Abstract
Intestinal samples and/or lymph nodes of two Iberian pigs from two different farms were submitted for histopathologic examination. Both pigs had proliferation of ileal and/or cecal crypts with almost complete absence of goblet cells. Infection by Lawsonia intracellularis was demonstrated by immunohistochemistry and polymerase chain reaction assay. The mesenteric lymph node of one pig had moderate lymphocyte depletion with granulomatous inflammation of the lymph node parenchyma. Histiocytes and multinucleated giant cells from the lymph node of one pig contained L. intracellularis antigen within the cytoplasm. This pig had also porcine circovirus type 2 (PCV-2) infection, but nucleic acid and antigen of this virus were not demonstrated in the lymph node. The second pig had lymphocyte depletion and marked granulomatous inflammation in Peyer's patches. Histiocytes and multinucleated giant cells in areas of granulomatous inflammation contained L. intracellularis antigen; no PCV-2 nucleic acid or antigen was detected in the tissues of this pig. This is the first description of granulomatous ileitis and lymphadenitis associated with L. intracellularis infection.
The proliferative enteropathy complex of pigs, caused by the bacterium Lawsonia intracellularis, includes a number of acute and chronic conditions manifested as hemorrhagic diarrhea, soft feces, and unthriftness. 6 Syndromes included are porcine intestinal adenomatosis (PIA), proliferative hemorrhagic enteropathy (PHE), necrotic enteritis, and regional ileitis (RI). All of these conditions involve a characteristic lesion consisting of thickening of the mucosa of the small intestine (mainly ileum), cecum, and/or proximal colon. The basic histologic lesion (PIA) is adenomatous hyperplasia of intestinal crypts, with immature epithelial cells and almost complete absence of goblet cells. 1 Immature enterocytes contain large numbers of the intracellular L. intracellularis in their apical cytoplasm. 6 Here, we describe atypical pathologic and immunopathologic features associated with L. intracellularis infection resulting in one case of granulomatous enteritis and one of granulomatous lymphadenitis in Iberian pigs.
Affected pigs came from two different farms located in the southwest part of Spain, where Iberian pig farms are relatively common. The first farm (A) was a 450-sow, farrow-to-finish operation, in which 2-month-old pigs exhibited yellowish green soft to liquid diarrhea, growth retardation, dehydration, and death. The affected group (703 pigs) included 106 sick animals (15.0% morbidity), and 91 of these animals died (85.8% lethality). At necropsy, performed by a veterinary practitioner, one pig had thickening of ileal, cecal and colonic walls, necrotizing colitis, and enlargement of mesenteric and inguinal superficial lymph nodes. The second farm (B), geographically very close to farm A, was a 400-sow, farrow-to-finish operation experiencing a disease outbreak similar to that on farm A but affecting 3-month-old pigs. The affected group (300 pigs) included 32 sick animals (10.7% morbidity), and four of these animals died (12.5% lethality). At necropsy, thickening of ileal and colon wall, terminal necrotizing ileitis, and a Trichuris suis nematode in the distal colon were observed in one pig. The veterinary practitioner responsible for both farms sent formalin-fixed samples to the Pathology Department of the Veterinary School of Barcelona (Spain). Samples consisted of mesenteric lymph node, ileum, and cecum from one pig on farm A (pig No. 1) and jejunum and ileum from one pig on farm B (pig No. 2). Terminal ileum and cecum of the same pigs were also submitted to the Infectious Diseases Unit of the Veterinary School of Leon (Spain) to detect L. intracellularis, using a previously described polymerase chain reaction (PCR) method. 3
Microscopically, pig No. 1 had marked proliferation of ileal and cecal crypts, with intense epithelial hyperplasia and almost complete absence of goblet cells (Fig. 1). Most of the intestinal crypts were dilated and contained mucus, inflammatory cells (mainly neutrophils), and cellular debris. Peyer's patches appeared slightly to moderately depleted of lymphocytes. There was necrosis of the superficial mucosa, hemorrhage, and fibrin exudation. Degenerating inflammatory cells were present in the ileum and in the cecum. There was a marked lymphoplasmacytic inflammatory infiltrate, and there were small to moderate numbers of macrophages and eosinophils in the cecal lamina propria. A mild inflammatory infiltrate of similar character was observed in the ileal lamina propria. The mesenteric lymph node had moderate lymphocyte depletion and slight to moderate histiocytic infiltration of the lymph node parenchyma. Rare multinucleate giant cells were also detected in the medulla of the lymph node. A few foci of epithelial cells were seen in medullary sinuses of the mesenteric lymph node. Pig No. 2 showed marked blunting of villi and markedly dilated intestinal crypts. These ileal crypts were almost devoid of goblet cells and contained degenerate inflammatory cells. A marked lymphoplasmacytic inflammatory infiltrate, together with small to moderate numbers of macrophages, neutrophils, and eosinophils, was present within ileal mucosa. Peyer's patches were markedly depleted of lymphocytes, and a marked granulomatous inflammatory infiltrate consisting of histiocytes and multinucleate giant cells was present (Fig. 2). Small to moderate numbers of neutrophils were present within the granulomatous infiltrate. A similar lesion was also seen in ileal mucosa to a lesser degree. The lamina propria of the jejunum from this pig showed mild lymphoplasmacytic inflammation.
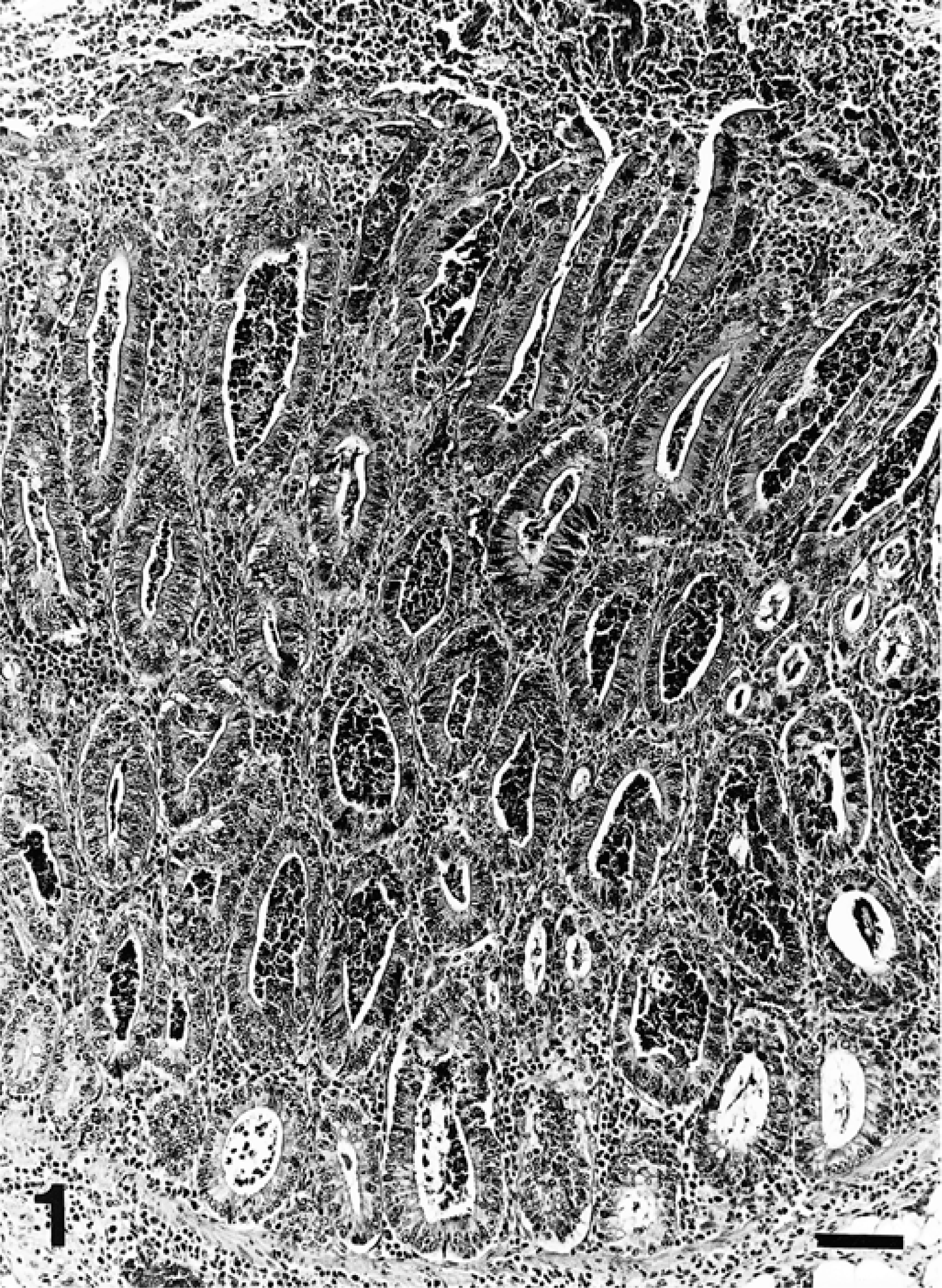
Ileal mucosa; pig No. 1. Marked proliferation of immature crypts, with superficial necrosis. Crypts are dilated and filled with necrotic and inflammatory debris. HE. Bar = 100 µm.
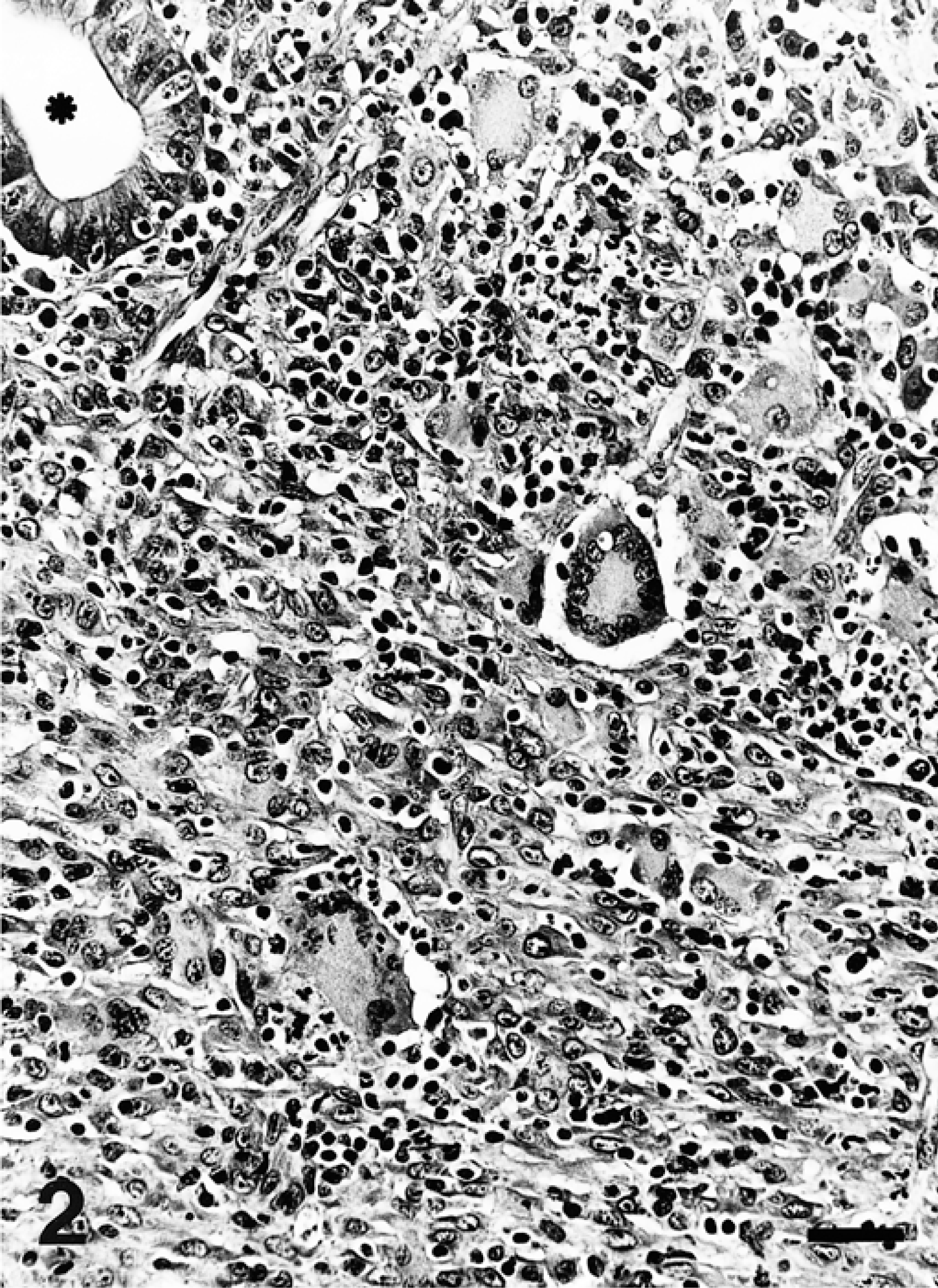
Peyer's patches; pig No. 2. Granulomatous inflammatory infiltrates diffusely distributed in the lymphoid tissue. An ileal crypt is also seen (asterisk). HE. Bar = 50 µm.
Based on the histopathologic changes, submitted tissues were evaluated by immunohistochemistry to detect L. intracellularis, 5 in situ hybridization to detect porcine circovirus type 2 (PCV-2), 8 immunohistochemistry to detect porcine reproductive and respiratory syndrome virus (PRRSV), 2 and special stains (Warthin–Starry, Grocott, Gram, and Ziehl–Neelsen). Large amounts of L. intracellularis antigen were detected in the apical cytoplasm of enterocytes located in hyperplasic crypts and within the cytoplasm of macrophages in the inflammatory infiltrates in the lamina propria in the ileum and cecum of pig No. 1 and in the ileum of pig No. 2. Antigen also was detected in the apical cytoplasm of epithelial cells in the medullary sinuses of the mesenteric lymph node of pig No. 1, in the cytoplasm of macrophages and multinucleate giant cells in the mesenteric lymph node of pig No. 1 (Fig. 3), and in the cytoplasm of macrophages and many multinucleated giant cells in the Peyer's patches of pig No. 2 (Fig. 4). These findings were corroborated by positive Warthin–Starry staining in the same locations as antigen detection. Pig No. 1 had PCV-2 nucleic acid and antigen in the cytoplasm of those macrophages of the lamina propria of ileum and cecum located close to the submucosa and in the center of Peyer's patch follicles. The mesenteric lymph node of pig No. 1 and all samples from pig No. 2 lacked PCV-2 nucleic acid and antigen. All samples were negative for PRRSV by immunohistochemistry, and for bacteria or fungi with Ziehl–Neelsen, Gram, and Grocott stains. PCR results showed 319-bp bands specific for L. intracellularis from samples of both pigs.
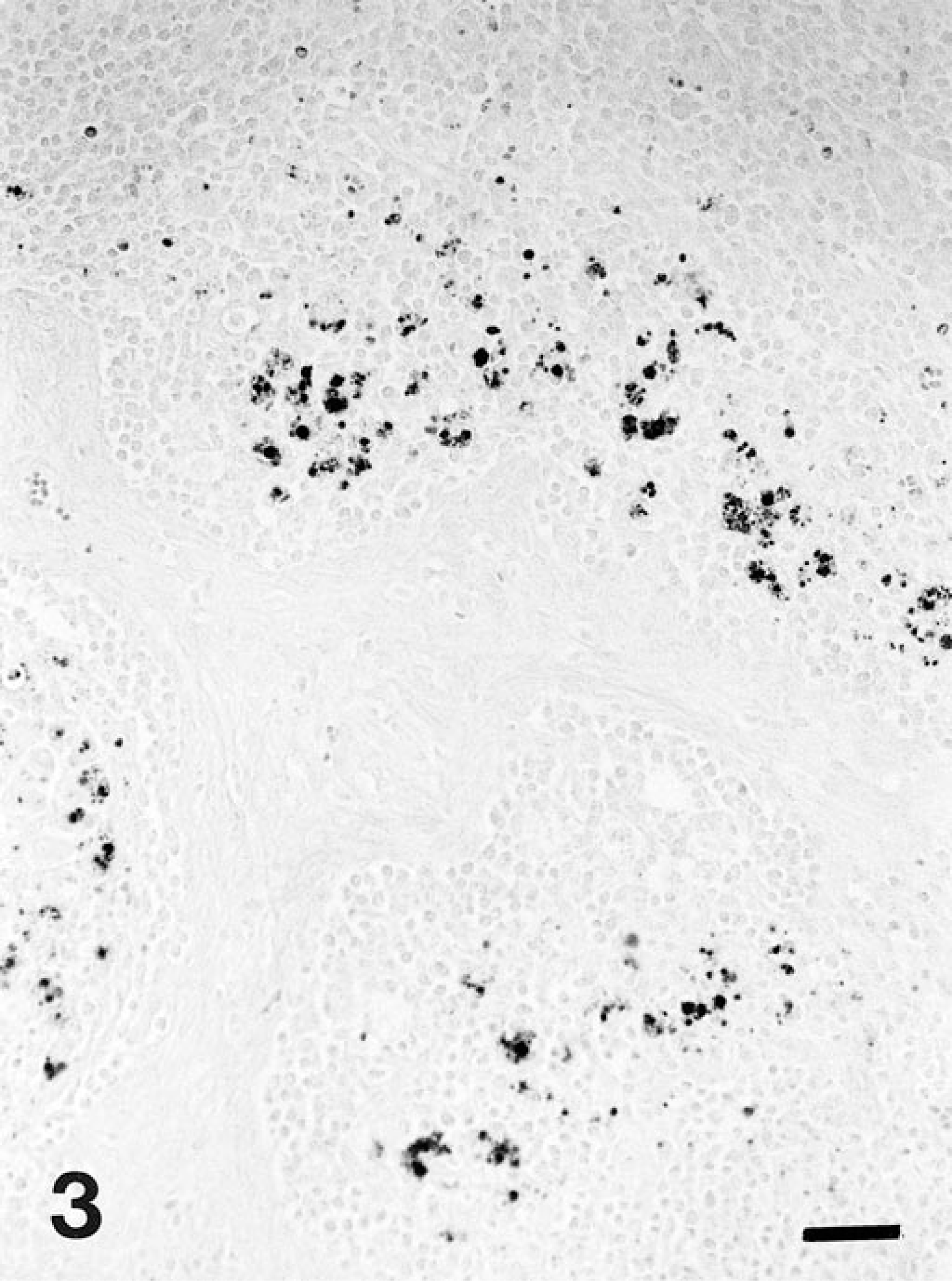
Mesenteric lymph node; pig No. 1. Immunostaining for Lawsonia intracellularis reveals the presence of antigen within cells of lymphoid follicles, close to medullary traveculae. Avidin–biotin–peroxidase complex method, fast green counterstain. Bar = 50 µm.
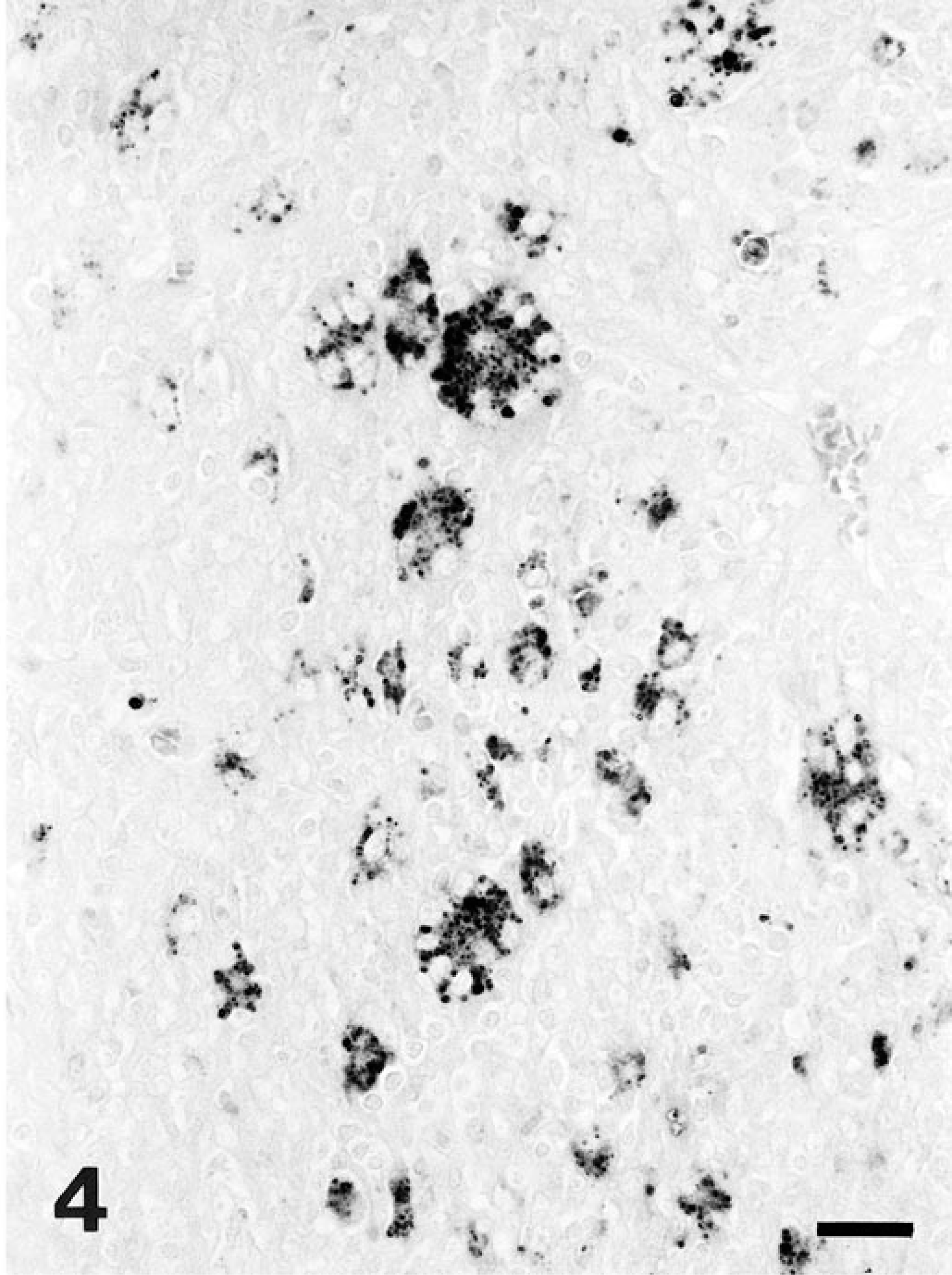
Peyer's patches; pig No. 2. Immunostaining for Lawsonia intracellularis reveals marked presence of bacterial antigen within the cytoplasm of multinucleated giant cells and macrophages. Avidin–biotin–peroxidase complex method, fast green counterstain. Bar = 25 µm.
Histopathologic features, immunohistochemistry, PCR, and in situ hybridization studies of these pigs led to the diagnosis of PHE and PCV-2 infection in pig No. 1 and PIA in pig No. 2. Proliferative enteropathies have been previously characterized pathologically, 7,9 but to our knowledge this is the first description of granulomatous enteritis (ileitis) and mesenteric lymphadenitis associated with L. intracellularis infection. The large amount of L. intracellularis antigen within multinucleated giant cells and macrophages in the Peyer's patches and the smaller amount of antigen in the mesenteric lymph node suggested a potential role for this bacterium in causing granulomatous inflammation. This suggestion is bolstered by the absence of other possible etiologic agents, including fungi, bacteria (Rhodococcus equi, Salmonella choleraesuis), foreign bodies, Mycobacterium spp., or viruses (PCV-2 and PRRSV). The pig showing granulomatous lymphadenitis was also infected with PCV-2, but viral nucleic acid and antigen were not detected in the mesenteric lymph node, although they were present in Peyer's patches and the lamina propria of the intestine. Granulomatous RI with mesenteric lymphadenitis in swine has only been described once, in Finland. In that report, the authors concluded that the granulomatous lesions were not associated with porcine intestinal adenomatosis or tuberculosis. 1 Foci of epithelial cells within mesenteric lymph nodes, rather infrequent in animals with PIA, have also been previously described. 4 Although the mechanism is unknown, it has been hypothesized that the proliferating epithelial cells may enter an afferent lymphatic vessel and be carried to a regional lymph node, where these cells retain their proliferative capacity. 4
L. intracellularis should be included in the differential diagnosis of granulomatous enteritis and mesenteric lymphadenitis in swine. However, the possibility that these unusual lesions containing L. intracellularis antigen might be associated with concurrent infections by other etiologic agents cannot be ruled out. This is the first report of PIA complex and PCV-2 infection in Iberian pigs. Iberian pigs were traditionally reared in outdoor production systems, but in the last 5 years those systems have been changed to semiintensive and intensive production forms. This fact may have led to similar clinical disease presentation in conventional and Iberian pigs.
Footnotes
Acknowledgements
We thank B. Pérez, S. Usero, and P. Losada for technical assistance.
