Abstract
Immunohistochemistry using a commercial polyclonal antibody for lyssavirus was applied to 39 archival cases of rabies. Paraffin blocks from 13 different species were available, including 3 dogs, 4 cats, 1 pig, 6 cattle, 4 horses, 1 llama, 7 skunks (Mephitis mephitis), 7 raccoons (Procyon lotor), 1 bat (Myotis species), 1 white-tailed deer (Odocoileus virginianus), 1 bobcat (Lynx rufus), 2 gray foxes (Urocyon cinereoargenteus), and 1 red fox (Vulpes vulpes). All cases had previously been diagnosed as rabies using histopathology and/or fluorescent antibody testing. The immunohistochemistry technique successfully detected lyssavirus antigen in all cases. In species for which 3 or more samples were available, distributional trends were seen in 4 main brain regions: brainstem, cerebellum, hippocampus, and cerebrum. The best site for rabies virus detection in dogs and cats was the hippocampus. For cattle, viral antigen was most prominent in the brainstem, followed by the cerebellum. In horses, the cervical spinal cord and adjacent brainstem were the optimal sites for detecting rabies virus antigen. In raccoons and skunks, positive labeling was widely dispersed, so selection might be less important for these wildlife reservoir species. Immunohistochemistry should prove useful in enhancing the accuracy of rabies diagnosis through informed selection of brain sampling sites when composite sampling is not feasible. This immunohistochemical technique could provide reliable virus detection in formalin-fixed tissues in any potentially infected species.
Rabies is a neurologic disease characterized by acute, progressive, fatal encephalitis. 1,3,19,20,26 It is probably the oldest known zoonosis, recognized for more than 5,000 years. 9,19,20 The main viral reservoirs include animals of the orders Carnivora and Chiroptera, 8 but all mammalian species are susceptible at any time, and the disease is found on every continent except Antarctica. 2,13,19,25 Rabies virus, a lyssavirus of the family Rhabdoviridae, is a neurotropic virus. Inoculation is generally through a carnivore or bat bite; the virions travel retrograde along axons and across synapses to the central nervous system (CNS). Viral replication and dissemination occur widely throughout the CNS before the virus spreads centrifugally to the salivary glands. 3,6,13,22,25,26 It is not possible to detect rabies infection during the incubation period, which is generally 1 to 8 weeks. 6,13,20,22,26 Therefore, the diagnosis of rabies can only be achieved with 100% certainty through postmortem examination of the central nervous system or trigeminal ganglia. 6,19,20 It is essential that rabies diagnosis in animals be rapid and accurate to facilitate postexposure treatment decisions for exposed humans. 27
Histologically, rabies is characterized by a viral encephalitis with intracytoplasmic Negri bodies in neurons, but these changes can be mild or even absent. 13,18,20 Although the presence of Negri bodies has been considered pathognomonic for rabies, they are absent in 20% to 60% of rabies cases. 1,10,15,16,20,24,26 Because the encephalitis of rabies can be challenging to distinguish from other viral encephalitides, the diagnosis of rabies should include ancillary tests such as direct fluorescent antibody test (FAT) and intracerebral inoculation in suckling mice, which are the standard tests for rabies today. 10,16,18,20,24,27 Despite its efficiency in quickly detecting rabies, FAT has many drawbacks that can limit its usefulness, including equipment cost, potential exposure of laboratory staff to live virus, fading nature of fluorochrome dyes, and, in the developing world, necessity of getting fresh specimens to potentially distant laboratories in adequate condition for testing. 1,7,10 An estimated 55,000 humans die annually of rabies, with almost all of these in the developing world (http://www.who.int/rabies/en/). Improved diagnostic testing for this at-risk population is essential. Over the last 2 decades, immunohistochemistry (IHC) has become increasingly popular for detecting numerous antigens in fixed tissues. 7,10,20,26 Studies have shown that the sensitivity of IHC for rabies is equal to that of FAT. 16,27 IHC may be even more sensitive in early diagnosis of suspected cases when traditional histological and FAT techniques could not detect viral antigens or lesions; 1,11,26 however, the disadvantage of IHC, so far, is that most publications were based on the use of in-house antibodies, mostly monoclonal, and therefore with specificity for the regional strains of the virus. 1,11,16,26,27
Because rabies does not always infect all regions of the brain equally and viral dissemination varies among species, the erratic distribution of viral antigen can compromise the reliability of test results if all parts of the brain are not sampled or if a particular sample has low levels of viral antigen. The collection of optimal samples is therefore essential for optimizing diagnosis. 2 The main objectives of this study were to develop an IHC protocol with a polyclonal antibody suitable for rabies diagnosis in various mammalian species, examine the reliability of that technique, and develop a preliminary assessment concerning the regions of the brain most useful for diagnosis of rabies in various species.
Methods
Cases
All formalin-fixed, paraffin-embedded (FFPE) samples were obtained from the archives of the Department of Pathology, Athens Diagnostic Laboratory, and Southeastern Cooperative Wildlife Disease Study at the College of Veterinary Medicine, University of Georgia, Athens. These cases had been previously diagnosed as rabies by FAT and/or histopathological examination. Immunohistochemistry was performed on a total of 39 cases, encompassing 13 different mammalian species, and included 7 skunks (Mephitis mephitis), 7 raccoons (Procyon lotor), 6 cattle, 4 cats, 3 dogs, 4 horses, 1 pig, 1 llama, 2 gray foxes (Urocyon cinereoargenteus), 1 red fox (Vulpes vulpes), 1 bat (Myotis species), 1 white-tailed deer (Odocoileus virginianus), and 1 bobcat (Lynx rufus). Pertinent data are in Table 1.
Intensity of Rabies Antigen Immunoreactivity in Major Brain Areas of 13 Mammalian Species a
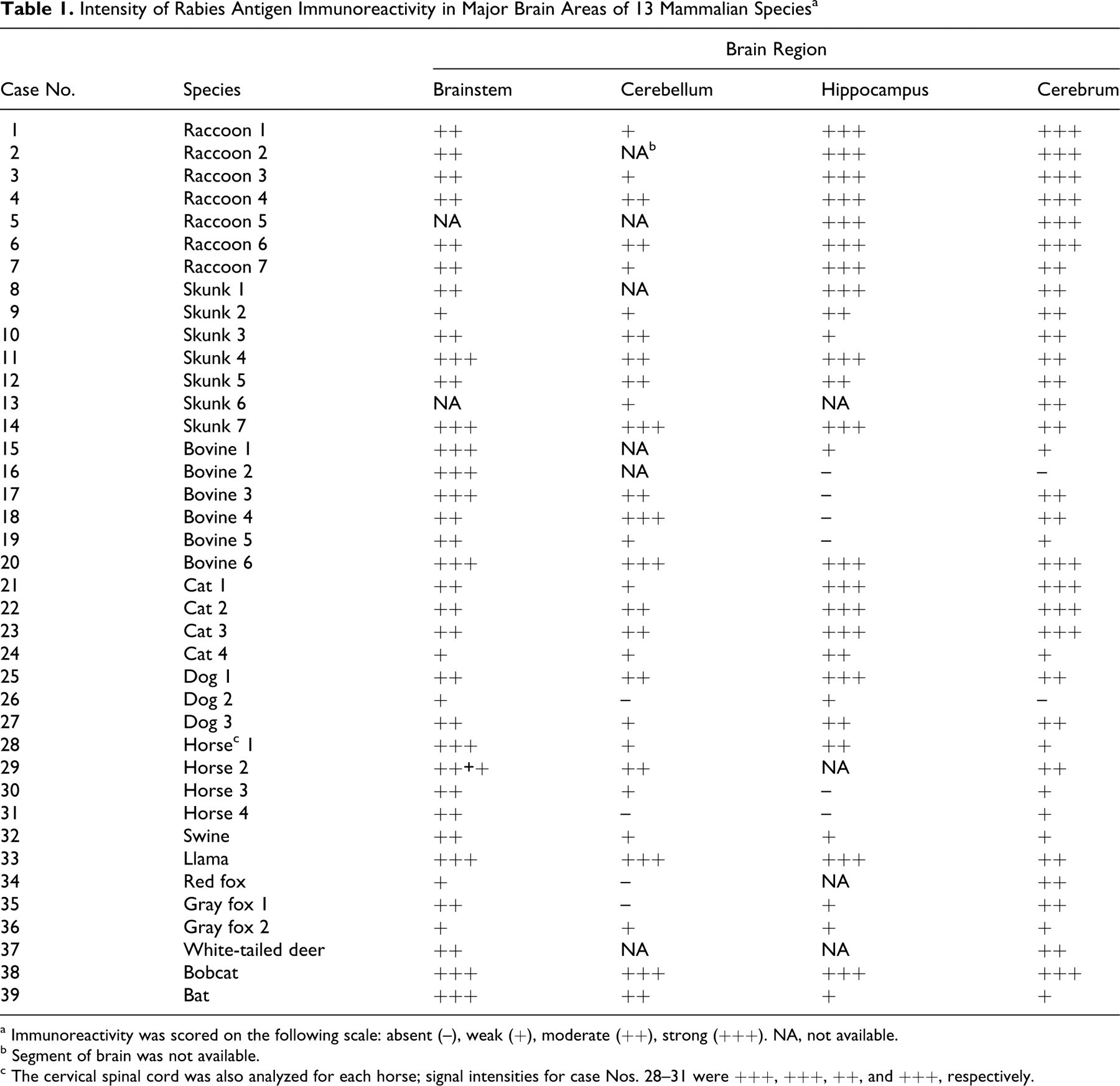
a Immunoreactivity was scored on the following scale: absent (–), weak (+), moderate (++), strong (+++). NA, not available.
b Segment of brain was not available.
c The cervical spinal cord was also analyzed for each horse; signal intensities for case Nos. 28–31 were +++, +++, ++, and +++, respectively.
Immunohistochemistry
All tissues were examined by immunohistochemistry using the following protocol to detect lyssaviral ribonucleoprotein. After deparaffinization in CitriSolv, tissue sections were quenched with 3% hydrogen peroxide and rinsed in water. The antigenic sites were exposed by microwaving in a citrate buffer solution (1X Vector Ag-Retrieval Unmasking Solution) for 10 minutes (4′-4′-3′ intervals), followed by blocking in 1:10 diluted Powerblock solution (Biogenex Universal Blocking Reagent) for 7 minutes. Primary antibody (rabies polyclonal DFA reagent, made in goat, Chemicon/Millipore, Billerica, MA) at a 1:2,000 dilution was incubated for 1 hour at 37°C followed by two 20-minute room temperature incubations: biotinylated link antibody (LSAB2 kit) and horseradish peroxidase–conjugated streptavidin (LSAB2 kit). The reaction was revealed in diaminobenzidine peroxidase substrate (Dako, Carpenteria, CA) for no longer than 5 minutes. The slides were counterstained with Mayer’s hematoxylin and coverslipped with Permount. Positive controls were a rabies FAT-positive brain. Negative controls included substituting primary antibody with PBS.
Viral Antigen Distribution
For each species, 4 anatomic locations in the brain were examined: brainstem, cerebellum, cerebrum, and hippocampus. All 4 equine cases also included cranial cervical spinal cord. The IHC reactivity was scored as follows: absent (–); weak (+), mean of 1 focus of positive signal/high-power field (hpf); moderate (++), mean, 2 to 5 foci of positive signal/hpf; and strong (+++), mean, more than 5 foci of positive signal/hpf.
Results
Immunohistochemistry results are in Table 1. Rabies virus antigen was detected in all 39 cases. The signal was observed almost exclusively in gray matter and characterized by sparse to diffuse granularity throughout the perikaryon of neurons and their axonal and dendritic processes. In some cases, inclusions appeared as brown oval homogeneous structures within the perikaryon of neurons (Fig. 1). The presence of viral antigen in the neuronal processes gave a granular appearance to the neuropil, with variable extension into axons (Fig. 2).
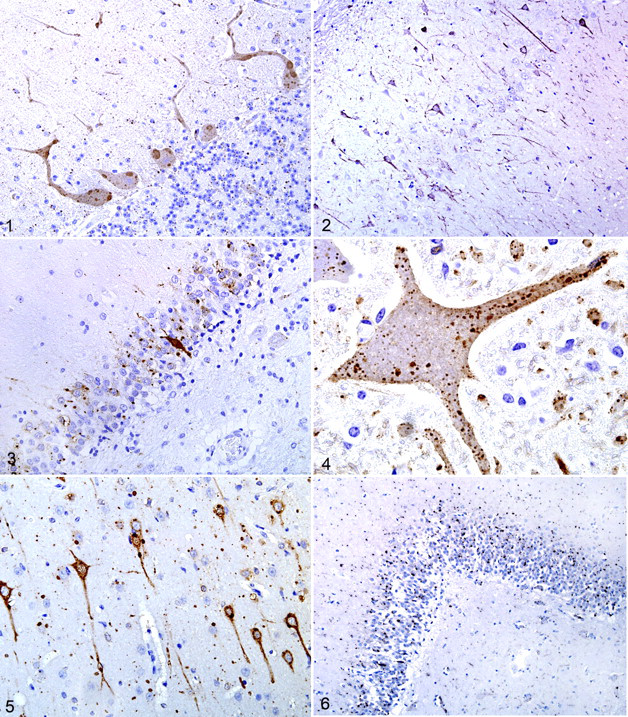
Some distributional differences of rabies antigen for each species were highlighted (Table 1). For the domestic carnivores (cats and dogs, case Nos. 21–27), results were very similar, with most intense signal in the hippocampus (Fig. 3), followed by the cerebrum, brainstem, and cerebellum in descending order of immunoreactivity.
In cattle (case Nos. 15-20), the most intense signal was in the perikaryon of large neurons of brainstem nuclei. In most cases, cytoplasmic reactivity was intense and diffuse. The cerebellum had the next most intense signal; Negri bodies in the Purkinje cells appeared as strongly labeled, oval, homogeneous structures (Fig. 1). Throughout the molecular layer of the cerebellum, dendritic processes had strong signal. There was patchy cytoplasmic labeling of neurons of the granular cell layer, including Golgi neurons.
For horses (case Nos. 28-31), the cervical spinal cord had the strongest signal, followed by the brainstem. In all 4 horses, the distribution of the viral antigen was diffuse throughout the neuraxis (Fig. 4), with sparing or even absence in sections of the hippocampus, cerebellum and cerebrum. In general, antigen granularity in the brain of horses, especially in the neuronal perikaryon, was less prominent than in the other species.
IHC was only applied to 1 pig (case No. 32), which had moderate positivity in the brainstem, with milder reactions in the other segments. The 1 llama studied (case No. 33) had intense signal in all segments.
In raccoons (P. lotor) (case Nos. 1-7) and skunks (M. mephitis) (case Nos. 8-14), IHC reactivity varied between moderate and strong in all examined segments (Fig. 5); however, in all cases, the hippocampus was the site where the signal was consistently very strong. In these 2 wildlife species, IHC of any brain segment yielded positive results.
Both red fox (V. vulpes) (case No. 34) and gray foxes (U. cinereoargenteus) (case Nos. 35 and 36) had the strongest immunoreactivity in the cerebral cortex, followed by the brainstem (Fig. 2). In the white-tailed deer (O. virginianus, case No. 37), bobcat (L. rufus, case No. 38), and bat (Myotis species, case No. 39), positive signal was detected in the available regions of the brain (Fig. 6).
Discussion
Immunohistochemistry demonstrated rabies viral antigen in all 39 cases examined. Because clinical manifestations of rabies in many species are unpredictable, the optimal rabies diagnostic test must detect viral antigen in any mammalian species. Some IHC studies for rabies only use monoclonal antibody preparations, which may not be suitable, especially if their activity depends on conformational epitopes. 19 The polyclonal antibody used in this study accurately detected rabies virus in all 13 tested species. Immunohistochemistry for rabies viral antigen detection in FFPE sections has been successfully applied in several countries, although most of the antibodies are not commercially available, 1,5,7,10,11,14,16,21 which makes introduction of the technique to other laboratories challenging. Although we did not evaluate the polyclonal antibody in cases from other parts of the world in this study, we have used it on cases of rabies in cattle in Brazil with positive results. Because the antibody is polyclonal, it should detect a wide range of rabies viral strains.
Although FAT is the standard technique for quick rabies diagnosis, it requires the use of expensive ultraviolet light microscopes 11 and fresh samples, which contain live virus and entail public health risk. 26 Transporting fresh samples is a problem in countries where diagnostic laboratories are not well established or where lack of refrigeration and high ambient temperatures can interfere with the FAT. 1,2,11 Field collection of brain samples often occurs far from diagnostic laboratories. Delays in sample collection and/or shipping in some countries may add 5 or more days from the death of the animal to the point of laboratory testing; the ensuing autolysis further hinders diagnostic accuracy. 6 In the formalin-fixed specimens used in IHC, the rabies virus is rapidly inactivated by formaldehyde, making the transport and laboratory processing of specimens much safer. 10 This is even more important when the diagnosis must be made overseas, requiring international transportation of samples with zoonotic or Office International Epizooties (OIE)−listed diseases. Recently, a new technique, known as direct immunohistochemical test (dRIT), has been evaluated under field and laboratory conditions to detect rabies virus antigen in glycerol-preserved, field-collected brain samples. 11 This technique uses touch-imprint brain samples that are further fixed in formalin with application of a monoclonal antibody. Diagnosis using the dRIT technique is rapid, with high sensitivity and specificity. 11 However, if the rabies test is negative with dRIT, the touch-imprint specimen is not suitable for additional diagnostic testing. In contrast, a major advantage of IHC is that the pathologic changes in the brain are clearly observable. Formalin fixation of brain preserves the tissue architecture and allows histologic evaluation to formulate a differential diagnosis. 10,19
Certain parts of the brain have historically been considered reliable for detecting Negri bodies or were routinely sent for FAT to confirm the diagnosis of rabies. These parts included the hippocampus in carnivores and the cerebellum in herbivores. 2,7,10,14,18,26,27 The initial diagnostic focus on hippocampus can be attributed to its high frequency of large antigen aggregates or inclusion bodies, which are easily identified by traditional histological stains. 10 The accuracy of laboratory diagnosis was inconsistent when based solely on the presence of Negri bodies, however, which are found only in 40% to 80% of rabies cases. 7 Along with such variable and inconsistent Negri body formation, some hematoxylin and eosin–stained inclusions in the cytoplasm of neurons resemble Negri bodies but might not be rabies-specific. Such pseudo-Negri bodies are protein-related inclusions and could lead to an initial false-positive diagnosis of rabies. These cytoplasmic inclusion bodies have been described in the neurons of nonrabid cats, cattle, moose, woodchucks, and skunks. 13,14,22,23,24 For these reasons, rabies diagnosis should not be based solely on the presence of Negri bodies in the hippocampus, especially in cases without inflammation, and IHC or FAT should be applied to rabies-suspect cases to confirm the presence of viral antigen.
In this study, the distribution of rabies virus antigen in the brain varied among species, demonstrating that sampling site in some species might be important. Numerous studies have analyzed the spread of rabies virus in the brain using sensitive diagnostic tools such as FAT. 2,3,17,24 A comprehensive study of multiple species using FAT concluded that when composite sampling of the brain is not possible, the thalamus should be the area of choice for testing. 2 However, considering results of our study, although supporting the collection of the whole brain, we also advocate choosing specific parts for the preliminary IHC. Knowledge of the optimal area of the brain for detection of rabies viral antigen will enhance the quality and efficiency of the diagnosis.
Dogs and cats, the 2 domestic carnivores studied, had very similar viral antigen distribution with strongest immunoreactivity in the hippocampus. Although rabies in dogs and cats has been greatly reduced in the United States, unvaccinated dogs and cats may contract rabies from wildlife and be the source of infection for humans. 6,9 Globally, most cases of rabies in humans are due to exposure to infected dogs. 7,19,21,23
In the 5 cattle examined, the brainstem proved to be the area of the brain with strongest IHC signal. Consequently, diagnostic testing should target this region along with the cerebellum, which also had moderate to strong immunoreactivity. The greatest economic and public health impact of rabies in cattle exists in Latin America. 6,17 In Brazil, it is estimated that more than 842,000 cattle deaths are caused by rabies each year, creating abundant potential for human exposure. 3,12,22
In this study, horses had the strongest immunoreactivity in the cervical spinal cord, followed by the brainstem, with a consistent positive signal present throughout the neuraxis but weak or even absent in sections of the hippocampus, cerebellum, and cerebrum. In horses, the variation in distribution of rabies viral antigen, reported from several studies using Negri body mapping or FAT, 3,12,17 emphasizes the difficulty in selecting the optimal sampling site for FAT diagnosis. Most studies used FAT, which has been shown to be less sensitive in horses than in other animal species. 3 In an FAT study of equine rabies, only 29% of samples were positive in all 4 CNS regions (cortex, hippocampus, brainstem, and medulla), 3 confirming this highly selective and limited viral distribution. Horses usually develop far fewer Negri bodies than any other species, sometimes in only 30% of cases, 4,12,17,25 perhaps paralleling the inconsistent and limited viral distribution. Many diagnosticians proclaim the cerebellum as the best tissue for FAT and detection of Negri bodies simply based on bovine rabies; this could lead to misdiagnosis in horses. 3 Given variable viral distribution and sparseness of Negri bodies in equine rabies, performing immunohistochemistry in the spinal cord and brainstem could increase diagnostic accuracy.
The application of IHC in the raccoon (P. lotor) and skunk (M. mephitis) indicated that the hippocampus is the most productive source of viral antigen in rabies. However, in these species, rabies antigen was abundant throughout the brain. In 2 domestic species (swine and llama) and some wildlife species [gray foxes (U. cinereoargenteus), red fox (V. vulpes), bat (Myotis species), white-tailed deer (O. virginianus), and bobcat (L. rufus)], only 1 or 2 cases of rabies from each species were available. Although the rabies antigen distribution in these cases cannot be assessed given the limited number of cases, this protocol proved accurate to detect rabies virus. Further IHC studies are necessary to evaluate the best sampling site for these species, but the information presented here could be used as a starting point for further assessment.
In summary, IHC for rabies detection using targeted sections of brain could enhance accurate diagnosis in various species. The public health implications of this disease warrant continued efforts to develop more accurate sampling and testing modalities. This IHC protocol provides an alternative to FAT and can be used safely, even in tropical and remote areas.
Footnotes
Acknowledgments
We thank Dr. M. K. Keel for providing the wildlife cases in this study. We also thank Jian Zhang, Kelley Gibson, and Trey Harden for valuable technical support.
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) received no financial support for the research and/or authorship of this article.
