Abstract
Epileptic seizures can lead to various reactions in the brain, ranging from neuronal necrosis and glial cell activation to focal structural disorganization. Furthermore, increased hippocampal neurogenesis has been documented in rodent models of acute convulsions. This is a report of hippocampal neurogenesis in a dog with spontaneous epileptic seizures. A 16-week-old epileptic German Shepherd Dog had marked neuronal cell proliferation (up to 5 mitotic figures per high-power field and increased immunohistochemical expression of proliferative cell nuclear antigen) in the dentate gyrus accompanied by microglial and astroglial activation. Some granule cells expressed doublecortin, a marker of immature neurons; mitotically active cells expressed neuronal nuclear antigen. No mitotic figures were found in the brain of age-matched control dogs. Whether increased neurogenesis represents a general reaction pattern of young epileptic dogs should be investigated.
Keywords
Epilepsy is the most common canine neurological disorder. The recurrent seizures are attributed to an excessive and hypersynchronous discharge of neurons in the brain. 2 A genetic predisposition for epilepsy has been described in several canine breeds, such as, Beagles, Golden Retrievers, Labrador Retrievers, and Vizslas. 2
The most frequent type of human epilepsy is temporal lobe epilepsy, which is defined as focal epilepsy arising from one or both temporal lobes. 5 In rodent models of this condition, abnormal hippocampal neurogenesis has been found in adult animals, and these new-born neurons are predominantly in ectopic hilar regions but also in the dentate granule cell layer. 8,10,13,14 Similar observations have been reported in children. 3
It is now widely accepted that neurogenesis occurs under physiological conditions in adult mammals, especially in the dentate gyrus of the hippocampus and the subventricular zone of lateral ventricles. 4,9,15 Adult neurogenesis is probably involved in learning tasks and in memory. Potential mechanisms contributing to increased neurogenesis after acute seizures have been discussed for rodent models. Release of mitogenic factors from dying neurons, deafferentiated granule cells, and reactive glia may enhance both the proliferation of neuronal precursor cells and the survival of newly formed neurons. 8 An increase in new-born neurons could be an anticonvulsive reaction pattern. 10 However, it could also be a key feature leading to chronic epilepsy, because the newly generated neurons might integrate improperly into local circuits leading to an unregulated discharge. 10
Other typical morphological alterations in human and canine temporal lobe epilepsy are activation of glia, neuronal necrosis, especially in the piriform lobe, and disorganization of granule cell neurons in the dentate gyrus. Hippocampal sclerosis (severe neuronal loss with concomitant gliosis) has been reported only in human cases. 16 Although epilepsy is the most common canine neurological disorder, increased or abnormal neurogenesis has so far not been reported in dogs. Here, we report extensive neurogenesis in the dentate gyrus of a young dog with epileptic seizures.
Case History
A 16-week-old, female German Shepherd Dog had a history of seizures over a period of 4 weeks, starting 1 week after initial vaccination against canine distemper virus, canine parvovirus, canine adenovirus-2, canine parainfluenzavirus, and leptospirosis with Nobivac DHPP1 and Lepto2. Hematological results before presentation at the referring hospital were within normal limits; serological tests for Toxoplasma and Neospora were negative. On neurological examination, the dog had a mild generalized tremor, reduced menace response, reduced startle reflexes (inability to negotiate an obstacle course), slow pupillary light responses bilaterally, and mild papilledema. Oral phenobarbitone administration (15 mg) did not control the seizures. No abnormalities were found in the cerebrospinal fluid (CSF) or on magnetic resonance imaging (MRI) of the brain. Three days before euthanasia, the dog seemed blind.
Methods
Necropsy was performed, followed by histological examination of the brain, cervical and thoracic spinal cord segments, the eyes, and specimens from most visceral organs. Tissues were fixed in 10% nonbuffered formalin for 2–3 days. After routine embedding in paraffin, 5-μm sections were stained with hematoxylin and eosin (HE) and, for brain sections, the periodic acid–Schiff reaction. Immunohistochemistry on brain sections, with appropriate positive controls, was performed to detect viral antigens of the following diseases: rabies, canine distemper, and parvovirus infection. Glial fibrillary acidic protein (GFAP) was the immunohistochemical marker for astrocytes, proliferating cell nuclear antigen (PCNA) for proliferating cells, and doublecortin (DCX) and neuronal nuclear antigen (NeuN) for migratory and generally postmigratory neurons, respectively. Positive controls were astrocytes in the brain sections for GFAP, lymph node for PCNA, fetal canine brain for DCX, and mature neurons as internal controls for NeuN. The antibodies and detection methods are detailed in Table 1.
Technique and Source of Antibodies for Immunohistochemistry
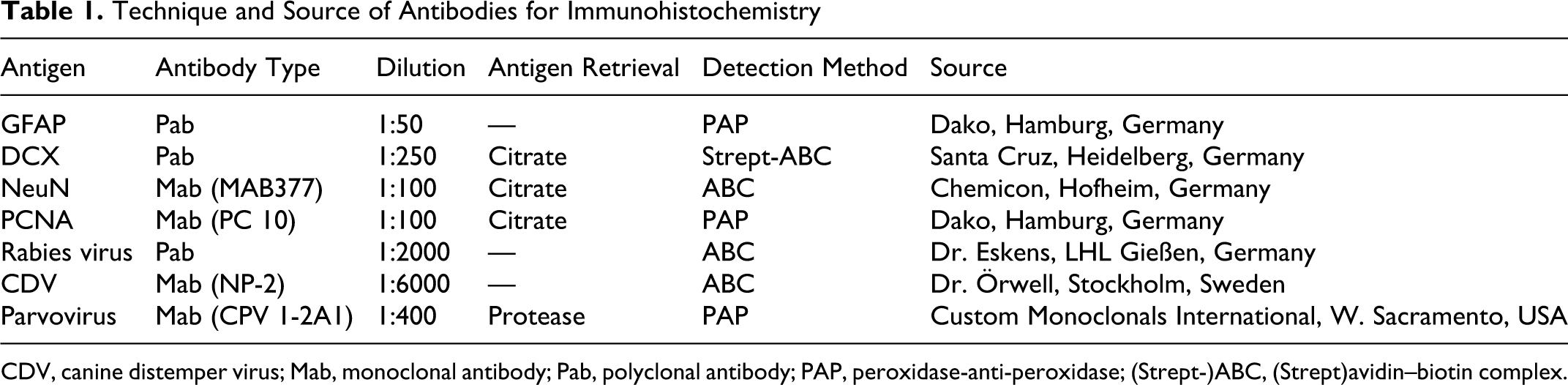
CDV, canine distemper virus; Mab, monoclonal antibody; Pab, polyclonal antibody; PAP, peroxidase-anti-peroxidase; (Strept-)ABC, (Strept)avidin–biotin complex.
The brains of 3 puppies without central nervous system lesions served as age-matched controls. In addition, the histopathological and immunohistochemical changes in the brains of 2 adult epileptic dogs (2 and 6 years old, respectively) were compared with those in the dog of this report. The 6-year-old epileptic dog suffered from chronic seizures; the clinical history of the other epileptic dog was not available.
Pathological Findings
At necropsy, the dog had moderate pallor and mild generalized skeletal muscle atrophy. The liver was normal in size with no evidence of an extrahepatic portosystemic shunt. No gross lesions were apparent in the brain, spinal cord, eyes, or other organs.
Histologically, brain lesions were most severe in the hippocampus. In other areas, especially in the frontal lobes of the cerebrum and in the spinal cord, there was mild multifocal microglial activation in the gray matter but no evidence of neuronal necrosis or mitosis. No histological lesions were detected in the piriform lobe, cerebellum, choroid plexus, or medulla oblongata. The periodic acid–Schiff reaction did not provide any evidence of intracellular storage of abnormal material in neurons or astrocytes in any brain area. Both eyes had reduced cellularity in the retinal ganglion cell and plexiform layers. Sections of heart, lung, liver, pancreas, stomach, intestine, spleen, mesenteric lymph nodes, bone marrow, kidneys, skeletal muscle, adrenal glands, thyroid gland, trigeminal ganglia, and optic nerves were unremarkable.
In the hippocampus, there was a diffuse increase in microglial cells, identified by their typical rod-shaped appearance. In addition, the granule cell layer of the dentate gyrus had marked proliferative activity, with up to 5 mitotic figures per high power field (Fig. 1). A few mitotic cells were in the molecular layer of the dentate gyrus, typical of ectopic granule cells.
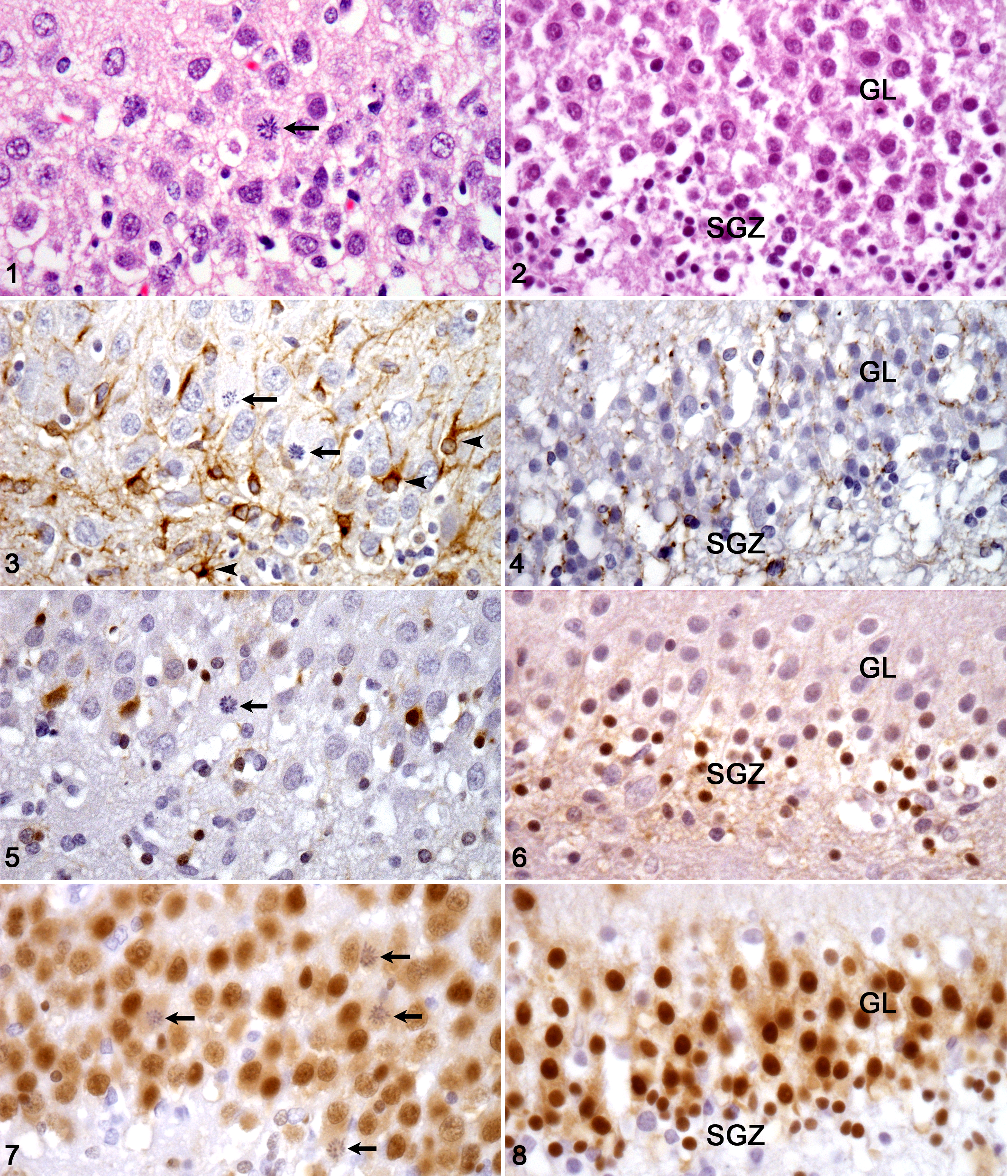
Mitotic figures were not found in the hippocampus or other areas of the brain in any of the 3 age-matched control dogs (Fig. 2) or in either of the 2 adult epileptic dogs. Nor were any structural changes detected in the dentate gyrus of the adult epileptic dogs. Instead, degeneration and necrosis of scattered neurons, mainly in the piriform lobe, and, in the 6-year-old dog, mild gliosis and satellitosis in the cerebral cortex were found.
The hippocampal changes were analyzed in more detail by immunohistochemistry for astrocytes, neuronal differentiation, and proliferation markers. Immunohistochemistry demonstrated an increased number of GFAP-positive cells with typical cytological features of activated astrocytes (interpreted as moderate diffuse astrogliosis) compared with the age-matched control brains (Fig. 3, 4). None of the mitotic cells, however, was GFAP-positive. Scattered individual cells in the granule cell layer of the dentate gyrus expressed PCNA, a protein that is associated with DNA synthesis, thereby indicating cellular proliferation. Few cells in the granule cell layer expressed DCX, a protein that is mainly expressed by immature migratory neurons (Fig. 5, 6). In comparison to age-matched controls with most DCX-positive cells in the subgranular zone, the DCX-positive cells in the epileptic dog were situated mainly within the granule cell layer, whereas those in the subgranular zone were fewer. All the mitotic cells were DCX-negative (Fig. 5, 6) but were weakly immunoreactive for NeuN (Fig. 7). Immunohistochemistry of the cerebellum for canine distemper, parvovirus, and rabies virus antigens was negative.
Discussion
Although the brain was once regarded as a postmitotic tissue, and it was presumed that neurons lost mitotic capability shortly after birth, neurogenesis is now a well-recognized physiological feature that lasts through adulthood in mammals. 7 Certain regions, namely the subventricular zone situated throughout the lateral walls of the lateral ventricles and the subgranular zone of the dentate gyrus, contain most newly formed neurons. 7,17 However, the neurogenesis is influenced by physiological and pathophysiological conditions. 1,6,11,12 In contrast to physiological neurogenesis, which enhances brain function, neurogenesis after acute seizures occurs also in ectopic regions and the newly formed neurons can develop morphological abnormalities, such as basal dendrites. 13 These altered neurons could contribute to the establishment of chronic epilepsy by forming local circuits that lead to spontaneous discharge (epileptic foci). 10 In the present case, only a few newly generated neurons were found to be ectopic (ie, in the molecular layer), and they did not have obvious morphological abnormalities. Nevertheless, dysfunction of those neurons could have resulted in an epileptic focus.
Interestingly, in chronic epilepsy in rats, the number of new-born cells decreases dramatically, presumably due to a milieu that is unfavorable for neurogenesis. 8 Dysfunctional neurons and disarranged circuits may persist and cause recurrent seizures. Whether this can also occur in chronic canine epilepsy remains to be determined.
The high mitotic index in the dentate gyrus of this dog is consistent with the increased neurogenesis observed after acute seizures in rodent models. However, even though hilar ectopic neurons are described after seizure-induced neurogenesis, the mitotic figures in the present case were predominantly within the granule cell layer of the dentate gyrus. Newly generated neurons were found in the granule cell layer in a rodent epilepsy model as in the present canine case. 14 In the dog, the increased mitotic rate was associated with few PCNA-expressing cells in the granule cell layer, faint expression of DCX, and a weak expression of NeuN, indicating increased mitotic activity of dentate granule cells. Whether these findings indicate a shift of neurogenesis from the subgranular zone to the granule cell layer or even a reentry of granule cells into the cell cycle remains speculative. However, DCX-positive cells were reduced in the subgranular zone of the epileptic dog in comparison to control dogs, which could indicate a transfer of neurogenesis to the granule cell layer. The expression pattern of mitotic cells (DCX-negative, NeuN-positive) is consistent with a phase beyond the immature DCX-expressing stage and at the onset of NeuN expression. NeuN usually serves as a marker of mature neurons. Von Bohlen and Halbach 17 reported, however, that the beginning of NeuN expression overlaps with DCX expression and therefore also labels still-immature dentate granule cells. 17 Thus, the NeuN-positive mitotic cells could be neurons at the interface between the migratory and postmigratory phase.
Interestingly, there was no evidence of neuronal necrosis in any brain area of this case, so the increased neurogenesis might reflect a disturbance in the synthesis of new neurons rather than a compensatory event. In addition, there was evidence of glial cell activation. This is a common reaction in epilepsy but often exacerbates seizure activity. 18 Mitotically active cells in the hippocampus were consistently GFAP-negative and lacked the morphological features of astrocytes or microglial cells. Therefore, astrogliosis and microglial activation might have contributed to seizure generation or could be merely the consequence of abnormal neuronal activity.
Sections of age-matched control dog brains had no mitotic figures in the hippocampus or elsewhere, which further emphasizes the findings in the epileptic puppy. Also, in the 2 adult epileptic dogs, no evidence of an increased mitotic rate was found in the hippocampus. Because the dog of this report was only 16 weeks old, and seizure-induced neurogenesis has only been reported in young children, the lack of neuronal proliferation in the adult epileptic dogs could be age-related. However, physiological neurogenesis is described in dogs up to 15 years of age, although with a drastic decrease in aged animals. 12,15 The 6-year-old epileptic dog had a history of chronic seizures, whereas the duration of seizures was unknown in the 2-year-old epileptic dog. Thus, the lack of hippocampal mitotic figures in these dogs could reflect the chronic stage of epilepsy when neurogenesis is decreased in rat models. 8 Loss of retinal neurons in this case could explain the blindness that developed in this dog. No histological lesions were detected in central optic areas of the brain.
In conclusion, increased neurogenesis was detected in the hippocampus of a young dog with epileptic seizures. Whether enhanced neurogenesis is a key feature of epilepsy in young dogs, as in rodent models and epileptic children, should be investigated in cohorts of epileptic dogs at various ages and stages of the disease. Should this be the case, the detection of mitotic neurons could be useful for the histological diagnosis of epilepsy in young dogs.
Footnotes
Acknowledgments
We thank the Histology Laboratory, Veterinary Laboratory Services, University of Liverpool, and Annette Artelt, Department of Veterinary Pathology, University of Gießen, for excellent technical support, and Dr. Udo Hetzel, Veterinary Pathology, School of Veterinary Science, University of Liverpool, for expert advice on ocular pathology. Many thanks to Dr. Manfred Henrich, Department of Veterinary Pathology, University of Gießen, for his assistance in adjusting the illustrations.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
