Abstract
Cyclooxygenase-2 (COX-2) has been shown to be the primary enzyme responsible for prostaglandin production during inflammation but is absent in most tissues under normal physiological states. High levels of COX-2 expression have been observed in the macula densa and thick ascending limbs of fetal kidneys; this expression declines to minimal levels during renal maturation. We hypothesized that the neoplastic cells of renal cell carcinoma (RCC) may revert to high expression of COX-2, and we evaluated its expression in three spontaneous cases of canine RCC by using immunohistochemical methods. The neoplastic cells of two of the three cases exhibited moderate to marked COX-2 immunoreactivity. These results suggest that some canine renal cell carcinomas express high levels of COX-2, which may play a role in the modulation of neoplastic cell growth.
Cyclooxygenase (COX) exists in two related but unique isoforms: cylcooxygenase-1 (COX-1) and cyclooxygenase-2 (COX-2). COX-1 is constitutively expressed and thought to function in normal cell physiology, whereas COX-2 is inducible, expressed in response to inflammatory stimuli, and a known mediator of inflammation and regulator of cell growth. 7 10 Several studies have demonstrated that treatment with nonsteroidal antiinflamatory drugs (NSAIDs) can result in the cessation of tumor growth in the acquired cancers of the urinary and colorectal region, likely by inhibition of COX enzymes. 2 6 8 Renal cell carcinoma (RCC) is a common primary renal neoplasm in dogs for which no information exists regarding expression of cyclooxygenase. Recently, we reported slight levels of constitutive expression of COX-2 in the macula densa (MD) and thick ascending limb (TAL) of the loop of Henle in the normal adult dog kidney. 5 COX-2 expression is several-fold higher in the immature nephrons of fetal kidneys and progressively declines to minimal levels prior to parturition. 4 This expression of COX-2 in the immature nephrons has been suggested to play a role in nephrogenesis. We hypothesize that the neoplastic cells of RCC may revert to high expression of COX-2 which may modulate tumor cell growth.
Dog kidneys with RCC were obtained from three adult dogs (one spayed female Golden Retriever; two intact males, mixed breed) from the oncology archives of the Purdue University Veterinary Teaching Hospital, West Lafayette, IN. The normal kidneys of adult Beagle dogs (n = 3) were collected at necropsy and retrieved from the archives of the Pathology Department at Searle, R&D, Skokie, IL.
All procedures performed in these studies were in compliance with the National Institutes of Health (NIH) guidelines for the care and use of animals in research (NIH guide) or received with prior permission from the Institutional Animal Care and Use Committee. Standard immunohistochemical procedures and commercially available assay kits were used for the immunolocalization of COX-1 and COX-2, as previously described with minor modifications. 8 In general, tissues were paraffin-embedded (Paraplast X-tra, Oxford Labware, St. Louis, MO), cut in 4–5-µm-thick sections, and mounted on positively charged slides (Superfrost Plus, Erie Scientific Co., Portsmouth, NH). Sections were then dewaxed, rehydrated in xylenes and descending alcohols, then blocked for endogenous avidin/biotin. All tissues were permeabilized in 0.3% triton/0.2% saponin/1% bovine serum albumin (BSA, Sigma, St Louis, MO) in phosphate-buffered saline (PBS), preblocked in PBS containing 10% normal goat serum (Vector Laboratories, Inc., Burlingame, CA), then incubated in primary antibody overnight at 4 C. The COX-1 and COX-2 primary antibodies were prostaglandin-H synthase-1 polyclonal rabbit anti-human (cat No. 160108, Cayman Chemical Co., Ann Arbor, MI) and PGHS-2 polyclonal rabbit anti-human (cat No. PG27, Oxford Biomedical Research, Inc., Oxford, MI) antibodies, respectively. These antibodies were diluted 1:100 in 1% BSA-PBS. Immunoreactive complexes were detected via an enhanced streptavidin–biotin affinity system (Omnitags Plus, Shandon-Lipshaw) and visualized with diaminobenzidine (Dako Corporation, Carpinteria, CA), which reacts with peroxidase to give a brown reaction product. Slides were counterstained briefly in hematoxylin-1 (Richard-Allan Scientific, Kalamazoo, MI). All control slides were treated without primary antibody and then incubated with either biotinylated goat anti-rabbit IgG (Vector) or biotinylated horse anti-rabbit IgG (Vector) at the appropriate dilution to accompany the positive slides.
Clinically, all three dogs with RCC presented with weight loss and were diagnosed to have renal masses via ultrasound. No metastases were identified by ultrasound or radiography (dogs Nos. OL-8140-1, −2, −4).
Expression of both isoforms of COX (COX-1 and COX-2) was evaluated in normal kidneys and in kidneys with RCC. In normal canine kidneys, COX-1 staining was observed in glomeruli, blood vessels, collecting ducts, and papillary interstitial cells, whereas COX-2 staining was isolated to the MD, TAL, and papillary interstitial cells (Fig. 1a, b). The distribution of COX-1 in the canine kidney is similar to that observed in the human kidney; however, the COX-2 staining pattern differs between these species in that COX-2 is not present in the MD or TAL in normal adult human kidneys. 5 In one animal, COX-2 expression was markedly increased in the MD and TAL of the normal kidney adjacent to the tumor. This increase in expression is thought to be induced in response to altered renal functions due to the renal neoplasia or an effect of cytokines released by the tumor cells.
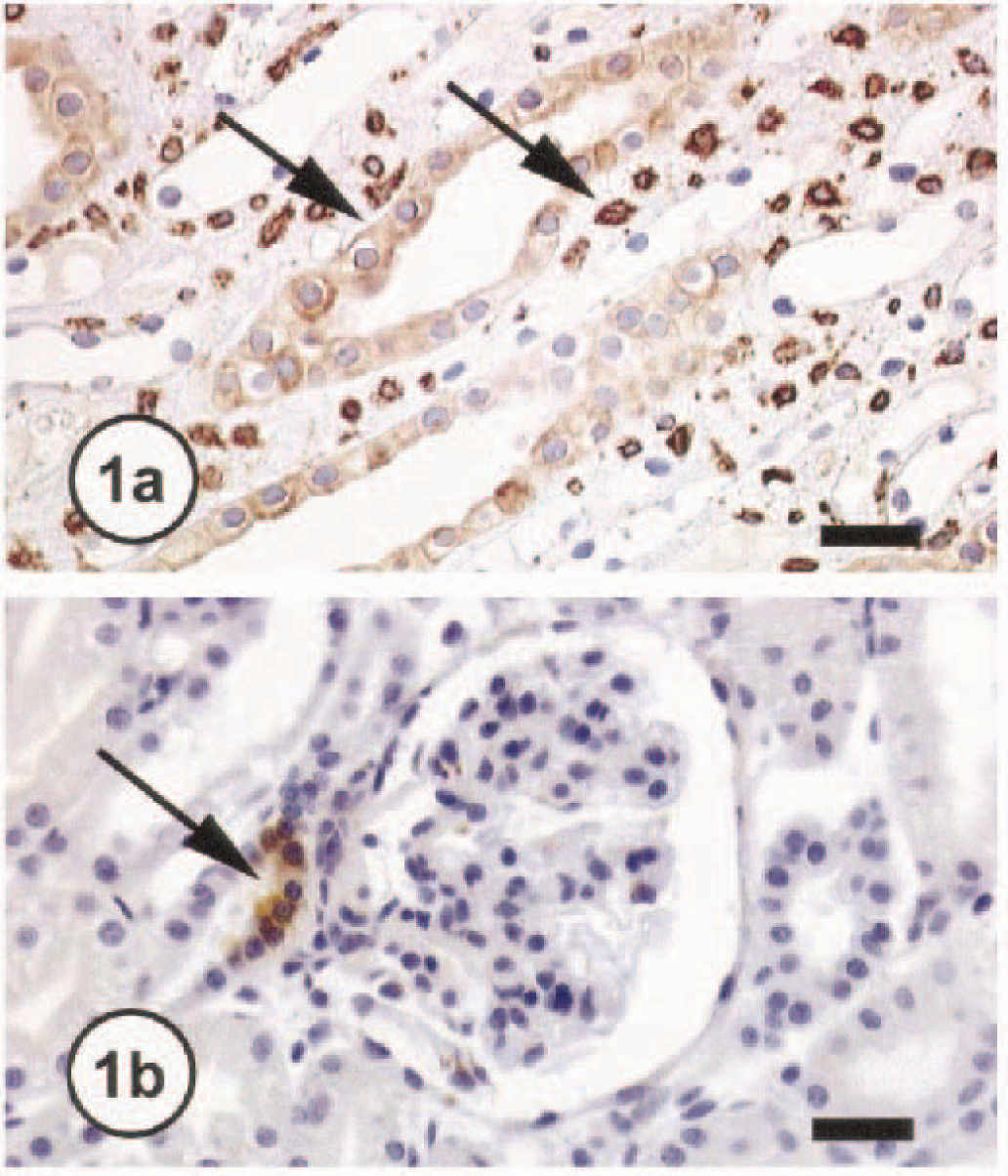
Kidney; normal dog No. 1701. Note COX-1 immunoreactivity (
All three tumors were poorly demarcated and comprised of sheets of large round to polygonal cells with large round nuceli, prominent single nucleoli, and approximately one mitotic figure per high-power field. Clear cells were not evident in any tumor examined. The neoplastic cells of RCC in two of three dogs showed moderate to marked cytoplasmic COX-2 staining (Fig. 2a, b), whereas no immunoreactivity was detected for COX-1 in the neoplastic cells (Fig. 2c). No COX-2 immunoreactivity was detected in the fibrovascular stroma of RCC. It is possible that the neoplastic cells of tumors expressing COX-2 in two of the dogs originated from the MD or TAL of the loop of Henle, whereas the tumor not expressing COX-2 from the third dog originated from a different region of the nephron. The high COX-2 expression observed in RCC is reminiscent of the prominent COX-2 expression observed in fetal kidneys, suggesting that COX-2 in RCC reverts to fetal levels. 1 9
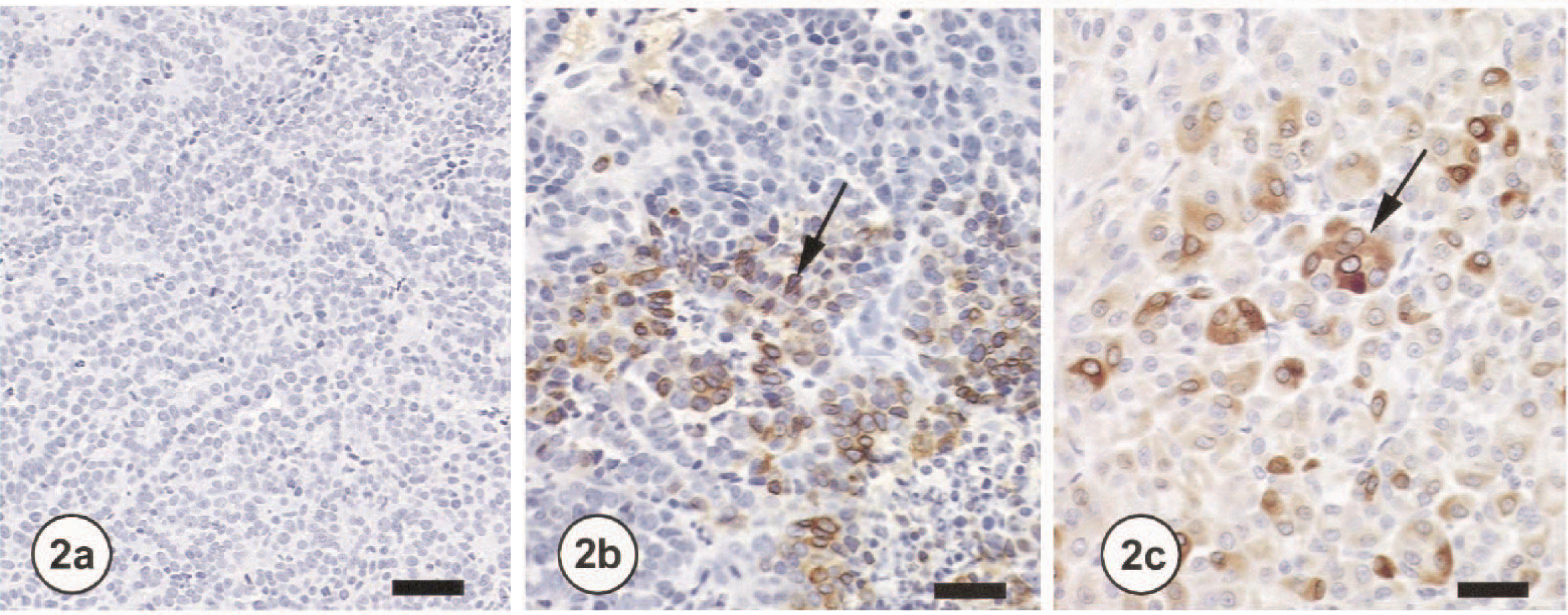
Kidney; dog No. OL8140-2. Note lack of COX-1 immunoreactivity (
The role of COX-2 in cell growth and differentiation is evident from several examples. COX-2 has been shown to be pivotal in the development and maturation of the fetal kidney, as demonstrated by the phenotype COX-2 knock-out mouse. Mice deficient in COX-2 enzymes exhibit severe disruption of kidney development and function. 1 9 These findings, in addition to the observations of elevated COX-2 expression in fetal kidneys, suggest a physiological role for COX-2 in immature nephron development. 3 Furthermore, the mitogenic activity of COX-2–derived prostaglandins is thought to contribute to growth of several malignant neoplasms and inhibition of these enzymes via NSAIDs has been demonstrated to slow, and in some cases stop, tumor growth. 6 11 These data, along with the current observations of COX-2 expression in RCC, suggest that COX-2–mediated prostaglandins may play a role in the modulation of neoplastic cell growth in some cases of canine RCC.
