and free of gross lesions of PMWS.
Histopathologic findings
Histologic lesions in piglets inoculated with PCV-2 were widespread and prominent in all immunized piglets and minimal or absent in nonimmunized piglets (Table 3). Control uninfected piglets had no histologic evidence of PMWS. Lesions in piglets infected with PCV-2 alone were similar to those reported previously, 20 in that modest lymphoreticular hyperplasia with germinal center formation was present in all lymph nodes examined (Fig. 2A, B). Multifocal lymphoplasmacytic inflammatory cell infiltrates were present in liver sections from all three piglets, and a similar lesion was seen in the myocardium on one of them.
A summary of salient histopathologic lesions in gnotobiotic piglets infected with PCV-2 and injected with KLH/ICFA∗ with or without thioglycollate (glycan) intraperitoneally (IP).
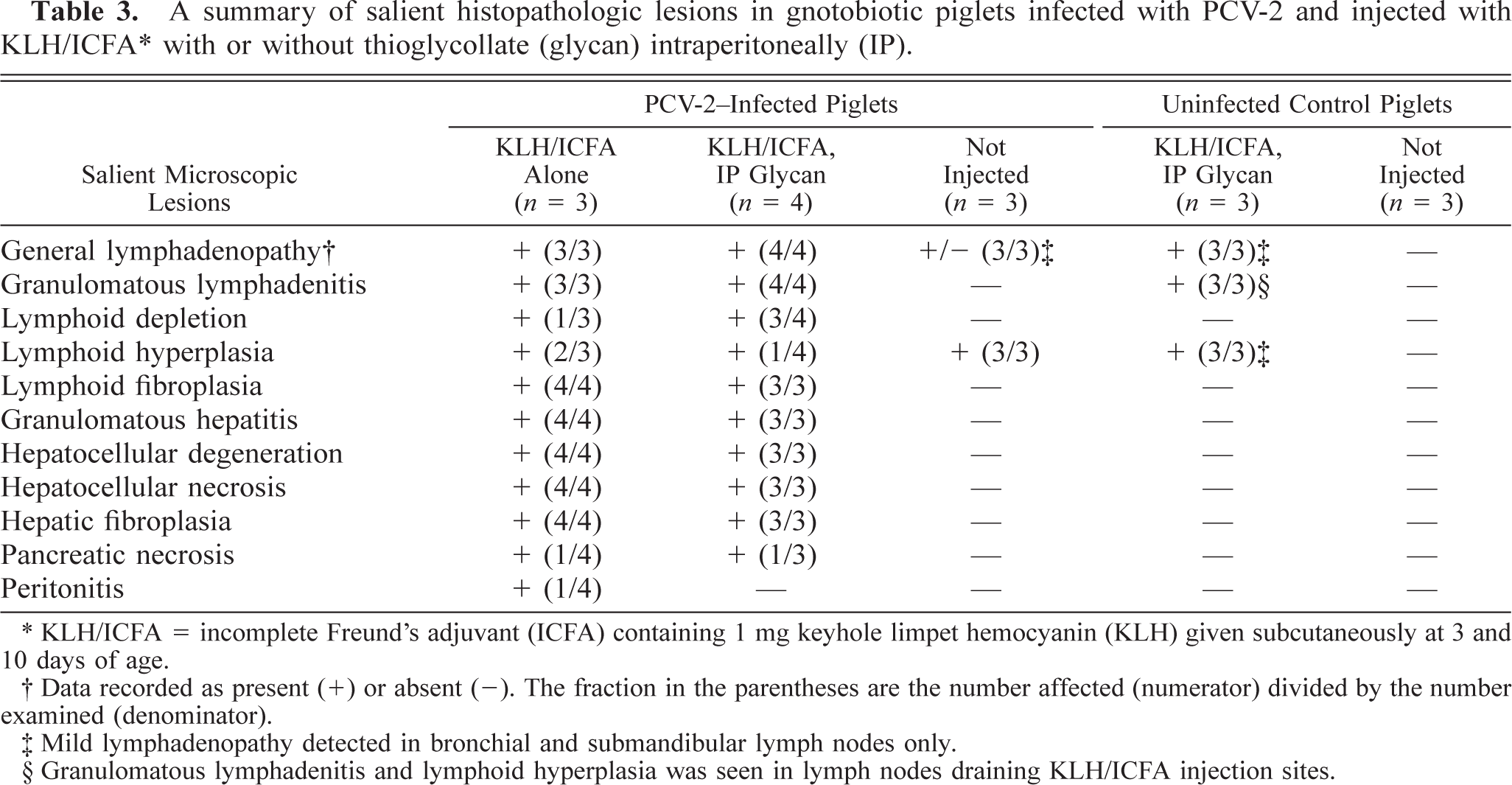

KLH/ICFA = incomplete Freund's adjuvant (ICFA) containing 1 mg keyhole limpet hemocyanin (KLH) given subcutaneously at 3 and 10 days of age.
Data recorded as present (+) or absent (-). The fraction in the parentheses are the number affected (numerator) divided by the number examined (denominator).
Mild lymphadenopathy detected in bronchial and submandibular lymph nodes only.
Granulomatous lymphadenitis and lymphoid hyperplasia was seen in lymph nodes draining KLH/ICFA injection sites.
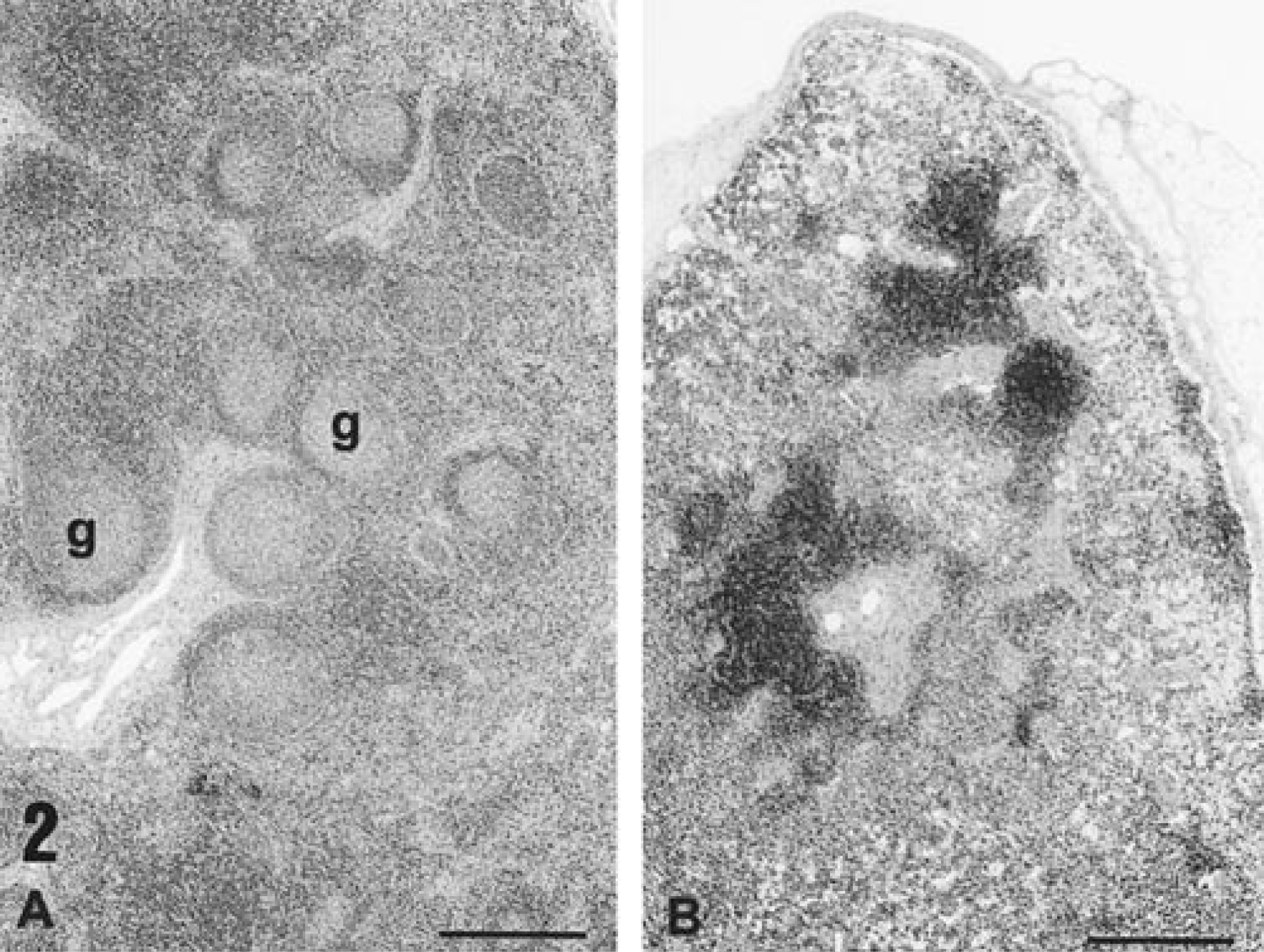
Histologic appearance of a lymph node from a piglet infected with PCV-2 alone 35 days previously. Note the germinal centers (g) and the increased cellularity of the lymph node (
In contrast and as documented previously in piglets coinfected with PCV-2 and PPV, 20 the primary inflammatory lesion of PMWS in PCV-2–infected gnotobiotes injected with KLH/ICFA or KLH/ICFA and IP glycan was widespread disseminated angiocentric lymphocytic granulomatous inflammatory cell infiltration of parenchymal tissues and lymph nodes. The range of PCV-2–induced granulomatous inflammation and associated lymphoid depletion, unaffected by ICFA-associated granulomatous lesions, is illustrated in sections from the spleens of two PMWS-affected piglets (Fig. 3A, B). Lymphoid depletion was present in each animal. The amount of histiocytic proliferation varied between these animals but was, in all cases, prominent when compared to uninfected controls or to spleens from piglets infected with PCV-2 alone.
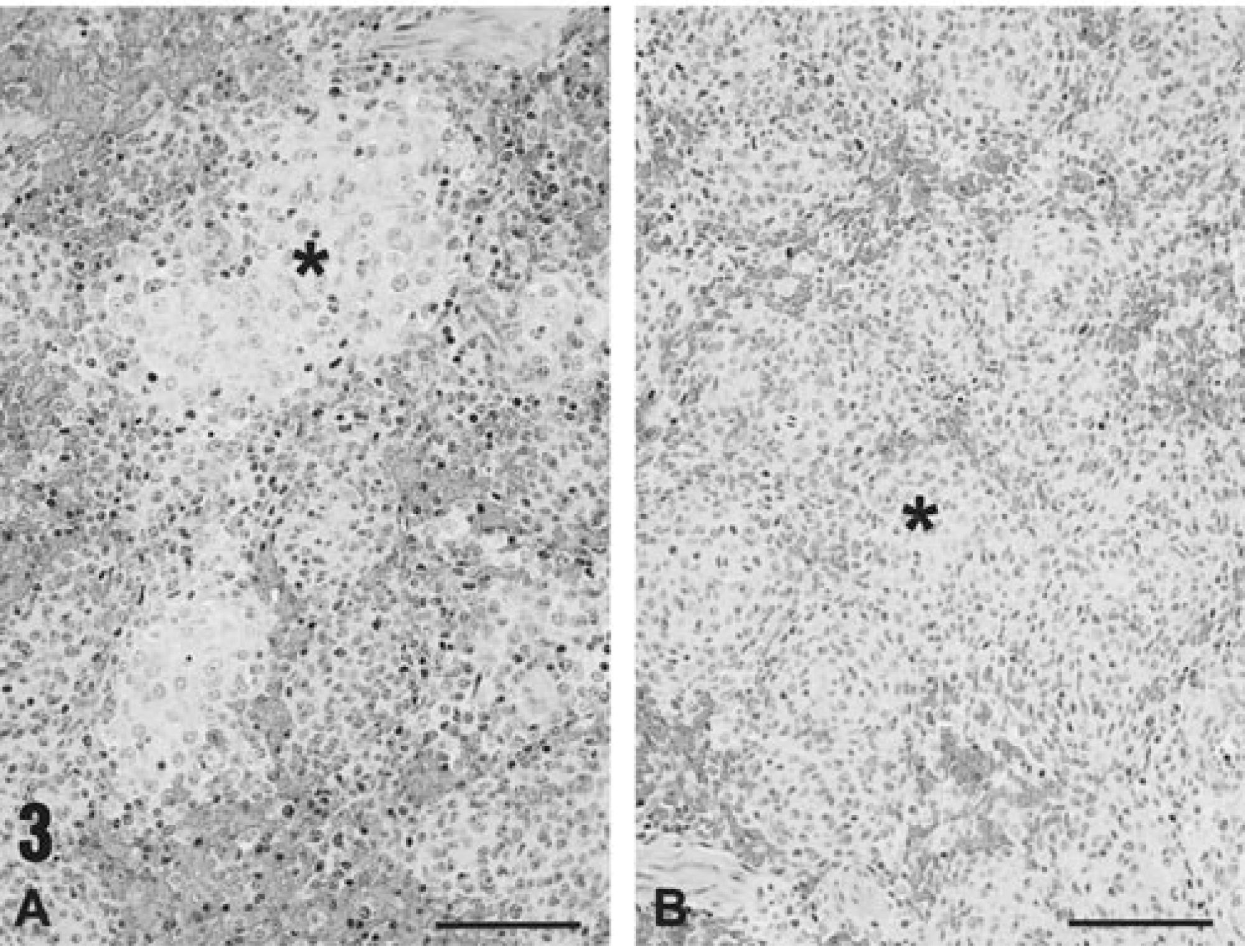
Histologic appearance of the spleens of PMWS-affected piglets.
Unlike many field cases of PMWS, 1 pulmonary involvement in affected gnotobiotes was minimal. Lymph nodes draining adjuvant injection sites still contained lipid adjuvant vacuoles and were largely depleted of mature lymphocytes (Fig. 4). Reactive macrophages, organized into either discrete granulomas or diffusely infiltrated into nodes, dominated the tissue sections. Stromal fibroblastic proliferation was prominent, and germinal centers were reduced or absent. Syncytial giant cells of macrophage origin were present, and mitotic figures in histiocytic cells were frequent. Macrophages contained numerous basophilic cytoplasmic viral inclusion bodies typical of PCV-2.
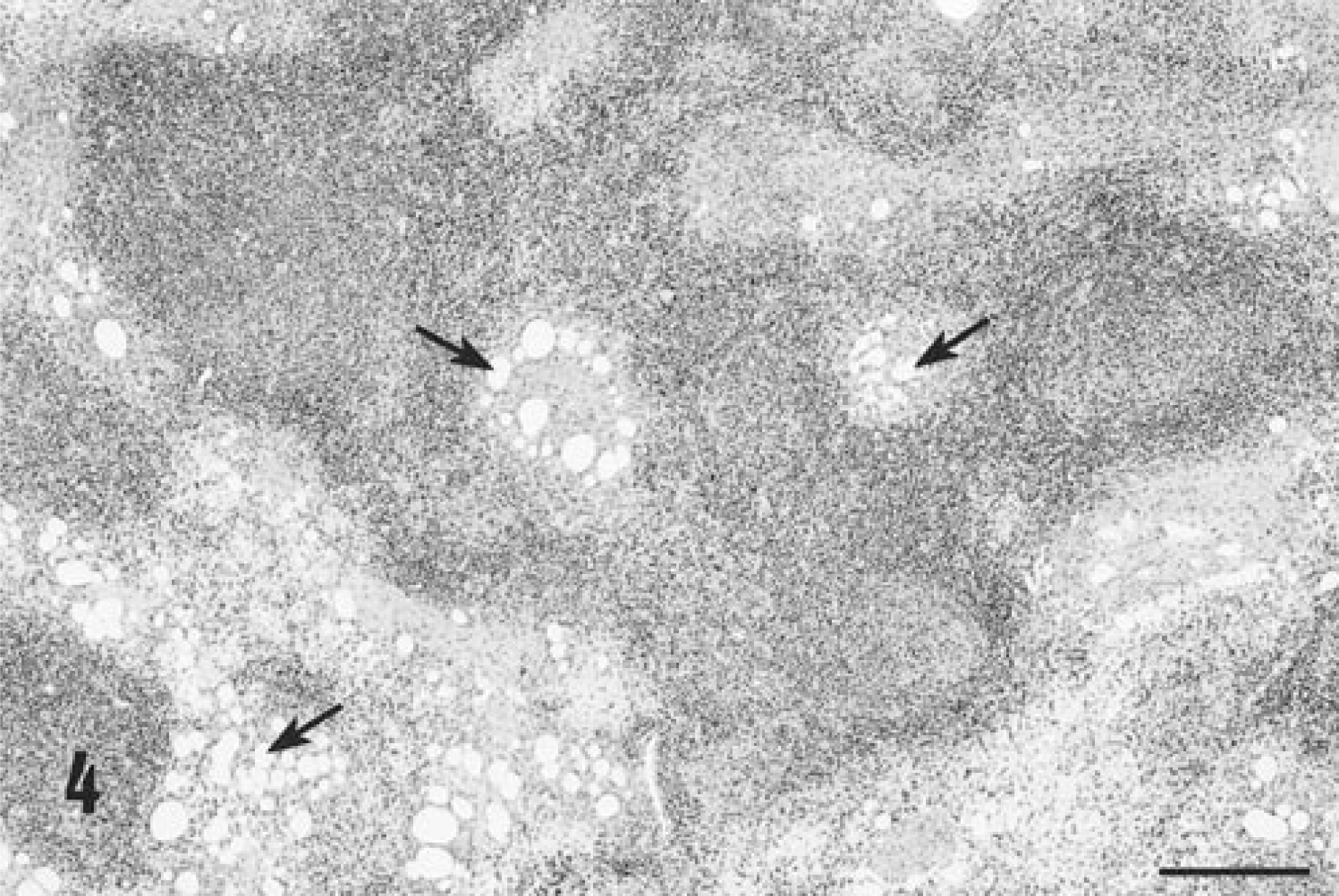
Histologic appearance of a lymph node from a KLH/ICFA-injected piglet affected with PCV-2–related PMWS on PID 35. Note the presence of adjuvant-associated lipid vacuoles contained within the section (arrows) and the localization of lipid vacuoles within the center of a nascent germinal center, presumably within follicular dendritic cells. Hematoxylin and eosin. Bar = 1,200 µm.
Inflammatory lesions were most severe and widespread in the liver. Diffuse hepatocyte damage, which involved some or the majority of hepatocytes identified in tissue sections, was present in all piglets (7/7). In some piglets, virtually every hepatocyte within involved lobules was necrotic or missing, and only a few severely degenerated hepatocytes remained (Fig. 5). Structural elements of the lobules were collapsed around the stromal elements, and the sinusoidal spaces were filled with blood. In less affected animals, individualization of hepatocytes (hepatic edema), acute cellular swelling, vacuolar degeneration, and individual hepatocyte necrosis was seen (Fig. 6). Accompanying this liver lesion was a moderate to severe diffuse granulomatous inflammatory response. Livers were diffusely infiltrated with macrophages, arranged in contiguous granulomas that displaced and replaced lost hepatocytes. In these sections, PCV-2 viral inclusion bodies, similar to those identified in lymph nodes, were detected within these macrophages, and an admixture of lymphocytes, neutrophils, and eosinophils were also present. Bile retention was prominent. Section replicates stained with Masson's trichrome stain for collagen accentuated the lobular pattern and emphasized the reduction in liver volume due to loss of hepatocytes.
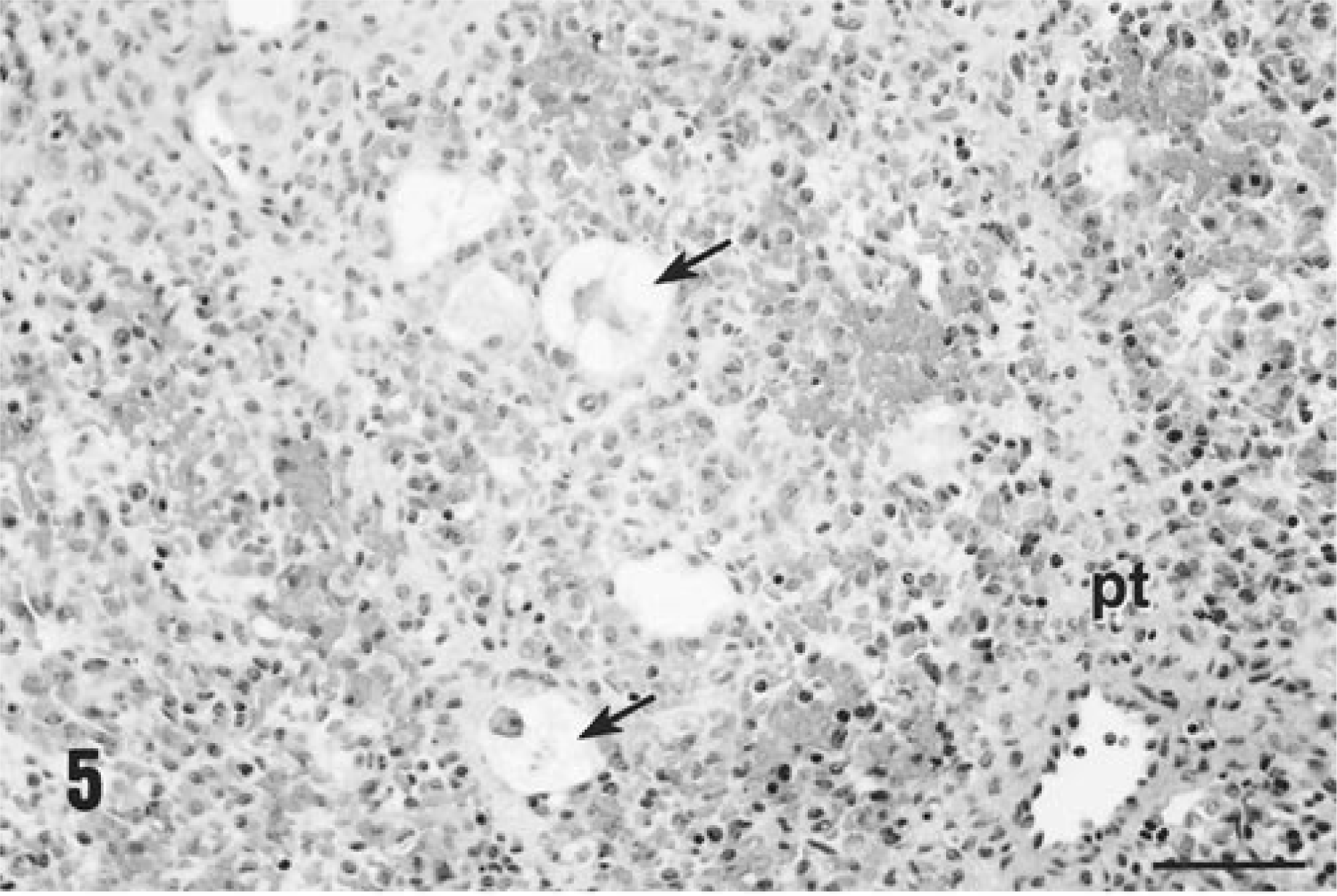
Histologic appearance of liver from a gnotobiotic piglet fatally infected with PCV-2–related PMWS, PID 32. Note the complete destruction of the hepatic cords and plates with preservation of the stromal connective tissue elements of the tissue, the presence of a portal triad (pt), and a few remaining necrotic hepatocytes (arrows). Hematoxylin and eosin. Bar = 300 µm.
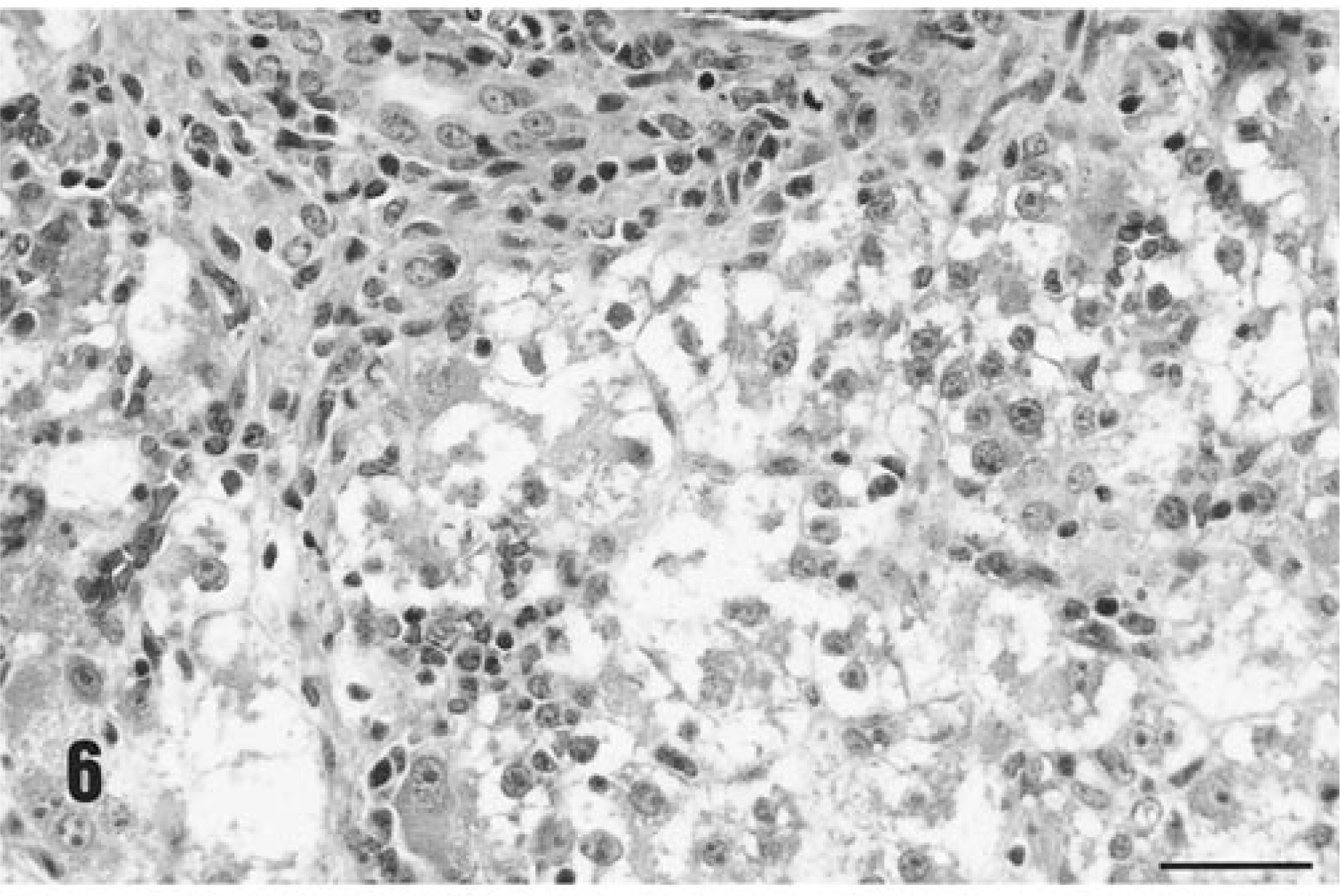
Histologic appearance of liver from an immunized piglet fatally infected with PCV-2–related PMWS, PID 35. Many hepatocytes are necrotic. There is granulomatous inflammatory cell infiltrates at the dorsal margin of this lesion. Hematoxylin and eosin. Bar = 165 µm.
The kidney and myocardium, aside from rare focal lymphoplasmacytic infiltrates, were normal. Acute diffuse pancreatic necrosis with associated peritonitis and peritoneal exocrine pancreatic enzyme–associated fat necrosis was present in the one piglet that died suddenly on PID 29. Focal pancreatic necrosis and associated granulomatous inflammation was seen in one other piglet. Five of seven piglets were labeled with BrDU prior to termination. As in the first group, extensive incorporation of label into replicating inflammatory macrophages and lymphocytes within the injection sites themselves, lymph nodes draining these sites (Fig. 7) and distant lymphoid tissues were seen. The BrDU label was strongly incorporated into the angiocentric granulomatous inflammatory lesions associated with PCV-2 as well. In addition, in those piglets with degenerate (but not necrotic) hepatocytes, incorporation of label into hepatocyte nuclear DNA was common.
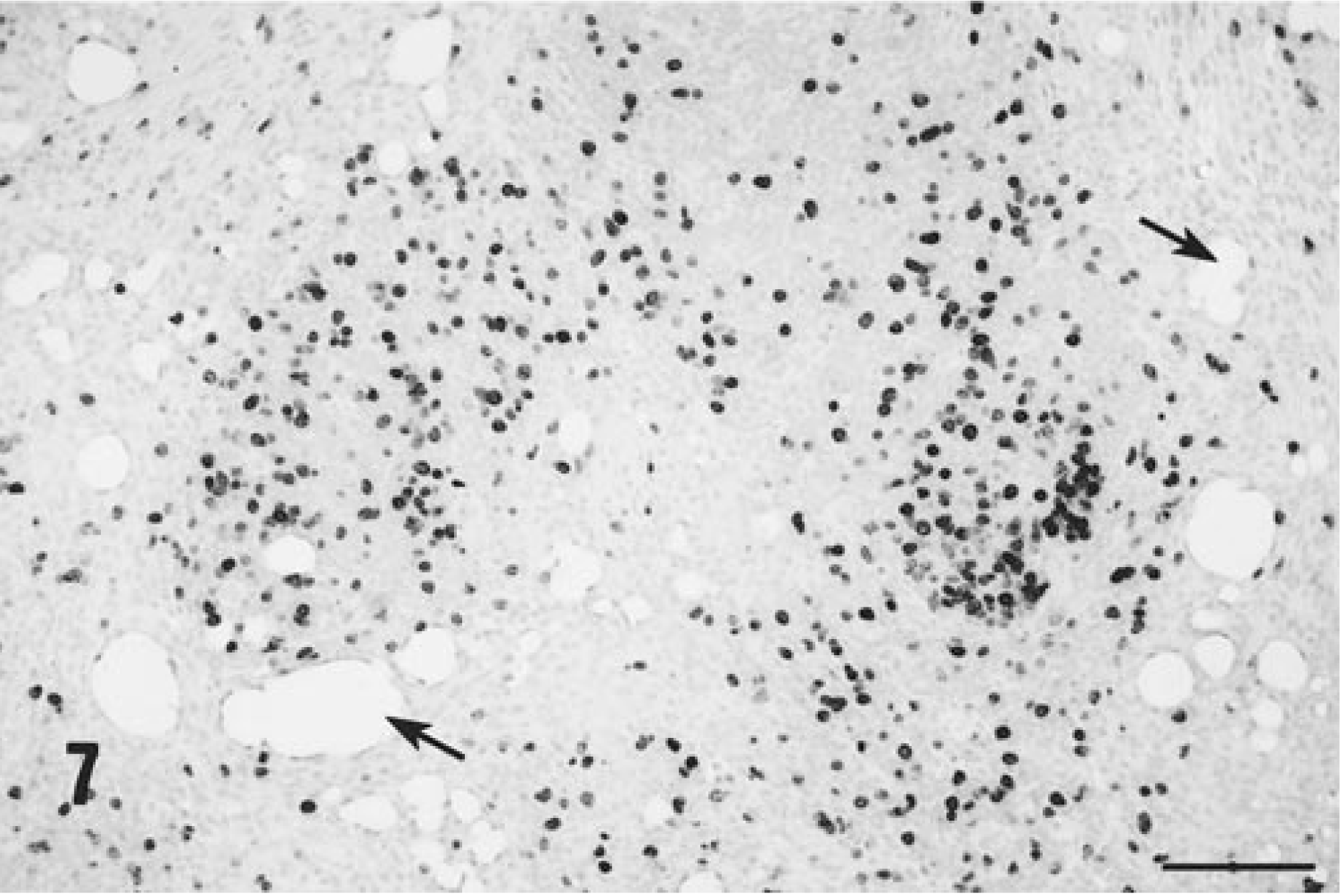
In situ BrDU incorporation into nuclear DNA in an axillary lymph node draining an injection site from a PMWS-affected gnotobiotic piglet, PID 32. Note that BrDU-positive cells are closely associated with lipid vacuoles (arrows) entrapped within the lymph node. Monoclonal antibody to BrDU, amplified by biotinylated and horseradish peroxidase conjugated secondary reagents, counterstained with hematoxylin. Bar = 450 µm.
Virology
Immunohistochemical staining for PCV-2 confirmed that the virus was widely disseminated in a pattern similar to that reported previously. 20 Virtually every macrophage contained within lymph nodes and liver (Figs. 8, 9) was viral antigen–positive. In addition, foci of virus-positive histiocytes were identified within the KLH/ICFA injection site granulomas (Fig. 10). Virology titrations were made from pooled lymphoid tissues (both inguinal axillaries/prescapular and submandibular lymph nodes, mesenteric, and bronchial lymph nodes and spleens) and liver from each PCV-2–infected gnotobiotic piglet, and the data are reported in Table 4. Both the liver and lymphoid tissues from PMWS-affected piglets contained large quantities of titratable infectious virus. Titers ranged from 106 to >108 infectious units per gram of tissue. In contrast, homogenates prepared from piglets infected with PCV-2 alone contained far less virus (range 104–106). Control uninfected piglets were virus-negative.
Titratable infectious PCV-2 in target tissues from piglets infected with PCV-2 and injected with KLH/ICFA with or without thioglycollate (glycan) intraperitoneally (IP).
TCID50 = tissue culture infectious doses that infected 50% of the microwell cultures.
Lymphoid tissues included superficial and deep inguinal, axillary, submandibular, bronchial, and mesenteric lymph nodes and the spleen.
GM = geometric mean.
ND = not done.
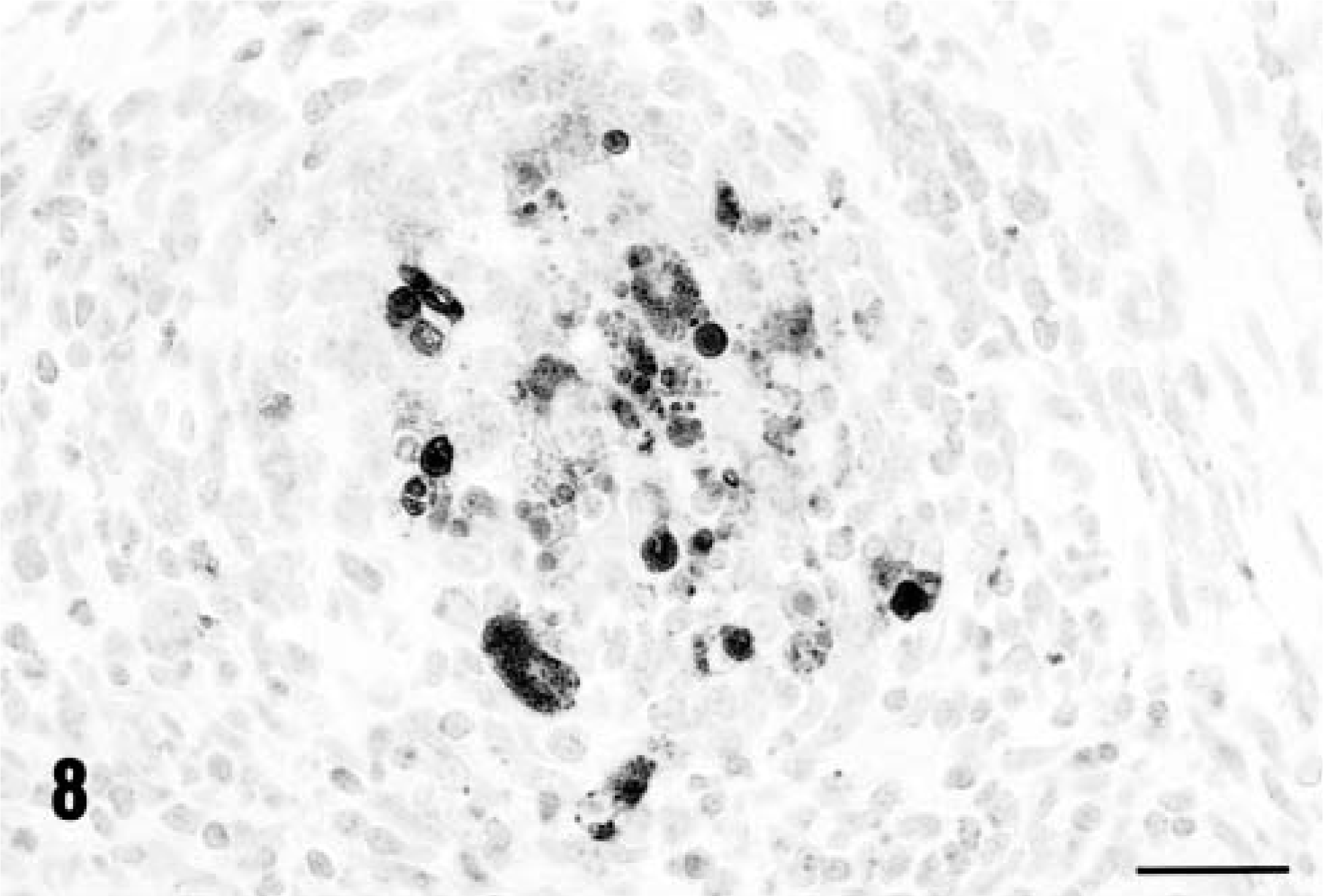
The distribution of immunoreactive PCV-2 antigen within a lymph node from a KLH/ICFA-injected piglet with PMWS, PID 35. Note that the antigen is within follicular dendritic cells in the germinal center. Anti–PCV-2 monoclonal antibody stained with biotinylated equine anti-mouse Ig, reacted with avidin peroxidase, developed with DAB, and counterstained with hematoxylin. Bar = 115 µm.
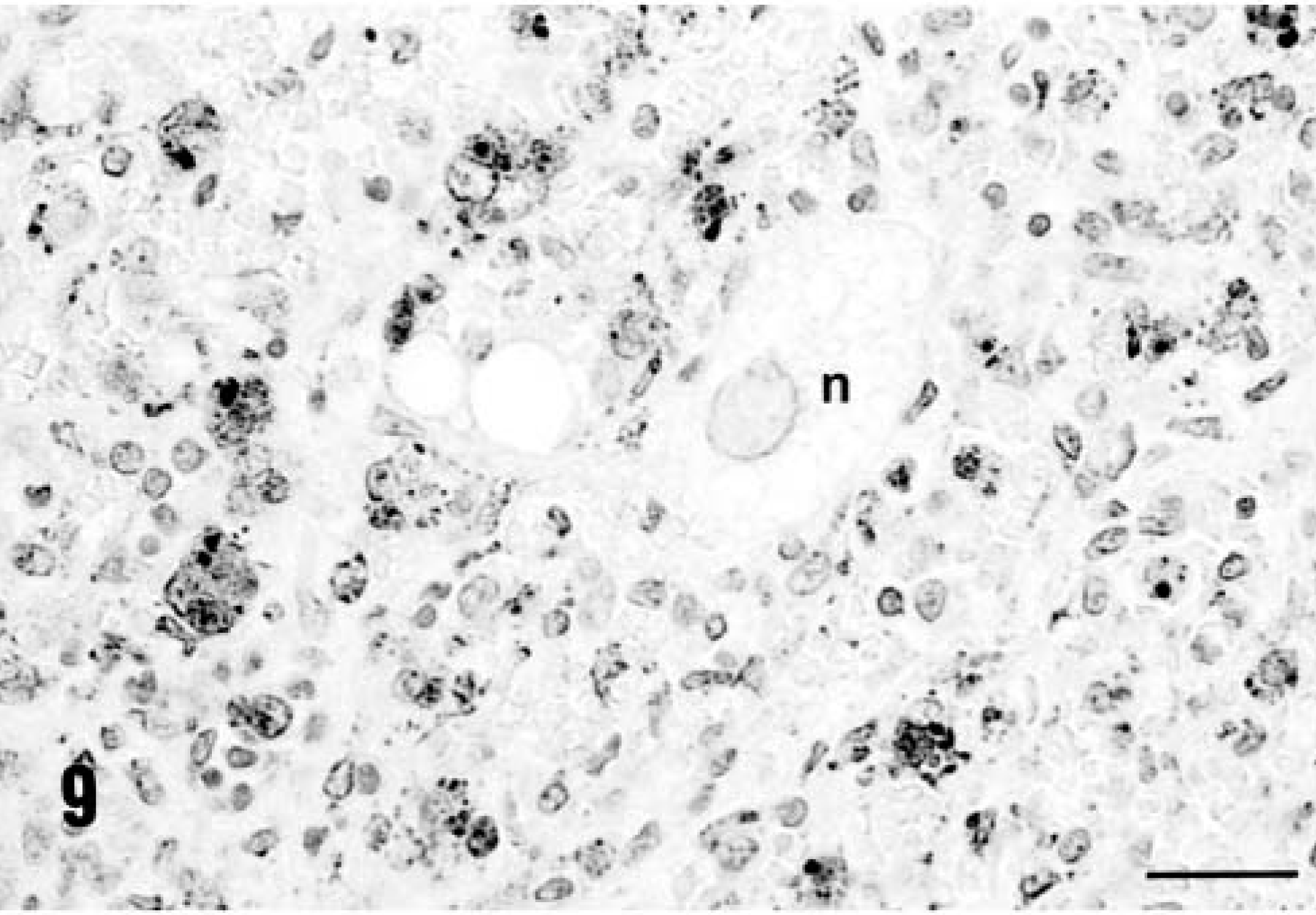
The distribution of immunoreactive PCV-2 antigen within infiltrating macrophages, Kupffer cells, and histiocytes within the liver of a piglet with PCV-2–related PMWS, PID 28. Note that the DAB reaction product is not contained within necrotic (n) and degenerating hepatocytes. Anti–PCV-2 monoclonal antibody stained with biotinylated equine anti-mouse Ig, reacted with avidin peroxidase, developed with DAB, and counterstained with hematoxylin. Bar = 125 µm.
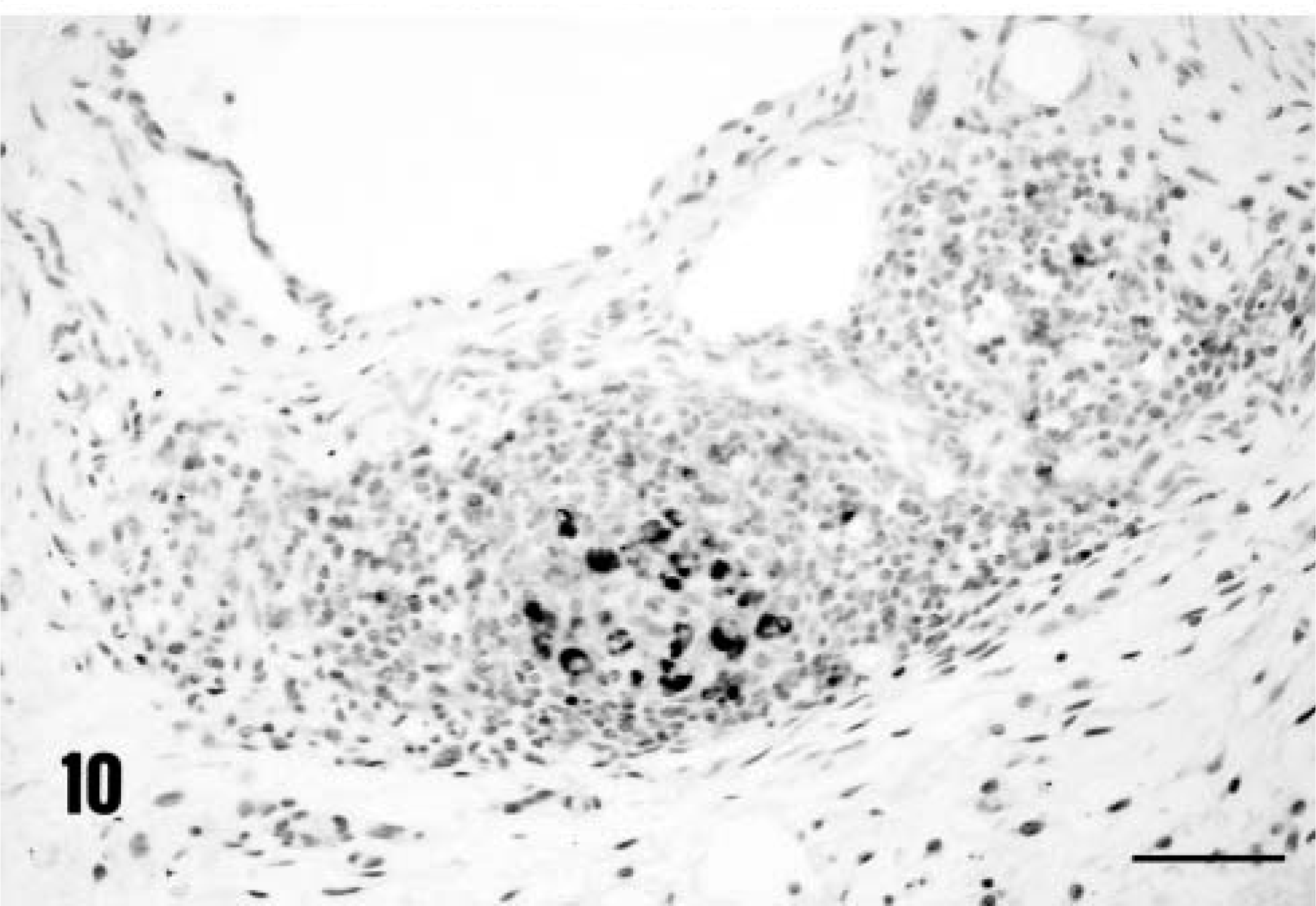
The distribution of immunoreactive PCV-2 antigen contained within a subcutaneous KLH/ICFA injection site in a piglet with PCV-2–associated PMWS, PID 32. Note the accumulation of DAB reaction product within a collection of histiocytes. Anti–PCV-2 monoclonal antibody stained with biotinylated equine anti-mouse Ig, reacted with avidin peroxidase, developed with DAB, and counterstained with hematoxylin. Bar = 300 µm.
Discussion
In the gnotobiotic experiments described here, we have fulfilled Koch's postulates for PMWS and have confirmed that PCV-2 alone is responsible for the wasting disease of swine known as postweaning multisystemic wasting syndrome. This complex, known by the acronym PMWS, has emerged as a new and potentially devastating viral infectious disease of swine. The primary organ system affected (pulmonary, hepatic, or renal) and mortality rate varies both within an infected herd and between infected herds. 1 9 15 The bulk of available evidence both in field cases and experimental studies generated in this study and others 1–3, 5–7, 12–14, 17 20 23 26 have established that PCV-2 is the primary viral infectious agent of PMWS. The constant association between viral inclusion bodies, viral protein, and viral DNA within the lesions of PMWS; the recovery of PCV-2 from field cases of PWMS; and the experimental infection experiments described here and in conventional colostrum-deprived 6 piglets support this conclusion.
Experimental reproduction of PMWS appears to be dependent upon the environmental status of virus-susceptible pigs. For example, as reported here and previously, 20 PCV-2 alone, when inoculated into gnotobiotic piglets, produces a clinically asymptomatic infection and very modest histologic evidence of inflammation. 20 In contrast, severe lesions of PMWS are observed in a proportion of conventional piglets 2 infected with PCV-2 but not knowingly coinfected with other infectious swine pathogens. Importantly, the mild and self-limiting infection characteristic of gnotobiotes is converted to fulminant and fatal PMWS when gnotobiotic piglets are coinfected with PPV, an innocuous viral infection in postnatal pigs. 14 20 Similar findings are reported when Caesarian-derived colostrum-deprived (CD/CD) piglets are coinfected with PRRS virus. 3 17 Thus, under conventional husbandry conditions, coinfections with other viruses and bacteria are common and likely account for the overt expression of PMWS in these animals.
In previous reports, 14 20 the role of a second agent (PPV) in the genesis of severe PWMS disease in gnotobiotes was not defined. Aside from reproductive failure and fetal deaths, PPV is not regarded as a significant cause of mortality in young swine, 10 19 even though the virus has a tropism for lymphoid tissues, specifically macrophages. 10 16 24 25 Data generated in the first study 20 and from quantitative viral recovery studies in dually infected piglets 6 suggest the following pathogenesis for PMWS: After coinfection, both PPV and PCV-2 localize in regional histiocytes of the oropharynx. PPV spreads rapidly beyond the inoculation site, chiefly by cell-associated viremia, and replicates in lymphoid tissues, thereby stimulating proliferative responses in both lymphocytes and macrophages and PPV-protective immunity. PCV-2 is also slowly replicating in macrophages, but effective immunity to PCV-2 is not established. During the immunoproliferative phase of PPV convalescence, activated macrophages and possibly other immune cells support heightened PCV-2 replication. PCV-2 virion production is upregulated and cell-to-cell spread is facilitated by the activation-mediated increased phagocytic and fusigenic capabilities of macrophages. Local compartmentalization barriers are breached and the virus is disseminated as a macrophage-dominant pantropic granulomatous inflammatory response expressed clinically as PMWS. This proposed pathogenesis, while consistent with what is known about PCV-2, does not constitute definitive proof of mechanism. Implied in the proposed pathogenesis is the concept that activation of the cells of the immune system during the early phase of the infection is a key host determinant for expression of virulence by PCV-2. If macrophage activation is an essential prerequisite for the development of PCV-2–mediated PMWS, then local/systemic activation by nonliving (PPV-free) agents should promote PMWS in PCV-2–infected gnotobiotic piglets. The data reported here support this concept and the proposed pathogenesis of disease.
In the first experiment, we tested the hypothesis that local immune activation of draining lymph nodes by subcutaneous injection with an antigen in adjuvant supported heightened replication of PCV-2. We capitalized upon the local stimulating properties of a strong adjuvant 8 with an irrelevant incorporated antigen (KLH) and also upon the fact that each piglet could serve, in effect, as its own control by performing one-sided injections in gnotobiotic piglets actively infected with PCV-2. In every instance, nodes draining adjuvant injection sites produced more infectious virus and were more strongly positive for PCV-2 antigen by immunochemistry than contralateral corresponding lymph nodes. The actual titers in activated infected nodes varied between piglets within a specific treatment group and ranged from <102 to 106. 5 (TCID50) 50% tissue culture infectious dose 0.1 g tissue. This variation is likely the result of the restricted (PID 13/14) examination interval and individual variation within the piglet groups. Upregulation of virus production was correlated to increased numbers of actively replicating cells in lymph nodes, as demonstrated by incorporated BrDU, and to histologic findings of lymphoreticular hyperplasia and germinal center formation.
That increased virion production in locally activated lymph nodes could result in PMWS was demonstrated in the second experiment. Here, experimental systemic immunostimulation, achieved either with bilateral injections of KLH/ICFA or bilateral injections of KLH/ICFA combined with IP thioglycolyate, an irritant known to produce sterile macrophage-dominant peritonitis, provided sufficient immunostimulation such that systemic PMWS was achieved in all (7/7) piglets within 35 days after infection. This disease outcome endpoint was reached and correlative data (distribution of viral antigen, recovery of high titers of infectious virus from lymph nodes and liver, etc.) indicated that our expectations for reproducing PMWS in gnotobiotes were met. That said however, the concept that new cellular DNA synthesis per se is the only in vivo prerequisite for PCV-2 replication should be modified since many tissues that were virus-negative by immunohistochemistry strongly incorporated BrDU label. For example, prominent BrDU labeling of epithelial cells of the renal cortex and glomeruli was present in all piglets, yet these BrDU-positive cells were routinely negative for PCV-2. Thus, it appears that new DNA synthesis, mitosis, and proliferation of histiocytes and macrophages induced by injected adjuvant provided the optimal conditions for virus replication and dissemination in vivo. The apparent tropism of PCV-2 for cells of histiocyte or macrophage lineage 1 20 was confirmed in this study. In fact, subcutaneous injection sites frequently contained foci of virus-infected cells in the dermis and associated granulomatous inflammatory cell infiltrates. In several instances, intraepithelial and subepithelial cells overlying the immunization sites also contained virus antigen. Follicular histiocytes occupying the centers of developing lymphoid follicles in lymph nodes were routinely virus antigen–positive, as were infiltrating macrophages and histiocytes in areas of PCV-2–associated granulomatous inflammation.
As observed previously in PCV-2/PPV-infected piglets, 20 hepatocyte necrosis and liver failure associated with severe widespread granulomatous hepatitis was the proximate cause of death in all cases of PMWS in gnotobiotic swine. Clinical manifestations of liver disease, including icterus, generalized edema, and hepatocellular degeneration and necrosis, all support this conclusion. In one piglet, liver disease was so dramatic as to result in prolonged bleeding and extensive subcutaneous bruising—presumably secondary to thrombocytopenia and/or reduction in plasma proteins associated with liver insufficiency, coagulation, and hemostasis—after routine venipuncture 5 days prior to death. The mechanism(s) of hepatocyte necrosis identified in PMWS-affected gnotobiotic piglets is not apparent in that a direct correlation between the extent of hepatocyte necrosis and the distribution of virus antigen could not be made. In several of the piglets that were terminated with moderate signs of PMWS on PID 35, many viable hepatocytes were identified in tissue sections. If hepatocyte necrosis was a direct consequence of virus replication in them, large numbers of viral antigen–positive hepatocytes should have been seen. They were not. There is no doubt that PCV-2 will replicate in hepatocytes in vivo, 1 and occasional virus-positive hepatocytes were present in all PMWS-affected piglets in this study. However, as emphasized previously, 20 viral antigen was largely confined to histiocytic cells contained within the granulomatous inflammatory cell infiltrates of the liver and most hepatocytes were conspicuously devoid of PCV-2 antigen.
Expression of PMWS in gnotobiotes differs somewhat from clinicopathologic presentation in field cases 1 or in experimental disease as a result of coinfection with PRRS virus 3 17 in that pneumonic lesions were not a significant feature of the disease. Aside from generalized lymphadenopathy, PMWS may present as respiratory, enteric or liver disease, renal failure, or even congestive heart failure. 9 15 It is likely though not yet proven that pathogenic cofactors that preferentially target different organ systems may be indirectly responsible for the expression of PCV-2–related lesions and associated inflammation in them. For example, CD/CD piglets coinfected with PRRS virus develop PMWS-associated respiratory insufficiency. 17 The lung is a known target organ for the PRRS agent, even though both virus infections were widely distributed in tissues of these piglets. 17
Limited serologic data indicate that PCV-2 has been present in certain hog populations since at least 1973. 27 It is a mistake to believe that this is a “new” or recently introduced viral infectious disease. What is new, however, is clinical expression of this virus infection as PMWS. Local or systemic immunostimulation with a foreign antigen emulsified in a strong irritating adjuvant such as ICFA has relevance in the reality of disease prevention through vaccination. As expected, 8 histologic evaluation of lymph nodes draining the site(s) of injection demonstrated lipid droplets from the immunogenic formulation imbedded within organized collections of activated macrophages and lymphocytes within draining lymphoid nodes. In this context, the larger question then is what is the magnitude of immunostimulation needed to convert PCV-2 infection from a limited, self-contained and subclinical infection into fulminant PMWS in weanling conventional piglets? Perhaps it was no accident that PMWS first occurred in “high health” herds in western Canada. 1 9 15 Reduction of potentially pathogenic microflora in these herds combined with intentional exclusion of known pathogens by isolation and prophylactic vaccination with modified live products may have unintentionally resulted in understimulated, immunologically “naive” pigs. In this scenario, any introduced immunostimulus, intentional or inadvertent, combined with preexistent subclinical and endemic PCV-2 infection may have resulted in conditions conducive to development of fulminant PMWS. All of this is speculative, of course, and other explanations for the sudden world-wide surge in the incidence of PMWS may well be forthcoming. Nonetheless, these observations are consistent with available field and experimental data and suggest that evolving management practices may inadvertently contribute to the genesis of PMWS in swine.
Footnotes
Acknowledgements
This research was supported by a research grant from The National Pork Producers Council, Ames, IA, USA, and USDA formula funds, The Ohio State University. The excellent technical assistance of Judith Younger and Michelle Newkirk is appreciated.
