Abstract
Actinobacillus equuli is a rare cause of peritonitis in adult horses. Septicemia and peritonitis due to A. equuli were diagnosed at necropsy in an 8-year-old Saddlebred mare. The origin of the infection was not known; however, small necrotic colonic mucosal lesions presumed to have been caused by phenylbutazone treatment may have allowed bacterial invasion. A good response to antimicrobial treatment has been documented in the small numbers of previously reported acute cases of peritonitis. Because it is potentially treatable, it is important for pathologists and clinicians to identify horses with A. equuli peritonitis.
Keywords
Actinobacillus equuli is a small, nonmotile, gram-negative rod. Certain strains of A. equuli are normal inhabitants of the oral cavity and intestinal tract of horses. 4 A. equuli is a common cause of septicemia in newborn foals, but infections in adult horses are less common and generally more localized. Cases of arthritis, endocarditis, orchitis, periorchitis, abortion, pleuropneumonia, and enteritis have been reported. 1 5 9 12 A few cases of acute and chronic peritonitis caused by A. equuli infection have also been documented in adult horses. 6 7 Death from A. equuli peritonitis is infrequent because of rapid response to antimicrobial and supportive therapy. 6 7 Here, we describe gross, cytologic, and histologic lesions in an adult horse with septicemia and peritonitis due to A. equuli infection.
The body of an 8-year-old Saddlebred mare was presented to the Livestock Disease Diagnostic Center, Department of Veterinary Science, University of Kentucky, for necropsy examination. The horse had developed laminitis 9 days previously and had been treated with 3 g of phenylbutazone (6 mg/kg) three times daily per os (PO) for 4 days, followed by 2 g (4 mg/kg) PO three times daily for 5 days. The following morning, the horse was found dead in the field. No signs of peritonitis or colic had been noted.
At necropsy, numerous 0.1–0.5-cm-diameter caseous white foci were scattered within the pulmonary parenchyma, mesenteric lymph nodes (Fig. 1), and renal cortices. The peritoneal cavity contained approximately 4 liters of red, opaque fluid, and serosal surfaces were granular. The mucosa of the small intestine, cecum, and large colon was slightly expanded by edema fluid. Small numbers of scattered pinpoint red foci of erosion were observed in the colonic and cecal mucosa. The third phalanx of both right and left forelimbs was slightly separated from the dorsal hoof wall but was not rotated. Pure cultures of A. equuli were isolated from lung, liver, kidney, and mesenteric lymph node. Numerous colonies of A. equuli and a few colonies of Escherichia coli were grown from abdominal fluid. Anaerobic bacterial culture was not performed. Smears of fluid from the peritoneal cavity were fixed and stained using both a Romanowsky-type method (Diff-Quik) and Brown and Hopps Gram stain (BH). Representative tissue samples were fixed in 10% neutral buffered formalin, routinely processed, and embedded in paraffin. Five-micrometer-thick sections were mounted on glass slides and stained with haemotoxylin and eosin (HE). Sections of lung, liver, small intestine, colon, mesentery, and mesenteric lymph node were also stained with BH.
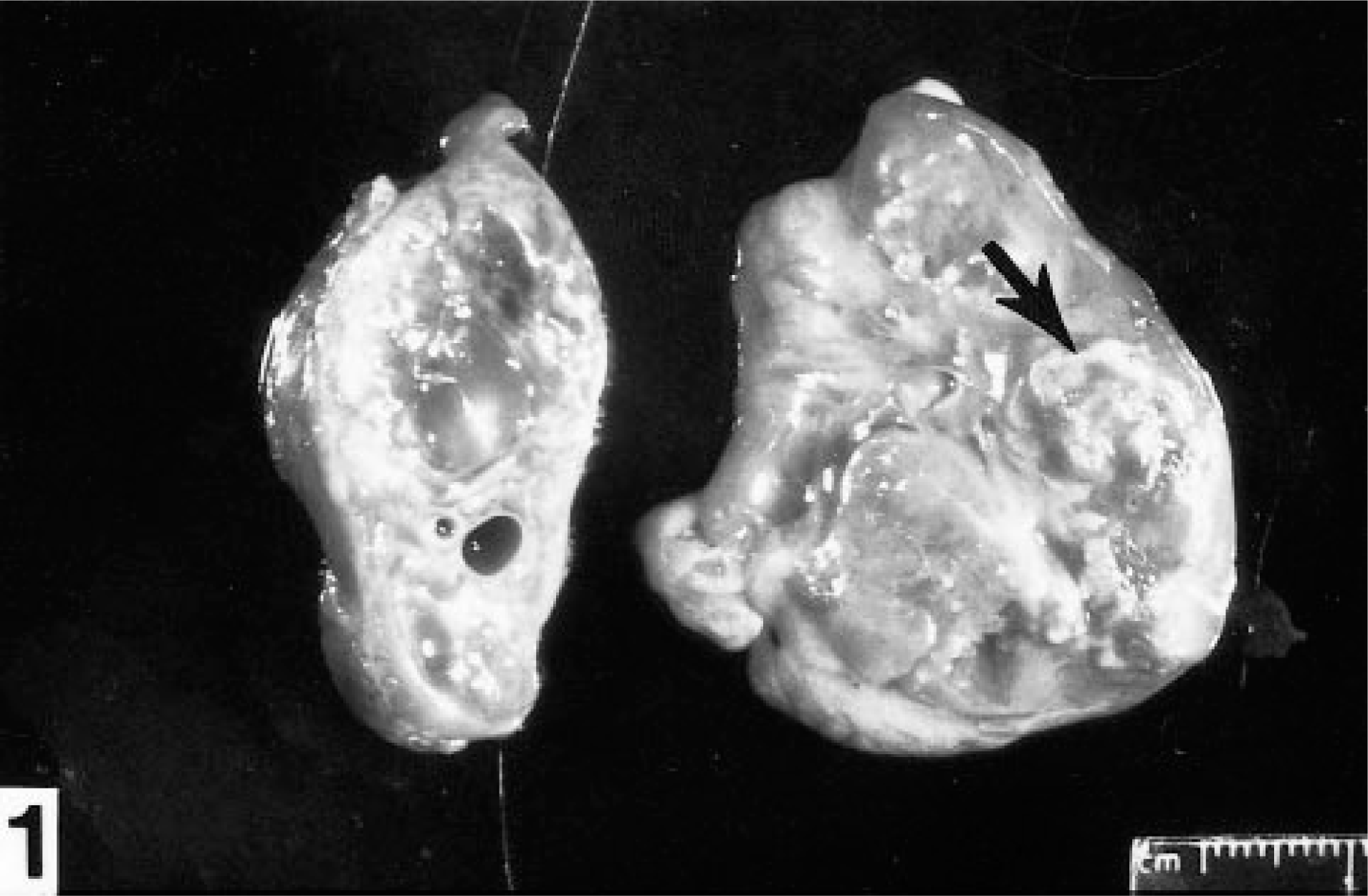
Mesenteric lymph node; horse. The parenchyma is expanded by caseous necrotic foci (arrow).
Smears of abdominal fluid revealed moderate numbers of polymorphonuclear and mononuclear leukocytes with vacuolated cytoplasm, among which were scattered moderate numbers of chains of short gram-negative bacilli (Fig. 2). Short chains of medium-size gram-negative rods were also observed and were presumed to be anaerobic contaminants (Fig. 2).
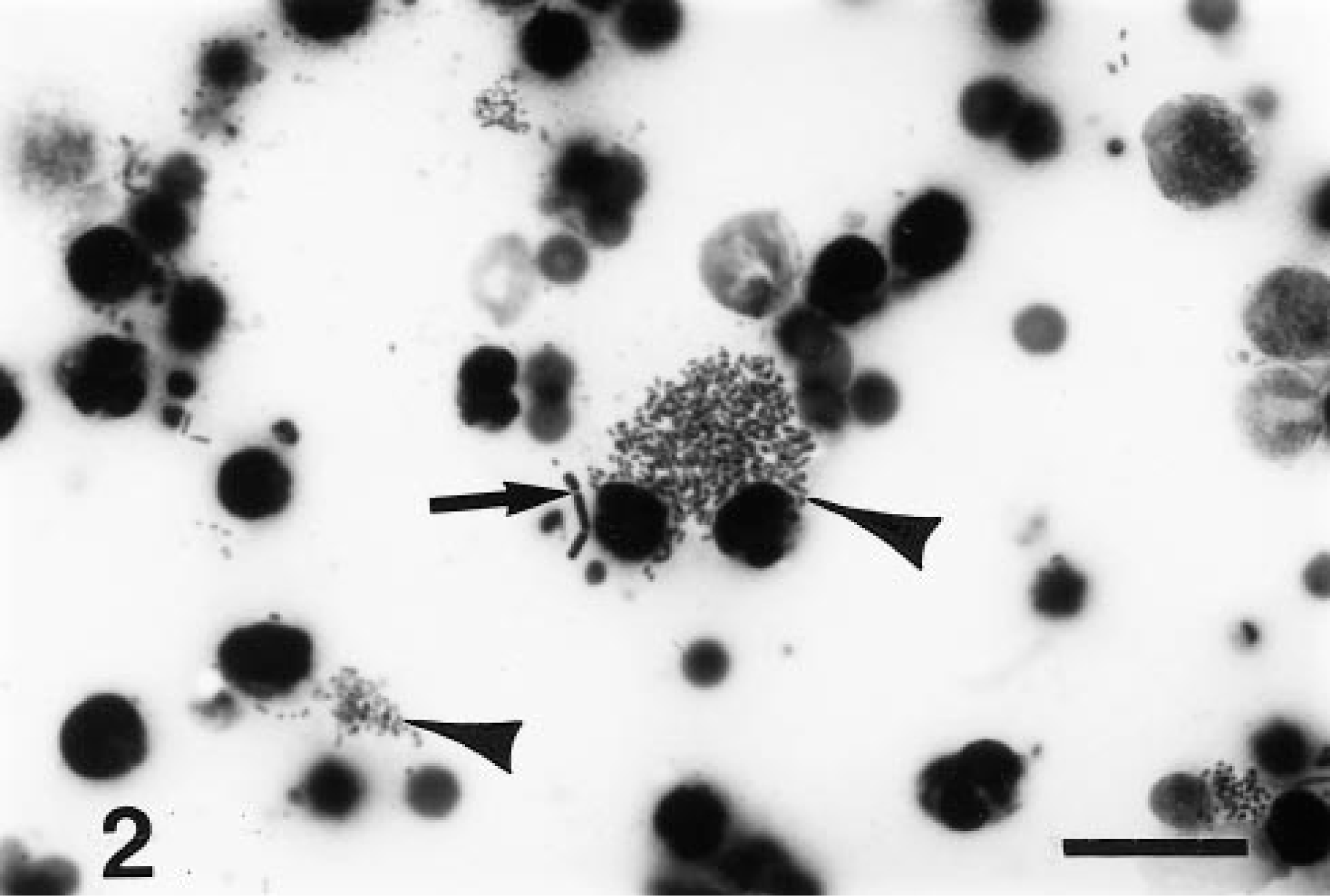
Smear of peritoneal fluid; horse. Clusters of gram-negative bacilli (arrowheads) are scattered among degenerate leukocytes. Chains of gram-negative rods (arrow) are presumed to be anaerobic contaminants. BH. Bar = 20 µm.
Microscopically, groups of pulmonary alveolar spaces adjacent to medium-size or large blood vessels contained moderate numbers of neutrophils and macrophages around large colonies of gram-negative bacilli (Fig. 3). Most pneumocytes lining the alveolar spaces were necrotic. Foci of similar inflammatory cells and bacteria were observed in sections from mesenteric lymph nodes, with caseous necrosis of the lymphoid tissue and intrasinusal fibrin thrombi. The intestinal mesentery and intestinal subserosal tissue were markedly expanded by proliferative fibrovascular tissue (Fig. 3) admixed with small numbers of scattered plasma cells, macrophages, and lymphocytes. Mesothelial cells were swollen and hyperplastic, with extensive erosion. Numerous degenerate neutrophils and large colonies of gram-negative bacilli were observed on the serosal surfaces and within the subjacent stroma. In some sections of colon, small eroded regions of the mucosa were replaced by a moderately thick coagulum of granular eosinophilic debris containing numerous entrapped gram-negative bacterial rods. Fibrin thrombi were observed in blood vessels subjacent to and within the necrotic mucosal tissue. The submucosa was expanded by edema.
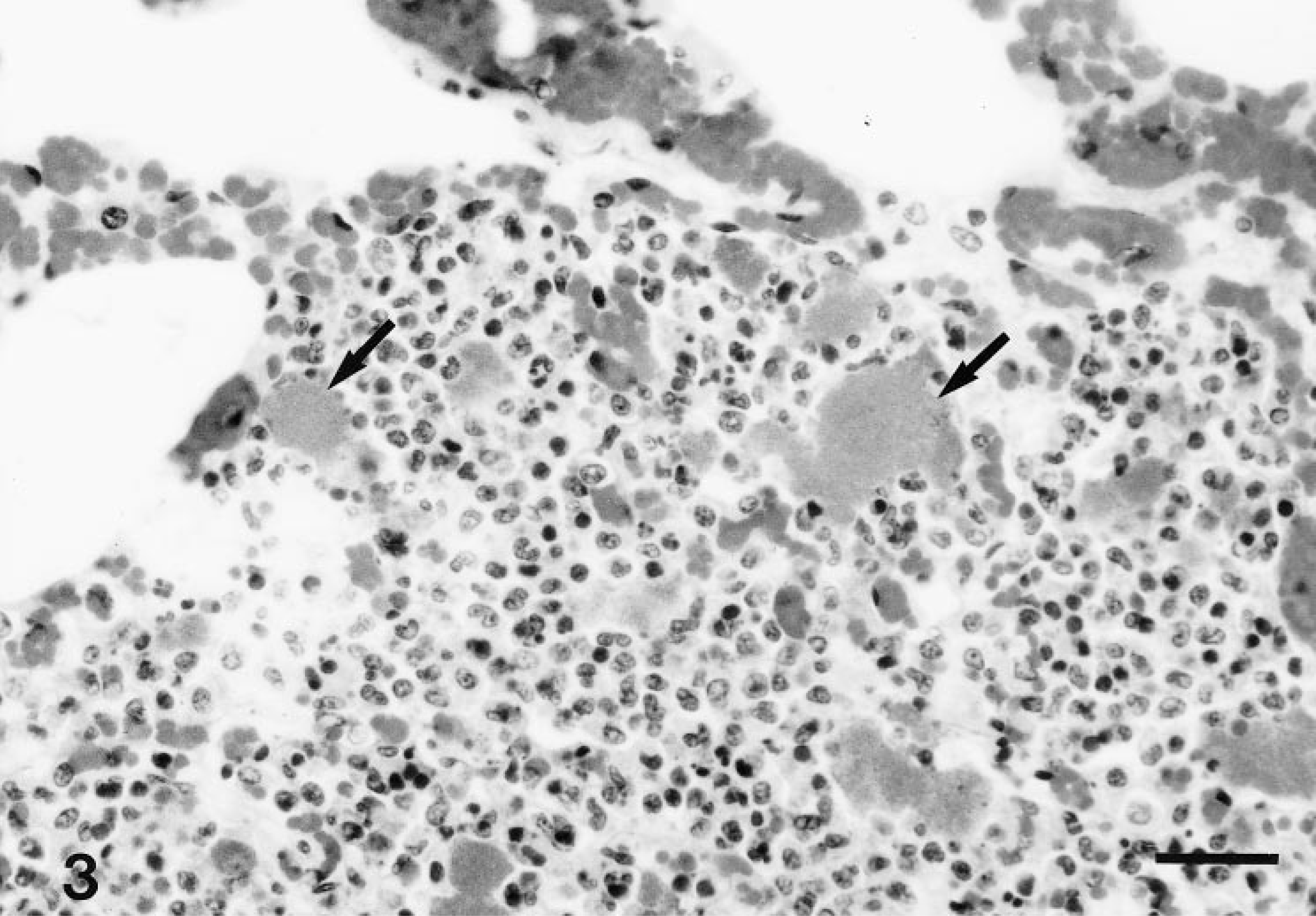
Lung; horse. Alveolar spaces are occupied by neutrophils, macrophages, and lymphocytes, with scattered large colonies of bacilli (arrows). HE. Bar = 30 µm.
Septicemia due to A. equuli infection was diagnosed. Morphologic changes included embolic suppurative pneumonia and necrosuppurative mesenteric lymphadenitis, with intralesional gram-negative bacilli.
A. equuli appears to be a common inhabitant of the oral cavity of horses, despite the low prevalence of infectious disease caused by this bacterial species in adults. In a previous study, 28 of 30 foals and adult horses harbored equine strains of A. equuli in the oral cavity. 3 The prevalence of isolation of A. equuli from the oral cavities of adult horses and foals on individual farms in another study ranged from 12% to 88%. 10 There is a high degree of strain variability among horses on individual farms and within individual horses over time. 10 Results from ribotyping and biochemical fingerprinting of A. equuli isolates from horses in Sweden were not useful for distinguishing between isolates from healthy horses and those from clinically ill cases. 11 Therefore, it is not known whether there are specific strains of A. equuli with greater virulence for foals and/or adult horses or whether such strains are common inhabitants of the equine intestinal tract.
In previously reported clinical cases of peritonitis due to A. equuli infection, there was no evidence of intestinal lesions. 6 7 In one case, peritoneal fluid from affected horses contained neutrophils undergoing degeneration and pleomorphic gram-negative bacilli. 7 In another study, peritoneal fluid samples contained high numbers of nondegenerate neutrophils, but bacteria were not observed. 6 It was suggested in both reports that infection of the peritoneal cavity was due to carriage of A. equuli by strongyle larvae from the intestinal tract. 6 7 A. equuli has been isolated from verminous aneurysms of the cranial mesenteric arteries caused by Strongylus vulgaris larvae. 7 Mesenteric and cecocolic verminous arteritis lesions were observed in one horse with chronic A. equuli peritonitis. 6 No such lesions were observed in the horse of the present study nor were there other possible sources of infection, such as periodontal disease.
In this horse, microscopic erosive lesions were observed in the large colon, with associated thrombosis of superficial submucosal blood vessels. This mare had been treated with phenylbutazone following the development of acute laminitis, the cause of which was not indicated. The dosage of 6 mg/kg three times daily PO for the first 4 days of treatment was higher than the typical recommended dosage for laminitis (4.4 mg/kg twice daily PO or intravenously[IV]). 2 Experimentally, some horses treated with phenylbutazone at 4.4 mg/kg three times daily IV for 12 days developed colonic mucosal erosions associated with thrombosis and disruption of blood vessel walls in the lamina propria. 8 The colonic erosions in this horse may have been caused by the phenylbutazone treatment, and the lesions may then have compromised mucosal integrity sufficiently to allow infection by resident luminal A. equuli. The erosive colonic lesions themselves were small and should not have in themselves caused death. Renal damage consistent with phenylbutazone toxicity was not noted.
Both acute and chronic forms of A. equuli peritonitis have been described. 6 7 Horses that are acutely affected show signs of abdominal pain, intestinal stasis, lethargy, and inappetance. Horses with chronic cases show weight loss over a period of several weeks. In one study, A. equuli isolates from horses with peritonitis were consistently susceptible to penicillin, in contrast with isolates from septicemic foals and extraperitoneal sites in adult horses. 7 All 15 horses in that study responded clinically to antimicrobial treatment, and no deaths were associated with the peritonitis. 7 Gross and microscopic lesions were unusually severe in the horse in the present report. Clinical signs of peritonitis were not recognized, and in the absence of antimicrobial and supportive therapy, the disease progressed unabated. The septicemia and peritonitis are presumed to have been the cause of death of this horse. Laminitis is not a direct cause of death of horses, and there was no evidence of renal failure or marked gastrointestinal ulceration due to phenylbutazone toxicity.
This is the first detailed description of gross and microscopic lesions of septicemia and peritonitis due to A. equuli infection in an adult horse. A. equuli peritonitis is a rare diagnosis in adult horses. Because appropriate antimicrobial treatment in small numbers of horses with acute A. equuli peritonitis has resulted in clinical improvement and a return to original activity, pathologists and clinicians examining smears of abdominal fluid from equine colic cases should be aware of this diagnosis.
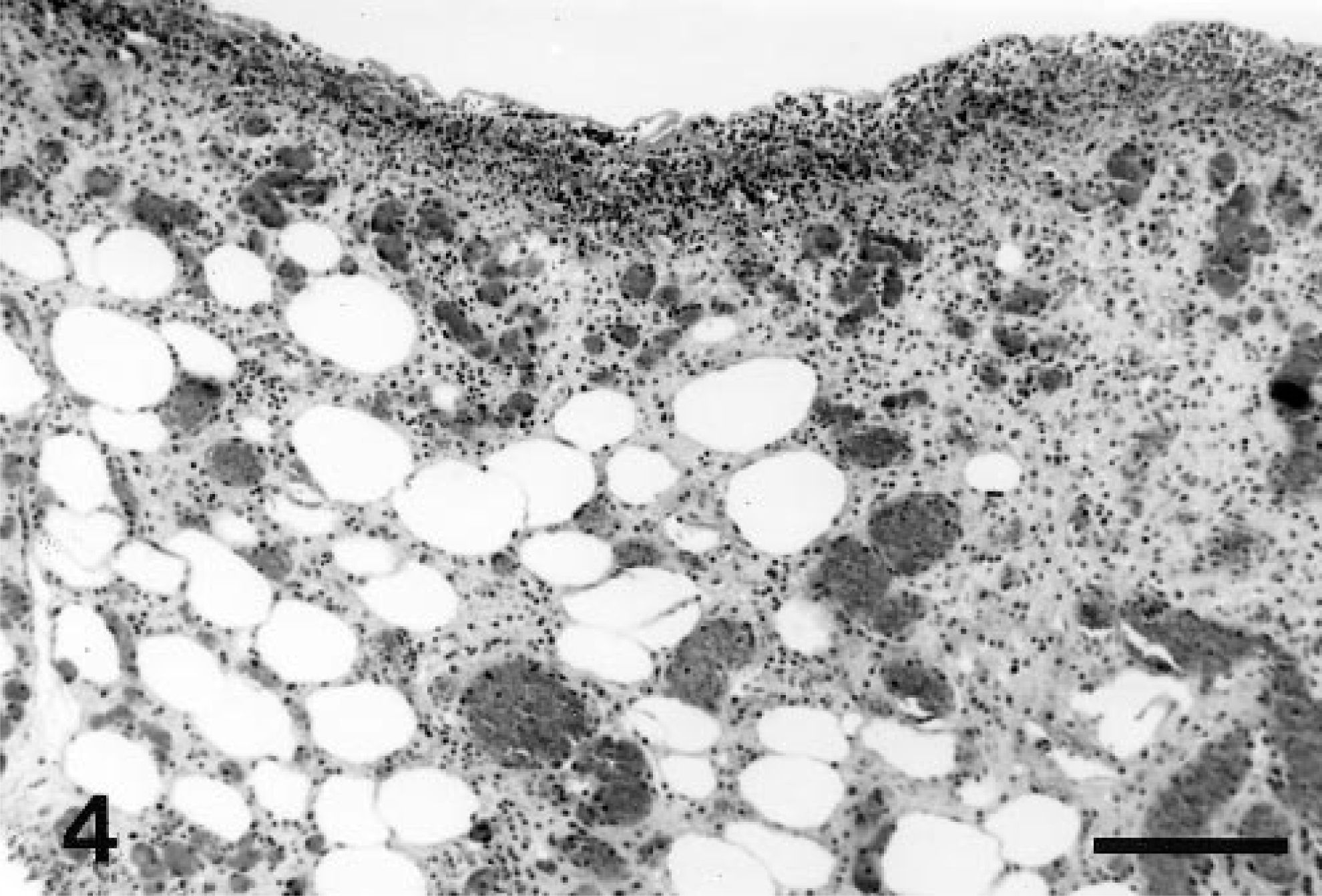
Intestinal mesentery; horse. The mesenteric membrane is markedly expanded by fibrovascular tissue and mononuclear inflammatory cells. HE. Bar = 200 µm.
Footnotes
Acknowledgements
This work is published as College of Agriculture, University of Kentucky, Publication No. 99-14-175. We thank Dr. L. McKinney of the Department of Pathology and Infectious Diseases, The Royal Veterinary College, University of London, for reviewing this manuscript.
