Abstract
A 4-year-old female pigtailed macaque (Macaca nemestrina), experimentally coinfected with simian immunodeficiency virus (SIVmac251) and Mycobacterium bovis (bacillus Calmette-Guerin), was euthanatized 1 year after infection because of weight loss and labored breathing. On gross examination, both kidneys were found to be markedly enlarged (right: 54.7 g and left: 51.7 g; normal < 20 g). Renal lesions were evaluated by histopathologic, immunohistochemical, and ultrastructural methods. Light microscopy revealed that the glomeruli were diffusely hypercellular with expansion of the mesangial matrix, and crescent formation affected approximately 60% of the glomeruli. By immunohistochemical evaluation, it was found that the crescents were composed principally of macrophages, as seen by CD68 (KP1), MRP8, MAC387, and HAM56 expression. Electron microscopic examination of the glomeruli revealed extensive intramembranous, subendothelial, and mesangial electron-dense deposits and multifocal fusion of the visceral epithelial foot processes. Immunofluorescence, used to determine the composition of the electron-dense deposits, revealed diffuse granular mesangial and capillary staining for immunoglobulin A (IgA). The renal changes described in this case report are most consistent with the findings of crescentic gloerulonephritis with IgA immune complex deposition in the glomerular basement membrane and mesangium as described in humans with IgA nephropathy.
Keywords
Humans
Mesangioproliferative glomerulonephritis (MesPGN) with crescent formation is a diffuse form of glomerulonephritis (GN) characterized by capillary thrombi with capillary wall necrosis and crescent formation in 60–80% of glomeruli. 14 In humans, this form of GN often has an acute onset and is classified as rapidly progressive glomerulonephritis (RPGN). 2,14,17 Clinically, it is characterized by rapid and progressive loss of renal function associated with severe oliguria and death within weeks to months unless dialysis is instituted. 2,14,17 In most cases, the prognosis for RPGN is poor.
RPGN is not associated with a specific etiology and may be idiopathic or secondary to multisystemic diseases (Goodpasture syndrome, Wegener's granulomatosis, polyarteritis nodosa, systemic lupus erythematosus, and Henoch-Schonlein syndrome). 2,8,17 In addition, other renal diseases including MesPGN and immunoglobulin A nephropathy (IgAN) may progress to RPGN. 2,9,13,17
In humans, crescentic glomerulonephritis (CGN) is characterized by morphologic abnormalities in both the glomerular tufts and the cells lining Bowman's capsule. Accentuated lobulation and cellular proliferation of the tufts are seen. Crescent formation is often considered to be due to extensive hyperplasia of the cells lining Bowman's capsule. However, podocytes and inflammatory cells (monocytes and lymphocytes) may also participate in crescent formation. Crescents may be multiple cell layers thick, and mitoses are commonly found. 3 Some crescents become organized by fibroplasia and collagen deposition. 10,14,15 Therefore, the morphologic features of CGN may vary in both the distribution and the composition of the crescents.
Nonhuman Primates
Naturally occurring renal disease has been reported more frequently in New World primates than in Old World primates. 4–7,12,16,18,19 The most common types of GN include proliferative, MesPGN, and membranous GN. 19 Interstitial lesions include periglomerular lymphocytic infiltrates, focal to diffuse infiltrates of lymphocytes and plasma cells in the interstitium, and linear to diffuse fibrosis of the renal parenchyma. 4–7,12 However, macaques experimentally infected with simian immunodeficiency virus (SIV) appear to have a higher incidence of MesPGN or focal segmental glomerulosclerosis (or both). 1,11 To our knowledge, this is the first report of CGN and MesPGN associated with IgA deposition in a macaque.
Materials and Methods
Five pigtailed monkeys that were experimentally coinfected with 106 tissue culture infective dose (50%) of SI-Vmac251 and 107 colony-forming units of bacillus Calmette-Guerin (BCG) by intravenous inoculation were used as an animal model to study the pathogenesis of tuberculosis in patients with acquired immunodeficiency syndrome. The subject of this case report was a female, pigtailed macaque (Macaca nemestrina) experimentally infected at 3 years of age. One year after infection, this monkey lost weight and had pale mucous membranes, lymphadenopathy, and labored breathing. Euthanasia was elected, and a complete necropsy was performed. Specimens of all major organs were fixed in 10% neutral buffered formalin and embedded in paraffin. The kidneys were sectioned at 3 µm and stained with hematoxylin and eosin, periodic acid–Schiff, periodic acid–Schiff silver methenamine (PASM), Masson trichrome, and Ziehl-Neelsen acid fast. Immunohistochemical staining was done using multiple monoclonal and polyclonal antibodies from BioGenex (San Ramon, CA), Immunotech (Miami, FL), and Accurate Chemical & Scientific Corp. (Westbury, NY) (see Table 1). All kidney sections were incubated with the primary antibody for 1 hour at room temperature, followed by biotinylated anti-mouse or anti-rabbit secondary antibodies (Dako, Carpinteria, CA) for 30 minutes. Finally, sections were incubated with avidin–biotin complex (ABC) for 30 minutes, and the reaction was visualized with 3,3′-diaminobenzidine (Dako) as the chromogen. To evaluate the presence of Ig, frozen sections of unfixed kidney were cut and fixed in 4% paraformaldehyde. Direct and indirect immunofluorescence procedures with anti-monkey IgM-fluorescein isothiocyanate, anti-monkey IgG-Texas red, and anti-monkey IgA-biotin–conjugated antibodies (Research Diagnosis Inc., Flanders, NJ) were used. Sections were washed for 15 minutes with 1× phosphate-buffered saline (PBS) in doubly distilled H2O + 0.2% fish skin gelatin, blocked with Dako protein block, and incubated with the labeled antibody for 30 minutes at room temperature. Anti-monkey IgA-biotin–conjugated antibody was detected with streptavidin conjugated with Alexa 488 (Molecular Probes, Eugene, OR). To-Pro3 (nuclear dye, Molecular Probes) at 1 µg/ml was used in some sections to differentiate between individual cells. To-Pro3 was incubated on tissue sections for 5 minutes and washed in PBS. Confocal microscopy was performed using a Leica TCS SP2 confocal microscope equipped with three lasers (Leica Microsystems, Exton, PA). The fluorescence of individual fluorochromes and the differential interference contrast (DIC) were captured separately. Specimens of the kidney were also postfixed in 2.5% glutaraldehyde and osmium tetroxide and embedded in Epon araldite for examination using a Jeol 1010 transmission electron microscope.
Antibodies used for immunohistochemistry to evaluate kidney lesions in this case report.∗
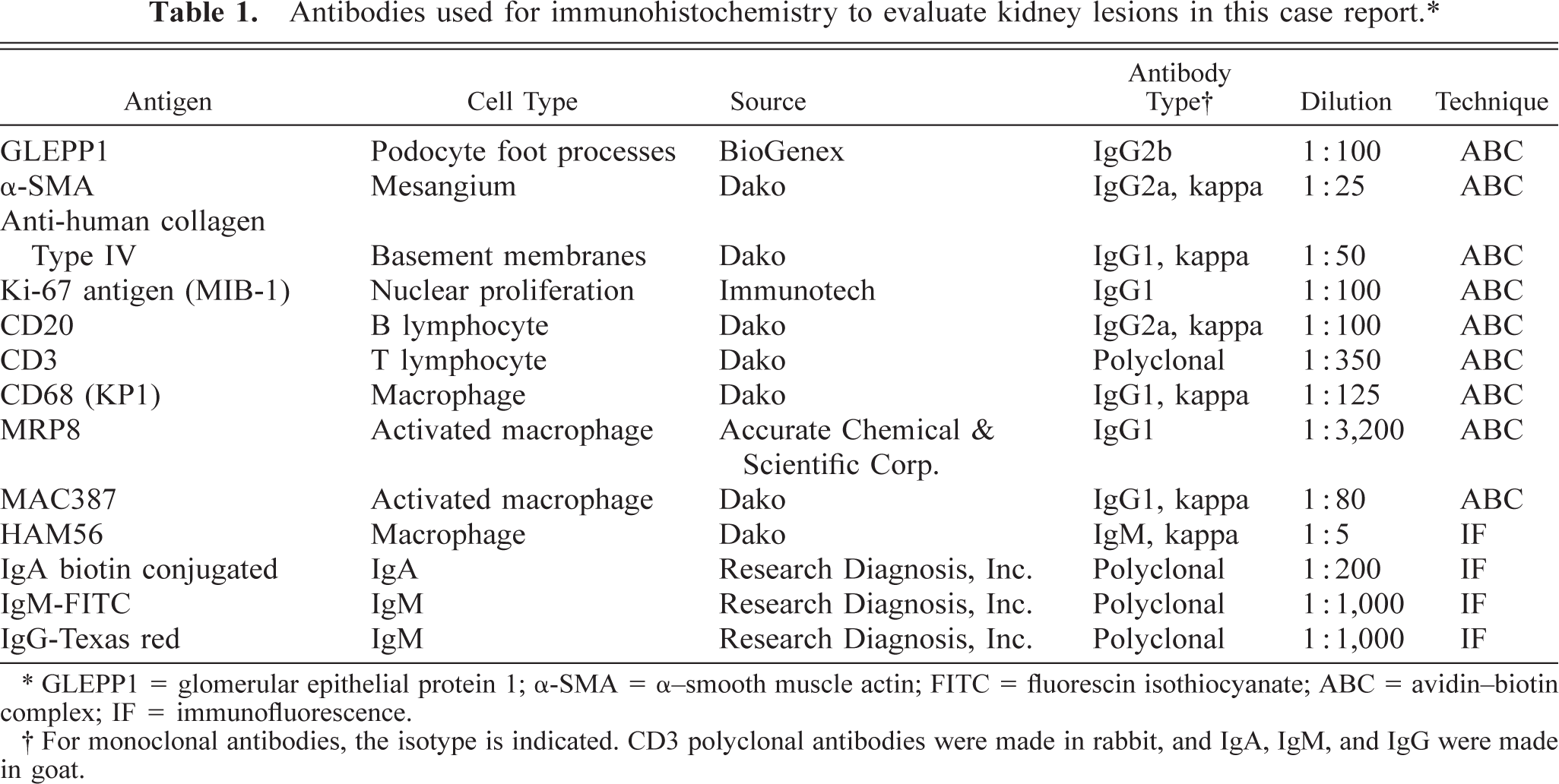
∗ GLEPP1 = glomerular epithelial protein 1; α-SMA = α-smooth muscle actin; FITC = fluorescin isothiocyanate; ABC = avidin-biotin complex; IF = immunofluorescence.
† For monoclonal antibodies, the isotype is indicated. CD3 polyclonal antibodies were made in rabbit, and IgA, IgM, and IgG were made in goat.
Gross pathology
At necropsy, 150 ml of clear pale yellow fluid was present in the abdominal cavity. Both kidneys were markedly enlarged (right: 54.7 g and left: 51.7 g; normal <20 g). The right kidney had a well-demarcated pale yellow infarct in the posterior pole. The capsular and cut surfaces of both kidneys had multiple pinpoint red foci. The spleen was moderately enlarged, with prominent lymphoid follicles on cut section. Axillary, inguinal, and mesenteric lymph nodes were enlarged.
Light microscopy
Microscopic examination revealed that renal corpuscles were enlarged because of cellular proliferation and infiltration in Bowman's space. Approximately 60% of the glomeruli had crescent formation in Bowman's space, which, in some instances, completely filled the space and compressed the glomerular tuft (Fig. 1). Increased mesangial matrix, proliferation of mesangial cells, diffuse thickening and wrinkling of basement membranes, and pseudotubule formation were seen with PASM staining (Fig. 2). A diffuse infiltrate of mononuclear cells, predominantly lymphocytes and plasma cells, was seen in the cortical interstitium.
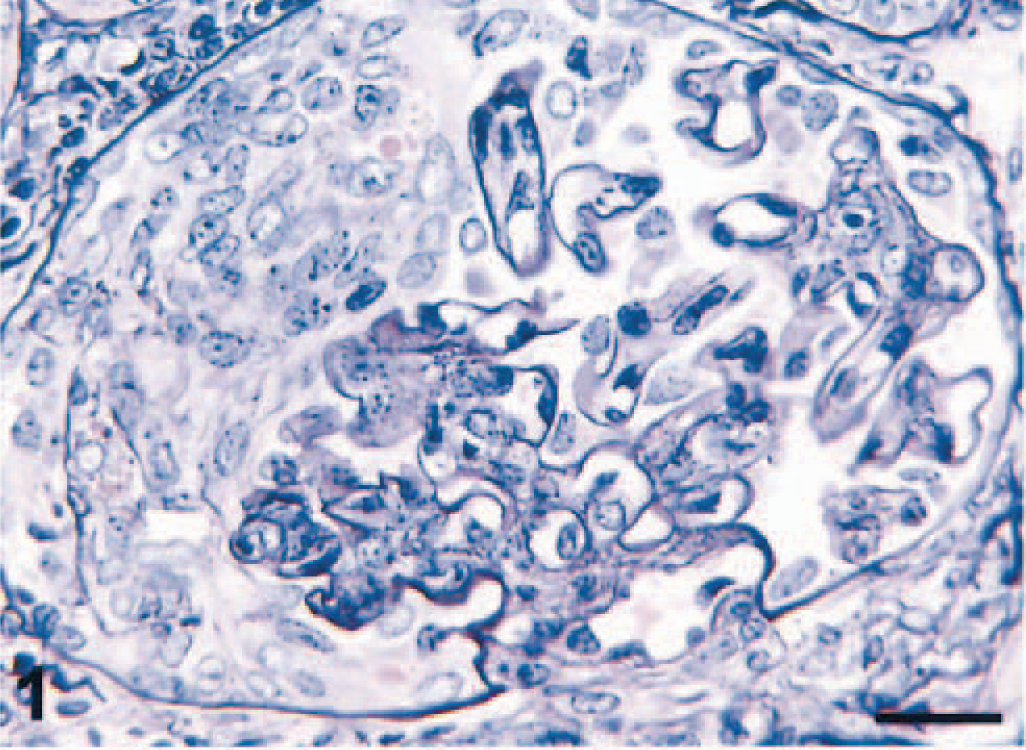
Kidney; pigtailed macaque. Note the cellular crescent formation virtually obliterating Bowman's space. PASM. Bar = 25 µm.
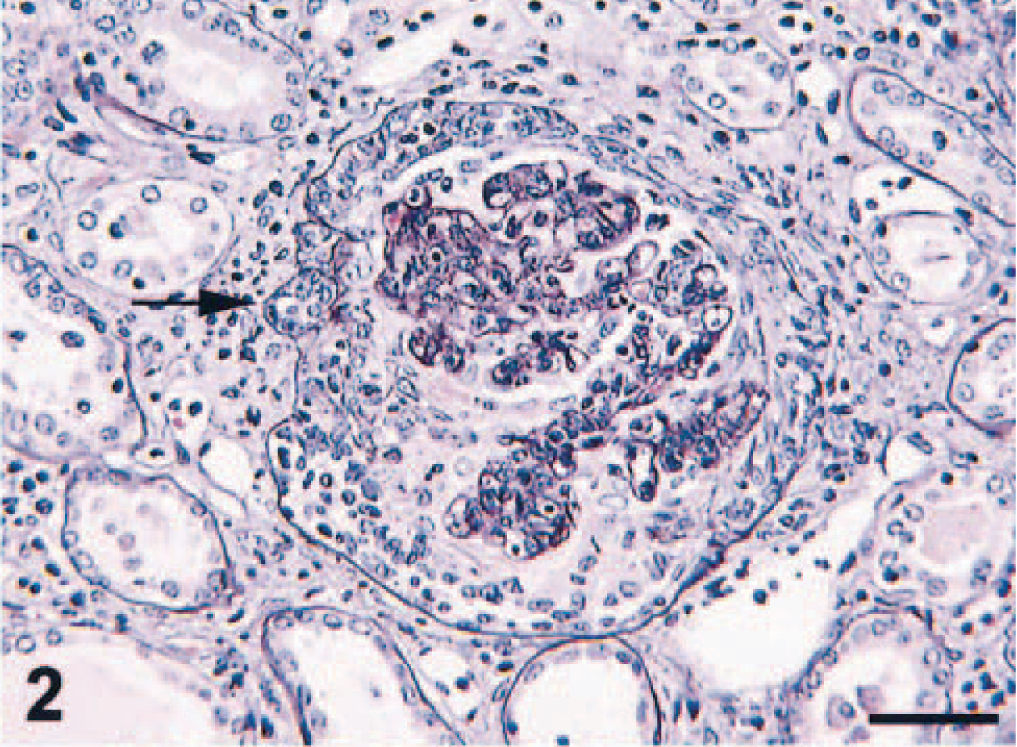
Kidney; pigtailed macaque. Thickened glomerular basement membranes, fibrin exudation in Bowman's space and pseudotubule formation (arrow). PASM. Bar = 50 µm.
Most tubules were lined by hyperplastic epithelial cells with small numbers of mitotic figures and occasional papillary projections into the lumina. Hyperplasia and proliferation of tubular epithelial cells were corroborated by positive staining with Ki-67 antibody (MIB-1). However, some tubules were dilated and lined by attenuated epithelial cells. Moderate numbers of macrophages and red blood cells or hyaline casts (or both) were present in most tubular lumina. No acid-fast bacilli were identified in kidney sections stained with Ziehl-Neelsen.
Immunohistochemistry
CD68- (KP1), MRP8-, MAC387-, and HAM56-positive macrophages were present in both the capillary lumina of the glomeruli and the crescents (MRP8 and MAC387 are markers for activated macrophages) (Fig. 3). Labeling with glomerular epithelial protein 1 (GLEPP1) was discontinuous, specifically in glomeruli with crescent formation (Fig. 4), whereas intense α–smooth muscle actin (α-SMA) expression was seen in the mesangial area (not shown). Increased expression of collagen Type IV was observed in the glomerular and parietal basement membranes as compared with normal kidney (not shown). Ki-67 antigen (MIB-1) was expressed in glomeruli and crescents (Fig. 5). Rare CD3 T lymphocytes were identified in the glomeruli or the crescents. Diffuse granular mesangial and capillary staining for IgA was observed by immunofluorescence (Fig. 6), whereas staining for IgM and IgG was minimal.
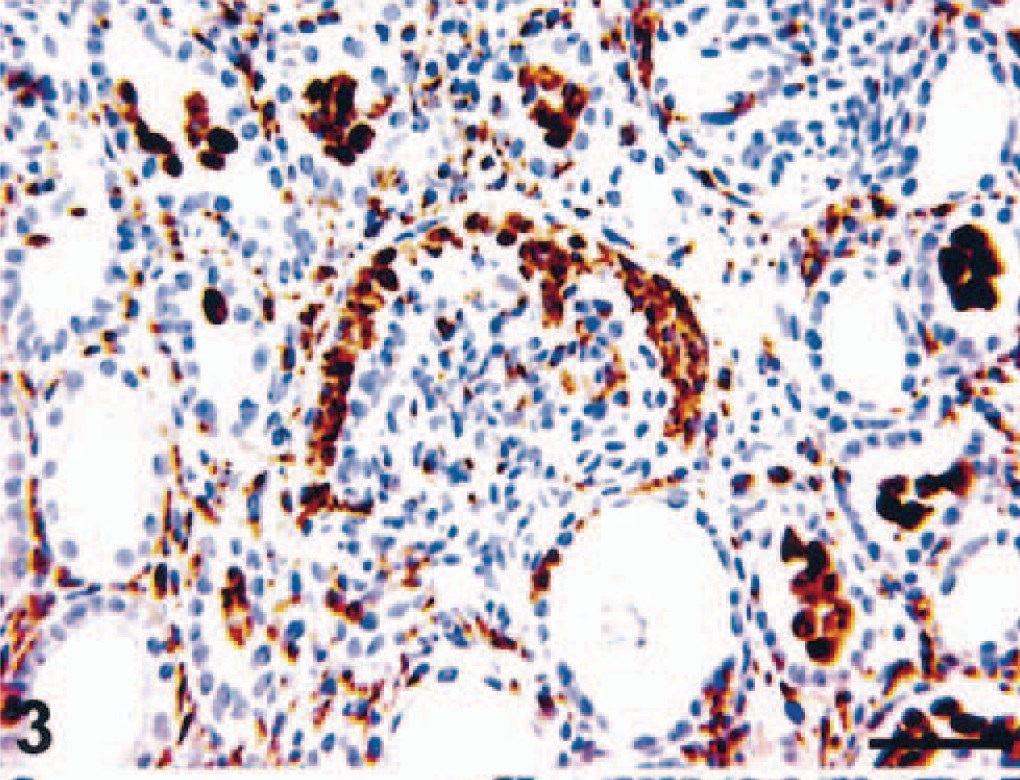
Kidney; pigtailed macaque. Macrophages are present both in the crescent and in the tubular lumens. MRP8. ABC peroxidase method, Mayer's hematoxylin counterstain. Bar = 50 µm.
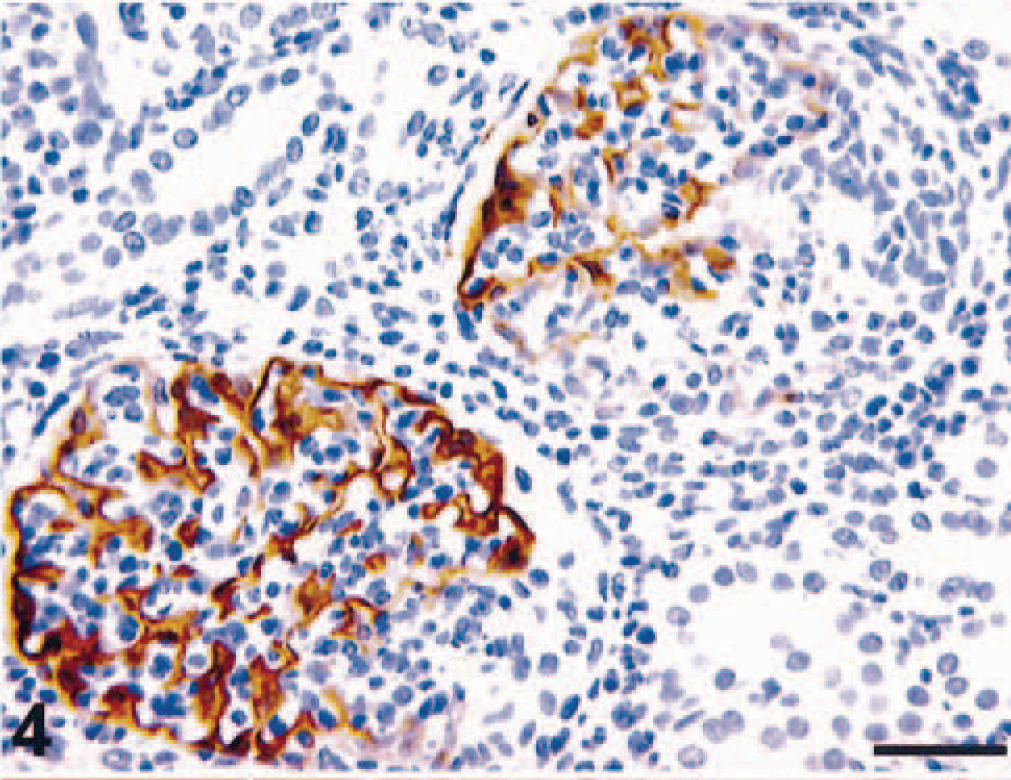
Kidney; pigtailed macaque. Discontinuous expression of GLEPP1 in injured glomerulus. Compare glomerulus with crescent on right with glomerulus on left. ABC peroxidase method, Mayer's hematoxylin counterstain. Bar = 50 µm.
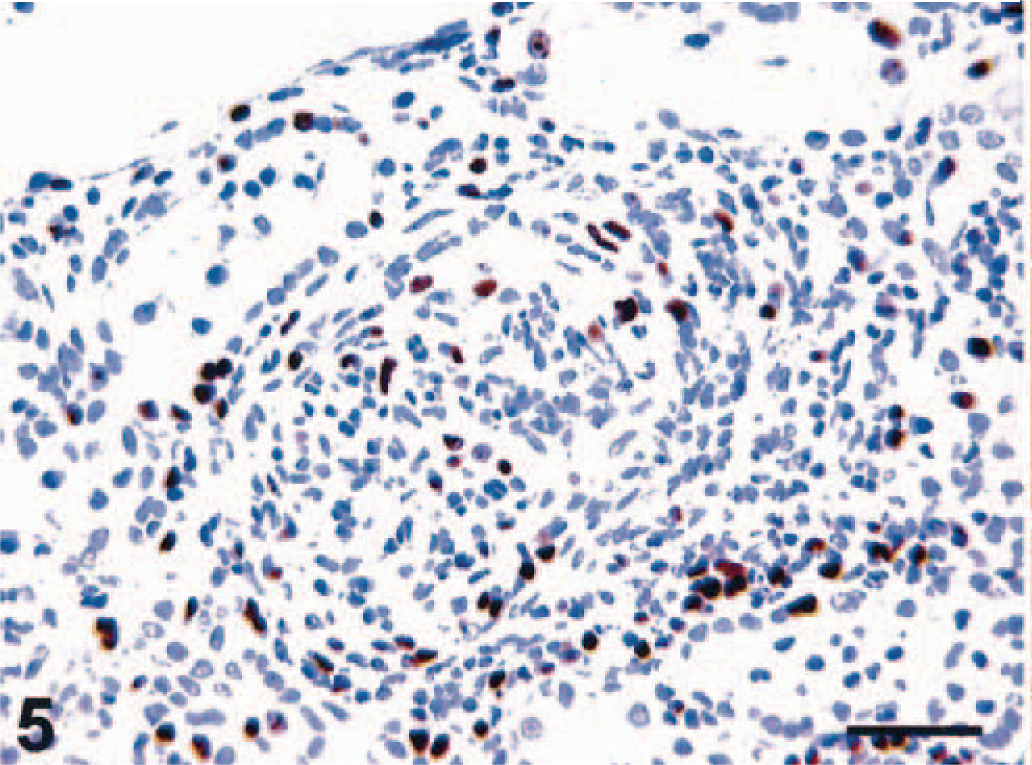
Kidney; pigtailed macaque. Increased Ki-67 (MIB-1) expression in glomerulus, crescent, tubular epithelium, and interstitium. ABC peroxidase method, Mayer's hematoxylin counterstain. Bar = 50 µm.
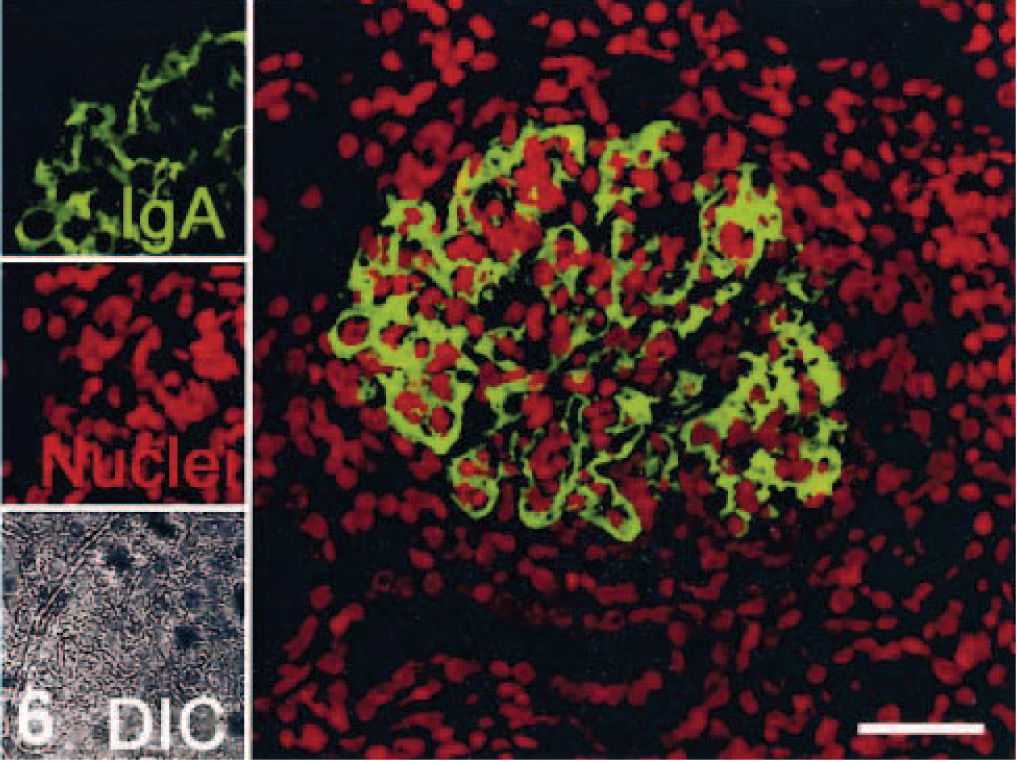
Kidney; pigtailed macaque. Localization of Ig deposition. Image for individual channels are shown on the left, significant IgA deposits in mesangial area, and glomerular basement membranes with Alexa 488 (green); nuclei with To-Pro3 (red), and DIC. A larger merged image containing two channels is shown on the right. Indirect immunofluorescence and confocal microscopy. Bar = 40 µm.
The interstitial infiltrate was composed principally of CD3+ T lymphocytes, CD68+ macrophages, and occasional CD20+ B lymphocytes. Increased collagen Type IV expression was also seen in the tubular basement membranes. Intense Ki67 antigen (MIB-1) expression was seen in the interstitial infiltrate and in the tubular epithelial cells.
Electron microscopy
Electron microscopic examination of the glomeruli revealed that crescents were composed of large cells with round to oval nuclei and abundant cytoplasm containing many organelles including mitochondria, lysosomes, and phagocytic vacuoles. These cells exhibited characteristics of macrophages (Fig. 7). Parietal epithelial cells were generally well preserved. Multifocal fusion of the visceral epithelial foot processes was seen. Extensive intramembranous, sub-endothelial, and mesangial electron-dense deposits were observed within glomeruli (Fig. 8). Increased numbers of mesangial cells with expansion of the mesangial matrix were also observed. Moderate numbers of fascicles composed of fibrin and red blood cells were observed in Bowman's space.
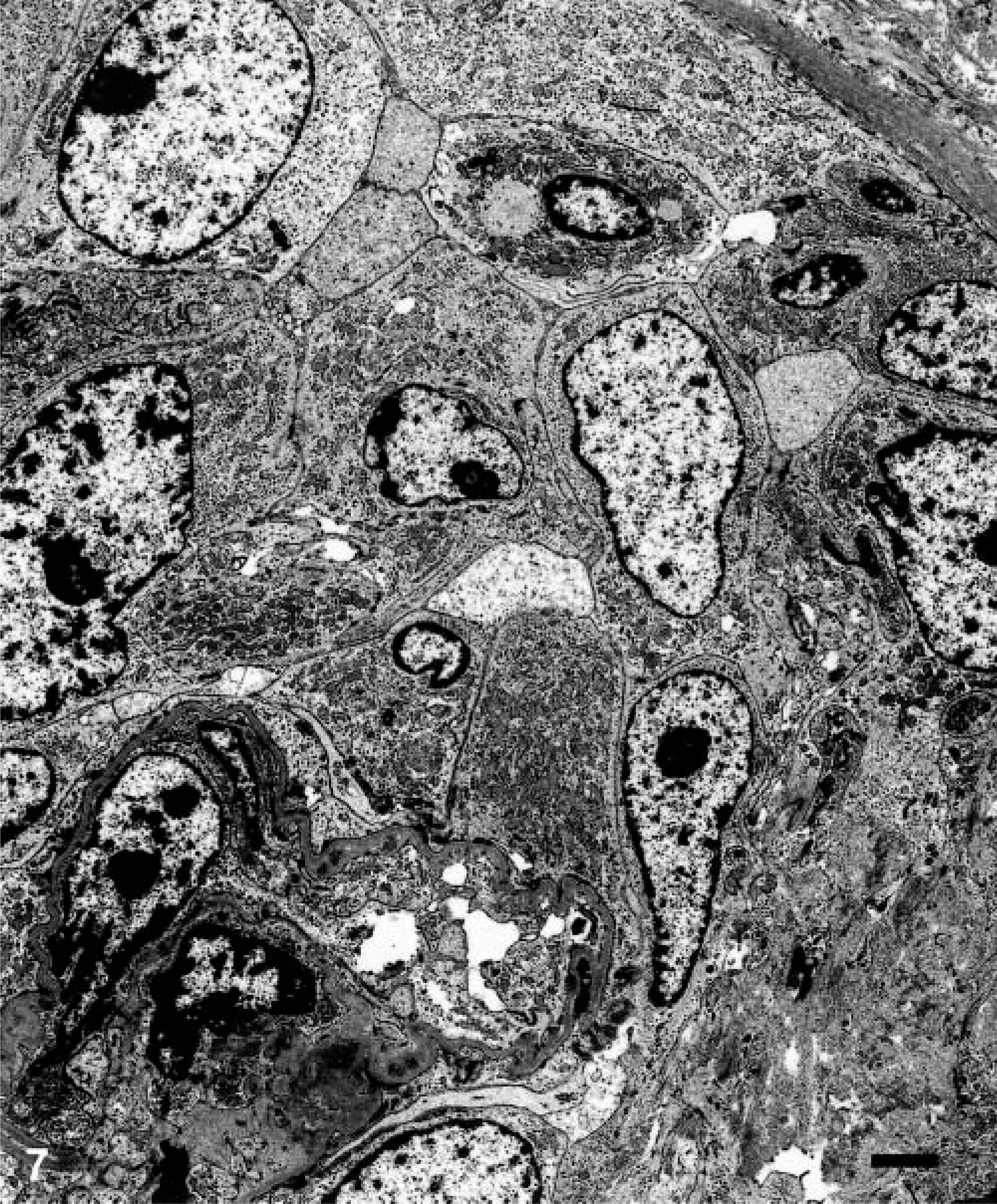
Transmission electron micrograph. Renal glomerulus; pigtailed macaque. Note increased numbers of macrophages in Bowman's space in CGN. Crescents in Bowman's space are composed of macrophages. Bar = 2 µm.
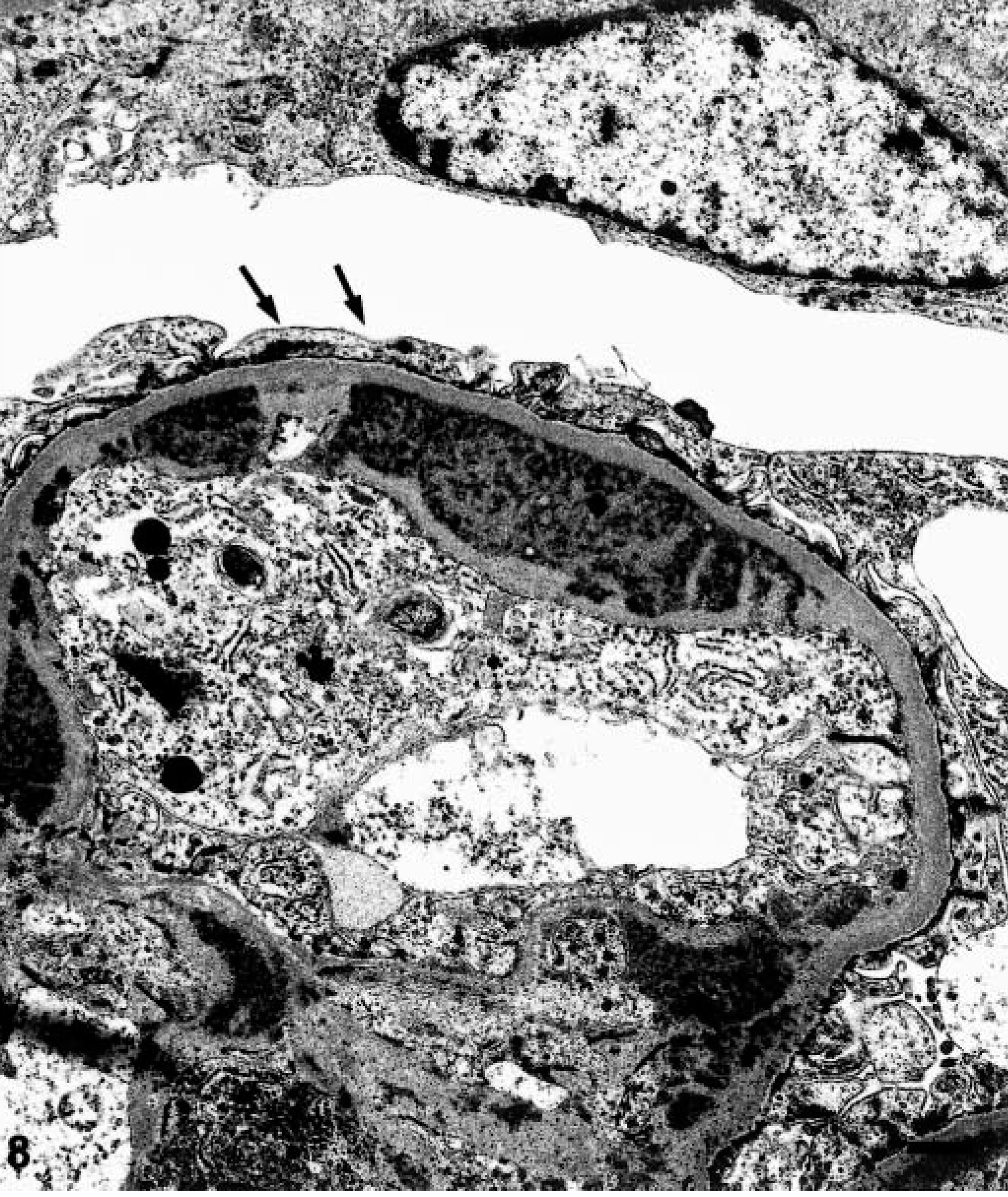
Transmission electron micrograph. Renal glomerulus; pigtailed macaque. Glomerular capillary loops containing electron-dense deposits within the basement membrane, subendothelial area, and mesangium. Foot processes are fused (arrows). Bar = 1 µm.
Discussion
Although MesPGN has been described in both uninfected and SIV-infected macaques, the occurrence of CGN has been only briefly described, and, to our knowledge, this is the first report of IgAN in a macaque. 1,4,11 Features consistent with MesPGN include proliferation of mesangial cells as demonstrated by increased expression of Ki-67 antigen, expansion of the mesangial matrix as shown by increased expression of α-SMA, and fusion of the podocyte foot processes as illustrated by discontinuous staining with GLEPP1 in damaged glomeruli. In this case, crescents were primarily composed of macrophages rather than parietal epithelial cells as demonstrated by immunohistochemistry and electron microscopy. The cellular composition of the crescents may be representative of the stage of crescent formation or the initiating cause of proliferation. Macrophage accumulation and proliferation are not thought to be related to either SIV or BCG infection because in situ hybridization for SIV-infected cells (data not shown) was negative and no acid-fast bacilli were identified with Ziehl-Neelsen staining of the kidney. Last, IgA was the major component of the electron-dense deposits within glomeruli. CGN in humans can occur secondary to either MesPGN or IgAN.
Differential diagnoses in this case include antineutrophilic cytoplasmic autoantibody (ANCA)–associated nephropathy and lupus nephritis. In humans, CGN with intraglomerular macrophages is commonly seen with ANCA-associated nephropathy, but this form of nephropathy is pauci-immune, with few to no detectable glomerular immune complex deposits. 13,17 CGN also occurs in lupus nephritis, but IgG rather than IgA is the primary Ig with deposition in glomeruli, tubules, interstitium, and blood vessels. 8 In this case, the electron-dense deposits were primarily composed of IgA and were localized to the mesangial matrix and the glomerular basement membrane. The lesions observed are most consistent with IgAN with secondary CGN.
Although the monkey was experimentally coinfected with SIV and BCG, the IgAN with secondary CGN is thought to be a spontaneous glomerular disease unrelated to these experimental infections. Focal segmental glomerulosclerosis with IgM and IgG (but not IgA) deposition is the lesion associated with SIV and HIV infection in nonhuman primates and humans, respectively. 1,11,17 Although mycobacterial infection of the kidney can occur in both humans and nonhuman primates, especially in the setting of immunodeficiency, the characteristic lesions are granulomas in which acid-fast bacilli are typically seen. Morphologic features of focal segmental glomerulosclerosis or granulomas (or both) were not observed in this case.
Footnotes
Acknowledgements
This work was partially supported by National Institutes of Health grants RR000164, RR000168. We thank Dr. Norval W. King for his help with transmission EM studies (Department of Pathology, Tufts University School of Veterinary Medicine) and Dr. Andrew A. Lackner for helpful comments (Tulane National Primate Research Center).
