Abstract
Eight newborn calves showing ataxia were necropsied and examined histologically. Six of seven cerebrospinal fluid samples collected from these animals had neutralizing antibody for Akabane virus (AKV). All examined calves had nonsuppurative encephalomyelitis, localized mainly in the midbrain and spinal cord. Corresponding to the encephalitic lesion, AKV antigen was demonstrated in neuroglial cells in the brain stem and neuronal cells in the ventral horn of the spinal cord. This is the first study to demonstrate AKV antigen by immunohistochemistry in naturally infected newborn calves.
Akabane disease is caused by Akabane virus (AKV), an arbovirus isolated from mosquitoes in Japan and Australia. Virologically, it has been demonstrated that AKV infection in cows occurs in summer. The consequence of natural and experimental infections of fetuses with AKV in the summer is congenital abnormalities during the winter months, chiefly arthrogryposis and hydranencephaly. 2 5–8, 12 There are few reports on the pathogenesis of AKV infection in ruminants just before and/or after birth, although AKV antigen had been detected in experimentally infected ovine fetuses. 9 13 Previously, natural infection with AKV resulted mainly in abortion, and consequently few infected calves with neuroglial signs were available for examination. We report here the first demonstration of AKV antigen by immunohistochemistry in neuroglial cells in naturally infected newborn calves.
In August to September 1998, 12 aborted fetuses and 18 newborn calves showing ataxia were seen in Fukuoka Prefecture in the southern part of Japan. Twelve aborted fetuses and two of the newborn calves were not examined. Sixteen 1–20-day-old newborn calves from 15 different farms were taken to the Ryochiku Livestock Hygiene Service Center, Fukuoka Prefecture, showing ataxia and, in one case (calf 3), blindness. Akabane disease was diagnosed in eight of these calves based on immunohistochemical demonstration of AKV antigen as described herein. After systematic necropsy, tissues were fixed for 7 days in 10% neutral buffered formalin, embedded in paraffin wax, and stained with hematoxylin and eosin (HE).
AKV, Aino virus (AIV), bovine herpesvirus type 1 (BHV-1) and Neospora caninum (N. caninum) were evaluated in tissue sections using immunohistochemistry by the streptavidin-biotin (SAB) immunoperoxidase method using Histofine SAB reagents (Nichirei Corp., Tokyo, Japan). After deparaffinization, endogenous peroxidase activity was blocked by treatment with 3% H2O2 in absolute methanol, then the tissue sections were treated with 0.1% pronase in phosphate-buffered saline for 5 minutes at room temperature. The sections were then incubated with normal goat or rabbit sera, primarily antibody, SAB reagents, and 0.05% 3,3-diaminobenzidine tetrahydrochloride and 0.1% H2O2 in tris buffer, pH 7.6. They were then counterstained with methylgreen. Anti-AKV and AIV rabbit sera were provided by Dr. Y. Miura, National Institute of Animal Health, Japan, and used at dilutions of 1:4,000 and 1:6,000. Anti-BHV-1 goat serum (VMRD Inc., Pullman, WA) and anti-N. caninum goat serum (VMRD Inc.) were diluted 1:2,048 and 1:2,560, respectively. Mouse brain experimentally infected with AKV, AIV, and N. caninum and BHV-1-infected calf lung samples served as positive controls. Sera from nonimmunized rabbit and goat served as negative controls. Cerebrospinal fluid (CSF) samples were aspirated from seven of the eight calves at necropsy and antibody levels were measured. The viruses used to assay CSF antibody levels were the JaGAr39 strain of AKV, JaNAr28 strain of AIV, the K-47 strain of Chuzan virus (CHV), the Tokachi strain of bovine viral diarrhea and mucosal disease (BVD-MDV), and the Los Angeles strain of BHV-1 (provided by Dr. Y. Miura, National Institute of Animal Health, Japan). Neutralizing antibodies for AKV, AIV, CHV, BVD-MDV, and BHV-1 were measured by the microtiter method, and those for bluetongue virus (BTV) were measured by the immunodiffusion method. 3 Virus isolation with HmLu-1 cells was done from the brain stem and spinal cord of each calf.
No gross lesions were found in any of the calves. Microscopically, changes were distributed predominantly in the brain stem, including the midbrain, pons, and medulla oblongata, and in the spinal cord (Table 1). They were found to a lesser degree in the cerebellum and the cerebrum. Lesions were composed of perivascular cuffs of lymphocytes, plasma cells, and macrophages, diffuse and focal proliferation of glial cells (Fig. 1), and pyknosis of neuronal cells. In the midbrain, one calf (No. 4) had a small focus of necrosis with pyknosis of glia and neurons, loss of myelin, infiltration of glial cells and macrophages, and vacuolation of neuropils (Fig. 2). In the cervical, thoracic, and lumber spinal cords, perivascular cuffing, diffuse and focal proliferation of glial cells, pyknosis of neuronal cells, and decreased number of neurons were noted to be distributed bilaterally in the ventral horn. No histologic lesions or viral inclusion bodies were found in any other organs examined, including muscles.
Lesions, antibody titers, and immunohistochemical findings in neonatal findings in neonatal calves with Akabane disease.∗
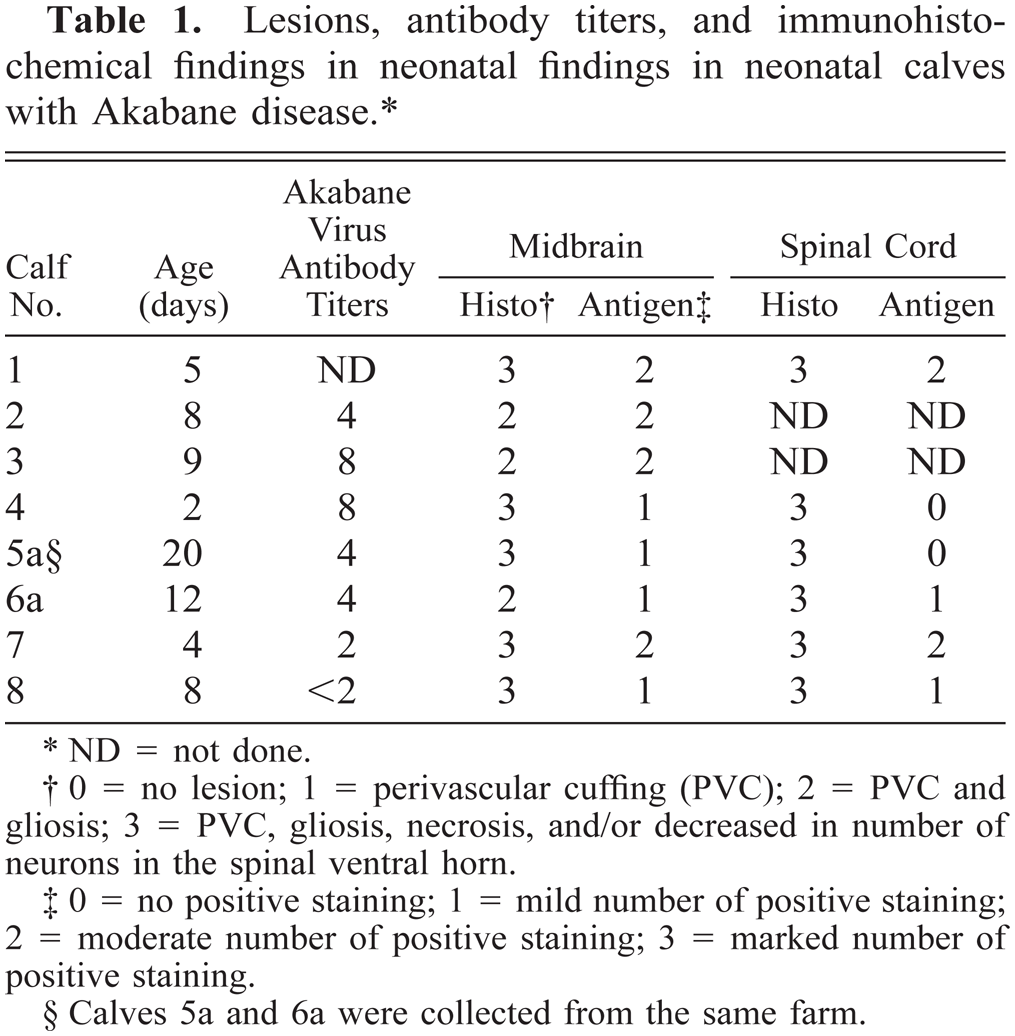
ND = not done.
0 = no lesion; 1 = perivascular cuffing (PVC); 2 = PVC and gliosis; 3 = PVC, gliosis, necrosis, and/or decreased in number of neurons in the spinal ventral horn.
0 = no positive staining; 1 = mild number of positive staining; 2 = moderate number of positive staining; 3 = marked number of positive staining.
Calves 5a and 6a were collected from the same farm.
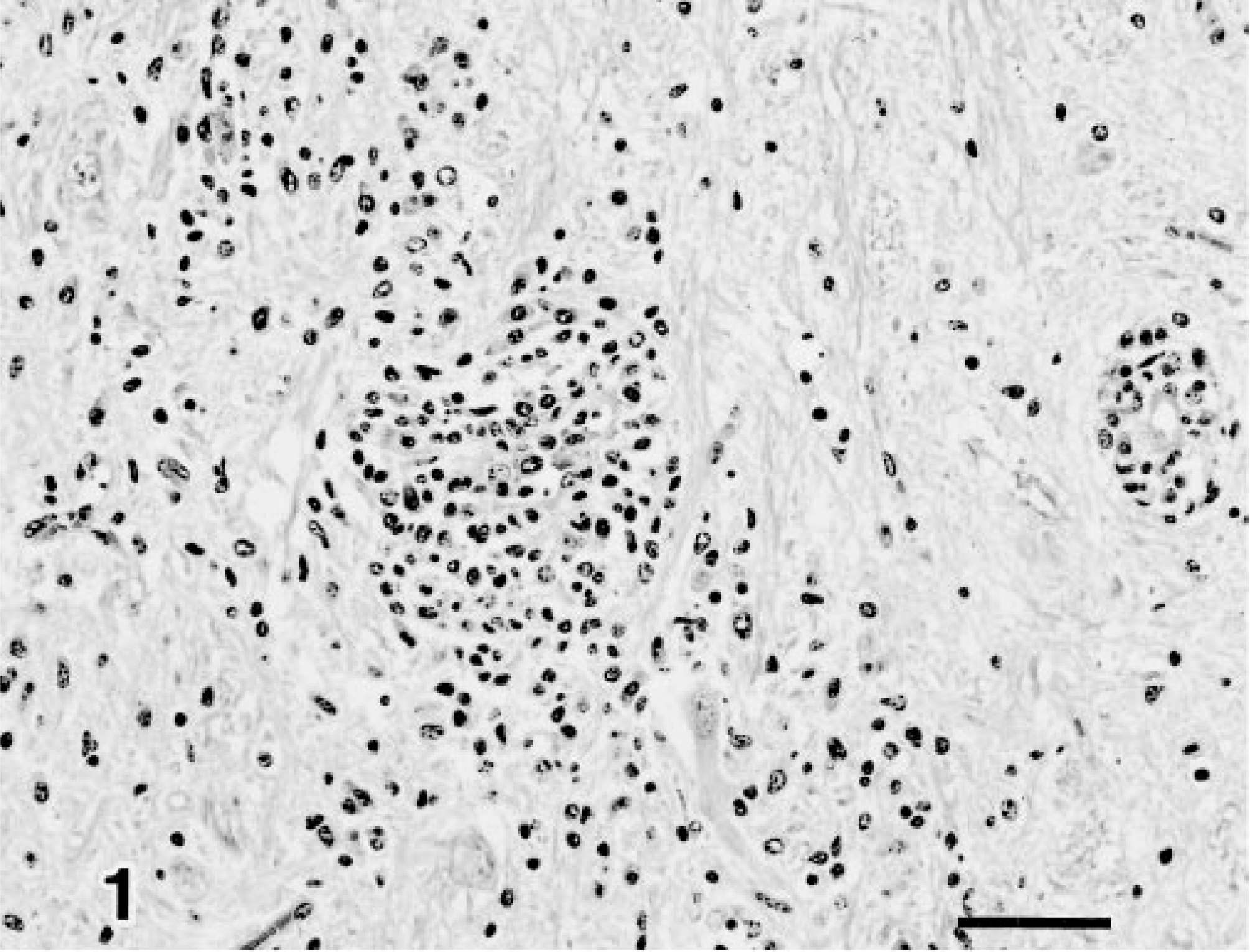
Midbrain; calf 10. Focal gliosis and perivascular cuffs in the midbrain. HE. Bar = 50 µm.
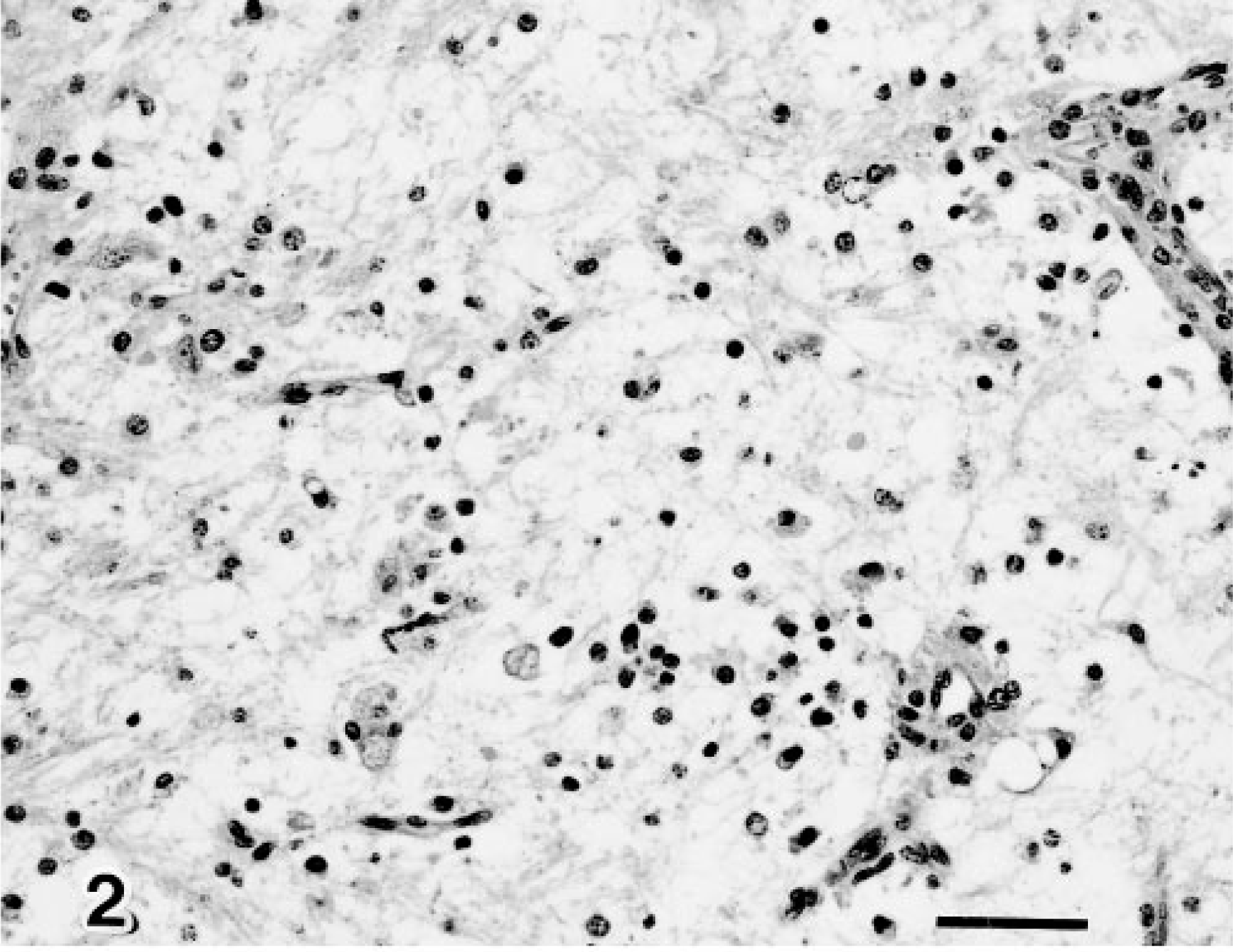
Midbrain; calf 4. Necrotic foci with demyelination and macrophages. HE. Bar = 25 µm.
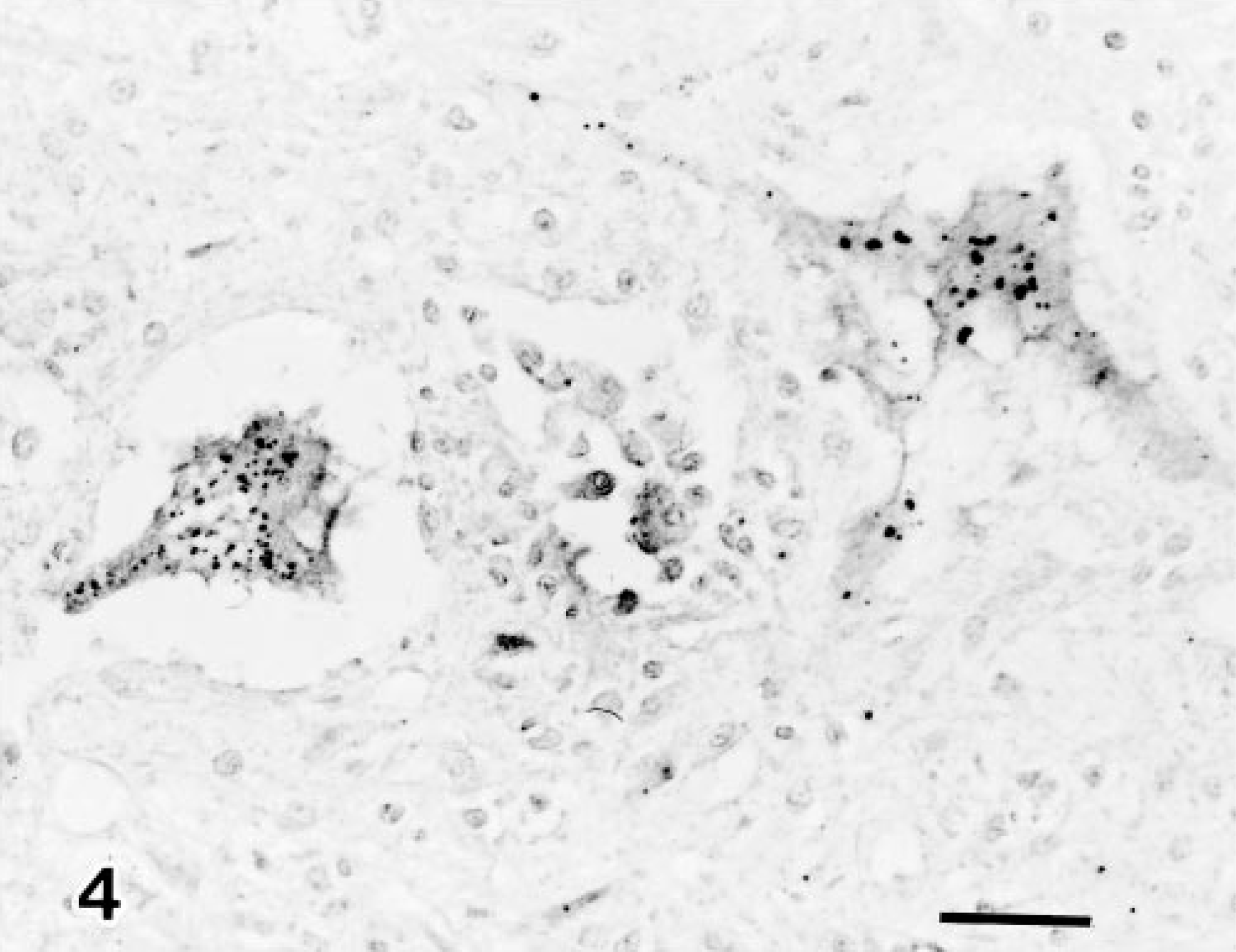
Immunohistochemically, AKV antigen was seen as granules in the cytoplasm of degenerated neuroglial cells in the brain stem (Fig. 3) as well as neuronal cells in the spinal ventral horn (Fig. 4), (Table 1). There was no evidence of AIV, BHV-1, or N. caninum antigen in the tissues examined.
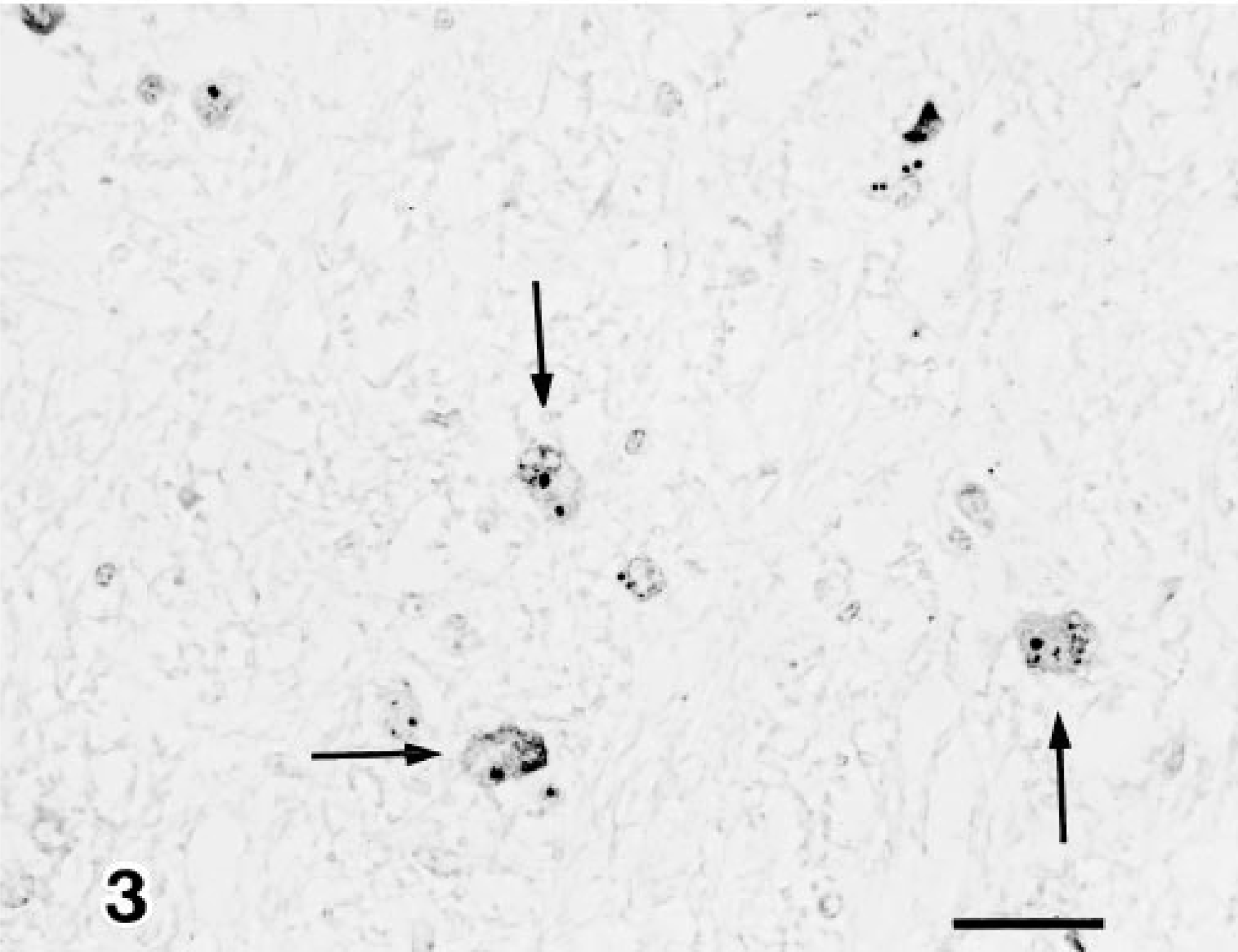
Midbrain; calf 10. Akabane virus antigen (arrows) is detected in the neuroglial cells. Anti-Akabane virus SAB. Bar = 25 µm.
Lumber spinal cord; calf 1. Akabane virus antigen is demonstrated in degenerated neuronal cells in the ventral horn. Anti-Akabane virus SAB. Bar = 35 µm.
Neutralizing antibody to AKV was found in the CSF from six of seven calves tested from the eight with microscopic lesions, and titers ranged from 1:2 to 1:8 (Table 1). No antibodies were detected against AIV, BHV-1, CHV, BVD-MD, and BTV. Moreover, no virus was isolated from the brain stem or spinal cord.
AKV is a predominantely seasonal disease in Japan. 5 It occurs from December through March and is associated with hydranencephaly and arthrogryposis. 5 7 In the present study, six calves with encephalomyelitis had neutralizing antibody for AKV but not for AIV, CHV, BVD-MDV, BHV-1, or BTV within CSF, suggesting that clinical abnormalities seen in these newborn calves might be due to AKV infection.
It is well known that many agents, such as viruses, 1 3 10 protozoa, 11 and bacteria, produce encephalitis in newborn calves and may be differentiated by the character of the brain lesions. In the present study, eight calves had nonpurulent encephalomyelitis, consisting of focal gliosis, perivascular cuffs, pyknosis of neuronal cells, and a decrease in the number of spinal ventral horn neurons. One newborn calf had small foci of necrosis with pyknosis of glia and neurons, infiltration of glial cells and macrophages, and vacuolation of the neuropil. These lesions were localized mainly in the midbrain and spinal cord with fewer in the cerebrum and cerebellum. No viral inclusion bodies were found in HE sections from any calves examined. The character of encephalomyelitis in the present cases, except for the focus of necrosis in one calf, closely resembled the description of the encephalitic lesion observed in the early stage of epizootic outbreaks of Akabane disease. 2 5 7
The distribution of AKV in infected fetuses has been investigated by culture 4 and immunofluorescence. 13 AKV antigen was demonstrated in neuroglial cells in the brain and spinal cord and in ganglion cells in cranial and abdominal ganglia in experimentally infected ovine fetuses. 9 13 In the present immunohistochemical study, AKV antigen was found in neuronal cells distributed in the spinal ventral horn and neuroglial cells in the midbrain of eight infected newborn calves. This is the first report to demonstrate AKV antigen by immunohistochemistry in naturally infected newborn calves.
Footnotes
Acknowledgements
We thank Mr. M. Kobayashi and Miss M. Shimada for preparing the pathological sections and Dr. Y. Ando and Mr. T. Fujisawa for preparing the photographs.
