Abstract
A 10-y-old spayed female Staffordshire Terrier dog was evaluated because of a cutaneous left ear base mass. Cytology revealed sheets of cells with anisocytosis and anisokaryosis, round-to-oval or plasmacytoid cytoplasm, and round, central, or eccentric nuclei; binucleate cells were present. Cytologic findings were consistent with a round cell tumor (plasmacytoma or agranular mast cell tumor), amelanotic melanoma, or anaplastic carcinoma. Histologically, neoplastic cells were polygonal to elongate, with round-to-oval nuclei and prominent nucleoli and arranged in sheets and nests on a fibrovascular stroma. Neoplastic cells with plasmacytoid morphology (round, glassy, eosinophilic cytoplasm with eccentric nuclei) were present in ~30% of the neoplasm. There were 18 mitoses in 2.37 mm2 (10 FN22/40× fields). Neoplastic cells had cytoplasmic immunolabeling for melan A and PNL2 and no immunolabeling for AE3/1 and MUM1, consistent with a dermal melanoma with plasmacytoid differentiation. The patient was re-evaluated ~1 mo after the first biopsy because of local recurrence of the original mass and new masses on the interscapular area and right elbow; these neoplasms were histologically identical to the original submission, plus scattered neoplastic cells in the new masses contained brown cytoplasmic pigment. The dog was euthanized because of swelling and hemorrhage of the tumors and right pelvic limb lameness. Our findings were consistent with a dermal melanoma with plasmacytoid features that were similar to human plasmacytoid melanoma, a rare variant of human melanoma that is diagnostically challenging as it may mimic a plasmacytoma.
Keywords
A 10-y-old spayed female Staffordshire Terrier dog was evaluated because of an ulcerated, 7 × 3 × 3-cm cutaneous mass located at the base of the left ear. Slides from a fine-needle aspirate were stained with aqueous Wright–Giemsa and evaluated by a board-certified veterinary clinical pathologist (CE Thorn). Cytologic evaluation revealed a highly cellular sample with a pale and slightly granular background (Fig. 1). Cells were arranged in loose sheets and had marked anisocytosis and anisokaryosis, with round-to-oval or plasmacytoid cytoplasm and fairly distinct cell borders. The cells had a moderate amount of pale basophilic cytoplasm, round, central, or eccentric nuclei with coarse chromatin, and multiple nucleoli. Binucleate cells were present (0–1 in each field), and rare mitoses were seen. Cytologic findings were consistent with a round cell tumor such as plasmacytoma, agranular mast cell tumor, amelanotic melanoma, or anaplastic carcinoma.
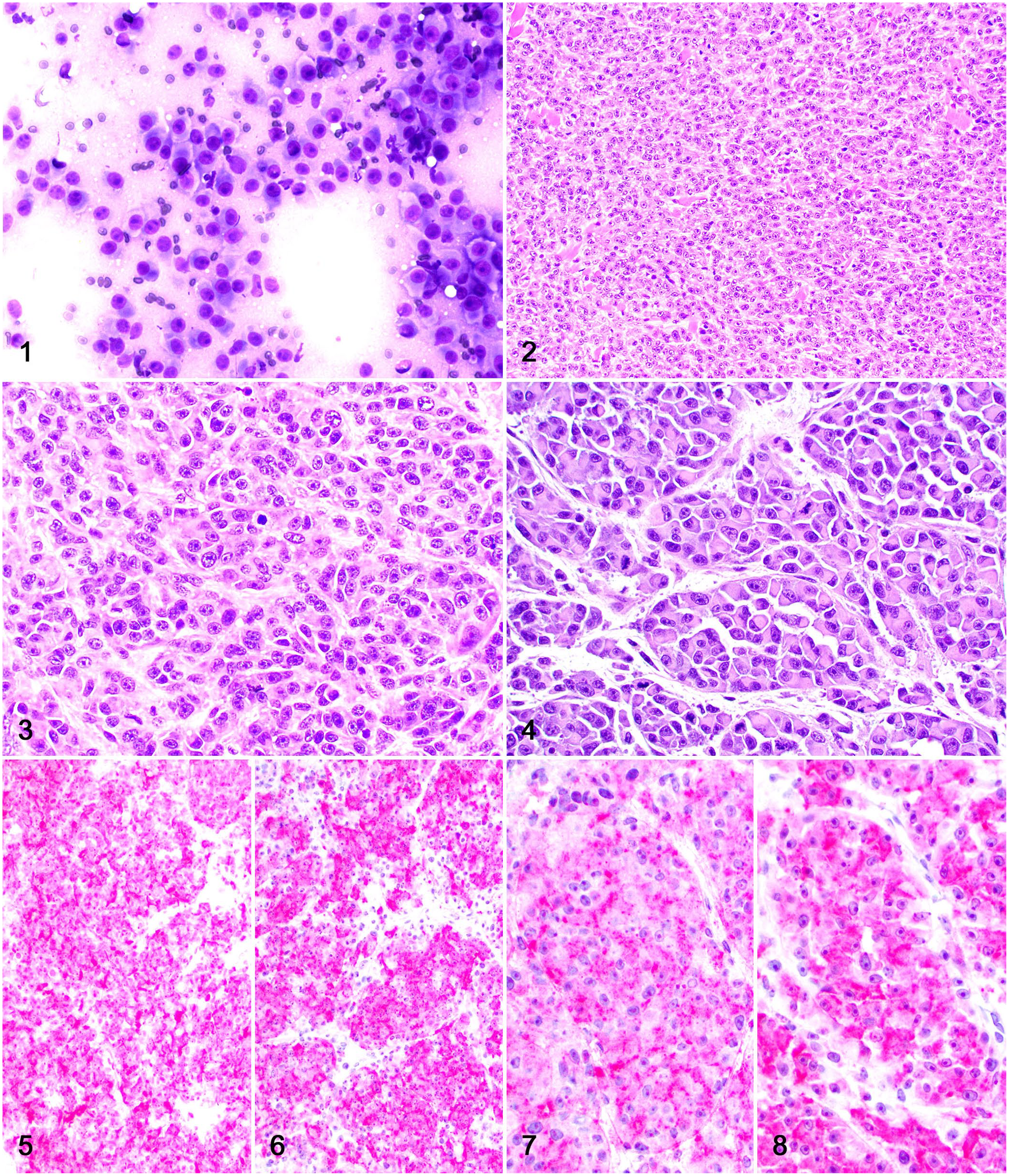
Dermal melanoma with plasmacytoid differentiation on the base of the left ear of a dog.
The mass was surgically resected and fixed in 10% neutral-buffered formalin for histologic evaluation at the Athens Veterinary Diagnostic Laboratory (AVDL; University of Georgia, Athens, GA, USA). Tissue sections were processed routinely for histology and stained with H&E. Histologically, the lesion consisted of a poorly demarcated, multinodular neoplasm that expanded and partially effaced the dermis and subcutaneous tissue. Neoplastic cells were polygonal to slightly elongate and arranged in closely apposed sheets and occasional nests separated by a scant fibrovascular stroma (Figs. 2, 3). The cytoplasm was abundant and eosinophilic, with indistinct margins. Nuclei were round to oval and had finely stippled chromatin with a prominent nucleolus. Multiple nests or packets of neoplastic cells with plasmacytoid morphology (round, glassy, eosinophilic cytoplasm with eccentric nuclei) were present in ~30% of the neoplasm (Fig. 4). There were 18 mitoses in 2.37 mm2 (10 FN22/40× fields).
Histologic findings were suggestive of a solid carcinoma of unknown origin, a dermal melanoma, and an atypical plasmacytoma. Immunohistochemistry (IHC) for melan A, PNL2, pancytokeratin AE1/3, and MUM1 was performed for diagnostic confirmation (Table 1). Neoplastic cells (including those with plasmacytic morphology) had widespread, robust cytoplasmic immunolabeling for melan A and PNL2 (Figs. 5–8) and no immunolabeling for AE3/1 and MUM1. Histologic and IHC findings were consistent with a dermal melanoma with plasmacytoid differentiation. Further IHC for Iba1 was performed to rule out the possibility that plasmacytoid cells were dendritic cells.
Details of the immunohistochemistry performed on a dermal melanoma in a dog.

NA = not applicable; RT = room temperature; RTU = ready to use. In all stain reactions, the chromogen used was diaminobenzidine (DAB), and the autostainer was IntelliPATH (Biocare).
The patient was re-evaluated ~1 mo after the first biopsy procedure because of local recurrence of the original mass, 2 new cutaneous masses on the interscapular area (~4 × 4× 3 cm) and right elbow (~2.5 × 3 × 2 cm), and right pelvic limb lameness. All masses were surgically resected and submitted for histologic evaluation at the AVDL. Radiograph of the thorax revealed no evidence of pulmonary metastases; a radiograph of the right pelvic limb was consistent with mild periosteal reaction on the stifle. Histologically, all lesions were identical to those observed in the original biopsy. However, scattered neoplastic cells with brown cytoplasmic pigment were observed in the interscapular and elbow neoplasms (Figs. 9–10). The pigment was black on melanin (Fontana–Masson) stain. The mitotic count was 59 (ear base mass), 26 (interscapular mass), and 42 (elbow mass) in 2.37 mm2. Neoplastic cells had widespread, robust cytoplasmic immunolabeling for melan A and PNL2, similar to the original submission. The dog was euthanized 2 mo later because of further swelling and hemorrhage of the shoulder and elbow lesions and progressive right pelvic limb lameness. No autopsy was performed.
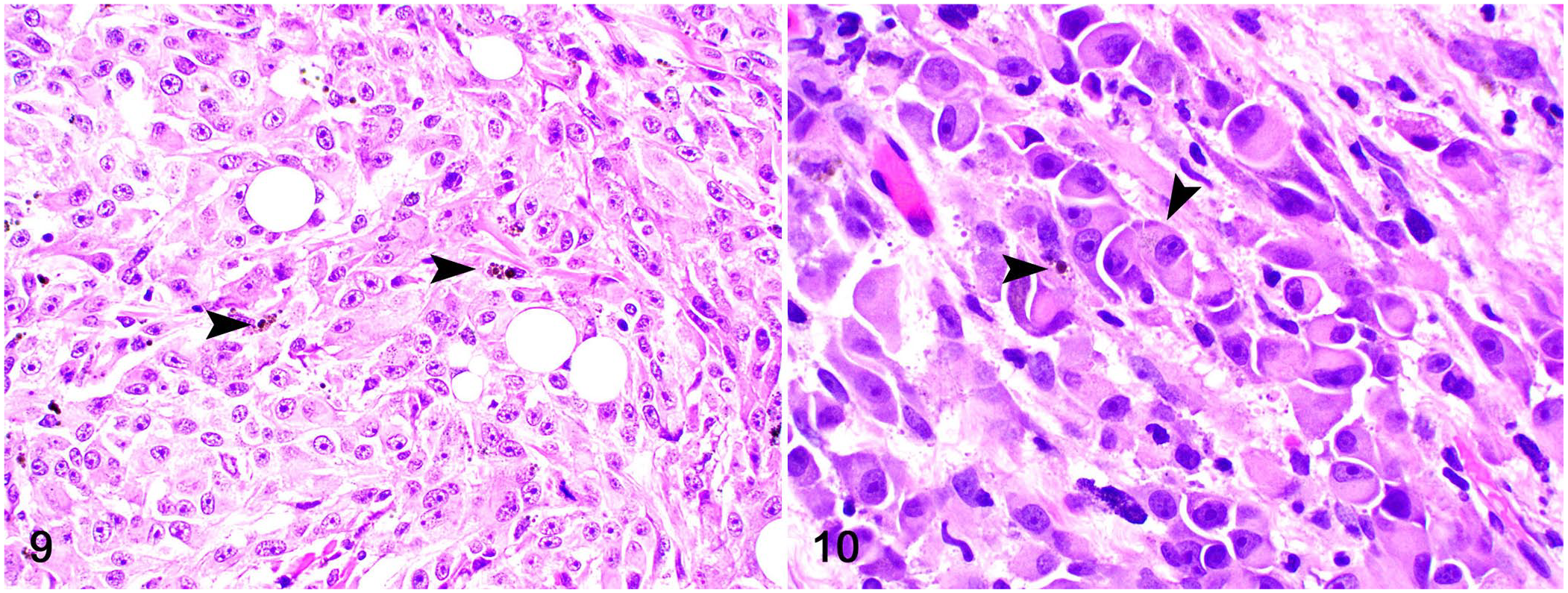
Dermal melanoma with plasmacytoid differentiation on the back of a dog.
Canine malignant melanoma is a common and locally invasive melanocytic neoplasm that occurs primarily in the oral cavity and skin. 6 Canine melanomas arising from the nail bed, mucocutaneus junctions, and oral cavity are more aggressive and have a worse prognosis than cutaneous melanomas, 6 which can be often cured by complete surgical excision.3,6 Cutaneous melanomas can be restricted to the dermis (dermal melanomas) or infiltrate the dermis and epidermis (compound melanomas), and most cases can be diagnosed based on cytology or histology. 6 The diagnosis is facilitated when neoplastic cell pigmentation and/or junctional activity (compound melanomas) are observed. 6
Melanomas typically consist of a combination of polygonal-to-elongate neoplastic cells, with variable amounts of cytoplasmic pigment, and arranged in sheets or bundles. 6 However, melanomas with atypical cell morphology, including round cell, small cell, giant cell, balloon cell, dendritic cell, adenomatous/papillary, and angiotropic/angiomatoid melanoma are also reported. 6 These distinct morphologic cell types that can be encountered within subsets of melanomas may pose a diagnostic challenge to the pathologist, particularly in amelanotic tumors. 7 In our first biopsy submission, neoplastic cells were confined to the dermis, and there was no detectable melanin within neoplastic cells, which, in association with the plasmacytoid differentiation of groups of neoplastic melanocytes, precluded a cytologic and histologic diagnosis of melanoma without the aid of IHC.
Amelanotic plasmacytoid melanoma is a rare morphologic variant of human melanoma that is diagnostically challenging as it may mimic a plasmacytoma.2,4 Although most neoplastic cells in plasmacytoid melanomas have a distinct plasmacytoid morphology, 2 only ~30% of the neoplastic cells in our case had such a feature, warranting the diagnosis of malignant dermal melanoma with plasmacytoid differentiation rather than plasmacytoid melanoma. 2 A search of Google, PubMed, CAB Direct, Web of Science, and Scopus for cases of plasmacytoid melanomas in dogs retrieved no results, suggesting that descriptions of canine melanomas with this morphologic feature have not been reported.
Similar to our case, in which neoplastic cell immunolabeling for melan A and PNL2 was supportive of a melanocytic neoplasm, most plasmacytoid melanomas in humans can be confirmed by the positive immunolabeling for melanocytic markers such as melan A, HMB-45, and tyrosinase. 2 However, rare cases with neoplastic cell immunolabeling for CD138, a plasma cell marker, have also been reported. 1 Because the plasmacytoid morphology of neoplastic melanocytes is thought to be associated with ultrastructural changes in neoplastic melanocytes, the significance of the reported immunolabeling in rare cases of plasmacytoid melanoma with plasma cell markers remains undetermined.1,2,4 In addition, plasmacytomas with melan A immunolabeling have been reported in dogs, suggesting that a comprehensive IHC panel should be used in cases with a questionable histogenesis. 5 Neoplastic cells in our case had no immunolabeling for MUM1, ruling out a plasmacytoma. Finally, plasmacytoid dendritic cells may be found within conventional human melanomas and should not be confused with neoplastic cells. 4 The lack of immunolabeling for Iba1 rules out a histiocytic immunophenotype in our case.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors declared that they received no financial support for their research and/or authorship of this article.
