Abstract
We evaluated the expression of vimentin, S100a, and Melan A/MART-1 (melanoma antigen recognized by T cells 1) in seven cell lines established independently from dogs with canine melanoma. We also compared routine immunostaining of 29 clinical specimens from melanoma cases using vimentin, S100a, and neuron-specific enolase (NSE) with staining for Melan A/MART-1 as part of a diagnostic panel. All the cell lines were positive for expression of vimentin and S-100a. MelanA/MART-1 expression was seen consistently in only two of the seven cell lines. Staining for Melan A/MART-1 was most intense near areas of heavy melanin pigmentation. All except one of the clinical specimens were positive for vimentin. S100a was expressed in the majority of both pigmented (15/20, 75%) and amelanotic (8/9, 88.8%) tumors. Seventeen of 29 (58.6%) tumors were positive for NSE. Melan A/MART-1 was expressed in 18/29 (62%) tumors, including 90% of pigmented tumors, but in no amelanotic tumors. Intensity of Melan A/MART-1 staining correlated positively with biologic behavior, with seven malignant tumors showing negative to weak staining and 10 benign tumors showing moderate to strong staining. Three malignant tumors showed moderate to intense staining for Melan A/ MART-1. Our results suggest that expression of Melan A/MART-1 may be unstable in cultured cell lines. Assessment of both S100a and Melan A/MART-1 expression is useful to confirm a diagnosis of canine melanoma, and Melan A/MART-1 may be especially informative regarding the biologic behavior of these tumors.
Dermal melanomas account for 9–20% of skin tumors in dogs and generally follow a benign course. Melanomas are the most common malignant tumor of the oral cavity and digits in dogs; 2,21,24,38 at least 90% of these tumors are malignant, 24 and many have metastasized by the time of diagnosis. However, few oral melanomas follow a benign course, and some dermal melanomas are malignant, making definitive prognostication for these tumors difficult. Melanocytes arise from embryonic neuroectoderm and, as such, retain the ability to differentiate into spindled or epithelioid cells, making a diagnosis of canine melanoma challenging in poorly differentiated amelanotic tumors. In these cases, determining the presence of constituent proteins whose expression is restricted to melanin-producing cells or cells arising from neuroectodermal tissues can assist in the diagnosis. Previous studies documented the use of vimentin and S100 to assist in the diagnosis of canine melanoma, but their potential prognostic significance was not examined in detail. 29,34,35 In a recent study examining immunohistochemical staining of oral canine melanomas, Melan A was considered a specific and sensitive marker for canine melanomas. 30
The present study was designed to define a panel of immunohistologic markers, based on the expression of vimentin, S100a, and Melan A/MART-1 (melanoma antigen recognized by T cells 1) that may offer diagnostic and prognostic information for canine melanoma. We examined seven canine melanoma cell lines representing tumors obtained from dogs with distinct presentations of disease (i.e., two primary nonmetastatic tumors, two primary metastatic tumors, a recurrent tumor, and two distant metastases) and a retrospective cohort consisting of 29 melanomas from 27 dogs admitted to the Texas Veterinary Medical Center (TVMC).
Vimentin is an intermediate filament that is expressed by mesenchymal and neuroectodermal cells in normal tissues. 19 Neuron specific enolase (NSE) is a glycolytic enzyme expressed by cells of neuronal or neuroectodermal origin. 11 S100a is an isoform of a calcium binding protein restricted to neuroectodermal cells, 37 and Melan A/MART-1 is a protein of unknown function that is expressed mainly by melanocytes. 8
Our objective was to define the usefulness of these five proteins to confirm cellular ontogeny, to compare the immunostains routinely used to diagnose melanomas (vimentin, S100, NSE) with Melan A/MART-1, and to identify any consistent staining properties that may predict malignancy. Our hypotheses were that 1) well-differentiated melanomas consistently express vimentin, S100a, NSE, and Melan A/MART-1, 2) poorly differentiated melanomas and melanomas with high metastatic potential lack expression of S100a, NSE, or Melan A/MART-1, and 3) Melan A/MART-1 is a more specific indicator of melanocytic differentiation than S100 and NSE.
Materials and Methods
Cell lines
The cell lines utilized were derived from primary tumors of dogs with oral melanoma (n = 5), a lymph node metastasis of cutaneous melanoma (n = 1), and a lung metastasis of oral melanoma (n = 1) and have been described previously 12,32,39 (Table 1). CML-2, CML-6MC2, and CML-13 had been in culture for several hundred passages at the time of this study. SHADOW cells were at passage 75, JENNY cells were at passage 81, SCOOTER cells were at passage 20, and TLM-1 cells were passed less than 30 times (16–27) at the time of the study.
Characteristics of canine melanoma cell lines.
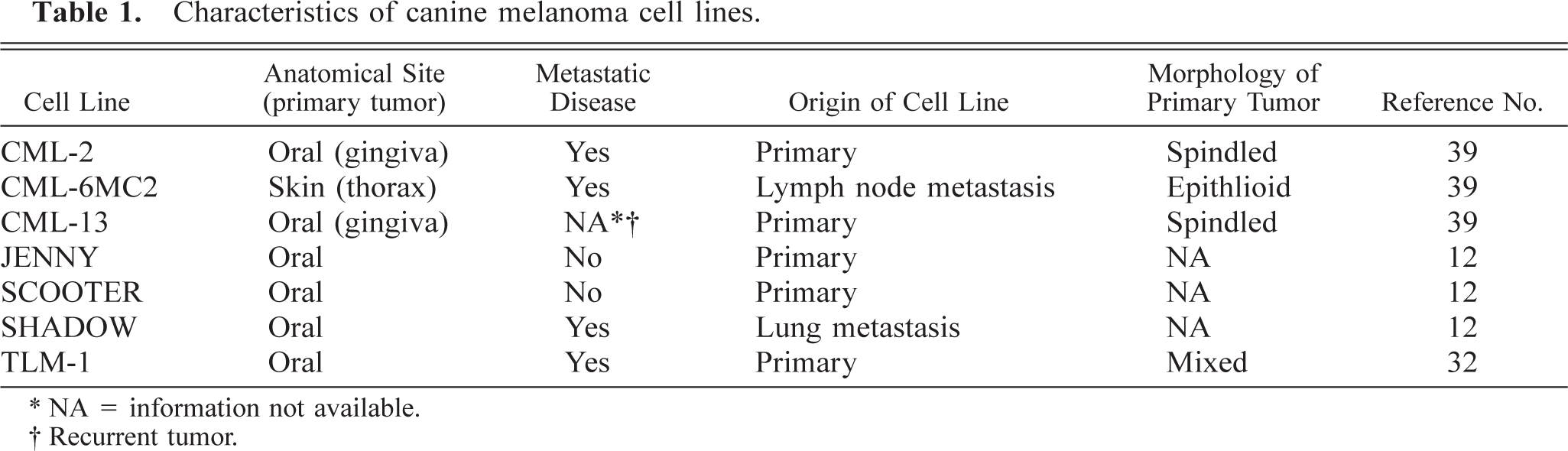
∗ NA = information not available.
† Recurrent tumor.
Clinical specimens
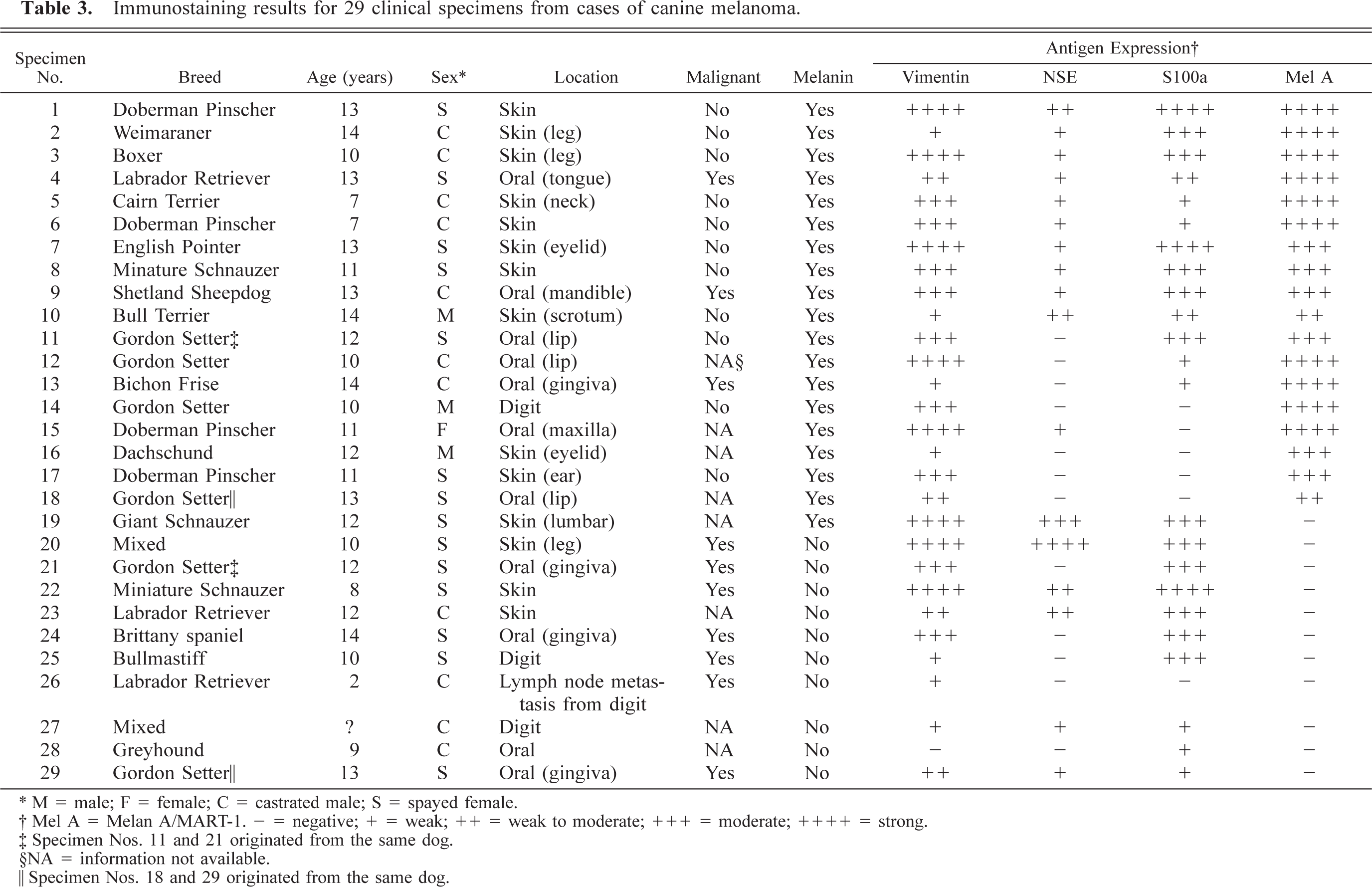
Primary cases were retrieved from the medical records database of the TVMC. Every case that had a histologic diagnosis of melanoma between 1994 and 1997 was reviewed for eligibility; only tumors with formalin-fixed archival material suitable for immunostaining were utilized. These consisted of 11 primary oral tumors (one tongue, three lip, seven gingiva), 17 dermal melanomas (three digit, three leg, two eyelid, two trunk, one ear, one scrotum, five unspecified), and one lymph node metastasis from a digital tumor. Twenty of the tumors were pigmented, and nine were amelanotic. The tumors were categorized as malignant or benign based on pleomorphism, degree of proliferation, local invasion, and documentation of metastasis. Pleomorphism and proliferation were not considered adequate indicators of malignancy in the absence of known recurrence, invasiveness, or documentation of metastasis. Using these criteria, nine tumors were identified as malignant, 11 were called benign, and the behavior of eight was not evident from the histology or history. The cases represented 27 dogs and 17 different breeds, with a distribution as follows: four Gordon Setters, four Doberman Pinschers, three Labrador Retrievers, two mixed breed dogs, two Miniature Schnauzers, and one each of Giant Schnauzer, Brittany spaniel, Bichon Frise, Cairn Terrier, Bull Terrier, Weimaraner, Boxer, Bullmastiff, Dachshund, Shetland Sheepdog, English Pointer, and Greyhound. The dogs ranged in age from 2 to 14 years, with an average age of 11 years. The age of one dog was unknown. Eleven of the dogs were neutered males, three were intact males, 12 were spayed females, and one was an intact female.
Cell culture
Tissue culture materials were obtained from Nalge Nunc (Naperville, IL). Cells were cultured in Dulbecco's modified Eagle medium (Gibco BRL, Grand Island, NY) containing 10% heat-inactivated fetal calf serum (Hyclone, Logan, UT) in a humidified atmosphere containing 5% CO2 at 37 C. Cells (5 × 104 in 100 µl) were grown in double-chamber slides until they reached 70–80% confluence. All the cell lines grew as monolayer cultures and were maintained by passage when they reached >90% confluence.
Immunocytochemistry and immunohistochemistry
At the end of the culture period, plastic chambers were removed, and slides were rinsed in phosphate buffered saline, pH 7.4, fixed in acetone at 4 C for 5 minutes, and air dried. For archival tissues, 5 µm serial sections from paraffin-embedded blocks were mounted on positively charged slides (Probe-on, Fisher Scientific, Pittsburgh, PA). Immunostaining was performed using a modified streptavidin–biotin–alkaline phosphate (ABC) method as described. 31,32 For clinical specimens, citrate, pH 6.0 microwave antigen retrieval was used for vimentin immunostaining, and ethylenediaminetetraacetic acid, pH 8.0, microwave antigen retrieval was used for Melan A/MART-1 immunostaining. Antigen retrieval was not used for NSE immunostaining or for any antigen in the cell lines. Slides were incubated with primary antibodies against vimentin (mouse monoclonal, clone V9, Dako, Carpinteria, CA) diluted 1:1,000, S100a (rabbit polyclonal, Dako) diluted 1:1,000, NSE (mouse monoclonal antibody, clone BBS/NC/VI-H14, Signet Pathology Systems, Dedham, MA) diluted 1:1,000, and Melan A/MART-1 (mouse monoclonal, clone A103, supplied by Novocastra, UK, through Vector Laboratories, Burlingame, CA) diluted 1:50 for 1 hour at room temperature. The “a” designation in the S100a antibody denotes the clone and not the A antigen. This antibody is reported by the manufacturer to label determinants common to the alpha and beta chains of S100a (α/α) and S100b (β/β). As per the manufacturer's recommendation, this antibody does not require antigen retrieval, and expression of this isoform appears to be restricted to neuroectodermal cells. 14,37 Secondary antibodies consisted of goat anti-rabbit IgG or goat anti-mouse IgG conjugated to biotin (Kirkegaard & Perry Laboratories, Gaithersburg, MD). The presence of the relevant antigens was detected using streptavidin conjugated to alkaline phosphatase. Following a rinse in Tris buffer, the color reaction was accomplished using the Histomark red kit (Kirkegaard & Perry Laboratories). Negative controls were prepared by using irrelevant isotype matched antibodies in place of the primary antibodies. Sections obtained from canine tissues (liver, kidney, brain, spleen, gut, skin) served as positive controls for the immunostains.
Cell cultures and cell lines were graded for intensity and percentage of positive cells. Samples were considered negative if staining was no greater than that of negative controls. Staining that was fine, diffuse, and not always visible at low magnification (40–100×) was considered weak. Staining that was punctate or regional but prominent enough to be seen at low magnification was called considered moderate. Staining that was diffuse, prominent, and easily visualized at low magnification was considered strong.
Results
Expression of vimentin
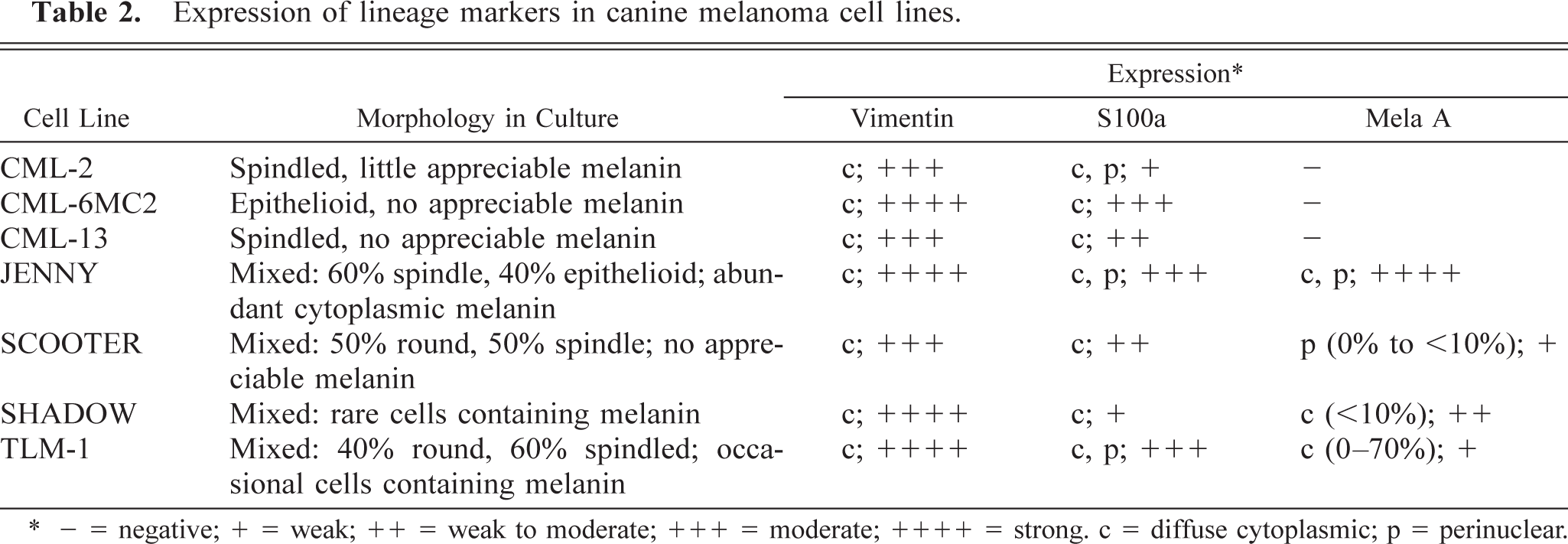
Each of the cell lines expressed vimentin (Table 2). CML-2 and CML-13 cells showed uniform cytoplasmic distribution of vimentin staining, whereas the remaining cell lines showed focal clumping that was mostly perinuclear in CML-6MC2 and TLM-1 cells, polar in JENNY cells, peripheral in SHADOW cells, and a combination of perinuclear, peripheral and polar in SCOOTER cells. All but one of the clinical specimens (27/28) expressed vimentin.
Expression of lineage markers in canine melanoma cell lines.
∗ - = negative; + = weak; ++ = weak to moderate; +++ = moderate; ++++ = strong. c = diffuse cytoplasmic; p = perinuclear.
Expression of S100a
Each of the cell lines expressed S100a (Table 2). The distribution of S100a within the cytoplasm was even and granular, but some cells in the TLM-1, CML-2, and JENNY cell lines showed preference for polar to bipolar or perinuclear distribution (Fig. 1).
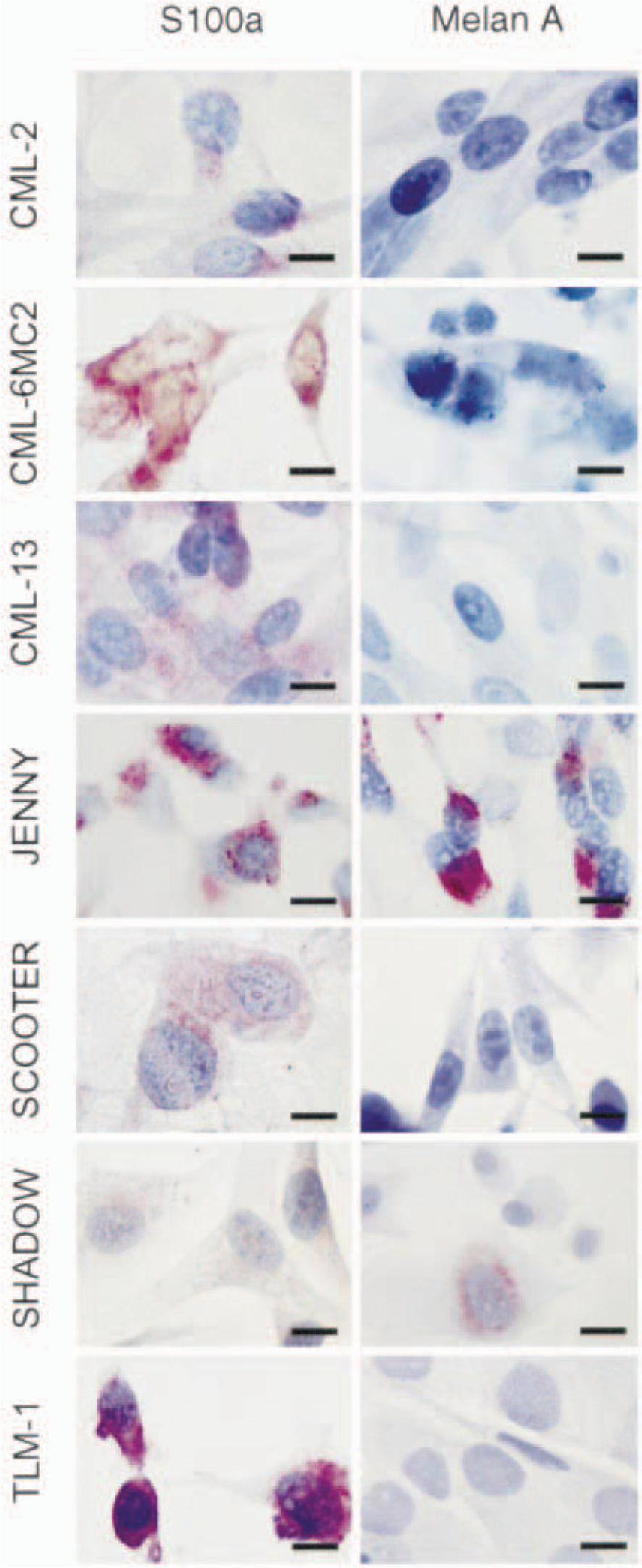
Melanoma cell lines CML-2, CML-6MC2, CML-13, JENNY, SCOOTER, SHADOW, and TLM-1; dogs. Cells were cultured in chamber slides and prepared for immunocytochemical staining. Each cell line was stained for expression of S100a or Melan A/MART-1 as indicated. An irrelevant rabbit IgG was used for the negative controls. ABC reaction. Bar = 10 µm.
Twenty-three of the 29 clinical specimens (79%) were positive for S100a (Table 3). Of those 23, 15 (65%) contained melanin. Seventy-five percent of the 20 pigmented tumors and 89% of the amelanotic tumors were S100a positive. Aproximately equal numbers of benign and malignant tumors were classified as having strong, moderate, and weak to negative staining (Table 3). Additionally, although the primary digital tumor from which specimen No. 26 (lymph node metastasis) was derived was not available for immunostaining at the time of this study, the original biopsy report revealed that the primary tumor also was positive for S100a.
Immunostaining results for 29 clinical specimens from cases of canine melanoma.
∗ M = male; F = female; C = castrated male; S = spayed female.
† Mel A = Melan A/MART-1. - = negative; + = weak; ++ = weak to moderate; +++ = moderate; ++++ = strong.
‡ Specimen Nos. 11 and 21 originated from the same dog.
§NA = information not available.
¶ Specimen Nos. 18 and 29 originated from the same dog.
Expression of Melan A/MART-1
Approximately 70% (40–100%) of JENNY cells stained for Melan A/MART-1 (Fig. 1, Table 2). Half of these cells stained vividly in polar perinuclear clumps that extended out into the cytoplasm, especially in spindled cells. The other half had pale Golgilike perinuclear uptake. Stain uptake in both instances appeared to be associated with melanin granules. Approximately 10% (5–25%) of SHADOW cells were positive for Melan A/MART-1 expression; the antigen was localized to the cytoplasmic and perinuclear compartments with a Golgi-like distribution (Fig. 1, Table 2). CML-2, CML-6MC2, and CML-13, three of the cell lines that had no visible melanin granules, were negative for Melan A/MART-1 expression (Fig. 1, Table 2). SCOOTER and TLM-1 cells showed variable results (Table 2). TLM-1 cells ranged from approximately 60–70% weakly positive cells to completely negative for Melan A/MART-1 expression (Fig. 1). SCOOTER cells ranged from approximately 10% positive cells to completely negative (Fig. 1).
Melan A/MART-1 was expressed by 18/29 (62%) clinical specimens; all 18 positive specimens were derived from pigmented tumors (Table 3). Among the pigmented tumors, 18/20 (90%) were positive for Melan A/MART-1 expression, and 2/20 (10%) were negative. All the amelanotic tumors were negative for Melan A/MART-1 expression. Of the 20 tumors for which behavior (malignant vs. benign) was known, seven malignant tumors were negative for Melan A/MART-1 expression, 10 benign tumors and three malignant tumors were classified as moderate or strong (3–4+), and one benign tumor was classified as weak to moderate (2+).
Expression of NSE
NSE was expressed in 18/29 (62%) of the tumors (Table 3). NSE was expressed in 12/20 pigmented tumors and 5/9 amelanotic tumors.
Discussion
Various lineage markers are routinely used to diagnose and stage human malignant melanoma, 36 but the application of such markers for diagnosis of canine melanoma and to provide prognostic information has been limited. 23,29,30,34,35 The present study was designed as a preliminary investigation to begin to define a panel of immunohistologic markers that may offer diagnostic and prognostic information for canine melanoma. We evaluated the expression of vimentin, S100a, and Melan A/MART-1 in seven canine melanoma cell lines and the expression of vimentin, S100a, NSE, and Melan A/MART-1 in 29 clinical specimens obtained retrospectively from the archives of the TVMC. An important advantage of these markers and of the antibodies used is that antigenic reactivity is preserved or can be retrieved after routine tissue fixation in 10% formalin. Retrieval after fixation has been confirmed for each antibody in one of our laboratories (J. Wojcieszyn), making it possible to extend these studies retrospectively or prospectively to clinical cases.
Cultured cells have been used extensively for evaluation of tumor markers. 10,20,25,33 Cultured cells were chosen as an in vitro model of canine melanoma for this study because of the various advantages they offer. Cell lines provide reliable and reproducible sources of tissue to develop and refine the application of immunohistologic markers for melanoma. They also allow manipulation of genes and proteins in living cells and could be useful for gene manipulation in further studies. However, there are inherent disadvantages associated with using cultured cells, such as in vitro selection that can influence gene expression. 31 Several issues were considered in building the panel of four lineage markers chosen for this study. First, evaluation of patterns of structural and “housekeeping” protein expression may offer diagnostic and prognostic information. S100 proteins and NSE are useful in the diagnosis of neuronal and neuroectodermal tumors. In addition, survival time is inversely related to the intensity of S100 staining in malignant melanomas of the facial skin and oral cavity in human patients. 18 S100 staining characteristics have also been useful in the diagnosis of canine amelanotic melanomas. 29,34 The use of a broad S100 immunostain requires antigen retrieval, which also unmasks the S100 antigen normally expressed in the epidermis, dermis, and sweat glands, making examination more difficult. However, the S100a antibody does not require antigen retrieval, and expression of this isoform appears to be restricted to neuroectodermal cells. 14,37
Each of the cell lines examined in the present study expressed vimentin and S100a. The CML-6MC2 cell line, which was derived from a metastasis, also stained most intensely for S100a. Aside from this observation, there were no consistent differences in the magnitude or location of vimentin or S100a expression in the cell lines. Thus, for this small sample size, these markers did not consistently predict behavior of these cell lines (growth rate, morphology, or melanin production) or the parental tumors (location, recurrence, or metastasis) whether used alone or in combination. All but one of the clinical specimens (No. 28) expressed vimentin, ranging from weak to strong intensity. Although vimentin-negative melanomas have been reported, 3 they are uncommon, and specimen No. 28 may have been a poorly differentiated melanoma or another type of tumor.
S100a identified 79% of all clinical specimens and 75% of pigmented tumors. These findings are similar to those from another study of immunostaining of canine melanomas. 30 In humans, polyclonal S100 is expected to stain nearly 100% of all melanocytic tumors, 5,26,28 but S100 negative tumors have been identified. 1 Of the amelanotic tumors, which would pose the greater diagnostic dilemma, 89% were positive for S100a. These results were similar to findings in other studies of human and canine melanomas. 27,34
Only two of seven cell lines tested in the present study were consistently positive for Melan A/MART-1 expression. Although lack of melanin production has been reported not to be related to Melan-A/MART-1 staining, 15 the two positive cell lines (JENNY and SHADOW) were the two cell lines with the most melanin, and stain uptake for Melan A/MART-1 was in close proximity to the melanin granules. Similarly, the CML-2, CML-6MC2, and CML-13 cell lines that had no visible melanin were consistently negative for Melan A/MART-1. Two of the cell lines, SCOOTER and TLM-1, had variable staining for Melan A. Loss of melanin granules in melanoma cell lines has been associated with prolonged passage in vitro. 32,39 CML-2, CML-6MC2, and CML-13 have the longest history in culture (several hundred passages). SHADOW cells were at passage 75, JENNY cells were at passage 81, SCOOTER cells were at passage 20, and TLM-1 cells had been passed less than 30 times at the time of the study. All cells had been stored in liquid nitrogen rather than maintained in continuous culture since their establishment. Therefore, it does not appear that number of passages alone is correlated with melanin production or Melan A/MART-1 expression in these cell lines.
Loss of Melan A/MART-1 expression in vitro also was described in a recent report where one of three short-term cell cultures derived from Melan A/MART-1–positive tumors did not express Melan A/MART-1 mRNA. 13 In the present study, some derivatives of the TLM-1 cell line had no visible melanin and also lacked expression of Melan A/MART-1 in culture. Nonetheless, the significance of this finding regarding canine melanoma is unclear. Melan A/MART-1 may be important for melanin synthesis in pathways that are distinct from tyrosinase; some pigmented tumors lack immunoreactivity against tyrosinase 10 or other melanoma-specific antigens. If Melan A/MART-1 were important for pigment production, its absence from cells that lack melanin would be predictable. Additionally, melanin production can be reinduced in canine melanoma cell lines that have lost melanin expression by growing the cells on soft agar, 39 but promotion of Melan A/MART-1 expression under these same conditions has not been examined. Because Melan A/MART-1 is an immunodominant antigen, loss of its expression could be a way for melanomas to evade the immune system. Among the cell lines that did not express Melan A/MART-1 were CML-2, which was derived from a tumor that had metastasized at the time of diagnosis, CML-6MC2, which was derived from a lymph node metastasis, and CML-13, which was derived from a recurrent tumor. However, SHADOW cells, which were derived from a metastatic tumor expressed Melan A/MART-1, albeit the frequency of positive cells was low. There does not appear to be an absolute correlation between melanin pigment or metastatic potential and Melan A/MART-1 expression; Melan A/MART-1 expression has been documented in cytologic preparations from lymph node metastases of amelanotic canine melanomas (J. F. Modiano, J. Wojcieszyn, unpublished data).
In the retrospective analysis of sporadic canine melanoma, Melan A/MART-1 identified 62% of all clinical specimens, 90% of the pigmented tumors, and none of the amelanotic tumors. Melan A/MART-1 has been described as a sensitive and specific marker for cells of melanocytic origin. 7 In one study, 89.1% of canine melanomas were positive for Melan A/MART-1. 30 In humans, results of immunostaining for Melan A/MART-1 seem to vary widely depending on the type of lesion. 4,13,15,22 In multiple studies, normal melanocytes exhibited more homogeneous staining and a higher percentage of Melan A/MART-1-positive cells than did malignant melanocytes or cells in melanoma metastases. 9,10,16,20 Melan A has also been reported to stain epithelioid melanomas more intensely and consistently than spindloid variants, 6,15 but this differential staining was not evident in the present study (data not shown). Melan A is reported to have a higher sensitivity for human amelanotic melanomas than was demonstrated here. 17 A higher percentage of immunoreactivity has also been demonstrated in melanoma metastases of both humans (81%) 17 and dogs (71%). 30 The reasons for the lower percentage of Melan A-positive tumors in the present study are unclear but, as for the cell lines, may be related to issues of pigment production, selection of variants with high proliferative potential and low immunogenicity, the ABC detection method (which may be less sensitive than other methods), or other phenotypic features of these tumors that cannot be classified at this time. For example, the more detailed classification scheme for human melanocytic lesions may help explain some of the disparity in immunostaining between canine and human melanomas. Alternatively, the tumors could represent other neoplasms of neuroectodermal origin that were not fully committed to the melanocyte lineage.
Based on the data presented here, S100a appears to be more sensitive then Melan A/MART-1 for the diagnosis of canine melanoma, especially for identification of amelanotic variants; however, Melan A/MART-1 may offer valuable prognostic information. Approximately equal numbers of benign and malignant tumors were classified as strong, moderate, and weak to negative in staining for S100a. In contrast, seven malignant tumors were classified as negative or weak (− to 1+) for Melan A/MART-1 expression, and only three malignant tumors were classified as moderate or strong (3–4+) for Melan A/MART-1 expression. Conversely, 10 benign tumors were moderate or strongly positive for Melan A/MART-1 expression (3–4+), and none were classified as negative or weak (− to 1+). The positive correlation between intensity of Melan A/MART-1 expression and benign behavior in this clinical cohort was significant (P < 0.03).
These results support inclusion of S100a and Melan A/MART-1 in a diagnostic panel for canine melanoma. S100a appears to be the more sensitive indicator of melanomas, especially for amelanotic tumors. Melan A/ MART-1 is a specific but less sensitive marker than S100a for melanoma. Despite its lower sensitivity, the intensity of its expression may be a reliable indicator of malignant potential, thus justifying its use in an immunohistochemical panel. Additional studies to further examine the sensitivity of Melan A/MART-1 and S100a in canine melanoma, especially as they relate to prognosis and diagnosis of equivocal and amelanotic tumors, are warranted.
Footnotes
Acknowledgements
We thank Dr. Steve Dow and Dr. Lauren Wolfe for providing cell lines, Stacie Bianco for technical help, Dr. Gary Cutter and Dr. Margaret Slater for assistance with statistical analyses, Dr. Kenita Rogers, Dr. Jennifer Thomas, and Dr. Michelle Ritt for critical review of the manuscript, and John Roths for assistance with preparation of the composite photograph. This work was supported in part by grant 1626 from the Canine Health Foundation of the American Kennel Club and grants 98PT–16 and 98CA–34 from the Morris Animal Foundation.
