Abstract
Omphalitis and the resulting septicemia contribute to perinatal mortality in several animal species. In foals, the most important causes of omphalitis are Escherichia coli and Streptococcus zooepidemicus. However to date, no information has been published about the role of Clostridium sordellii in these infections. In this paper, we describe 8 cases of perinatal mortality in foals associated with internal umbilical remnant infection by C. sordellii. The foals studied were between 12 and 21 days old at the time of death, and various breeds were represented in the group. Five of the foals were male and 3 were female. The diagnosis was established on the basis of the detection of C. sordellii by 3 methods (culture, fluorescent antibody test, and immunohistochemistry) and on gross and histopathologic findings. All foals had acute peritonitis, and the internal umbilical remnant was thickened by edema, hemorrhage, and fibrosis. A moderate amount of serosanguinous fluid with fibrin strands was present in the pericardial sac and pleural cavity. Histopathologically, the urachus and umbilical arterial walls were thickened by edema and exhibited hemorrhage, fibrin, and leukocytic infiltration. Gram-positive bacterial rods were observed in subepithelial areas of the urachus, the adventicia of umbilical arteries, and interstitium of the internal umbilical remnant. On the basis of these findings, we suggest that C. sordellii should be considered in the differential diagnosis for infections of the internal umbilical remnant in foals.
Omphalitis and the ensuing septicemia contribute to perinatal mortality in several animal species.3,10,12,13,29 The microorganisms most frequently associated with omphalitis in domestic animals are Escherichia coli, Proteus spp., Staphylococcus spp. and Arcanobacterium (Actinomyces) pyogenes.38 In addition, several clostridial species have been associated with omphalitis in lambs,29 humans,4,11,15,44 and a llama.43 Of these clostridial species, Clostridium sordellii has been frequently associated with omphalitis in newborn human babies.2,23,28
In foals, the most important causes of omphalitis are E. coli and Streptococcus zooepidemicus.3,9,35,41 Clostridium botulinum type B has also been isolated from internal umbilical remnants of foals which had neurologic signs and navel abscesses,45 and Clostridium perfringens has been associated with urachitis and uroperitoneum in neonatal foals.22 However, there is currently no published information about the role of C. sordellii in omphalitis in foals.
Clostridia are gram-positive anaerobic rods that are a significant cause of disease and death in farm animals. C. sordellii, Clostridium septicum, C. perfringens, Clostridium chauvoei, and Clostridium novyi, alone or in concert, are responsible for malignant edema of animals. C. sordellii has been associated with malignant edema of cattle and sheep and wound infections in humans.1,29,39,46 Although malignant edema has been described in horses, it has never been attributed to C. sordellii in this species.33,34 This microorganism has also been associated with hyperammonemia in horses;18 emphysematous abomasitis in lambs;24,25 hemorrhagic enteritis in foals,21 sheep,5 and cattle;6 and the so-called sudden death syndrome in cattle, a condition about which information on pathogenesis is scant.17
C. sordellii produces 2 major toxins: a lethal toxin and a hemorrhagic toxin.27,40 The lethal toxin produced by C. sordellii is causally involved in enteritis of domestic animals and in wound infections of humans.20,37 The hemorrhagic toxin is not well characterized,40 but it has been suggested as a cause of dermal and intestinal necrosis in guinea pigs.37 The toxins of C. sordellii are often compared with toxins A and B of Clostridium difficile, and it has been reported that a specific antiserum against the lethal toxin of C. sordellii successfully neutralized the cytotoxic and lethal activities of C. difficile toxin B.7,19,26,31,37,47
In this report, we present 8 cases of perinatal mortality in foals associated with navel infection by C. sordellii.
Materials and Methods
Case selection and clinical history
Eight foals were selected from the archives of the San Bernardino branch of the California Animal Health and Food Safety Laboratory System on the basis of isolation of C. sordellii from tissues of foals with omphalitis.
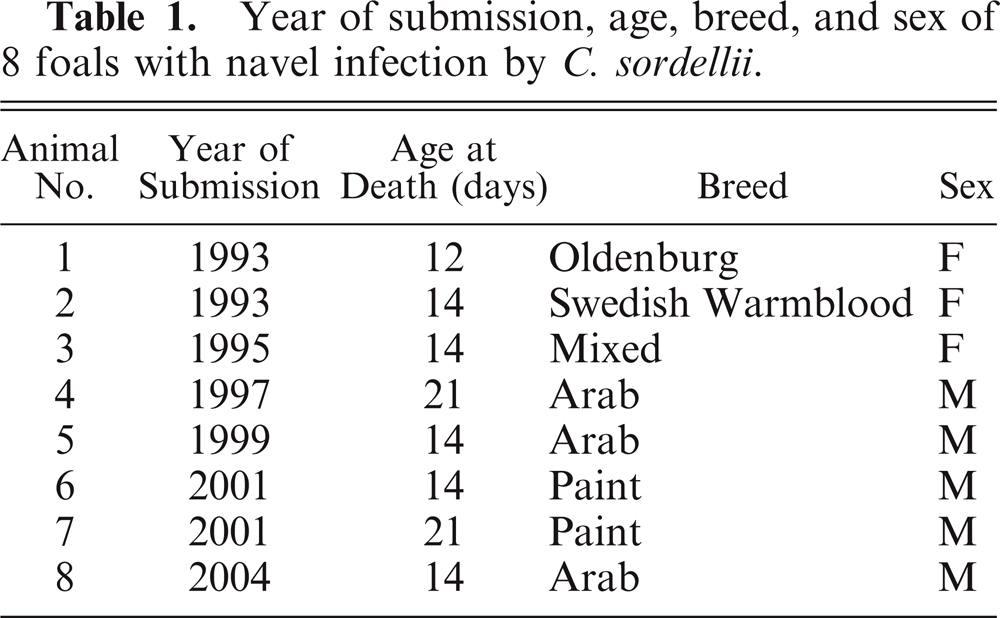
Foals studied were between 12 and 21 days old at the time of death (average 15.5 days, median 14 days), and 5 were male and 3 female. Several breeds were represented amongst these animals. These cases occurred between 1993 and 2004 on different properties throughout Southern California (Table 1).
Year of submission, age, breed, and sex of 8 foals with navel infection byC. sordellii.

Clinical histories indicated that all the foals were normal at birth. In most foals, (1, 2, 4, 5, 7, and 8), no clinical signs were observed before death; they were thought to be clinically normal at night and found dead the next morning. However, foal No. 3 neither drank nor defecated for 24 hours before death, and foal No. 6 was hyperexcited and showed respiratory distress for a few hours before death. No treatment was attempted on any of the animals in this study.
Internal umbilical remnants from 2 foals (7 and 10 days old) that died of causes unrelated to umbilical infection were also included in this study as controls for the microscopic examination.
Gross and microscopic pathology
A full necropsy was performed on all foals within 8–24 hours of death, and samples of brain, lung, liver, kidney, spleen, urinary bladder, muscle, internal umbilical remnant, and small and large intestine were fixed by immersion in 10% buffered formalin, pH 7.2, for 24 hours. All tissues from the 8 foals and the internal umbilical remnant tissues from the 2 control foals were processed by standard techniques for the production of 4 μm-thick HE sections. Internal umbilical remnant sections were also Gram-stained.
Bacteriology
Samples of internal umbilical remnant and peritoneal fluid from all affected foals were aseptically collected and inoculated onto 5% sheep blood agar and incubated both aerobically and anaerobically at 37°C for 48 hours. Subsamples of the same specimens were inoculated onto cooked meat medium and incubated anaerobically at 37°C for 48 hours. Samples of intestinal content and internal umbilical remnant from all animals were inoculated onto MacConkey agar and incubated aerobically at 37°C for 24 hours. All isolates were identified by conventional biochemical techniques.
Direct fluorescent antibody test
Paraffin sections of internal umbilical remnant of the 8 affected foals were subjected to a direct fluorescent antibody test (FAT) for C. sordellii, C. chauvoei, C. septicum, and C. novyi with the use of a slight modification of the technique described by Sterne and Batty.42 Briefly, deparaffinized and hydrated sections were incubated in darkness with fluorescent conjugates prepared against the above-mentioned clostridia in a moist chamber at 37°C for 30 minutes. The slides were washed with phosphate-buffered solution (PBS), pH 7.2; counterstained with a 0.2% Evan Blue (Sigma, St. Louis, MO) solution in PBS for 20 seconds; washed in PBS; mounted with buffered glycerol, pH 8.0 and examined under ultraviolet light in a fluorescent microscope. International reference strains of the clostridial species mentioned above were used as control for each fluorescent preparation. The fluorescent antibodies were from VMRD (Pullman, WA).
Immunohistochemistry
A streptavidin–biotin kit was used according to the manufacturer's instructions (LSAB-peroxidase K675; Dako, Carpinteria, CA). Primary rabbit polyclonal antibodies (20 mg/ml) against C. sordellii were prepared as previously described and used diluted 1:6,500 in PBS, pH 7.4.8 Sections 4 μm-thick were deparaffinized and incubated overnight with the primary antibody. After washing, the sections were incubated for 15 minutes with an anti-rabbit biotinylated antibody, washed again, and incubated with a streptavidin–peroxidase complex for a further 15 minutes. The sections were then developed with a hydrogen–peroxidase–diaminobenzidine solution, counterstained with Harry's hematoxylin and coverslipped. The incubations were carried out at room temperature. Negative controls consisted of sections incubated with normal rabbit serum instead of the primary antibody. Positive controls consisted of sections of muscle of guinea pigs that had been inoculated with C. sordellii.
Results
Gross pathology
Gross lesions were similar in all animals. Ventral abdominal muscles and subcutaneous tissue were expanded by yellow and gelatinous edema, which spread radially from the navel for 4–5 cm. The parietal peritoneum around the navel and the mesentery were intensely red. The parietal peritoneum caudal to the navel was diffusely emphysematous. There was a moderate excess of bloody, slightly cloudy, and foul-smelling peritoneal fluid. The serosal surfaces of the abdominal organs had multifocal paintbrush hemorrhages that were most marked on the colonic serosa. The internal umbilical remnant was thickened by edema, hemorrhage, and fibrosis. The apex of the urinary bladder and the urachus were thickened, edematous, and hyperemic. A moderate amount of serosanguinous fluid with fibrin strands was in the pericardial sac and pleural cavity. No other significant abnormalities were observed in any of the animals.
Microscopic pathology
In the sections from the 8 affected animals, the lumen of the urachus was obliterated by desquamated epithelial cells, hemorrhage, fibrin, cell debris, and neutrophils (mostly degenerate) and fewer lymphocytes, plasma cells, and macrophages. In most foals, the epithelium was completely lost, and in only 2 foals could a partially preserved epithelium be observed. The subepithelial layers of the urachus revealed diffuse edema, hemorrhage (Fig. 1), and a mostly neutrophilic infiltrate with fewer lymphocytes, plasma cells, and macrophages. The external layers of the urachus contained immature connective tissue and dilated lymphatic vessels, which were engorged by neutrophils and fibrin. The arteries of the internal umbilical remnant were thrombosed with abundant neutrophils (mostly degenerated), red blood cells, and laminated fibrin and fewer lymphocytes, plasma cells, and macrophages (Fig. 2). The endothelial cells of the intima were hypertrophic, and multifocal areas of necrosis and mineralization were seen in the tunica intima and media. Diffusely, the tunica muscularis was edematous, causing separation of the smooth muscle fibers. The adventitia of arteries and interstitium of the internal umbilical remnant were also expanded by edema, fibrin exudation and hemorrhages. Gram-positive bacterial rods were observed in subepithelial areas of the urachus (Fig. 3), the adventicia of umbilical arteries, and interstitium of the internal umbilical remnant. These bacteria were large (5–7 × 0.8–1 μm), with parallel borders and round ends and were observed singly or in clusters. Many of the bacteria exhibited cylindrical or spherical spores in a central or subterminal location, which in most cases did not deform the rods.
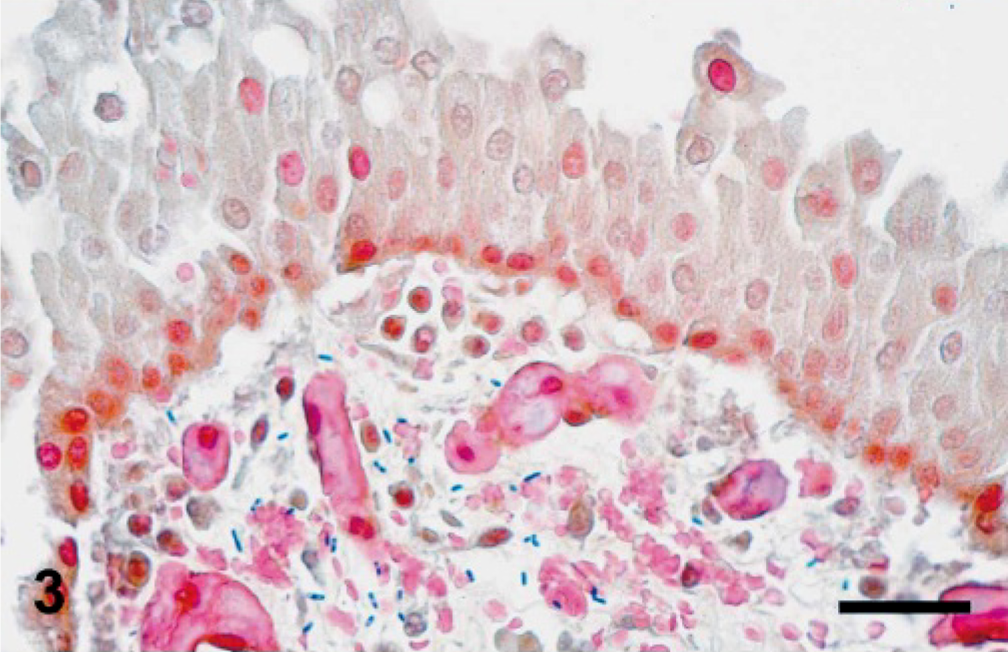
Urachus, foal No. 3. Higher magnification of Fig. 1. Observe the large number of Gram-positive rods under the urachus epithelium. Many of these bacteria have terminal or subterminal spores. Gram. Bar = 60 μm.
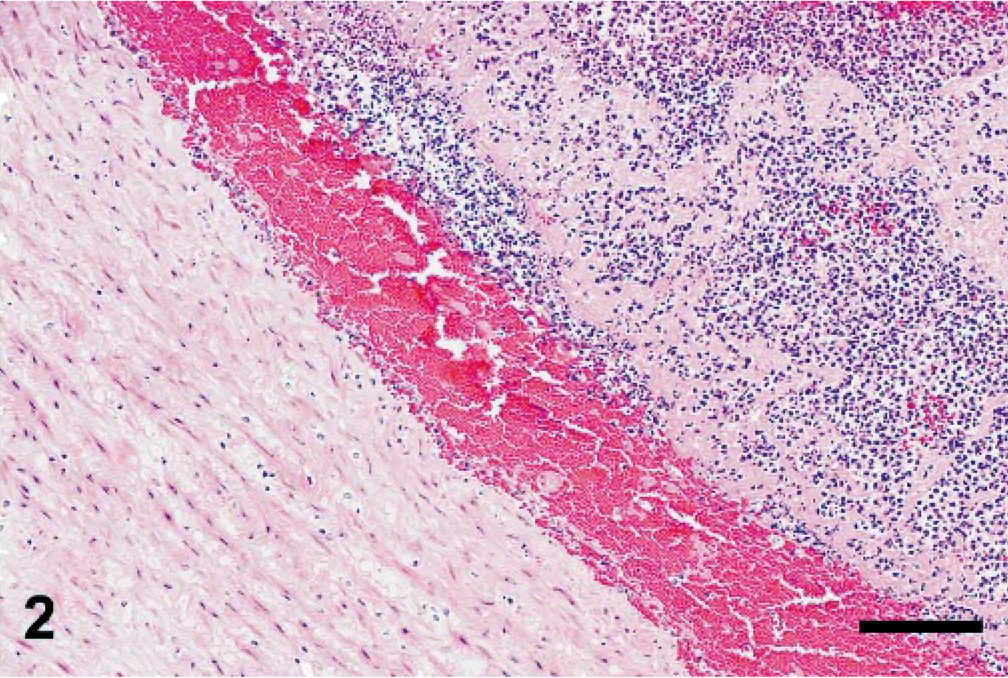
Umbilical artery, foal No. 1. The artery is thrombosed and infiltrated by abundant neutrophils, red blood cells, and laminated fibrin and fewer lymphocytes, plasma cells, and macrophages. HE. Bar = 120 μm.
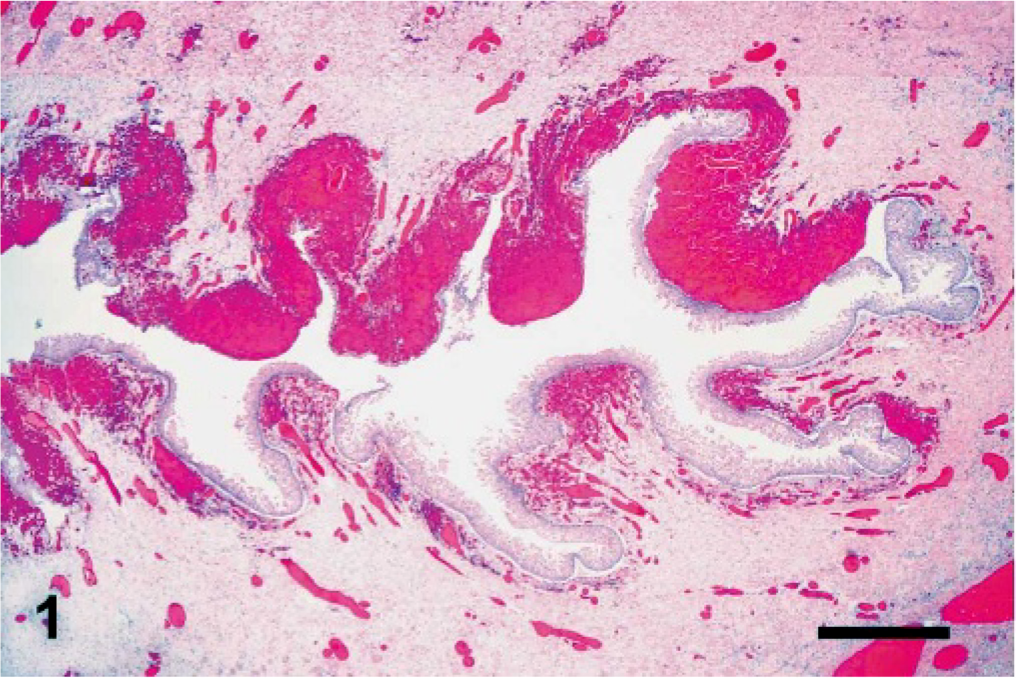
Urachus, foal No. 3. Observe the desquamation of the urachus epithelium and focally extensive subepithelial hemorrhage. HE. Bar = 300 μm.
The gastric and intestinal serosas demonstrated diffuse edema, capillary congestion, multifocal hemorrhage, and multifocal neutrophilic infiltration. No significant histologic abnormalities were observed in the other organs examined.
The internal umbilical remnants of the 2 control foals showed only minimal diffuse lymphoplasmocytic infiltration.
Bacteriology
A summary of bacteriologic findings is shown in Table 2. C. sordellii was isolated from internal umbilical remnant, peritoneal fluid, or both in all 8 affected foals. In all samples, the cultures were moderate or rich. However, only 2 foals (case Nos. 1 and 6) yielded a pure culture of C. sordellii from the internal umbilical remnant, whereas in the other 6 foals, this microorganism was isolated together with other bacteria (Table 2). No Salmonella spp. were isolated from any of the animals.
Bacteriologic findings in 8 foals with navel infection.
L = large; M = moderate; R = rare; NI = no isolation.
+ = positive for fluorescent antibody test (FAT) or immunohistochemistry (IHC).
FAT
The FAT on formalin-fixed, paraffin-embedded tissues of internal umbilical remnant was positive for C. sordellii in all foals, whereas FAT for C. chauvoei, C. novyi, and C. septicum was negative. Each fluorescent conjugate produced positive staining with the corresponding microorganism but not with any of the other clostridial species tested. Positively-stained sections revealed many rods, singly or in clusters, that had apple-green fluorescence on a dark red background (Figs. 4, 5). Morphology and location of these bacteria were similar to those described for Gram-stained sections.
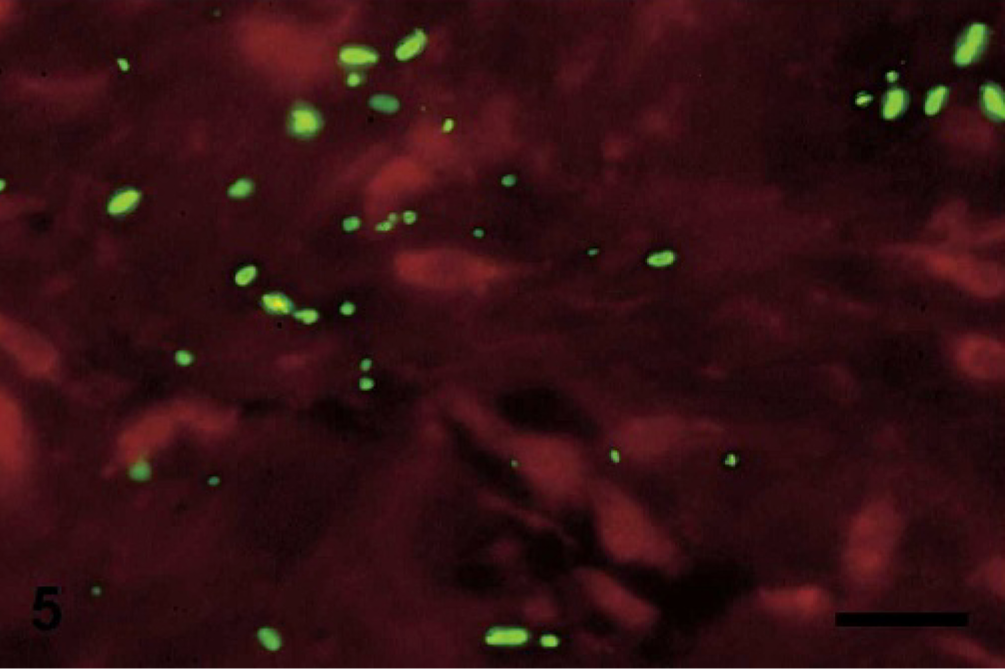
Urachus, foal No. 4. Observe the central and subterminal spores in positively stained C. sordellii. FAT–Evans blue counterstain. Bar = 25 μm.
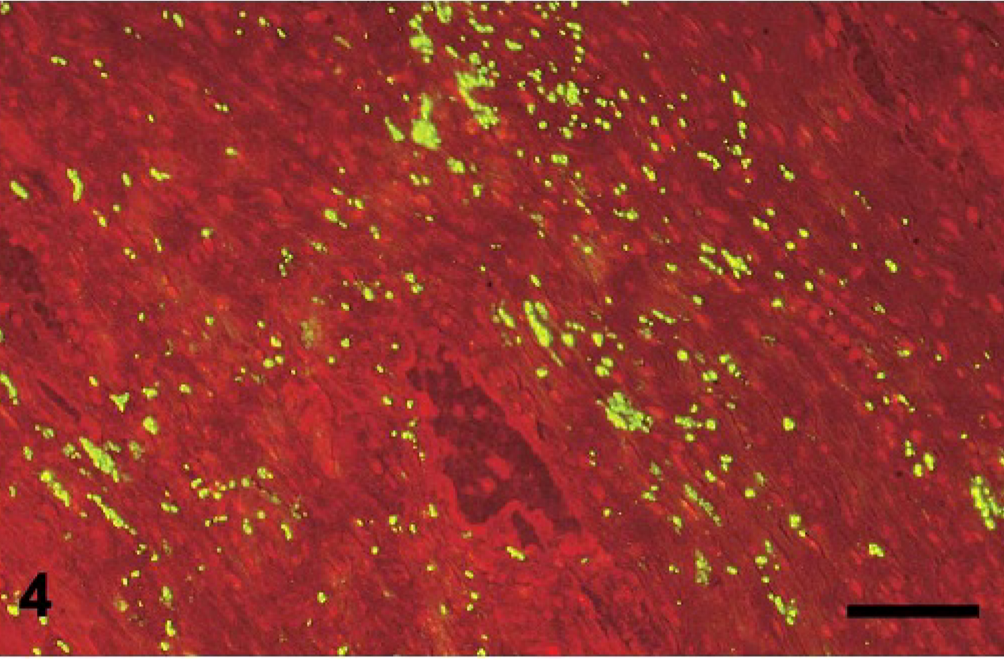
Urachus, foal No. 4. Observe the large number of fluorescent C. sordellii. Fluorescent antibody test–Evans (FAT–Evans) blue counterstain. Bar = 120 μm.
Immunohistochemistry
Internal umbilical remnant sections processed by the streptavidin–biotin technique were positive when tested with C. sordellii primary antibodies (Fig. 6). The positive-stained bacteria were in the same locations and had similar morphology to those described for the sections stained with Gram and FAT. Positive control tissues from guinea pigs stained positively with this technique. No staining was observed in any of the negative controls.
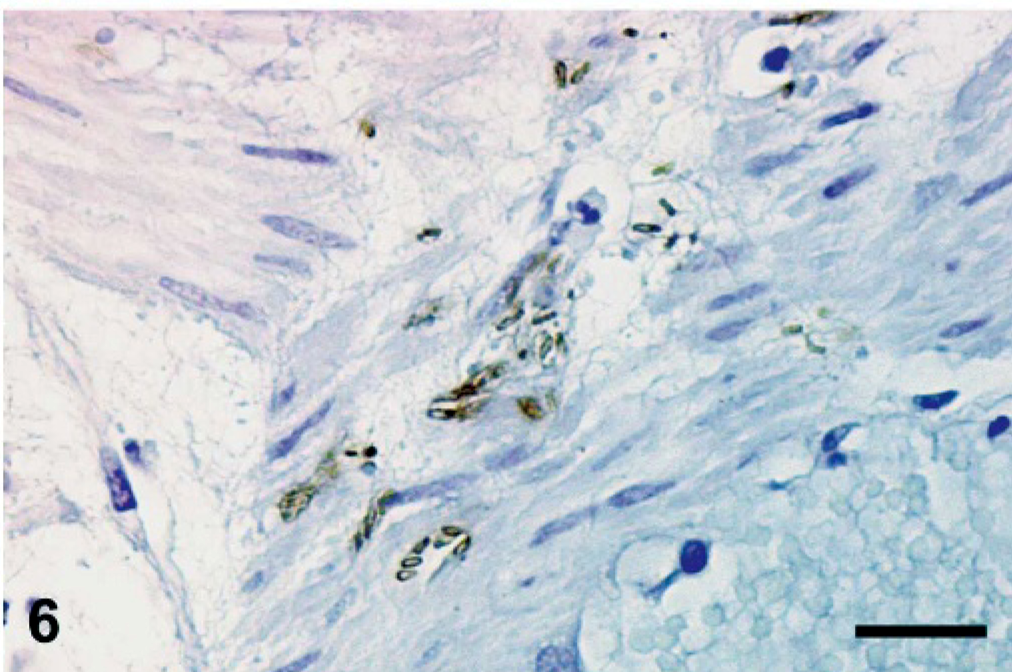
Internal umbilical remnant, foal No. 7. Observe the rods with central and subterminal spores positively stained by a streptavidin–biotin technique with C. sordellii primary antibodies. Bar = 25 μm.
Discussion
In this study, a diagnosis of internal umbilical remnant infections by C. sordellii was established on the basis of gross and histopathologic findings coupled with specific detection of this microorganism by 3 methods: culture, FAT, and immunohistochemistry.
C. sordellii was isolated from the internal umbilical remnant, peritoneal fluid, or both in all animals. Also, the isolation of C. sordellii in purity from the internal umbilical remnant of 2 foals suggests that this microorganism was the primary pathogen in at least these 2 foals. Internal umbilical remnant sections of foal No. 4, from which C. sordellii was isolated in only peritoneal fluid, were intensely positive to C. sordellii FAT and immunohistochemistry, thus confirming the presence of this bacterium in the internal umbilical remnant of the mentioned animal. All the animals in this study showed systemic changes, including excess of pericardial fluid with fibrin strands and hemorrhages of serous membranes, suggesting that systemic spread of C. sordellii or its toxins could have occurred. Also, C. sordellii in pure culture was isolated from lungs and heart blood of foal No. 2 (data not shown), confirming that, at least in this foal, bacteremia was present. The role of the other bacteria isolated from the internal umbilical remnant is difficult to evaluate. However, some of the microorganisms isolated from the internal umbilical remnant in the animals of this study have been previously implicated in omphalitis of foals, and it is possible that they acted synergistically with C. sordellii to cause omphalitis.22 We cannot, however, rule out the supposition that at least some of these microorganisms were secondary invaders.
The internal umbilical remnant consists of the urachus, 2 umbilical arteries, and 1 umbilical vein, which are encased in a sheath of connective tissue.32 Tearing of the umbilical cord at birth opens a potential path for infection. Poor hygiene of the navel area and the surrounding environment at the time of birth often predisposes animals to internal umbilical remnant infections. These infections frequently serve as a source for bacterial sepsis, secondary infections, or both in other parts of the body.14,35,36
Most foals in our study died suddenly, suggesting that the infections were acute or hyperacute. Probably, the organisms had been present for a while and eventually breached the limitation of the umbilical remnant to cause acute peritonitis.
Internal umbilical remnants from 2 foals that died of causes unrelated to infection of these tissues were included in this study as normal controls. The lack of significant inflammatory infiltrate in the tissues of the 2 foals lends credence to the belief that the inflammatory reaction observed in the affected foals was indeed produced by C. sordellii, acting alone or in combination with the other microorganisms demonstrated in those tissues.
In adult sheep, C. sordellii, together with other clostridial species, is responsible for malignant edema, an infection of subcutaneous and muscular tissue predisposed by wound infections.29,46 In human babies, C. sordellii has been responsible for several cases of fulminant necrotizing omphalitis.2,23,28 Also, metritis in postparturient sheep has been associated with C. sordellii, and it has been postulated that the uterine infection ascended from the vulva when it came into contact with the ground during lambing or abortion.16 Similarly, several deaths by endometritis and toxic shock syndrome associated with C. sordellii have occurred in women after medically induced abortions.30 Malignant edema in animals is considered an “exogenous infection,” in which the organisms responsible for the infection or its spores are present in the environment and gain access to the tissues via skin or mucosal lacerations. Dissimilarly, blackleg is an “endogenous infection” of mostly cattle produced by C. chauvoei alone, in which the spores of this microorganism are latent in the muscle of clinically healthy animals and germinate when predisposing factors such as trauma produce tissue necrosis in an anaerobic environment.39 In the foals presented here, it is most likely that the infection was exogenous, with C. sordellii gaining entrance to the tissues via the torn umbilical cord. Also, the local necrosis, the leftover blood, and decreased oxygen tension produced in this area by the breaking of the cord could have provided anaerobic conditions for the proliferation of C. sordellii.
Information about the distribution of C. sordellii in the environment is scant. However, the wound infections described in animals and humans mentioned before1,2,23,46 and the navel contamination of the foals in our study suggest that this microorganism is present in the environment and is, therefore, a potential risk for wound contamination.
According to gross, histopathologic, and microbiologic findings, we consider that the foals presented in this study died of a combination of peritonitis and septicemia originating from a internal umbilical remnant infection by C. sordellii.
Footnotes
Acknowledgements
We thank S. J. Uzal and R. Barker for reviewing this manuscript and E. J. Hurley and S. Kwiek for excellent technical assistance. This work was performed with financial support from California Animal Health and Food Safety Laboratory. J. Ortega was supported by the “Becas dirigidas a la movilidad del profesorado” of the Fundacion Universitaria San Pablo-CEU, Madrid, Spain.
