Abstract
Causes of canine juvenile hydrocephalus have been well documented. However, the effects of hydrocephalus on periventricular white matter have been only partially described. The present report shows that hydrocephalus-associated lesions of the periventricular white matter, i.e., formation of diverticula, clefts, and tears, are prevalent. Marked hydrocephalus was identified at necropsy in 20 juvenile dogs between 1990 and 1999. The severity grade was based upon a ratio of lateral ventricular dimensions to cortical thickness. All animals exhibited ependymal lesions consisting of attenuation, with or without abortive attempts of ependymal regeneration, and ulceration. In 10 dogs (50%), unilateral or bilateral periventricular diverticula and cleft formation in the region of the caudate nucleus were observed. The diverticula were formed at the caudal pole of the caudate nucleus, communicated with the ventricular lumen, and were associated with ependymal denudation. Loss of the ependymal lining probably contributes to a bulk shift of cerebrospinal fluid from the ventricular lumen to the periventricular white matter, leading to diverticulum formation. Clefts were observed within the parenchyma at the border of the internal capsule and putamen, consistent with an ischemic insult. Occasionally tearing with separation of the caudate nucleus from the subcortical white matter was found, representing unification of expanding clefts and diverticula. In one of the few clinically well-documented cases, tearing was correlated with a sudden decline in neurologic function, culminating in euthanasia. However, tears and clefts may exhibit a chronicity of several days, as indicated by the presence of astroglial scars along the lesion margins.
Hydrocephalus refers to an increase in volume of cerebrospinal fluid (CSF) in the ventricular system of the brain and is a common finding in dogs. 14 Most commonly, hydrocephalus is caused by impaired CSF circulation through the mesencephalic aqueduct or lateral apertures of the fourth ventricle. 14 Hydrocephalus may result in compression atrophy of the surrounding nervous tissue parenchyma and the generation of diverticula in the periventricular white matter of the brain. The generation of these diverticula has been attributed to inflammation due to bacterial, parasitic, or viral infection. 1–3, 7 15 Here, inflammation is thought to disrupt a proposed ependymal barrier to CSF movement into periventricular tissue or periventricular cysts form in response to inflammation with subsequent extension to the dilated ventricle.
Here, we report a series of cases with a common pattern of hydrocephalus. These cases are associated with compression atrophy of the cerebral cortex and a high incidence of diverticula formation at the margins of the caudate nucleus, cleft formation in the internal capsule and ultimately tearing of the internal capsule. However, unlike in previously reported cases, the incidence and severity of periventricular encephalitis were minor. Inflammation apparently is not required for the development of periventricular diverticula, but rather ependymal loss and periventricular ischemic damage are fundamental to the pathogenesis of these changes.
Materials and Methods
Twenty cases of marked hydrocephalus in juvenile dogs were identified in the necropsy accessions of the Applied Veterinary Pathology section of the Ohio State University between 1990 and 1999. An animal was classified as juvenile if it was less than 12 months of age, regardless of breed, and exhibited sexual and/or skeletal immaturity. The dorsal aspect of the calvarium was removed, followed by in situ immersion fixation of brain in 10% neutral buffered formalin. Brain tissue from all cases was fixed in this manner for a minimum of 1 week at room temperature. For gross evaluation of ventricular and periventricular changes, the dorsal aspect of the cerebral hemispheres was removed. Hydrocephalus was defined as marked when the width of the lateral ventricle at the level of the pars centralis was greater than the thickness of adjacent cortical tissue by a factor of ≥2 when measured in a horizontal plane. For histologic evaluation, brain tissue was embedded in paraffin using standard methods, and 5-µm sections were cut, mounted on glass slides, and stained with hematoxylin and eosin (HE).
Results
Of the 20 juvenile dogs with marked hydrocephalus, 10 were female and nine were male. In one dog, the sex was not recorded (Table 1). All dogs were ≤8 months of age (Table 1). Eighteen of the 20 dogs were euthanatized; the remaining two dogs died spontaneously.
Sex, age, breed, prevalence of lesions with disruption of the periventricular white matter (PWML), and periventricular encephalitis in 20 juvenile dogs with marked hydrocephalus.
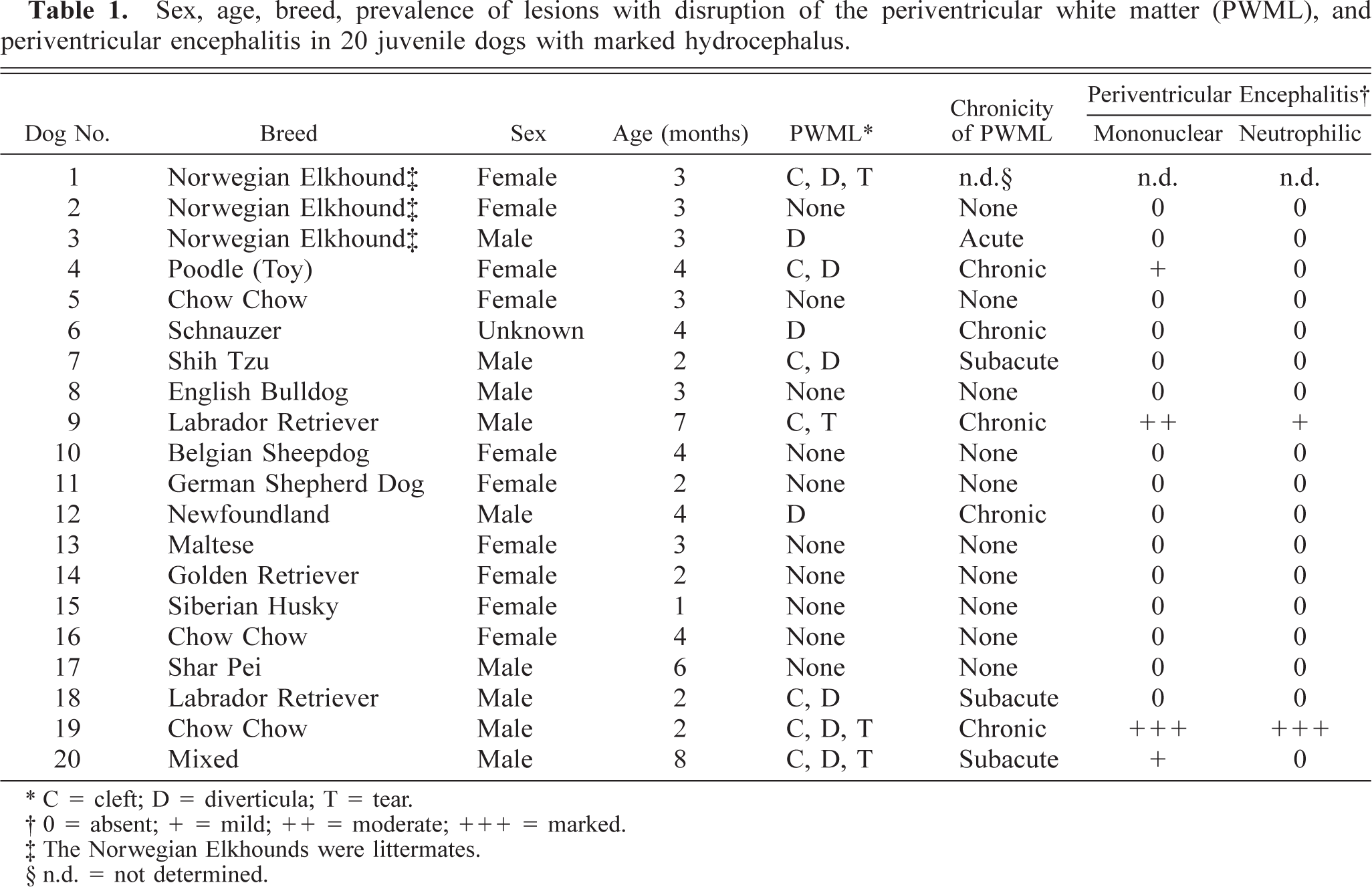
C = cleft; D = diverticula; T = tear.
0 = absent; + = mild; ++ = moderate; +++ = marked.
The Norwegian Elkhounds were littermates.
n.d. = not determined.
Gross neuropathologic findings
In all dogs, marked dilation of the lateral ventricles was associated with loss of the septum pellucidum, marked atrophy of the corpus callosum, and marked atrophy of the periventricular white and gray matter. The parietal cerebral cortex was attenuated to a width of <1 cm (Figs. 1, 2A). In 17 dogs (Nos. 1–8, 10–13, 16–20), both lateral ventricles were dilated, and two dogs (Nos. 14, 15) had unilateral hydrocephalus. The hydrocephalus was attributed to stenosis of either the mesencephalic aqueduct (dog Nos. 1–8, 10–13, 16–20) or the interventricular foramen (dog Nos. 14, 15). In one dog (No. 9), the entire ventricular system of the brain and the central canal of the spinal cord were dilated, consistent with either a stenosis of the lateral apertures or a communicating hydrocephalus.
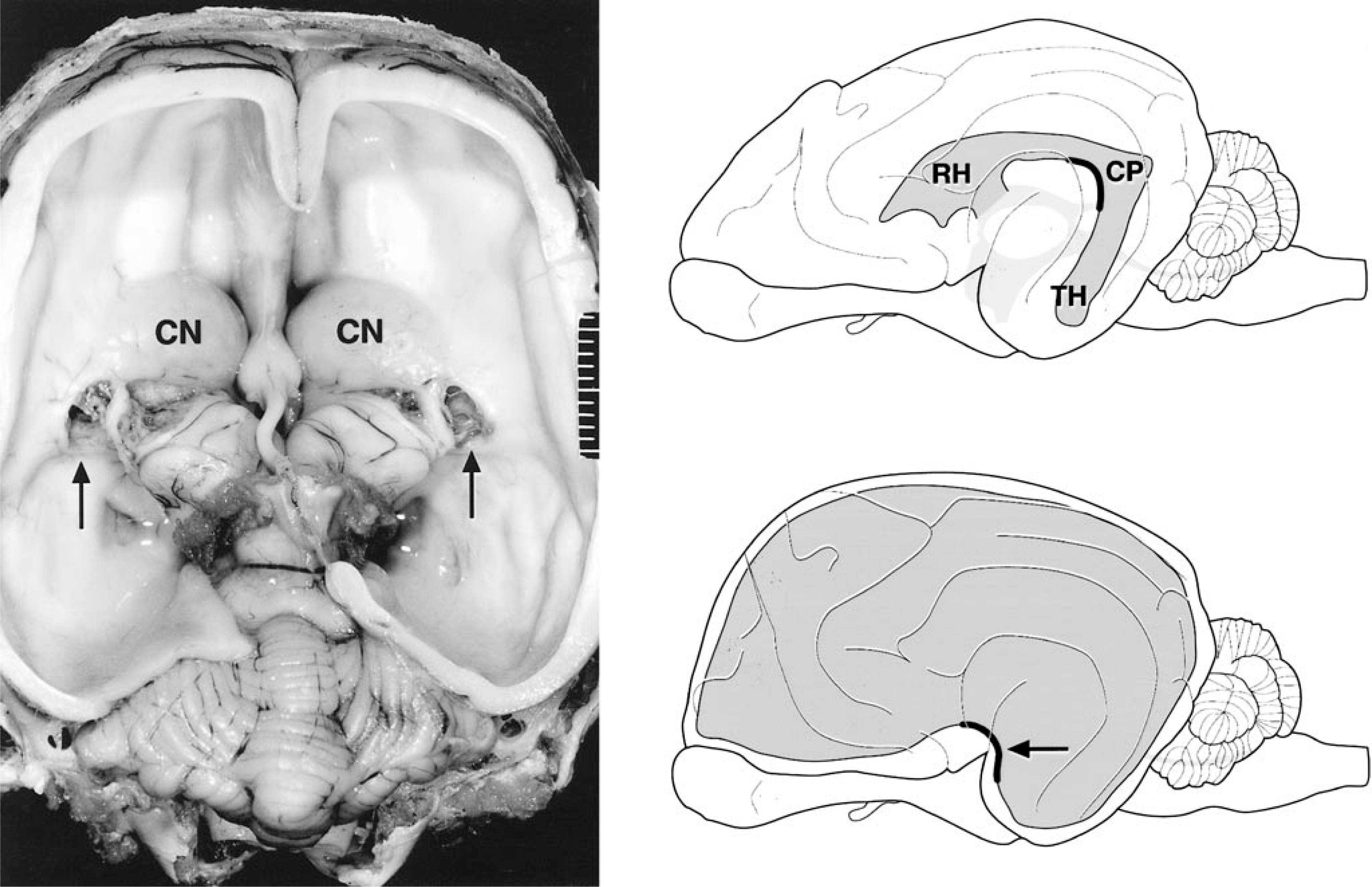
Left. Brain (vault and cerebral cortex removed); dog No. 4. Marked internal hydrocephalus with bilateral diverticula (arrows) at the caudolateral aspect of the caudate nucleus (CN). Formalin fixation. Right. Schematic illustration of the ventricular system of the canine brain with special emphasis on the lateral ventricles. Top right. Normal anatomy of the lateral ventricle with rostral horn (RH, cornu rostralis), central part (CP, pars centralis), and temporal horn (TH, cornu temporale). The concave arch of the central part is highlighted. Bottom right. Brain with marked dilation of the lateral ventricle (internal hydrocephalus). The arrow at the concave arch of the central part is indicating the point of diverticula formation.
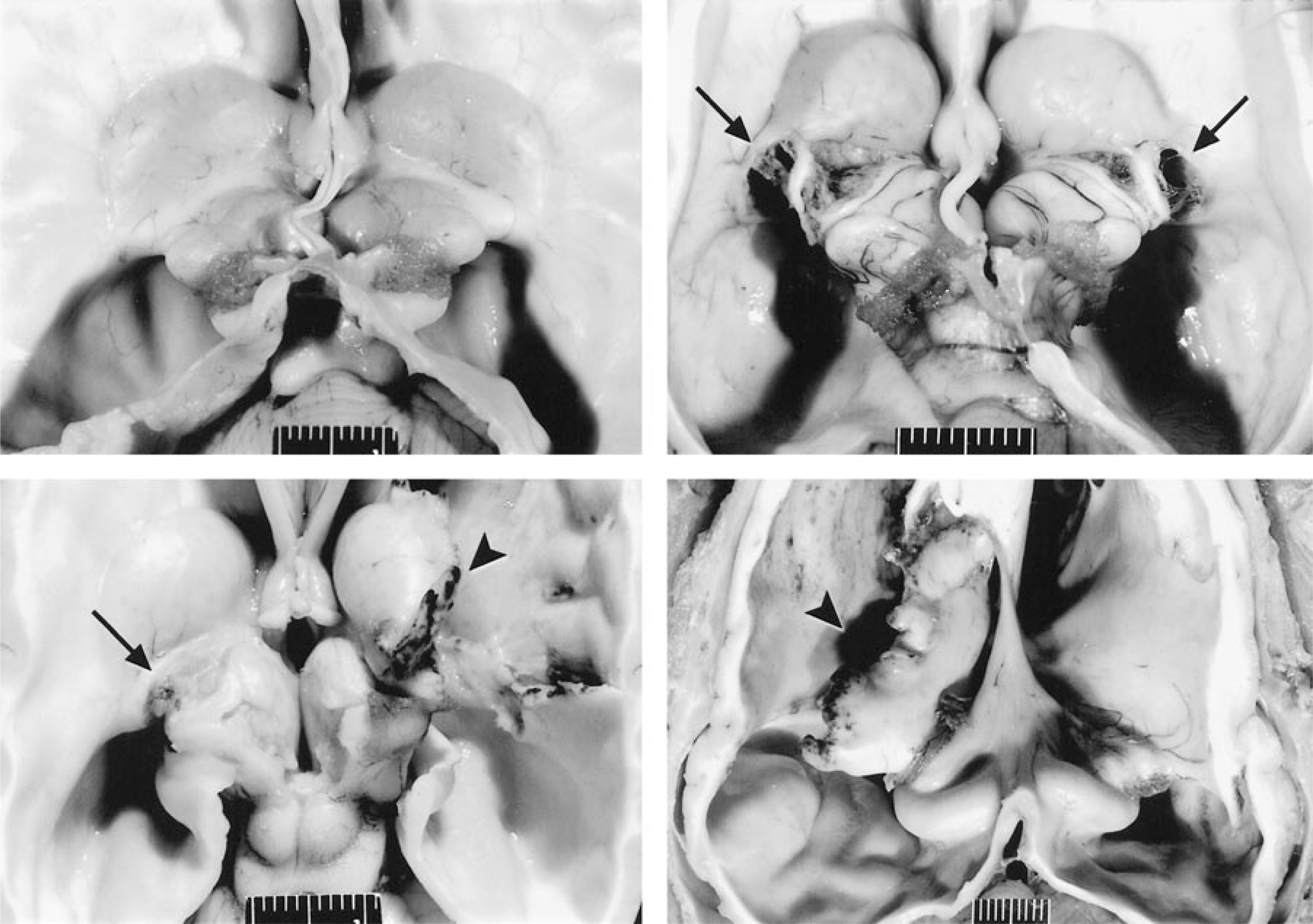
Brain (vault and cerebral cortex removed); dog. Marked hydrocephalus without and with different types of disrupting periventricular white matter lesions. Formalin fixation.
Three types of lesions characterized by disruption of the periventricular white matter were found in 50% of the affected animals, namely diverticulum and cleft formation with or without tearing. In six dogs (Nos. 1, 4, 7, 18–20), unilateral or bilateral diverticula of the periventricular white matter adjacent to the caudal pole of the caudate nucleus were found. The diverticula were located at the central part of the lateral ventricle at the caudolateral aspect of the caudate nucleus adjacent to the thalamus and hippocampus (Figs. 1, 2B). Diverticula extended inwards from the lateral ventricle and penetrated the periventricular white matter for a distance of up to 5 mm. In five dogs (Nos. 1, 4, 18–20), unilateral or bilateral cleftlike separations of the internal capsule and the putamen were present (Fig. 3). The clefts were not connected to the diverticula. In four dogs, unilateral tearing consistent with communication of cleft and diverticulum resulted in partial or total separation of the caudate nucleus from the underlying internal capsule. Three dogs (Nos. 1, 19, 20) with unilateral tearing had diverticula and cleft formation on the contralateral side (Figs. 2C, 3). In one dog (No. 9), the tear extended dorsally into the cerebral cortical white matter, including the marginal and coronal gyrus of the dorsal parietal and occipital cerebral cortex. In two dogs (Nos. 9, 19), the margins of the tears were rounded and bulged (Fig. 2D). Rounding of tear margins was correlated with histologic evidence of extensive astroglial scarring.
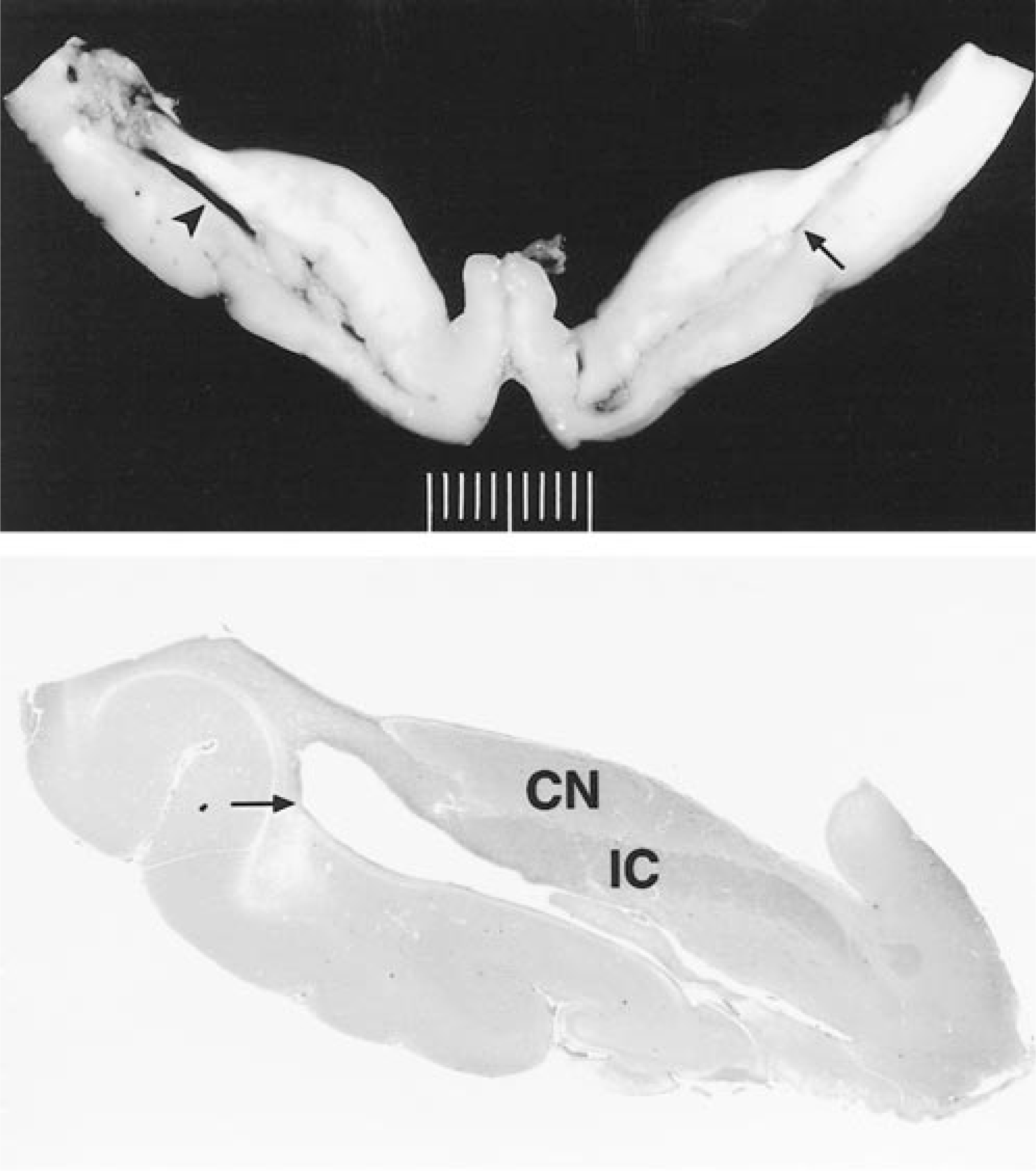
Brain; dog. Cross sections showing marked internal hydrocephalus. Top. Dog No. 19. Unilateral cleft formation (arrow) and contralateral tear (arrowhead). The tear in the internal capsule is characterized by communication between the lateral ventricle and the former cleft. The tissue bordering the dorsolateral aspect of the tear is necrotic. Formalin fixation. Bottom. Dog No. 18. Subgross illustration of the anatomic localization of a cleft (arrow); caudate nucleus (CN) relative to the internal capsule (IC). The cleft forms at the transition of the internal capsule to the putamen.
Histologic findings
Except for one dog (No. 1), all dogs were examined histologically. In all dogs, attenuation and segmental loss of the ependymal lining with mild diffuse subependymal astrogliosis were present. These changes were pronounced over the caudate nucleus and its margins. Occasionally, focal ependymal rosettes were associated with areas of ependymal attenuation and loss, consistent with an abortive regenerative response (Fig. 4A). The epithelium of the choroid plexus of the lateral ventricles was atrophied. Active axonal degeneration within the subependymal white matter and neuronal necrosis were rarely detected. Diverticula were localized to areas of ependymal denudation, extending from the ventricles into the periventricular white matter (Fig. 4B). In three dogs (Nos. 3, 6, 12), diverticula were detected histologically that were not grossly apparent. Parenchymal hemorrhage, hemosiderosis, and endothelial hypertrophy were present along the margins of the diverticula, and vascular thrombosis, vascular necrosis, and necrosis of the parenchyma could be observed at the leading edge (dog Nos. 6, 7, 20). A minimal neutrophilic response was observed in areas of more extensive parenchymal necrosis. A mild lymphoplasmacytic inflammation was associated with the diverticula in one dog (No. 4) where proliferative astroglial processes lined the margins. A similar astroglial response was observed in two additional dogs (Nos. 7, 18), indicating a prolonged course. The presence of an astroglial lining was confirmed by immunohistochemical staining of glial fibrillary acidic protein (data not shown).
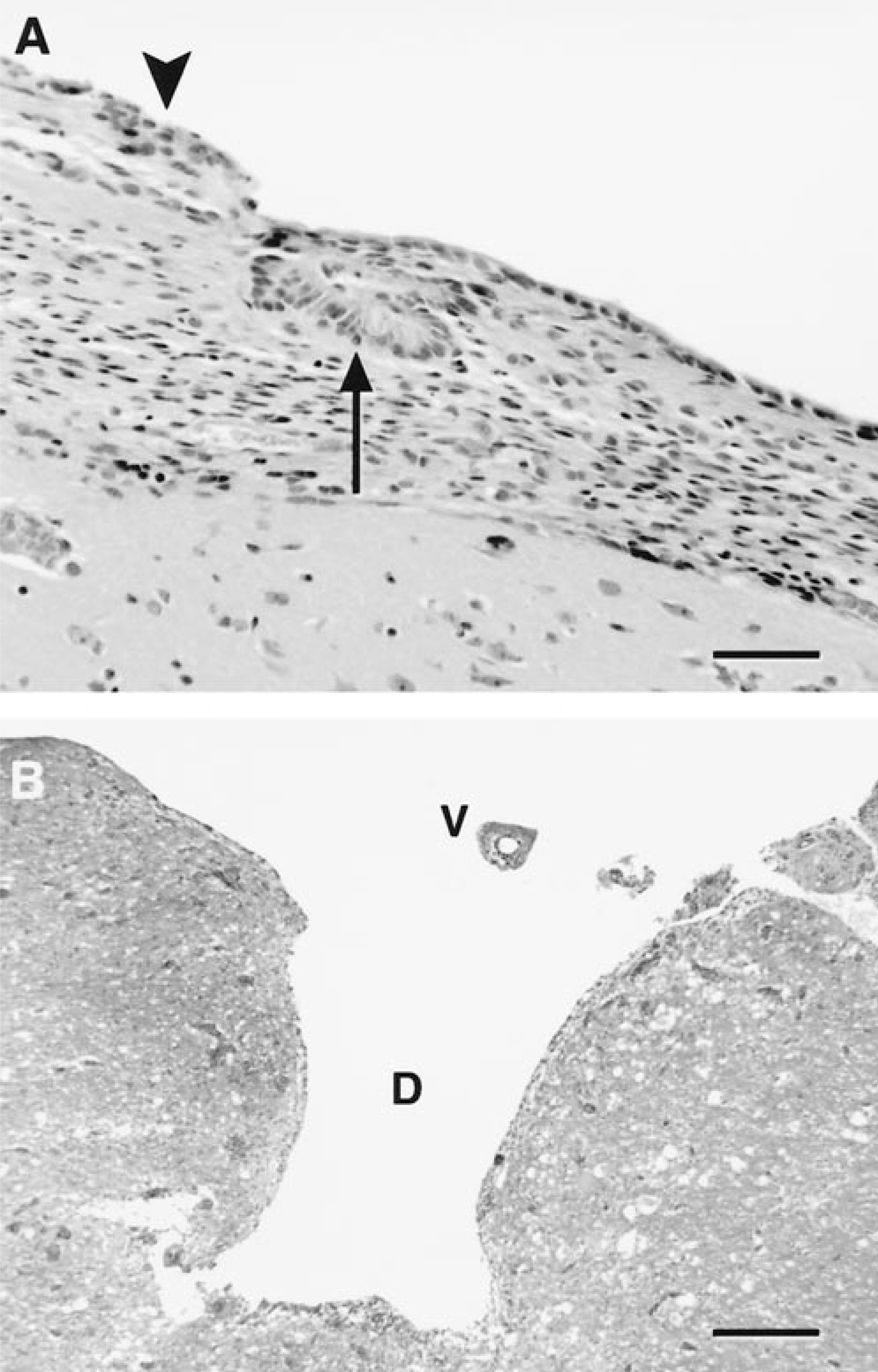
Brain; dog. Ependymal changes associated with diverticulum formation.
Isolated clefts were present at the border of the internal capsule and putamen (dog Nos. 4, 7, 18; Fig. 3). The cleft of one dog (No. 7) was seen only microscopically. Tissue surrounding clefts exhibited hemorrhage, endothelial hypertrophy, axonal swelling, axonophagia, and mild microglial activation (Fig. 5). In addition, astroglial hypertrophy and hyperplasia characterized by the occurrence of protoplasmic and fibrous astrocytes were found. Astroglial processes segmentally bordered the clefts in two dogs. Occasional small and intact vessels traversed the clefts.
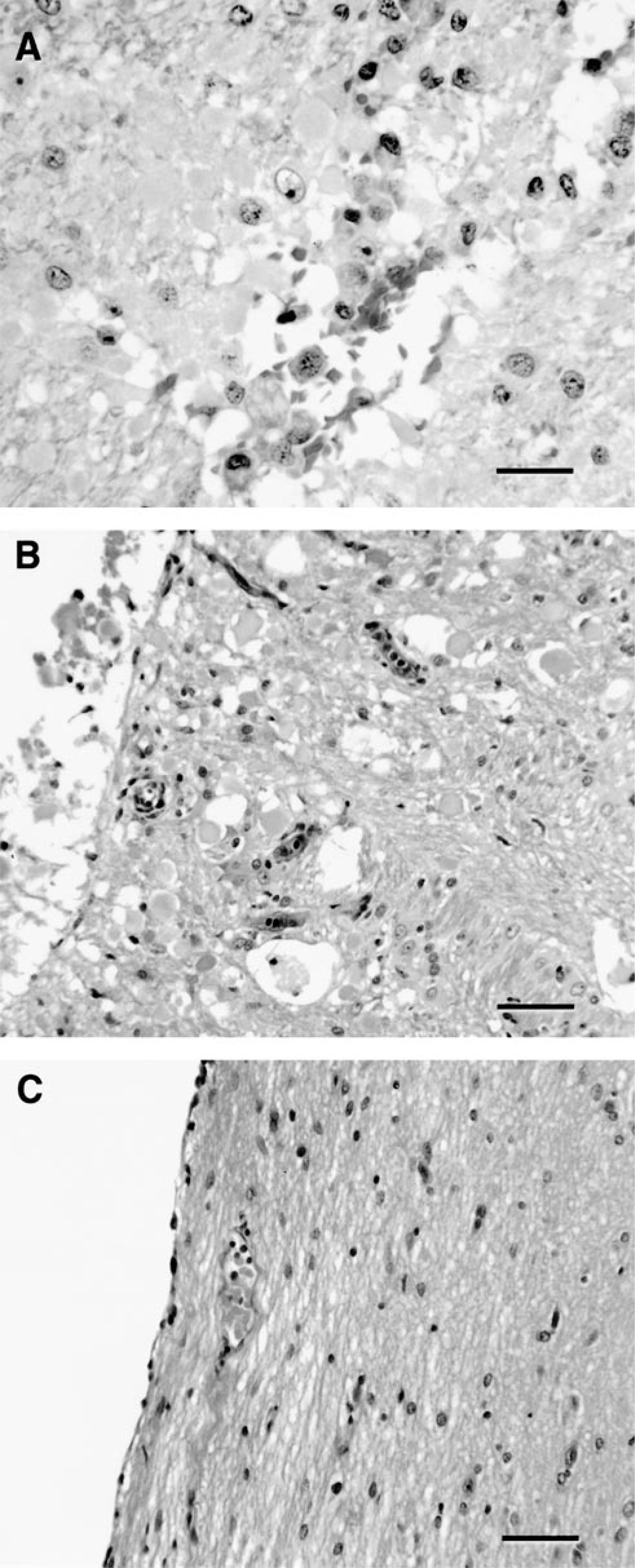
Brain; dog. Different stages of cleft formation.
Tears extended far into the internal capsule and separated the caudate nucleus from the underlying putamen. There was marked hemorrhage, vascular necrosis and thrombosis, and parenchymal necrosis of the surrounding tissue with abundant gitter cells. These areas of malacia were most extensive in tissue most isolated from the leading margin of the tear, being conspicuous in tissues from a dog where the tear extended dorsally (dog No. 9; Fig. 6). Reendothelization of thrombi and vascular proliferation were indicators of chronicity in this dog. Vascular endothelial hypertrophy and neovascularization were also present along the tear margin in dog No. 19, together with fibrous astrogliosis. Although one dog (No. 19) had marked neutrophilic and lymphoplasmacytic periventricular encephalitis, the other dogs (Nos. 9, 20) exhibiting tearing had only mild to moderate lymphoplasmacytic inflammation, with only few neutrophils restricted to the areas of active necrosis (dog No. 9).
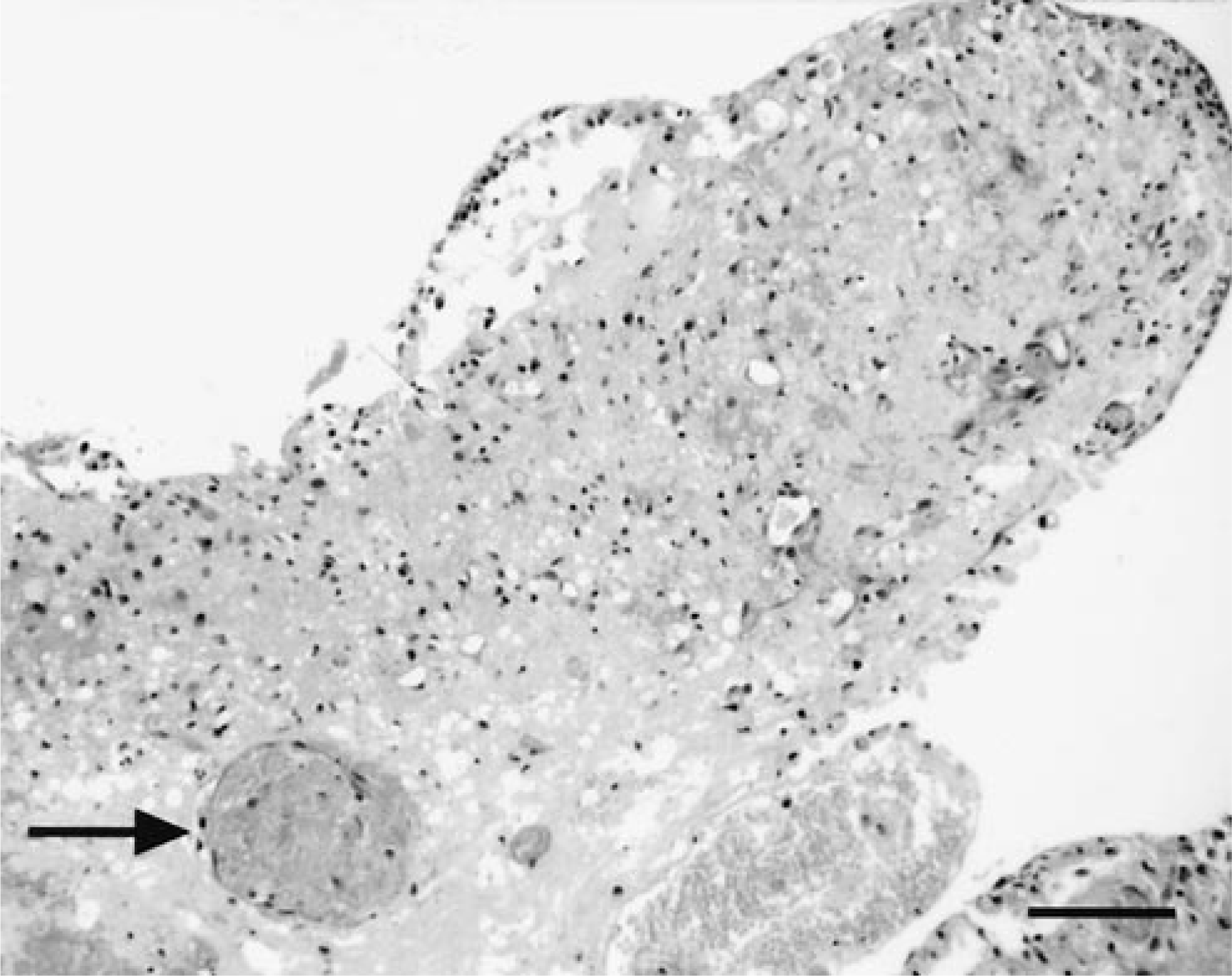
Brain; dog No. 9. Infarcted tissue fold of the cerebral cortical white matter with vascular thrombosis (arrow) after separation from the overlying cortical tissue. HE. Bar = 80 µm.
Syringomyelia was present in the cervical (dog Nos. 9, 19) and lumbar (dog No. 9) spinal cord segments in two of the three dogs with tearing that were investigated histologically. In these cases, a dilated central canal communicated with a nonependymal lined tract extending into the dorsal funiculi.
Discussion
A total of 20 juvenile dogs with marked hydrocephalus were identified in the present retrospective study. Hydrocephalus was attributed in the majority of cases to congenital CSF outflow obstructions, which is a common cause of hydrocephalus in young dogs. 14 The dog with generalized ventricular enlargement (i.e., lateral, third, and fourth ventricular involvement) and cervical and lumbar syringomyelia was described in detail elsewhere. 11 Although this animal was described as having a communicating hydrocephalus, where malresorption of CSF into the cerebral venous system is postulated, defects in the lateral apertures as a cause of internal hydrocephalus cannot be excluded. 10 The percentage of cases with periventricular diverticula, clefts, and tears is high (50%), exceeding the incidence of such changes reported in the literature.
Progressive cerebral cortical atrophy leads to a progressive enlargement of the lateral ventricles. Thus, ependymal attenuation and ulceration in hydrocephalus likely represent an attempt of ependymal cells, cells known to have a limited proliferative capacity, to cover a progressively enlarging surface. 12 Although the complete range of ependymal functions in postnatal life are currently under debate, damage may result in loss of barrier functions or impaired secretory or absorptive capacity, resulting in fluid shift from the ventricles into the periventricular white matter. This shift in CSF may appear initially as periventricular (i.e., transependymal) edema, ultimately giving rise to formation of diverticula. 4 12 The concept of a barrier function of ependyma is supported by the present finding of diverticula restricted to locations of compromised ependymal integrity. Initial formation of diverticula may be caused by repetitive increases in hydrostatic intracranial pressure during systole or paroxysmal increases during coughing, events proposed in the pathogenesis of syringomyelia. 6 The fact that two of the three hydrocephalic dogs with periventricular tearing, the most severe expression of periventricular regressive changes, also had syringomyelia supports the concept of a common pathogenesis.
The pathogenesis of cleft formation likely involves increased intracranial pressure with stretching and compression of periventricular parenchyma, causing regional reductions in venous blood flow and ultimately resulting in infarction of tissue between the internal capsule and putamen. 5 8 9 A reduction of capillary density and caliber has been reported in the caudate nucleus and internal capsule of dogs with hydrocephalus. 13 Progressive expansion of diverticula and clefts most likely results in their union, forming a tear that separates the caudate nucleus from underlying tissue. Ischemic damage likely drives the expansion of clefts and diverticula, resulting in tearing. Given this model, we cannot exclude the possibility that similar events contribute to the initiation of diverticula formation.
From a clinical perspective, special interest was focused on a Norwegian Elkhound litter. At the age of 10 weeks, four littermates were donated to the Department of Veterinary Clinical Sciences. All puppies were blind and had strabismus. Three days prior to donation, the neurologic symptoms of one puppy (No. 1) had deteriorated, and the animal had to be euthanatized 12 days after the donation. As described, a marked internal hydrocephalus with pronounced tearing was present at necropsy. Two other puppies (Nos. 2, 3) were maintained 4 days longer prior to euthanasia. These animals did not exhibit clinical deterioration despite a degree of hydrocephalus similar to that described for the first littermate. Both animals did not exhibit tearing, although one of the two puppies (No. 3) had microscopic diverticula. Necropsy findings in the fourth puppy included mild hydrocephalus, and for this reason the animal was not included in the present study. Tissues from this dog were evaluated histologically, and diverticula and clefts were not observed (unpublished observation), providing support for the hypothesis that formation of diverticula and clefts reflects the magnitude of ventricular enlargement.
Although tearing of periventricular white matter was associated with acute clinical deterioration in one dog, evidence of chronicity in diverticula, clefts, and tears in eight dogs indicates that the changes are survivable. Chronicity was indicated by astroglial scarring and vascular proliferation in the adjacent nervous tissue parenchyma.
Periventricular encephalitis was present in four of the dogs with diverticula, clefts, and/or tears. However, the severity of inflammation appeared disproportionately mild compared with the degree of tissue destruction, with the exception of one dog. In three of the four dogs, periventricular lymphoplasmacytic encephalitis was associated with chronic disruption of the periventricular white matter, and in one dog with neutrophilic inflammation, the neutrophils were restricted to areas of tissue necrosis. In all but one dog discussed here, inflammation was considered a response to tissue disruption and devitalization with breakdown of the blood–brain barrier and not the cause of periventricular diverticula, clefts, and tearing. This conclusion is in contrast to those of other researchers, who emphasized the importance of inflammation in the generation of diverticula. 3 7 15
In addition to the 20 described cases of marked juvenile canine hydrocephalus, six cases of marked hydrocephalus were identified in dogs ≥12 months of age during the same interval (i.e., 1990–1999). As in the juvenile cases, marked hydrocephalus was defined as a degree of lateral ventricular enlargement in which the width of the ventricle at the level of the pars centralis was greater than the thickness of subjacent cortical tissue by a factor of ≥2. However, gross and histologic evidence of periventricular diverticula, clefts, and tearing was not observed, in contrast to the 50% incidence of these changes in juvenile dogs with a similar magnitude of hydrocephalus. Further investigation is warranted to determine whether the absence of periventricular white matter disruption in adults reflects an age-dependent difference in the response to or development of increased intracranial pressure.
Footnotes
Acknowledgements
We thank clinicians, pathology residents, and faculty of the Ohio State University for the submission of quality case material over the years. We are indebted to Tim Voijt for his assistance in preparing the illustrations.
