Abstract
Glanders is caused by the gram-negative bacterium Burkholderia mallei. In this study, we investigated the histopathology and immunohistochemical localization of B. mallei in natural cases of equine glanders. Four horses showing clinical signs of nasal discharge and multiple cutaneous nodules or papulae in the hindlimbs and abdomen were reported in Mongolia. They tested positive for B. mallei infection on complement fixation, Rose Bengal agglutination, and mallein tests. Gross and histological lesions observed in these cases were similar to those previously reported in equine glanders. Immunohistochemistry using a monoclonal antibody to B. mallei BpaB showed localization of the bacterial antigen in the cytoplasm of neutrophils, macrophages, epithelioid cells, and multinucleated giant cells in the pyogranulomas and abscesses in target organs. Some alveolar type II cells and bronchiolar epithelial cells also contained the antigen. These results suggest that the anti-BpaB antibody is useful for identifying B. mallei–infected cell types in naturally infected horses.
Glanders is a fatal zoonotic disease caused by the gram-negative, immotile, aerobic rod-shaped bacterium Burkholderia mallei, a facultative intracellular bacterium. Natural B. mallei infections occur predominantly in donkeys, mules, and horses. B. mallei is a zoonotic pathogen that was used as a biological weapon during World Wars I and II, and the Russian invasion of Afghanistan. 6,7,14 Therefore, B. mallei infection, or glanders, is classified both as a category B infection by the Centers for Disease Control and Prevention and as a Tier 1 biological select agents and toxins by the US Department of Health and Human Services and the US Department of Agriculture. Glanders has been eradicated from many Western countries. However, it is still endemic in the Middle East, Asia, Africa, and South America. Additionally, outbreaks of glanders have recently been reported in Turkey, 2 Brazil, 4 Iran, 6 India, 7 Pakistan, 9 Bahrain, 12 and Iraq. 14
The major clinical signs of glanders in horses are nasal discharge and skin nodules. The gross and histologic lesions of equine glanders have been reported previously from many countries. However, limited information is available on the in situ localization of B. mallei in naturally occurring glanders in horses.
The 4 cases of glanders analyzed in this study occurred in Arabian horses in 2018 in Mongolia. Cases 1 and 2 were from a small farm in the Ulziit district of Töv province, and cases 3 and 4 were from a small farm in the Nalaikh district of Ulaanbaator City. Cases 1 and 4 were female, and cases 2 and 3 were male. The cases had nasal discharge and multiple cutaneous nodules on the hindlimbs and abdomen. Horse owners detected nasal discharge (cases 2, 3, and 4) and skin nodules (cases 1 and 2) for approximately 5 months before euthanasia. These horses had been imported from Russia 3 years before at the age of 2 to 5 years. Other native Mongolian horses in the herds did not show clinical signs. The 4 glanderous horses tested positive for B. mallei in complement fixation, Rose Bengal plate agglutination, and mallein tests.
The infected horses were euthanized and necropsied at the Institute of Veterinary Medicine in Ulaanbaatar, Mongolia. Tissue samples were collected from the mucous membranes of the nasal cavity (septum and conchae), lung, liver, spleen, kidney, inguinal lymph nodes, axillary lymph nodes, mediastinal lymph nodes, heart, and skin. These were fixed in 10% neutral-buffered formalin and then embedded in paraffin. Three-micrometer-thick sections were stained with hematoxylin and eosin for histopathological examination. Serial sections from the skin, nasal cartilage, mediastinal lymph nodes, and lungs were stained with phosphotungstic acid-hematoxylin to demonstrate fibrin. The slides were reviewed by 2 veterinary pathologists.
Indirect immunofluorescence staining was performed using the labeled streptavidin-biotin (SAB) technique (Histofine SAB-PO Kit, Nichirei). The sections were dewaxed and heated with 0.01 M citric acid buffer (pH 6.0) in a microwave for antigen retrieval. Subsequently, they were treated with 0.3% hydrogen peroxide in methanol and blocked with 10% normal rabbit serum. Anti–B. mallei BpaB antibody (clone BpaB#4; Kerafast, diluted 1:100 in phosphate-buffered saline [PBS]) was added, and the sections were incubated for 18 hours at 4 °C. These sections were washed with PBS and incubated with biotinylated rabbit anti-mouse immunoglobulin (Histofine SAB-PO Kit, Nichirei) for 20 minutes at room temperature. After further washing with PBS, the sections were incubated with streptavidin-conjugated peroxidase for 10 minutes at room temperature. The bound peroxidase was detected with 3,3′-diaminobenzidine (200 µg in Tris-HCl buffer [TBS], pH 7.6). The sections were counterstained with hematoxylin. As a negative control, sections were incubated without primary antibody.
At necropsy, all 4 horses were severely emaciated. They had moderate bilateral nasal discharge (purulent or hemorrhagic) with 0.5- to 1.0-cm-diameter round ulcerated nodules on the nasal mucosa. Two horses (cases 1 and 2) had multiple small cutaneous lesions on the stifle, thigh, gaskin, and hock of the medial hindlimbs as well as the ventral abdomen, which occasionally erupted to form 1- to 2-cm diameter yellowish nodules (Fig. 1). Numerous gray, hard, approximately 0.3-cm-diameter miliary nodules were present on the surface of the cranial, caudal, and accessory lobes of the lungs in all horses. Additionally, multifocal to coalescing, yellowish nodules surrounded by tan-white fibrous tissues (Fig. 2) were frequently found in the parenchyma of the lungs of all horses.
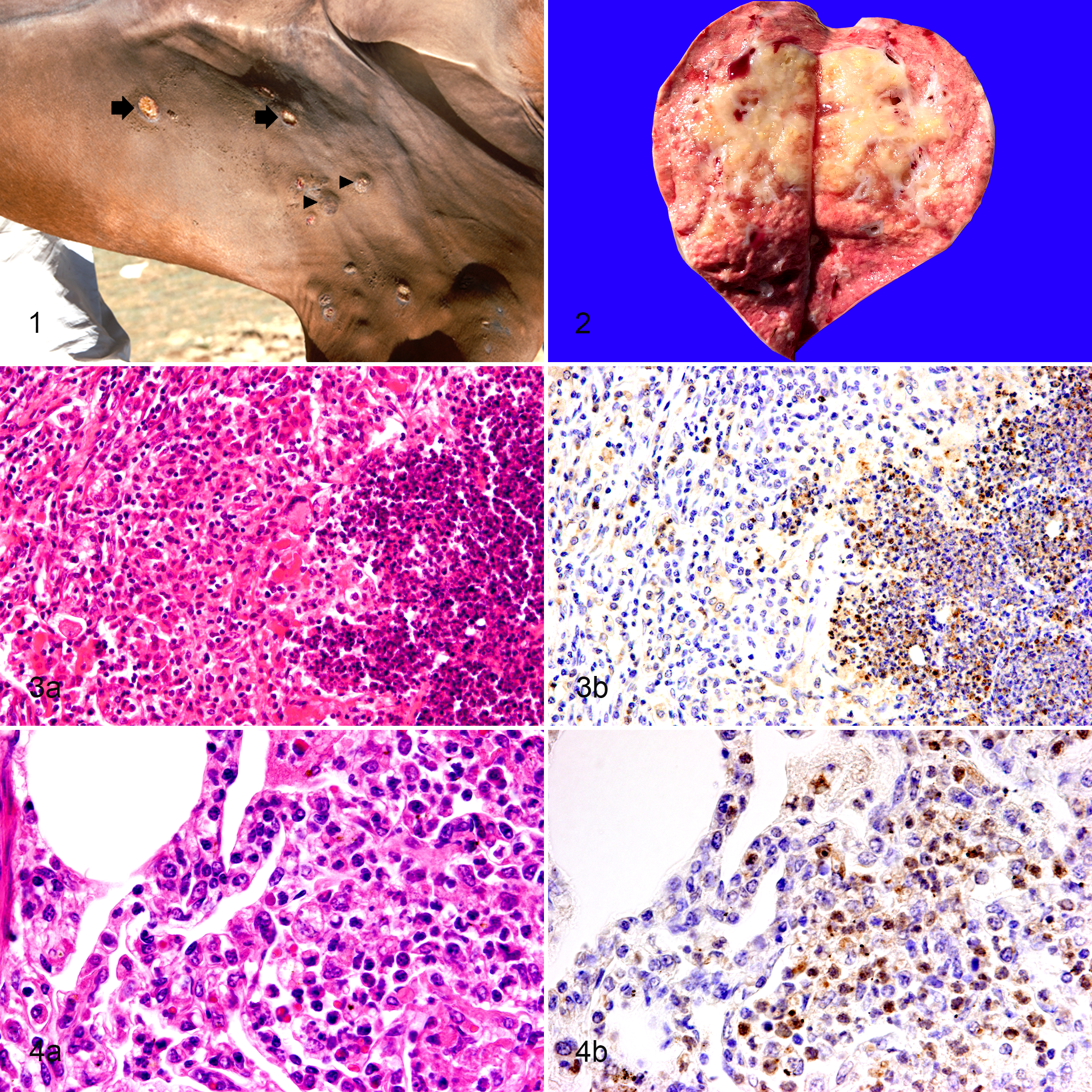
Glanders, horse.
Histologically, multiple pyogranulomas (Fig. 3a) and small abscesses (Fig. 4a) were frequently found in the lungs in all cases. In case 3, severe infiltration of neutrophils, macrophages, and lymphocytes in the lumen of bronchi and bronchioles was observed multifocally. In the other 3 horses, however, the bronchi and bronchioles were spared.
Pyogranulomas and small abscesses were sometimes observed in the liver (cases 1 and 2) and mediastinal lymph nodes (case 4), but their frequency was lower than that in the lung. Skin nodules in cases 1 and 2 consisted of dermal and subcutaneous abscesses (Supplement Figure S1); they were also occasionally ulcerated. Severe diffuse necrosis with multifocal ulceration was observed in the nasal mucosa of cases 2 and 4, associated with submucosal abscesses and cartilage destruction (Supplement Fig. S2). Fibrin exudation was frequently observed around the pyogranuloma in the lungs (Supplement Figs. S3 and S4), mediastinal lymph nodes, nasal mucosa, and skin, but not in the liver. No gross or histologic lesions were observed in the spleen, kidney, inguinal lymph nodes, axillary lymph nodes, or heart.
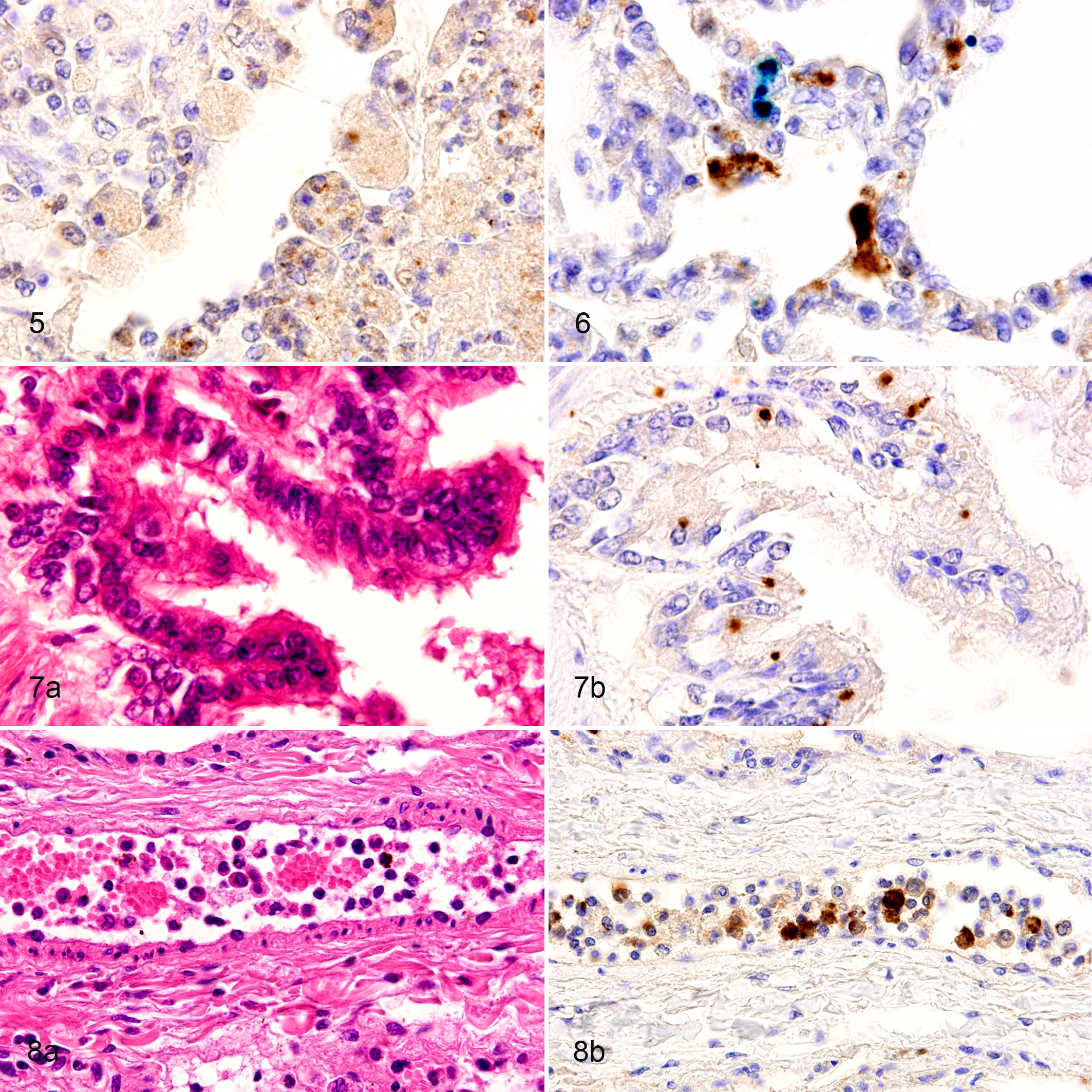
Immunohistochemistry (Table 1) revealed B. mallei BpaB antigen in the cytoplasm of neutrophils at the center of the pyogranulomas (Fig. 3b) and in small abscesses (Fig. 4b) in the lung of all horses, in the liver of 2 horses (cases 1 and 2), and in the mediastinal lymph nodes of 1 horse (case 4). Macrophages, epithelioid macrophages, and multinucleated giant cells within the pyogranuloma also contained the antigen (Fig. 5). Furthermore, B. mallei antigen was detected in the alveolar cells, morphologically suggestive of type II pneumocytes; these were distinguished from hemosiderin-laden macrophages by Berlin blue staining (Fig. 6).
Localization of Burkholderia mallei BpaB Antigen Using Immunohistochemistry in Horses With Glanders.
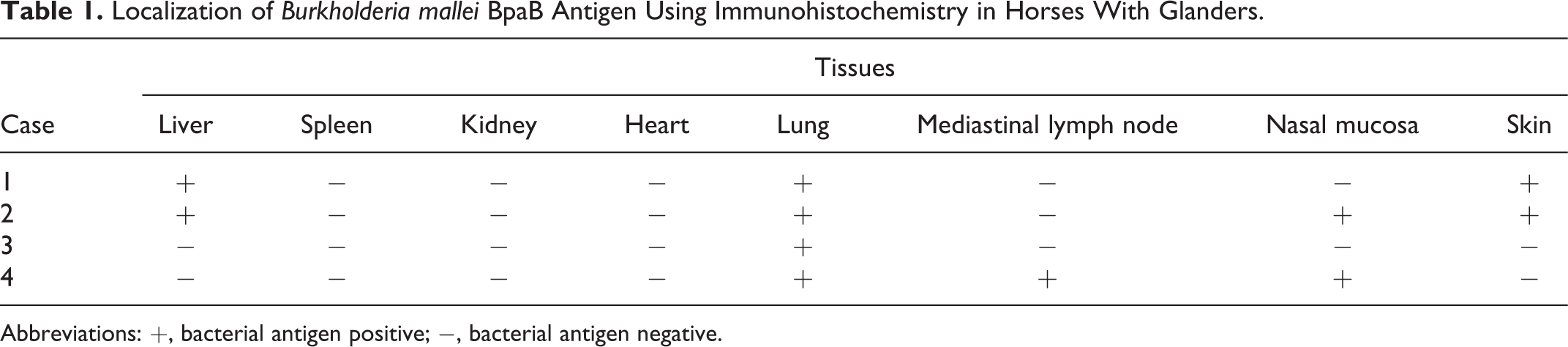
Abbreviations: +, bacterial antigen positive; −, bacterial antigen negative.
Glanders, lung, horses.
Punctate positive signals were occasionally detected in the cytoplasm of bronchiolar epithelial cells (Fig. 7a and b). In case 3, infiltration of B. mallei antigen-positive neutrophils and macrophages was observed in the lumen of bronchi and the bronchioles. In case 4, intravascular monocytes and neutrophils located in the pulmonary blood vessels were also positive for B. mallei antigen (Fig. 8a and b).
The B. mallei BpaB antigen was also detected in the cytoplasm of neutrophils in the dermal and subcutaneous abscesses in the skin (cases 1 and 2) and in the submucosal abscesses in the nasal mucosa (cases 2 and 4). BpaB antigen was not detected in the spleen, kidney, inguinal lymph nodes, axillary lymph nodes, or heart in any of the cases.
In this study, we have described the histopathological and immunohistochemical findings observed in 4 cases of naturally occurring equine glanders in Mongolia. Clinical, gross, and histological findings in the 4 glanderous horses are similar to those previously reported in chronic cases of equine glanders, 1,2,4,8 except for marked fibrin exudation surrounding the pyogranulomas and abscesses. Relatively active bacterial proliferation may correlate with increased vascular permeability and the resultant fibrin exudation and severe neutrophilic infiltration. Few studies have been published using immunohistochemistry for examining bacterial localization in natural cases of equine glanders. We used monoclonal antibody against the BpaB antigen, which is expressed on the surface of B. mallei bacteria. 5 Consistent with immunohistochemistry for BpaB in B. mallei–infected mice and marmosets, 13 BpaB was detected within macrophages, neutrophils, and respiratory epithelial cells of naturally infected glanderous horses. Localization of BpaB antigen-positive neutrophils and macrophages was well correlated with the localization of pyogranulomas and abscesses in the tissues.
Although the mode of in vivo dissemination of B. mallei is not fully understood, it is assumed that the bacteria can traverse the pharyngeal mucosa, and perhaps the intestinal mucosa after oral infection. 3,10,11 Subsequently, the bacteria are seeded into the internal organs, particularly the lung, where lesions almost always occur. 3,13 In the present study, one of the 4 horses (case 3) showed bronchitis and bronchiolitis. These characteristics of the infiltration of BpaB-positive neutrophils and macrophages in the airway lumen may indicate bacterial spread through the airway within the lung. However, transbronchial spread does not appear to be necessary for bacterial dissemination. The other 3 cases did not show bronchitis or bronchiolitis. The localization of pyogranulomas and abscesses did not coincide with that of bronchi or bronchioles.
Our results indicate that anti–B. mallei BpaB monoclonal antibody detects B. mallei antigen in formalin-fixed paraffin-embedded tissues from the field cases of equine glanders. Thus, the antibody is useful for pathological analyses.
Supplemental Material
Combined_supplemental_materials-Erdemsurakh_et_al - Pathological and Immunohistochemical Analyses of Naturally Occurring Equine Glanders Using an Anti-BpaB Antibody
Combined_supplemental_materials-Erdemsurakh_et_al for Pathological and Immunohistochemical Analyses of Naturally Occurring Equine Glanders Using an Anti-BpaB Antibody by Ochbayar Erdemsurakh, Baatarjargal Purevdorj, Khurtsbaatar Ochirbat, Altanchimeg Adilbish, Batbaatar Vanaabaatar, Keisuke Aoshima, Atsushi Kobayashi and Takashi Kimura in Veterinary Pathology
Footnotes
Acknowledgements
We deeply appreciate the Mongolian veterinarians who extended support during field activity of this research. We thank Dr Takashi Umemura (chief advisor of the JICA project) for his support and advice.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by the Livestock Promotional Subsidy from the Japan Racing Association.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
