Abstract
American canine hepatozoonosis is caused by Hepatozoon americanum, a recently described species of apicomplexan protozoan parasite. An immunohistochemical procedure using a polyclonal antibody to sporozoites of H. americanum clearly identified asexual stages of H. americanum in canine striated muscle. The method also detects hepatozoa present in naturally infected coyotes and raccoons and reacts with certain other apicomplexans. Use of this immunohistochemical procedure confirms the canine intermediate host-parasite relationships that were presumptively established using conventional histopathologic methods.
Canine hepatozoonosis is a tick-borne disease of dogs that occurs throughout much of the world. The infection was first recognized in India in 1905, 3 and the etiologic agent eventually was classified as Hepatozoon canis. 14 Canine hepatozoonosis was first reported in the USA in 1978. 1 Disease was characterized by signs that accompany systemic illness and skeletal and cardiac myositis. Particularly prominent were fever, myalgia, weakness, generalized wasting, pronounced mature neutrophilic leukocytosis, generalized periosteal bone proliferation, failure to respond to routine treatment, and a progressive course. When the disease was recognized in North America, it was assumed that the causative agent was H. canis and further that the definitive host and vector was Rhipicephalus sanguineus, the brown dog tick. 1 Critical examination of additional cases clearly identified prominent differences in the severity of clinical signs and gross and histopathologic changes between the US and Old World forms of disease. Based on those differences and differences in the morphology of gamonts in the canine intermediate host, the organism was reclassified and named Hepatozoon americanum in 1997. 13 An additional highly significant difference exists in the definitive host. Whereas R. sanguineus is a well-adapted host for H. canis, Amblyomma maculatum is a highly suitable definitive host for H. americanum. 6,7 Investigation of this “new” etiologic agent has been limited and superficial.
Until recently, the nature of the constituents of the so-called onion-skin cysts that are highly characteristic of American canine hepatozoonosis was unclear. 2,5,12 However, in recent studies using routine histologic and limited histochemical methods the “central mass” of earlier investigators has been identified as a dog cell (specific identity not known) whose cytoplasm harbors merogonous stages of the parasite. 10,11 This central mass is surrounded by lamellated, nonsulfated acid mucopolysaccharide within which may be occasional enmeshed fibroblasts and vasculature. 10 The morphology of various stages of asexual multiplication that develop within host cells that are located predominantly between striated muscle fibers of the dog have been characterized. 11 The earliest stage recognized is a zoite within a large modified host cell; this stage is followed by formation of a meront and production of merozoites within the host cell. Rupture of the cell and the onion-skin cyst encasement results in liberation of merozoites, and an acute and chronic granulomatous inflammatory reaction follows. The resulting discrete granulomas contain numerous intracellular parasites (merozoites and immature or mature gamonts) within both the extravascular and intravascular compartments of the granuloma. 10
The phylogenetic relationships between H. americanum and several other apicomplexans have been explored by means of 18S ribosomal RNA (rRNA) gene sequences. 9 Those studies suggest a very close relationship between the two canine hepatozoa and H. catesbianae, a parasite of the bullfrog (Rana catesbiana.) They also reveal sister-group relationships among Hepatozoon, Plasmodium, and Cryptosporidum. Other apicomplexans, including species of Cytauxzoon, Babesia, Toxoplasma, Neospora, Eimeria, Isospora, and Sarcocystis, are less closely related.
Here, we describe an immunohistochemical method for demonstration of H. americanum and present immunohistologic data that confirm earlier interpretations 10,11 regarding the host–parasite relationships that were based on conventional histopathologic methods. We also report results of attempts to immunostain several other apicomplexans and hepatozoa from hosts other than the domestic dog.
Materials and Methods
Experimental animals
A New Zealand White rabbit and one dog housed in the Laboratory Animal Resources Unit at Oklahoma State University were used in this study. The dog was naturally infected with H. americanum. 11 Animals were cared for in accordance with accepted laboratory animal practices.
Archived tissues of experimentally and naturally infected dogs and experimentally and naturally infected coyotes 4,11 were subjected to immunohistochemical staining. Archived tissues of various host species (Table 1) naturally infected with a variety of apicomplexan genera (Hepatozoon, Cryptosporidium, Cytauxzoon, Eimeria, Isospora, Sarcocystis, Besnoitia, Neospora, and Toxoplasma) were similarly prepared.
Comparative immunohistochemical staining of Hepatozoon and various other apicomplexans.
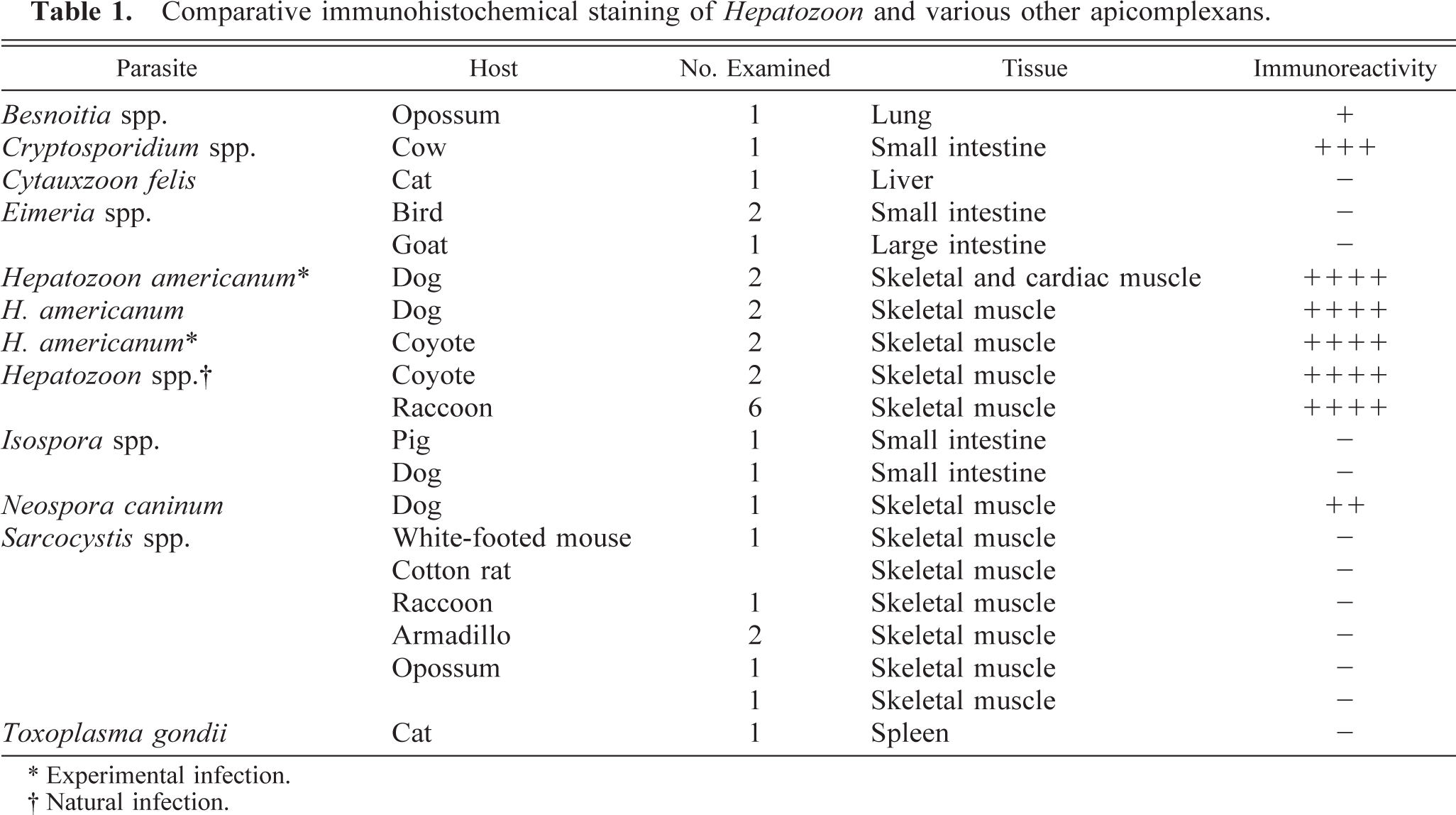
∗ Experimental infection.
† Natural infection.
Preparation of antigen and antiserum
Antigen was prepared from sporozoites of H. americanum. One thousand oocysts were collected from adult A. maculatum experimentally infected as nymphs by feeding on a dog naturally infected with H. americanum. Oocysts were mechanically ruptured by passing them through a 16-gauge needle, and the sporocysts were treated with canine bile to release sporozoites. 7 Sporozoites were washed three times with phosphate-buffered saline (PBS, pH 7.4) and mixed with equal amounts of PBS and Freund's incomplete adjuvant. Prepared antigen was injected intramuscularly at three sites in a young NZ White rabbit. Additional doses of adjuvanted antigen were administered intramuscularly 3 weeks, 5 weeks, and 30 weeks after the initial injection. Antibody titers were determined periodically using an indirect enzyme-linked immnosorbent assay. 8 Antiserum in this study was prepared from blood collected 8 days after the fourth exposure to antigen. (antibody titer = 1:3,200).
Immunohistochemistry
Tissues containing stages of the organism were stained with both hematoxylin and eosin (HE) and immunohistochemical techniques. Paraffin-embedded archival tissues were sectioned at 4 µm and mounted on positively charged slides. Sections were deparaffinized and hydrated in graded alcohols to distilled water. They were then subjected to heat-induced epitope retrieval by immersion in 0.01 M sodium citrate buffer, heated to 15 psi in a pressure cooker (Presto, 4 quart) for 9 minutes, and allowed to cool for approximately 10 minutes before removal from the cooker. Immunostaining was performed in a Dako Autoimmunostainer Model LV (Dako, Carpinteria, CA) as follows. Endogenous peroxidase was inactivated with hydrogen peroxide. Specimens were incubated in 10% goat serum for 20 minutes, placed directly in 1:200 rabbit anti-hepatozoon serum for 30 minutes, then rinsed in buffer. Sections were incubated in biotinylated goat anti-rabbit IgG (1:500), rinsed, and then incubated in 1:1,000 horseradish peroxidase–streptavidin conjugate for 20 minutes and rinsed in buffer. Chromogen (AEC) was applied for 10 minutes, and the sections were then rinsed in buffer, counterstained with Mayer's hematoxylin, rinsed, and coverslipped. For negative immunostaining controls the primary antibody applied to serial sections was replaced with normal rabbit IgG.
Results
Corresponding asexual stages of H. americanum, presumptively identifiable in HE-stained sections (Figs. 1a, 2a, 5), stained intensely and were clearly defined in immunostained specimens (Figs. 1b, 2b 6). Figures 3–5 and 7 illustrate progressive stages of parasite development.
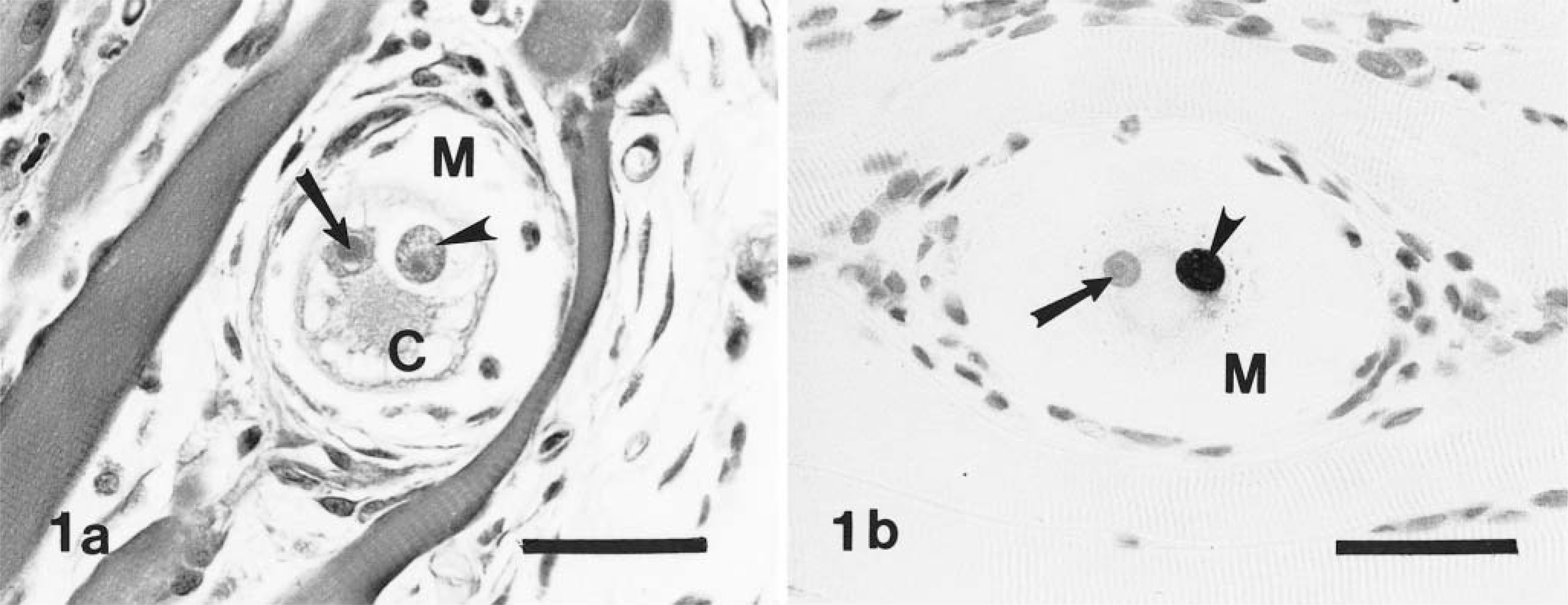
Skeletal muscle; experimentally infected dog. Fig. 1a Early stage of parasite (arrowhead) within an unidentified canine host cell (C). Nucleus contains a large nucleolus (arrow). The host cell is situated between skeletal muscle fibers and is surrounded by a narrow zone of mucopolysaccharide material (M) within which are enmeshed fibroblasts. HE. Bar = 40 µm. Fig. 1b Note host cell nucleus (arrow), the parasite (arrowhead), and clear zone of mucopolysaccharide (M) surrounding the host cell. Avidin–biotin immunoperoxidase, Mayer's hematoxylin counterstain. Bar = 40 µm.
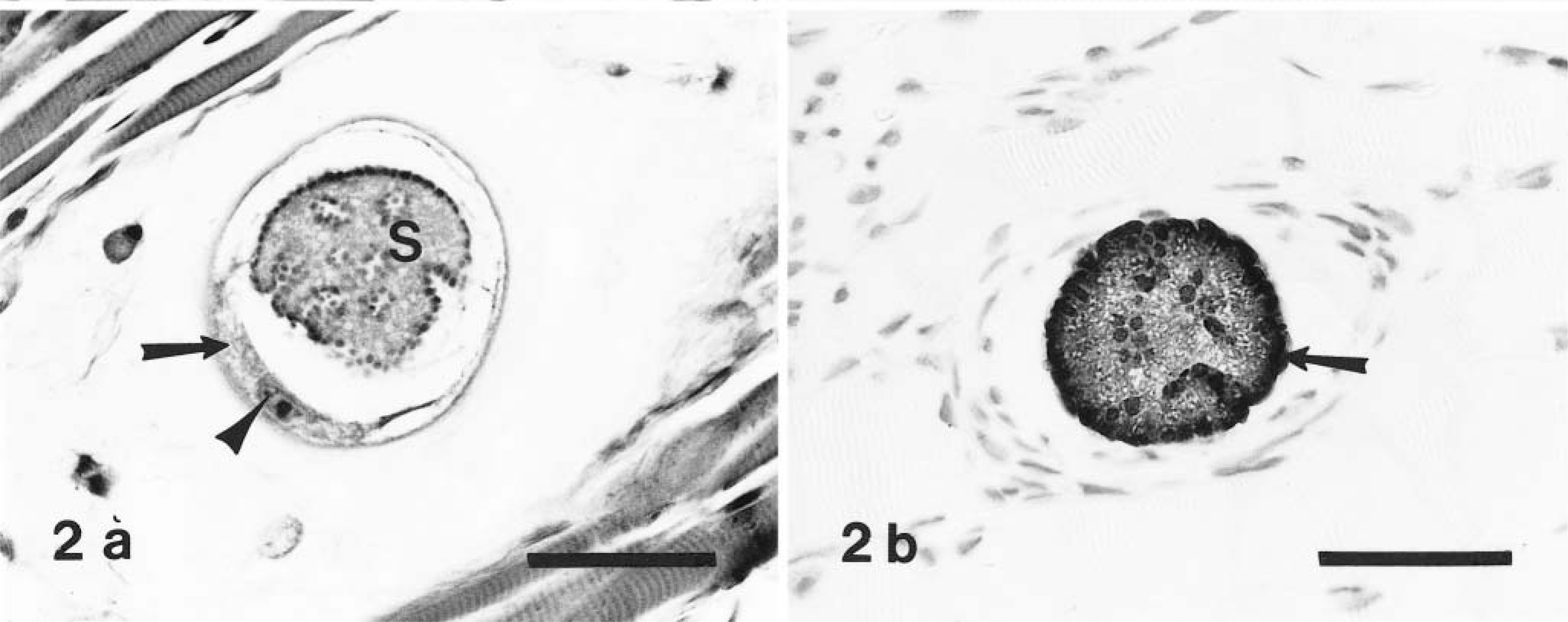
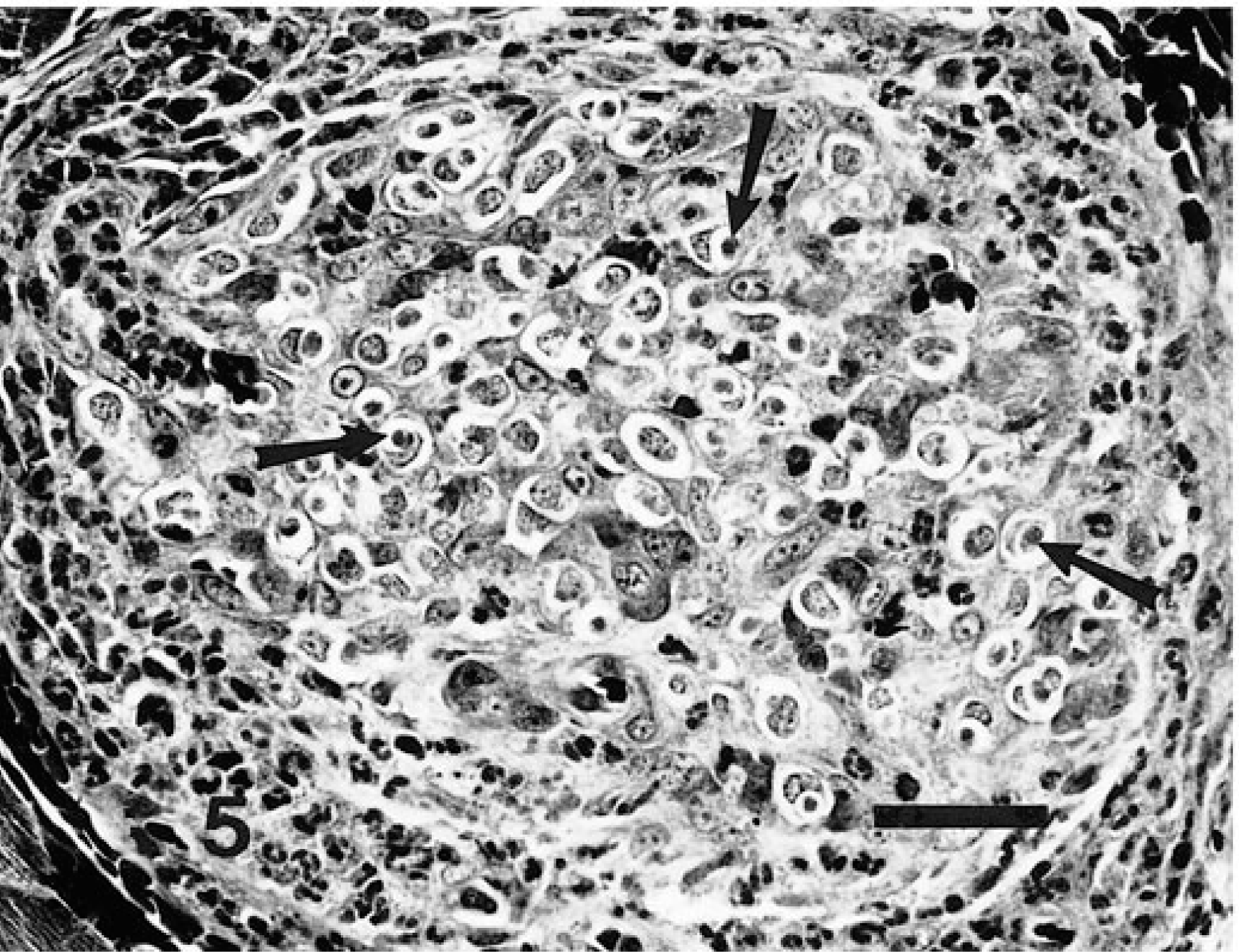
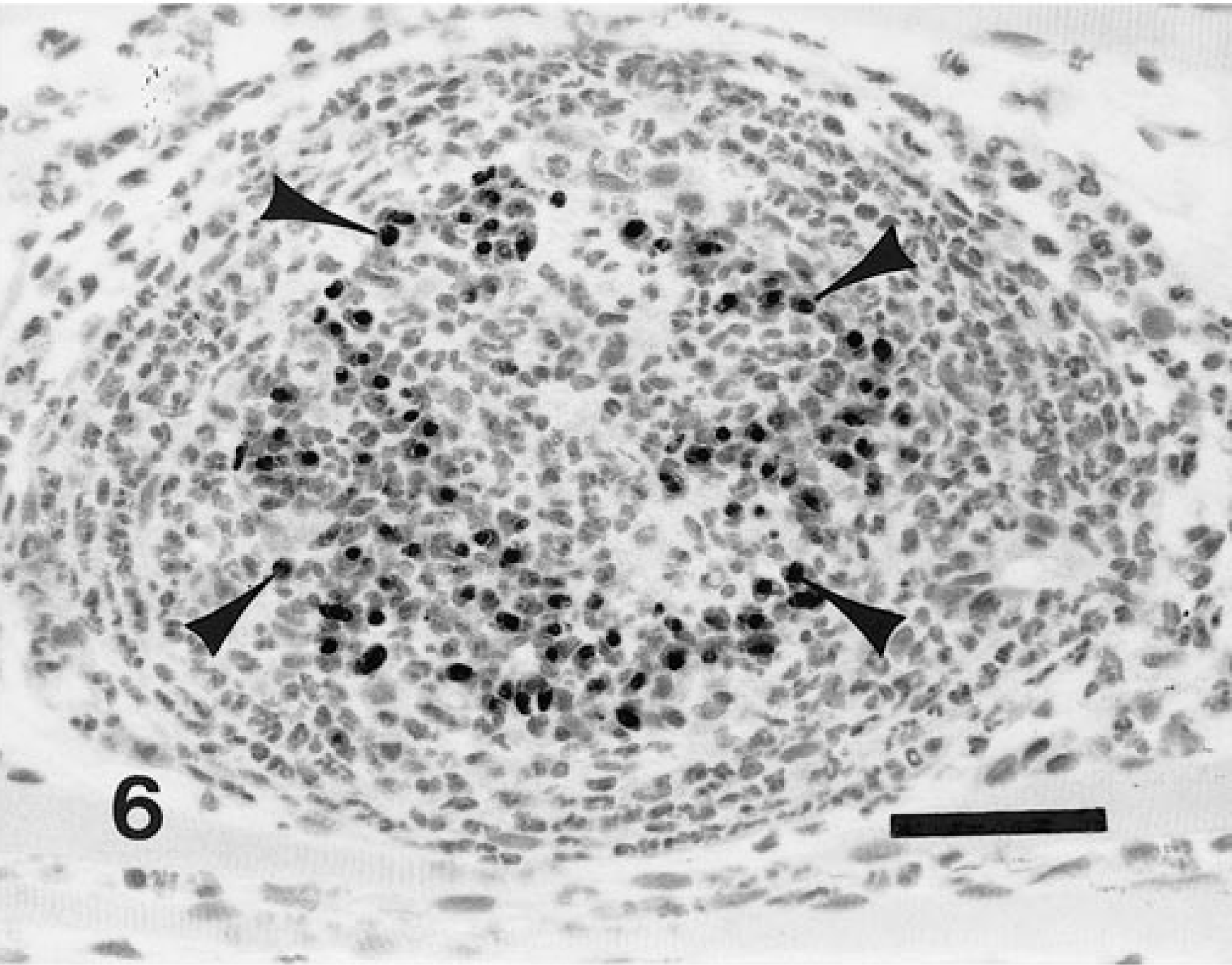
Skeletal muscle; naturally infected dog. Fig. 2a Meront (S) with parasitic nuclear material aggregated both at the periphery and along infolded areas of meront membrane. The meront is contained within a host cell whose nucleus (arrowhead), nucleolus, and crescentic segments of cytoplasm (arrow) are evident. HE. Bar = 50 µm. Fig. 2b Meront in stage of development comparable to that in Fig. 2a. Note intense staining at periphery (arrow) and within the body of the meront structure. Avidin–biotin immunoperoxidase, Mayer's hematoxylin counterstain. Bar = 40 µm.
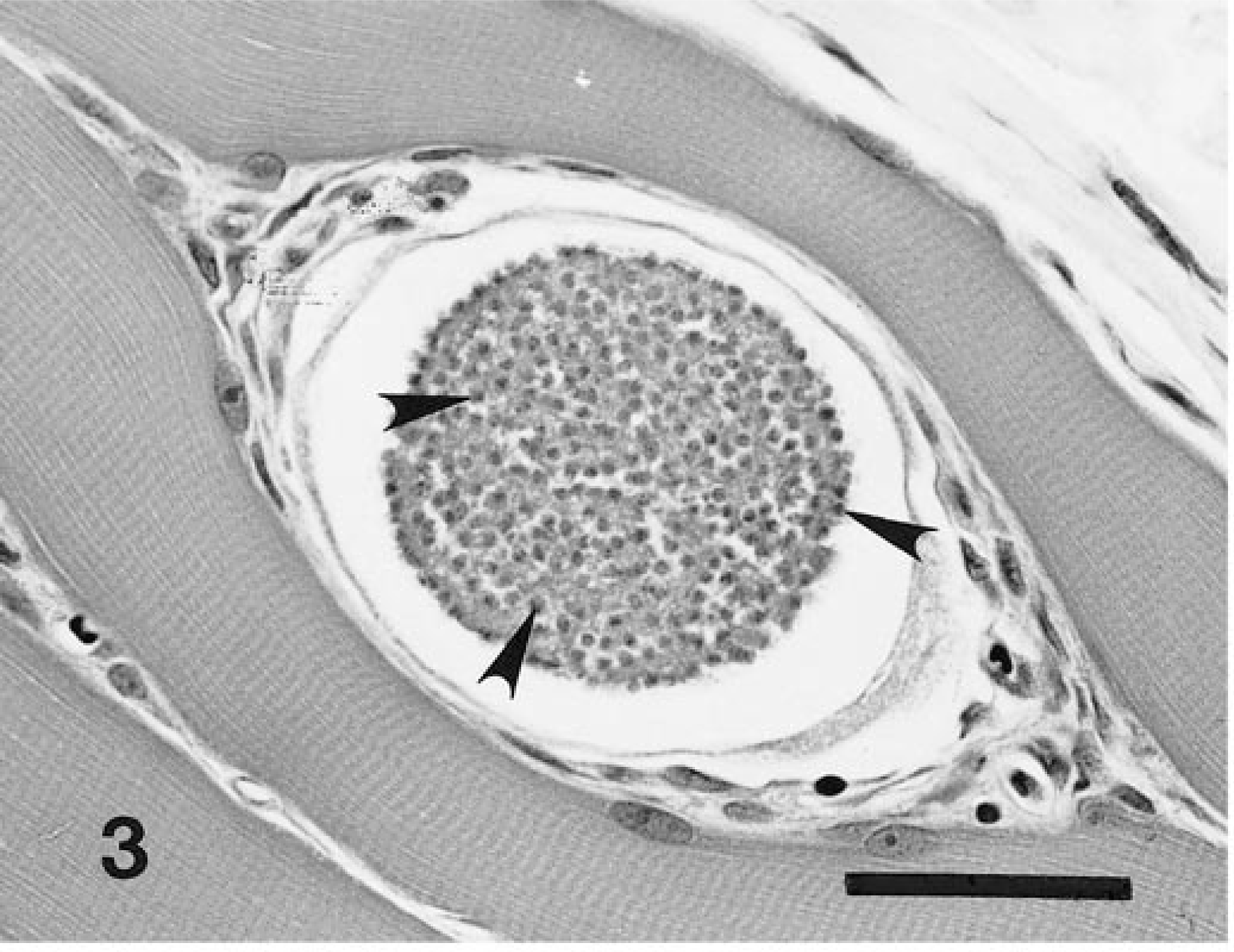
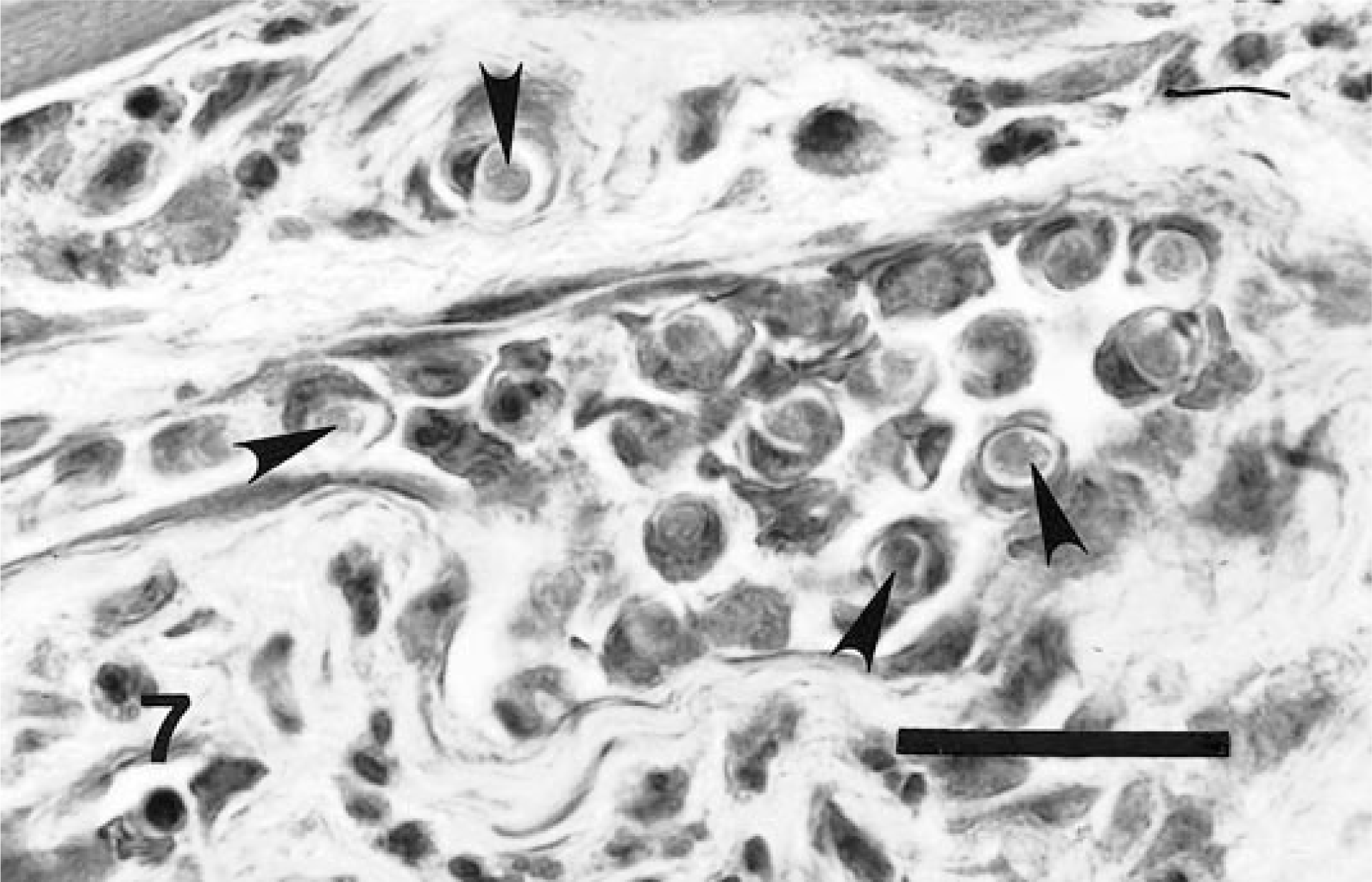
Skeletal muscle; experimentally infected dog. Immature merozoites (arrowheads) are well confined within a meront, which at this stage presumably still resides within an intact host cell. HE. Bar = 40 µm.
Skeletal muscle; experimentally infected dog. Acute inflammatory response following degeneration/rupture of meront and host cell with resultant liberation of merozoites into adjacent tissue. The inflammatory cell population at this stage is predominantly neutrophilic. HE. Bar = 50 µm.
Skeletal muscle; naturally infected dog. Discrete pyogranuloma in which the proportion of neutrophils is greatly reduced and many monocytes/macrophages harbor intracytoplasmic parasites (arrows). HE. Bar = 50 µm.
Skeletal muscle; experimentally infected coyote. Pyogranuloma which contains numerous positively stained intracellular organisms (arrowheads). Avidin–biotin immunoperoxidase, Mayer's hematoxylin counterstain. Bar = 50 µm.
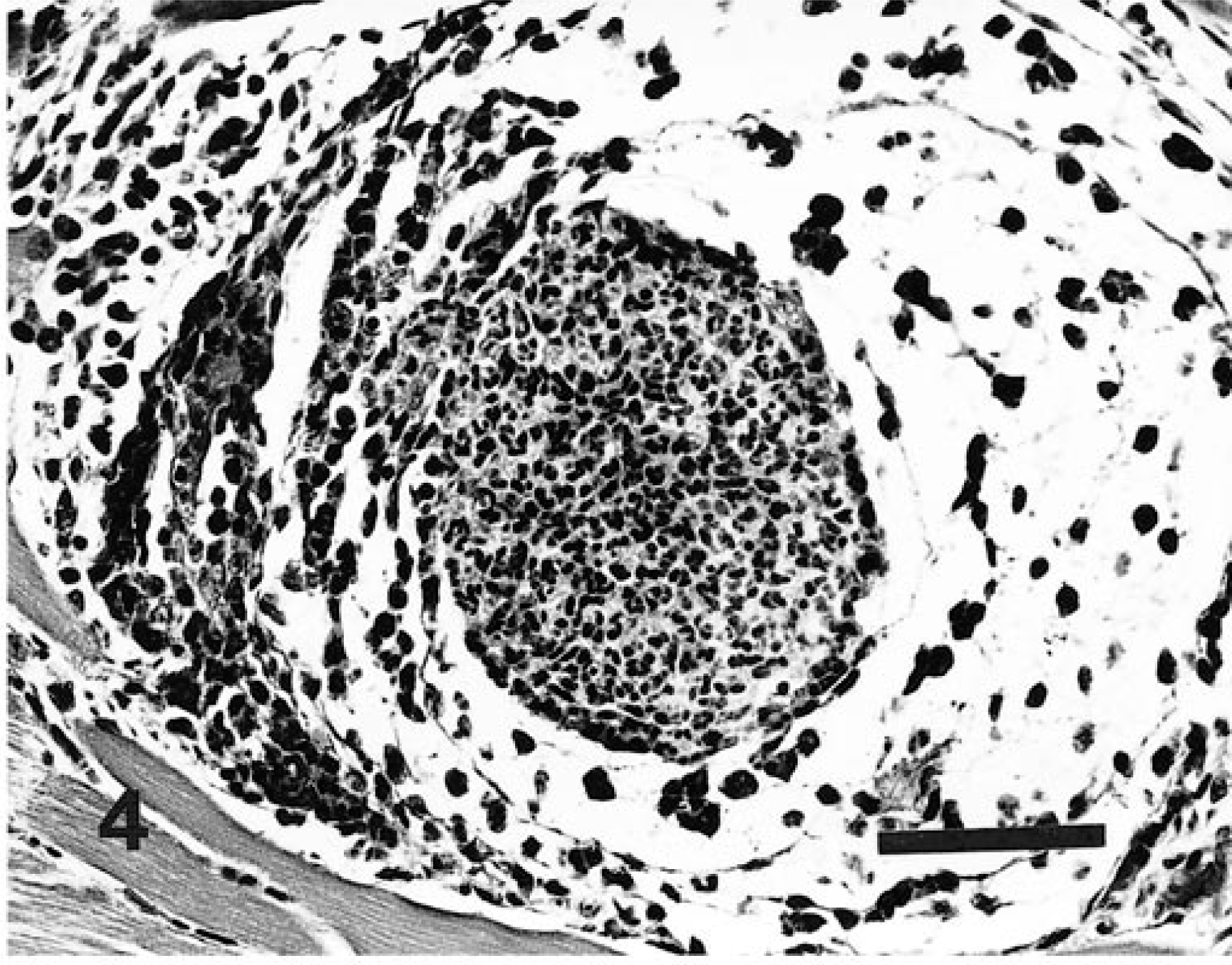
Skeletal muscle; naturally infected dog. Higher magnification of large sinusoidal vessel and adjacent connective tissue of granuloma. Both the extravascular and intravascular compartments contain host cells that harbor parasites (arrowheads), i.e., merozoites or immature or mature gamonts. HE. Bar = 20 µm.
The results of application of this immunostaining procedure to other unidentified species of Hepatozoon (naturally infected coyote, raccoon) and other genera of apicomplexans are listed in Table 1. Other hepatozoa stained intensely, Cryptosporidium spp. stained moderately intensely, Neospora spp. stained only moderately, and a thin membrane (the inner cyst wall) of Besnoitia spp. stained weakly. Other organisms tested failed to stain.
Discussion
Application of the avidin–biotin immunoperoxidase method using a polyclonal antibody produced in a rabbit in response to parenteral injection of sporozoites of H. americanum clearly and intensely demonstrated asexual stages of the parasite. Sections of infected muscle thus prepared clearly revealed stages of the parasite in the cytoplasm of an as yet unidentified host cell located between host myocytes. These observations confirm earlier findings and interpretations of the host–parasite relationship. 10,11
Although the primary antibody reacted strongly with H. americanum, it also reacted with other Hepatozoon species and, to a lesser extent, with certain other apicomplexans. Staining was particularly intense for Cryptosporidium, which appears relatively closely related to H. americanum on the basis of RNA analysis. 9 Thus, the primary antibody used in the immunostain was not absolutely specific for H. americanum. The degree of cross-reaction was not surprising given that the antibody used in the immunohistochemical staining procedure was polyclonal and was produced in response to injected antigen derived from whole sporozoites and sporocyst walls.
Although evidence obtained in these immunohistochemical studies confirms earlier conclusions regarding host–parasite relationships based on conventional (HE) histologic methods, 10,11 the utility of this immunostaining procedure for diagnostic purposes is likely limited. The infection is readily confirmed by routine histopathologic procedures applied to canine striated muscle. Although untested, the procedure may have diagnostic value in more readily demonstrating the sparsely distributed gametocytes in peripheral blood smears. It should also be useful in identification of individual zoites in their migration through host tissues. Because some Hepatozoon species in addition to H. americanum are stained with this method, it should have value in the demonstration of hepatozoa in other host species.
