Abstract
Palisading granulomatous dermatitis and panniculitis is recognized in various cutaneous inflammatory lesions secondary to presumed collagen damage. Cutaneous nodules with a palisading arrangement of histiocytes surrounding foci of collagen degeneration have been clinically termed palisading granuloma in dogs. Study aims were to characterize the cellular infiltrate of canine palisading granuloma and document salient clinical features. Inclusion criteria were met for 36 dogs and encompassed nodular dermal and subcutaneous histiocyte-predominant cellular infiltrates targeting and enveloping collagen fibers/necrotic foci with palisading configurations. Infectious causes were ruled out via standard histochemical stains and/or clinical data. Medical records were reviewed for signalment, clinical features, treatment, outcome, and comorbidities. Immunohistochemistry (IBA1, CD204, E-cadherin) and Masson’s trichrome stain were used to assess histiocytic populations and dermal collagen, respectively. The histiocytes had moderate or strong immunolabeling for IBA1 and CD204 in 36/36 dogs (100%) and mild positive immunolabeling for E-cadherin in 3/36 dogs (8%). Alteration of collagen was graded as moderate or strong in 32/36 dogs (89%) and mild in 3/36 dogs (8%). Large breeds predominated with 30/36 dogs (83%) being ≥23 kg. Focal nodules were identified in 31/36 dogs (86%). The head/face were involved in 19/36 dogs (53%) and the extremities in 18/36 dogs (50%). Lesions from the 5/36 dogs (14%) with multiple nodules contained prominent eosinophilic infiltrates. Following excision, there was no evidence of recurrence. In conclusion, palisading granulomas are a distinct, non-neoplastic, histiocyte-predominant inflammatory condition in dogs associated with altered dermal collagen and favorable prognosis.
Keywords
Palisading granulomatous dermatitis and panniculitis is a common histologic feature of numerous cutaneous disease processes described in humans including palisading granulomas, granuloma annulare, rheumatoid nodules, necrobiotic xanthogranulomas, lipoid necrobiosis, and interstitial granulomatous dermatitis. 4,13,23 The palisading component refers to the histologic arrangement of histiocytes often with elongated and spindle-shaped nuclei arranged roughly parallel to each other and perpendicular to the edge of a central necrotic zone in what has been described previously as a “fence-like” fashion. 20 While a variety of neoplastic or inflammatory conditions can contain foci of comparable histologic features, this study describes discrete nodules predominated by palisading histiocytes which envelope collagen fibers with no other identifiable underlying disease processes, termed palisading granulomas.
Primary peer-reviewed publications focused on palisading granulomas in dogs or any other domestic animals are lacking and, to the authors’ knowledge, are limited to one textbook description. 9 Palisading granulomas are categorized as a noninfectious nodular granulomatous dermatitis. Other noninfectious nodular to diffuse histiocytic dermatoses include sterile granuloma and pyogranuloma syndrome, reactive histiocytosis, cutaneous xanthoma, canine sarcoidosis, foreign body reactions, reactive fibrohistiocytic nodules, and sterile granulomatous dermatitis and lymphadenitis (juvenile and adult-onset cellulitis). 9,12 Additionally, though histologically distinct, histiocytic neoplasms in the skin may be nodular to diffuse and include canine cutaneous histiocytoma, Langerhans cell histiocytosis, and histiocytic sarcoma. 10,13,17,18
Histiocytes differentiate from CD34+ stem cell precursors of the bone marrow into circulating monocytes that mature into macrophages (CD11b+, CD11d+, CD18+, IBA1+, CD163+, CD204+, and CD1+/−) on exit into tissues. The CD34+ precursors also give rise to several dendritic cell lineages. 17,18 Dendritic lineages specifically important in the skin include intraepidermal Langerhans cells (CD1a+, CD11c+, CD18+, IBA1+, CD204−, and E-cadherin+) and interstitial dermal dendritic cells (CD1a+, CD11c+, CD18+, IBA1+, CD90/Thy-1+, CD11b+/−, CD163+/−, and CD204+/−) that are localized to perivascular and periadnexal regions throughout the dermis. 17,18 Interdigitating dendritic cells are found in lymph nodes and the spleen and are composed of both resident dendritic cells and migrating antigen-presenting cells including Langerhans cells and interstitial dendritic cells from the skin and mucosal surfaces (ie, respiratory and alimentary tracts). 17,18
The aim of this study was to better characterize palisading granulomas and to (1) define and characterize histologic features, (2) assess the degree of concomitant inflammatory cell types, and (3) identify any evidence of collagen alteration. Furthermore, medical records were reviewed to assess the clinical outcome and identify any correlative clinical diseases or other factors.
Methods and Materials
Histopathological Study
An electronic database search of the surgical biopsy service at Penn Vet Diagnostic Laboratories was performed for reports of dogs containing the term “palisading granuloma” from January 1, 2007, to January 1, 2019. Initially, 70 cases were identified and hematoxylin and eosin (HE) stained sections were reviewed with the following inclusion criteria: (1) canine species, (2) dermal or subcutaneous location, (3) nodular inflammatory infiltrate composed of histiocytes with or without other inflammatory cells oriented around dermal collagen bundles, and (4) lack of evidence/proof of underlying infectious etiology via negative Gram, Grocott’s methenamine silver, or periodic acid-Schiff, and Ziehl-Neelsen acid-fast stains. Inclusion criteria were met for 36 of the 70 cases. Reasons for exclusion consisted primarily of noncutaneous anatomic location (eg, oral mucosa), or histologic evidence of another concurrent disease process/entity (eg, neoplasia, sebaceous gland hyperplasia, seroma/granulation tissue, and/or furunculosis) strongly suggesting that the similar histologic features were secondary reactive processes and not a primary palisading granuloma.
HE-stained sections were further reviewed for each dog and assessed for evidence of the following: vasculitis, actinic dermatosis, multinucleated giant cells (ie, granulomatous inflammation), background mucinous debris, relative density of comingled lymphoplasmacytic and eosinophilic infiltrates, and collagen flame figures. Additionally, lesion margins were subjectively assessed for degree of demarcation.
Immunohistochemistry
Immunohistochemistry for IBA1, CD204, and E-cadherin was used for all 36 cases to better categorize the histiocytic infiltrates. Histologic preparation started with 5-µm paraffin sections mounted on positively charged ProbeOn slides (Thermo Fisher Scientific). The immunostaining procedure was performed using a Leica BOND RXm automated platform combined with the Bond Polymer Refine Detection kit (Leica #DS9800). Briefly, after dewaxing and rehydration, sections were pretreated with either the epitope retrieval BOND ER2 (Leica #AR9640) pH 9 buffer (CD204 and E-cadherin) or the epitope retrieval BOND ER1 (Leica #AR9640) pH 6 buffer (IBA1) for 20 minutes at 98 °C. Endogenous peroxidase was inactivated with 3% hydrogen peroxide for 5 minutes at room temperature. Nonspecific tissue-antibody interactions were blocked with Leica PowerVision IHC/ISH Super Blocking solution (PV6122) for 30 minutes at room temperature. The same blocking solution also served as diluent for the primary antibody. Individual rabbit polyclonal primary antibodies against IBA1 (DAKO #019-19741) and rabbit monoclonal antibodies against E-cadherin (Abcam #76319) were used at concentrations of 1/1500 and 1/1000, respectively. Individual mouse monoclonal primary antibodies against CD204 (Transgenic #KT022; CosmoBio USA) was used at concentration of 1/1500. Each were incubated on the sections for 45 minutes at room temperature. A biotin-free polymeric immunohistochemistry (IHC) detection system consisting of horseradish peroxidase (HRP) conjugated anti-rabbit IgG was then applied for 25 minutes at room temperature. Immunoreactivity was revealed with the diaminobenzidine chromogen reaction. Slides were finally counterstained in hematoxylin, dehydrated in an ethanol series, cleared in xylene, and permanently mounted with a resinous mounting medium (Thermo Scientific ClearVueTM coverslipper). Sections of canine lymph node were included as positive controls for IBA1 and CD204 with both demonstrating strong brown cytoplasmic and membranous immunostaining of histiocytes scattered throughout the section with a preponderance in medullary and cortical sinuses. A section of canine integument (lip) was included as positive control for E-cadherin with strong brown membranous immunostaining of epidermal and follicular keratinocytes, adnexa, and peripheral nerves in addition to the strong brown cytoplasmic and membranous immunostaining of intraepidermal and few dermal histiocytes. Negative controls were obtained either by respective omission of the IBA1, CD204, and E-cadherin antibodies or replacement with an irrelevant isotype-matched rabbit monoclonal antibody. Tissues from all 36 dogs were stained at the same time utilizing the same procedure for each of the respective immunostaining protocols. A 3-tier semiquantitative scoring system was developed to assess immunolabeling as follows: negative, <5% positive cells; mild, 5% to 50% cells positive; moderate, 50% to 75% positive cells; and strong, >75% positive cells.
Additionally, for a representative subset of 5 dogs with focal nodules, immunohistochemistry for CD3, CD79b, Mum1, and MHCII were performed. Histologic preparation started with 5-µm paraffin sections mounted on positively-charged ProbeOn slides (Thermo Fisher Scientific). The immunostaining procedure was performed using a Leica BOND RXm automated platform combined with the Bond Polymer Refine Detection kit (Leica #DS9800). Briefly, after dewaxing and rehydration, sections were pretreated with the epitope retrieval BOND ER2 (Leica #AR9640) pH 9 buffer for 20 minutes at 98 °C. Endogenous peroxidase was inactivated with 3% hydrogen peroxide for 5 minutes at room temperature. Nonspecific tissue-antibody interactions were blocked with Leica PowerVision IHC/ISH Super Blocking solution (PV6122) for 30 minutes at room temperature. The same blocking solution also served as diluent for the primary antibody. Individual rat monoclonal antibodies (Bio-Rad #1477) against CD3, mouse monoclonal antibodies (Abcam #234019) against CD79b, mouse monoclonal antibodies (DAKO #M7259) against Mum1, and mouse monoclonal antibodies (DAKO #M0746) against MHCII were used at concentrations of 1/600, 1/500, 1/30, and 1/100, respectively. Each were incubated on the sections for 45 minutes at room temperature. A biotin-free polymeric IHC detection system consisting of HRP conjugated anti-rabbit IgG was then applied for 25 minutes at room temperature. Immunoreactivity was revealed with the diaminobenzidine chromogen reaction. Slides were finally counterstained in hematoxylin, dehydrated in an ethanol series, cleared in xylene, and permanently mounted with a resinous mounting medium (Thermo Scientific ClearVueTM coverslipper). Sections of canine lymph node were included as positive controls for all 4 immunostaining protocols. The CD3 positive controls identified large numbers of strong diffuse brown cytoplasmic immunostaining of lymphocytes contained within paracortical regions. The CD79b positive controls identified large number of strong diffuse brown cytoplasmic immunostaining of lymphocytes specifically contained within follicles. Mum1 positive controls identified smaller numbers of scattered individualized plasma cells with strong brown intranuclear immunostaining predominately within the medullary and cortical sinuses. MHCII positive controls identified strong diffuse brown cytoplasmic and membranous staining of the majority of cells with preponderance for the medullary and cortical sinuses and follicles. Negative controls were obtained either by omission of the respective antibodies or replacement with an irrelevant isotype-matched rabbit monoclonal antibody. Tissues from all 5 dogs were stained at the same time utilizing the same procedure for each of the respective immunostaining protocols.
Masson’s Trichrome Staining
Masson’s trichrome blue variant histochemical staining was performed according to protocols previously described in the literature 21 on all 36 included dogs to identify and characterize intralesional collagen alteration (ie, bright red staining of collagen fiber cores). 7,8 Canine small intestine tissue was used for positive control. Additionally, the adjacent unaffected dermis of all skin lesions provided as internal controls for all 36 dogs. Tissues from all 36 dogs were stained contemporaneously.
A separate 3-tiered semiquantitative grading system was developed to assess the Masson’s trichrome histochemical staining of collagen. Scoring was based on the percentage of 20 fields of view at 400× magnification containing any evidence of bright red-staining collagen cores within the histiocyte-predominated inflammatory infiltrates and was organized into the following: negative, no identifiable collagen change; mild, <25% fields of view; moderate, between 25% and 50% fields of view; and strong, >50% fields of view. The microscope field of view diameter used for the semiquantitative grading was equivalent to a total area of 3.93 mm2 scanned for each lesion.
Signalment and Clinical Correlates
Signalment data and lesion information (location, size, and nodal involvement) were collected for each dog from histopathology submission forms. Questionnaires were sent to submitting veterinarians requesting information about lesion location, comorbidities, and/or clinical management and outcome. Complete medical records and/or questionnaires were obtained for 12/36 dogs (33%).
Results
Histopathological Study
Lesions from the 36 dogs contained a histiocyte-predominant inflammatory infiltrate composed of multifocal to coalescing arrangements of elongated to fusiform-shaped cells arranged in parallel sheets and streams roughly perpendicular to central collagen bundles (Figs. 1–8). In some foci the histiocytes formed parallel streams adjacent to collagen fibers or slight whorls as well (Fig. 7).
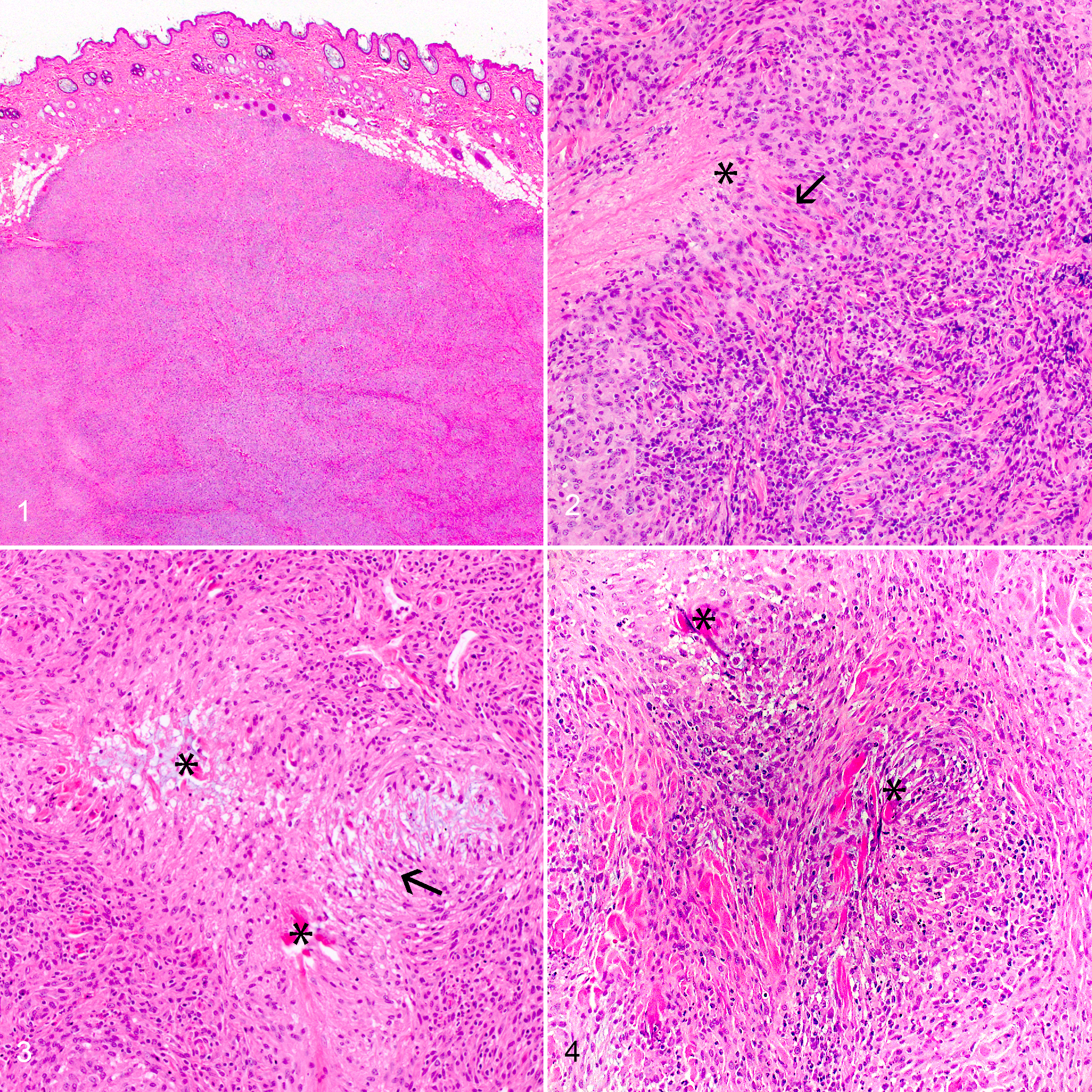
Palisading granuloma, haired skin, dog. Hematoxylin and eosin.
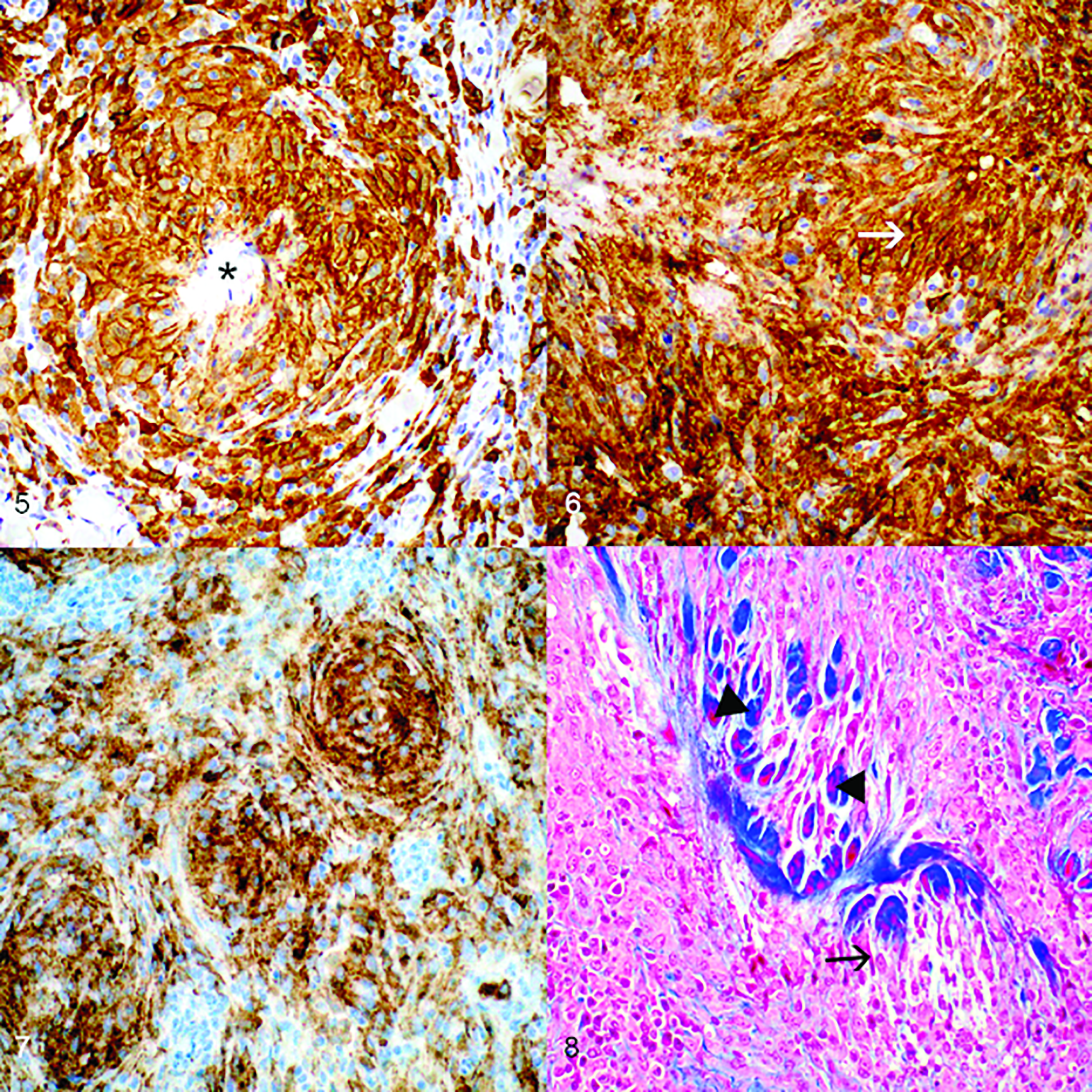
Palisading granuloma, haired skin, dog. Immunohistochemistry.
There was no evidence of vasculitis or actinic change (ie, actinic keratosis, solar elastosis, and actinic comedones) and multinucleated giant cells were not identified in any of the included lesions. No infectious agents were identified with Gram, Grocott’s methenamine silver, periodic acid-Schiff, or Ziehl-Neelsen stains. Moderate infiltrates of lymphocytes and plasma cells were identified in 14/36 dogs (39%; Fig. 2). Additionally, in 10 of these 14 dogs, there were mild to moderate numbers of eosinophils. None of the 36 dogs had collagen flame figures (ie, collagen fibers with an eosinophilic tinctorial quality lined by abundant free eosinophil granules and macrophages). 24 A pale gray-blue mucinous stroma was intermingled in the background matrix for 8/36 dogs (22%; Fig. 3). The inflammatory nodules were definitively circumscribed in 31/36 dogs (86%) and were poorly circumscribed in 5/36 dogs (14%).
Immunohistochemistry
The immunohistochemical labeling for IBA1, CD204, and E-cadherin were each graded according to the above-described 3-tier semiquantitative system based on percentage of inflammatory populations demonstrating positive immunoreactivity (Table 1). Immunoreactivity for IBA1 scored strong in 32/36 dogs (89%) and moderate in the remaining 4/36 dogs (11%; Fig. 5). Immunoreactivity for CD204 was a strong positive in 24/36 dogs (67%) and moderate in 12/36 dogs (33%; Figs. 6, 7). Immunoreactivity of E-cadherin was negative for 33/36 dogs (92%) and mild for the remaining 3/36 dogs (8%).
Scores for immunolabeling of histiocytes in 36 dogs with palisading granulomatous dermatitis and panniculitisa.
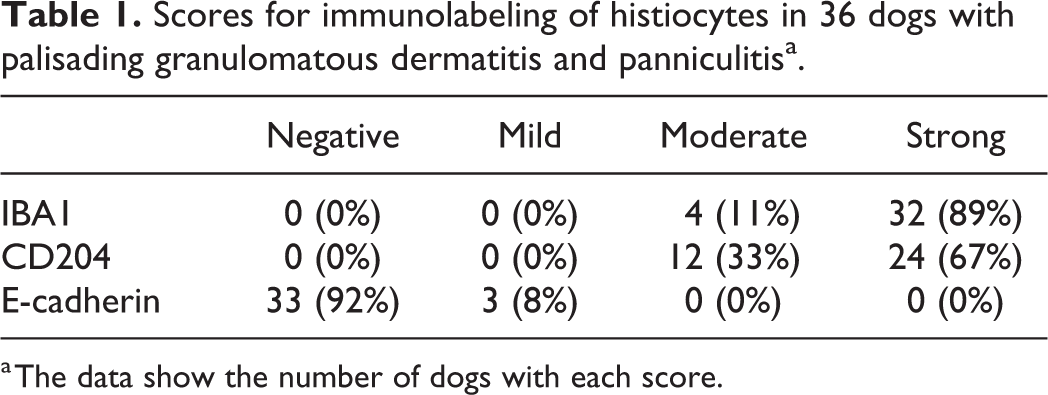
a The data show the number of dogs with each score.
In the smaller subset of 5 dogs examined, moderate numbers of CD3+ cells were scattered throughout each of the lesions among the histiocytic nodules (Suppl. Fig. S1). There were fewer CD79b+ cells scattered throughout the lesions (Suppl. Fig. S2). Additionally, CD79b+ cells were more numerous along the periphery of the nodules where they variably formed small aggregates in each lesion. Cells with strong diffuse nuclear Mum1 labeling were infrequently scattered throughout each of the lesions (Suppl. Fig. S3). Diffuse cytoplasmic immunopositivity for MHCII was strongly positive throughout all 5 lesions and further demonstrated the well-circumscribed margins of each of the nodules. Strong MHC II positive labeling was also identified in intraepidermal Langerhans cells and in cells interpreted as interstitial dendritic cells in the adjacent nonlesional dermis (Suppl. Figs. S4, S5).
Masson’s Trichrome Staining
Masson’s trichrome staining demonstrated evidence of dermal collagen alteration (ie, bright red staining of collagen cores) in nodules from 35/36 included dogs (97%; Fig. 8). Of the 36 cases, the scores for alteration of collagen were negative in 1 (3%), mild in 3 (8%), moderate in 11 (31%), and strong in 21 (58%) cases. These altered collagen bundles were multifocally surrounded by palisading arrangements of histiocytes positive for IBA1 and CD204. The lone negative scoring nodule was ultimately included in the study regardless of the inability to highlight altered collagen cores given all inclusion criteria were met and the histologic appearance carried a strong uniformity to other included nodules.
Clinical Correlates and Outcomes
Complete medical records and/or completed questionnaires were obtained for 12/36 dogs (33%). No sex predilection was identified for the 36 included dogs (18 males; 2 intact and 16 castrated; and 18 spayed females). Age at biopsy ranged from 1 to 12 years with a median of 7 years. Breed sizes were predominately large with 30/36 (83%) of the patients being ≥23 kg. The large breed dogs in the study were 7 large mixed-breed dogs (≥23 kg), 4 Labrador Retrievers, 3 German Shepherds, 3 American Pit Bull Terriers, 2 Golden Retrievers, and 1 of each of the following: Akita, Bernese Mountain Dog, Boxer, Briard, Bull Mastiff, Cane Corso, Husky, Old English Sheepdog, Rottweiler, Saint Bernard, and a nonspecified large breed. The 6 smaller dogs included 2 mixed-breeds and 1 of each of the following: Boston Terrier, Cocker Spaniel, American Eskimo, and Maltese.
The nodules were focal in 31/36 dogs (86%) and multifocal in 5/36 dogs (14%). The nodules in the 5 cases with multifocal distribution involved the lip and gingiva, the muzzle and eyelids, the prepuce and left thigh, the right stifle and both hocks, and the nasal bridge and both hindlimbs. Nodules from all 5 of the dogs with multifocal distribution had moderate comingled infiltrates of lymphocytes, plasma cells, and eosinophils. Of the 36 cases (ie, focal or multifocal distribution), 19 (53%) had nodule(s) on the head, muzzle, face, or nasal planum; 18 (50%) on the extremities; and 2 (6%) on other regions of the body (dorsum and prepuce; Suppl. Table S1).
The nodules ranged from 0.5-cm-diameter (0.07 cm3) to 4-cm-diameter (33.5 cm3) with a mean of 4.6 cm3 (approximately 2 cm diameter). Ongoing concerns and/or treatment for osteoarthritis was mentioned in 5/12 medical records, though there was no correlation between the location of the nodules (eg, head, muzzle or extremity) and site of suspected osteoarthritis (eg, hips, stifle, or vertebral column). Four dogs had a clinical history compatible with allergic skin disease prior to or concurrent with biopsy submission, and one of these dogs also had a history of intermittent flea infestation. One dog with multifocal nodules, some of which contained a moderate eosinophilic infiltrate, was previously diagnosed with a severe hypersensitivity reaction to a suspect arthropod bite one year prior to presentation.
The onset, progression, and clinical outcome were reviewed in biopsy submission forms (36/36 dogs) and available medical records and/or response to questionnaires (12/36 dogs). Only one of the dogs had a history of known traumatic events in the region of the lesions prior to lesion onset (this dog had a focal nodule on the head). In the 12 cases that had follow-up information in the medical records and/or responded to the questionnaires, 3 dogs had nodules of sudden-onset and fast initial-growth phase (<7 days), and 4 dogs had slow progression in size of the nodules prior to surgical removal. Lesion onset and changes/progression were not noted for the other 5 of these 12 dogs, and were not mentioned in the biopsy submission forms of the other 24 dogs. No nodal involvement and no recurrence following excisional biopsy was noted for any of the 36 dogs. All biopsy samples included in this study were excisional with clean histologic margins.
Discussion
The cutaneous entity of palisading granuloma in dogs has been largely anecdotal to date and based primarily on similarities of histologic lesions to cutaneous histiocyte-predominated inflammatory nodules in humans. The veterinary literature lacks case reports of palisading granulomas in dogs or other species and lesion description is limited to Skin Diseases of Dogs and Cats, 2nd Edition. 9 In the current study, the 36 dogs meeting all inclusion criteria had a distinct, non-neoplastic, histiocyte-predominant inflammatory lesion of palisading granuloma. Palisading granulomas are generally circumscribed infiltrates of histiocytes perpendicularly oriented to collagen fibers or foci of necrosis. These may include other inflammatory cell types (ie, lymphocytes, eosinophils, and fewer neutrophils) and, in some foci, histiocytes may also form streams adjacent and parallel to collagen fibers. Collagen fibers frequently demonstrated an altered staining quality with Masson’s trichrome. Though this alteration of collagen bundles (ie, central red staining cores) is not definitive proof of collagen degeneration, it does highlight a potential change in collagen structure that affects stain/counterstain penetration, suggestive of damage. 15
Palisading granulomatous dermatitis can also be identified histologically adjacent to other inflammatory and neoplastic processes, potentially as a result of the aforementioned altered collagen. This is highlighted by the 34 cases excluded from the present study that had distinct principal diagnoses (eg, sebaceous hyperplasia, trichoepithelioma, regressing histiocytoma, atypical spindle cell proliferation, soft tissue sarcoma, vascular proliferative lesion, seroma/granulation tissue, and folliculitis/furunculosis) and varying amounts of palisading arrangements of histiocytes. It is worth noting that a few lesions were identified within the oral cavity, but excluded from this study as they were from noncutaneous sites. For the 36 dogs in this study, there was an overall scarcity of any readily identifiable comorbidities (ie, autoimmune disease, vasculitis, endocrinopathy, or other causative lesions).
In humans, palisading neutrophilic and granulomatous dermatitis is observed in numerous cutaneous disease processes with lesions diagnosed as palisading granulomas, granuloma annulare, rheumatoid nodules, necrobiotic xanthogranulomas, lipoid necrobiosis, and more broadly as interstitial granulomatous dermatitis with substantial clinical and histological variants. 4,14,23 Lesions of interstitial granulomatous dermatitis, with or without histiocyte palisading, may occur in association with eosinophilic granulomatosis with polyangiitis (Churg-Strauss syndrome), granulomatosis with polyangiitis (formerly Wegener’s granulomatosis), systemic lupus erythematosus, psoriatic arthritis, scleroderma, sarcoidosis, rheumatoid arthritis, inflammatory bowel disease/ulcerative colitis, and multiple causes of systemic vasculitis. 4,11,14
Granuloma annulare is considered the most common of interstitial granulomatous dermatitis in humans to exhibit palisading arrangements of histiocytes surrounding dermal and hypodermal collagen with connective tissue degeneration. 14,23 The typical clinical manifestation involves skin-colored papulonodular lesions grouped in centrifugally distributed rings. 14 It is an asymptomatic and benign condition of unknown cause with an unpredictable course that often spontaneously regresses within a 2- to 3-year period. 23 Given the overall innocuous nature of granuloma annulare in humans, aggressive medical or surgical therapy is not recommended, but a few case studies have demonstrated response to cyclosporine therapy. 23 Some of the proposed causes for the collagen fiber degeneration include leukocytoclastic vasculitis, sunburn, insect bite, external trauma, borreliosis, and collagen destruction secondary to metabolic and microangiopathic effects of diabetes mellitus. 11,14
The 5 dogs with multifocal lesions in the present study suggest a potential for an underlying systemic inflammatory condition or more robust immune response to an inciting factor. Interestingly, all of these nodules had a more pronounced mixed inflammatory infiltrate of lymphocytes, plasma cells, and eosinophils compared to the single nodules in the other cases. A case with similar features with more robust lymphoplasmacytic inflammation was reported (AFIP Wednesday Slide Conference, 2007, contributed by Eckman) and was noted to have a bilateral distribution at the ischial tuberosities, for which one nodule responded to surgical removal and the other resolved with glucocorticoid therapy. The possibility of blunt force trauma as a contributing factor or cause was discussed in this case due to the lesion distribution at the ischial tuberosities. According to the data reviewed in this study, palisading granulomas are most commonly encountered in adult large breed dogs at extremities or the face, which is also suggestive of trauma as a potential inciting cause. This remains conjecture considering the overall rarity of palisading granulomas and perceived commonality of traumatic events. In contrast to human eosinophilic granulomatosis with polyangiitis (Churg-Strauss syndrome), no evidence of systemic disease or vasculitis was identified in any dogs in the present study. The lack of clinical history and/or histopathological findings supporting a concurrent systemic disease process (ie, autoimmune disease) or more diffuse collagen targeting further supports the hypothesis that localized trauma/damage to collagen bundles may be the primary inciting cause.
The restricted nodular palisading arrangement of histiocytes surrounding altered collagen fibers supports a reactive inflammatory process responding to the altered collagen, as opposed to the collagen fibers being altered by the CD204- and IBA1-positive histiocytes. The lymphocytic infiltrate documented in a subset of 5 cases suggests an immune response given the close association of predominately CD3+ T cells with fewer CD79b+ B cells and Mum1+ plasma cells with the histiocytes. The robust MHC II positive staining is not surprising of the histiocytic component as constitutive expression is expected in professional antigen presenting cells and this staining is strongly positive in adjacent, nonlesional histiocytes (Suppl. Fig. S5).
The varying degree of eosinophilic infiltrates in 10 dogs in this study, especially the 5 cases with multifocal distribution, warrants consideration of some overlap between eosinophilic granulomas and palisading granulomas. Though the clinical history was supportive of allergic skin disease in 4 dogs, none of these dogs had a significant eosinophilic infiltrate noted in their palisading granulomas. Interestingly, no collagen flame figures were identified in the dogs included in the current study either. None of these 5 dogs with more robust eosinophilic infiltrates had evidence of a hypersensitivity reaction in the clinical history, follow-up, or lesion description typical of, except for one dog that was presented 1 year prior to biopsy to an emergency service for shock and a potential arthropod bite reaction as an inciting factor was discussed.
Granulomatous dermatitis with some palisading features can also be seen in horses, particularly documented with equine eosinophilic granulomas. Equine eosinophilic granulomas most commonly present as multifocal nodules composed of dense populations of infiltrating eosinophils with intermingled and surrounding histiocytes, epithelioid macrophages, and multinucleated giant cells that form variably palisading arrangements. Multiple causes in horses have been proposed including hypersensitivity reactions to arthropod bites, irritation from trauma, silicone-coated needles, and atopy. 2,22 Nodules usually appear during the warmer months of the year. No apparent breed, age, or sex predispositions have been identified. 16 Most recent consensus on the pathogenesis of these eosinophilic granulomas is centered on massive eosinophil degranulation forming collagen flame figures that lack evidence of true collagen alteration identifiable by Masson’s trichrome stain or electron microscopy. 24 Interestingly, some lesions have been reported in locations likely subject to physical trauma or pressure (eg, saddle, harness, halter, or other tack). 16 This subset of cases highlights a potential overlap with canine palisading granulomas related to blunt trauma or irritation.
The overall histologic arrangement and makeup of inflammatory cells in addition to the immunohistochemistry results and associated clinical histories helps further differentiate palisading granulomas from other canine histiocytic diseases. Reactive cutaneous and systemic histiocytosis are multifocal in nature, and other than the variation in organ involvement, are histologically identical to one another. They are characterized by an angiocentric, cellular infiltrate composed of activated interstitial, myeloid, and/or perivascular dermal dendritic antigen-presenting cells admixed with variable numbers of lymphocytes, plasma cells, and/or neutrophils. 1,19 The deep dermis and subcutis are usually extensively involved. 20 Interstitial dendritic cells are expected to be IBA1-positive and E-cadherin-negative with variable CD204 labeling. 3,17,18 Canine sarcoidosis may be focal or multifocal and is histologically defined by dense aggregates of epithelioid macrophages lacking a peripheral cuff of lymphocytes (“naked” granuloma). 9,13 The absence of a palisading arrangement of histiocytes separates this entity from that of palisading granuloma, though a similar pathogenesis is not entirely ruled out. Positive immunolabeling with CD18 and IBA1, but not E-cadherin, is described in one case report of canine sarcoidosis. 13
Cutaneous histiocytic neoplasms differ from palisading granulomas in distribution, morphology, and cell of origin. Cutaneous histiocytoma and cutaneous Langerhans cell histiocytosis are derived from Langerhans cells that are expected to be E-cadherin and IBA1-positive and CD204-negative. 3,17,18 Histiocytomas are typically focal nodules with a top-heavy (ie, superficial dermis) histiocytic component often with epitheliotropism, epidermal abutment, and epidermal hyperplasia manifesting with multiple thin interdigitating rete ridges and/or ulceration. 9,17,18 There are often moderate to marked T cell infiltrates (cytotoxic CD8+ cells) at the base of the nodules in the deep dermis. 9 Langerhans cell histiocytosis is histologically similar to cutaneous histiocytomas, but is multifocal throughout the skin and may be more pleomorphic with lymph node, pulmonary, and other organ involvement. 17,18 Another histopathologic differential diagnosis in some instances may be an inflamed low-grade soft tissue sarcoma if the palisading features are less apparent. These can be further differentiated with immunohistochemistry: the histiocytes are immunolabeled for IBA1 and CD204 in palisading granulomatous dermatitis, whereas the neoplastic cells are negative for these markers and may potentially immunolabel with other more specific markers for respective mesenchymal cell origins (eg, smooth muscle actin, desmin, CD31, S100, and others).
The 3 dogs with mild positivity for E-cadherin among the infiltrate were also moderately to strongly positive for IBA1 and CD204, and important, the E-cadherin-positive cells did not demonstrate a distinct palisading appearance. This demonstrates the potential for migration of Langerhans cells into dermal and subcutaneous inflammatory lesions or en route to regional lymph nodes as antigen presenting cells.
The few dogs with documented fast-growing and/or slow-progressing nodules is similar to that described in humans. 5,6 The retrospective nature of this study did not allow for identification of any spontaneous regression of the nodules, but given the histopathologic resemblances to human and equine inflammatory nodules that do occasionally resolve without surgical removal, it is a possible conjecture for dogs as well. Excisional biopsy appears to be curative and supports the interpretation of these entities as benign localized inflammatory nodules.
In conclusion, palisading granulomas are a distinct, non-neoplastic, histiocyte-predominant condition in dogs associated with altered dermal collagen and favorable prognosis. Large breed dogs are overrepresented. Most lesions affected the face/head and limbs, which supports the hypothesis that physical trauma to dermal collagen may act as a primary inciting cause.
Supplemental Material
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211030525 - Palisading granulomatous dermatitis and panniculitis (palisading granuloma) of dogs
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211030525 for Palisading granulomatous dermatitis and panniculitis (palisading granuloma) of dogs by Michael Lund, Elizabeth A. Mauldin, Enrico Radaelli and Charles W. Bradley in Veterinary Pathology
Footnotes
Acknowledgements
The authors thank Brona Ranieri for assistance with the immunohistochemical staining and to the submitting veterinarians for providing additional follow-up information. The Penn Vet Comparative Pathology Core is supported by the Abramson Cancer Center Support Grant (P30 CA016520).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
