Abstract
Retrospectively, we demonstrated that seven pigs necropsied in 1989 had characteristic porcine circovirus 2 (PCV-2) lymphoid tissue lesions of severe lymphoid depletion, syncytial giant cell formation, and intracytoplasmic inclusion bodies in macrophages. Immunohistochemically, PCV-2 antigen was detected in these tissues and the distribution of positive staining corresponded to the distribution of inclusion bodies. Electron microscopy demonstrated viral particles compatible with porcine PCV-2. Therefore, the disease occurred in Japan as early as 1989.
In 1991, a new syndrome that affected fattening pigs in Canada, characterized by poor body condition and skin pallor, sometimes with icterus, was described by Clark and Harding. The disease was first referred to as postweaning multisystemic wasting syndrome (PMWS), but a positive diagnosis was not made until 1995. 4 7 After that, the disease was recognized in the USA, Spain, and Canada. 9 12 14 PMWS was diagnosed by the detection of characteristic circoviral inclusion bodies in affected tissues, viral antigen by immunohistochemistry, viral particles by electron microscopy, and/or isolation of the virus. 1 2 5 6 13 Retrospectively, we diagnosed PMWS in seven pigs collected in 1989, and we suspect that this report is the first case of porcine circovirus type 2 (PCV-2) infection in Japan.
During December 1989–June 1990, a total of 20 necropsies were performed of fattening pigs, aged 4–12 weeks, that were removed from a pig farm of 800 breeding sows and transported to the Livestock Hygiene Service Center, Mie Prefecture, Japan, for diagnosis of disease. They showed poor body condition, dyspnea, and sudden death. The mortality at the farm was 5–6%. After systemic necropsy, tissues were fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned, and stained with hematoxylin and eosin.
Immunohistochemical tests for porcine circovirus (PCV), porcine reproductive and respiratory syndrome virus (PRRSV), Aujeszky's disease virus (ADV), hog cholera virus (HCV), and toxoplasma antigens were performed with the streptavidin-biotin (SAB) immunoperoxidase method using Histofine SAB reagents (Nichirei Corp., Tokyo, Japan). After deparaffinization of the tonsil, mesenteric lymph node, spleen, ileum, and lung sections, endogenous peroxidase activity was blocked by treatment with 0.3% H2O2 in absolute methanol, and then the tissue sections were treated with 0.1% pronase in phosphate-buffered saline for 5 or 30 minutes at 22 C. The sections were then incubated with normal goat primary antibody, SAB reagent, and 0.05% 3,3-diaminobendine tetrahydrochloride (Dojido Corp., Kumamoto, Japan) and 0.1% H2O2 in Tris buffer, pH 7.6. Anti-PRRSV, 8 anti-ADV, 10 anti-toxoplasma rabbit sera (provided by Drs. S. Yamada and T. Imada, National Institute of Animal Health, Japan) were used at a dilution of 1:2,048 and were incubated for 30 minutes at room temperature. Anti-HCV monoclonal serum 11 (Central Veterinary Laboratory, UK) was used at a dilution of 1:100 and was incubated overnight at 4 C. In the case of PCV immunohistochemical staining, anti-PCV-1 rabbit serum 13 (provided by Dr. T. Imada, National Institute of Animal Health, Japan) and biotinylated anti-PCV porcine serum 13 that reacted with PCV-2 but not PCV-1 (provided by Dr. M. Haritani, National Institute of Animal Health, Japan) were used at a dilutions of 1:500 and 1:16, respectively. Sections were then counterstained with methyl green. Experimentally PRRSV, ADV, HCV, and toxoplasma-infected and naturally PCV-infected pig samples served as positive controls. Gnotobiotic piglet tissue samples and sera from a nonimmunized rabbit and mice served as negative controls.
Small parts of the samples were taken from paraffin blocks, deparaffinized, fixed in 1% osmic acid, embedded in epon mixture, sectioned, stained with uranyl acetate and lead citrate, and examined using a JEOL 100CX electron microscope.
Systemic lymphadenopathy was observed in 7 of 20 pigs (7/20), fibrinous polyserositis in 6/20, pulmonary edema in 4/20, and pulmonary abscesses in 2/20. Microscopically, characteristic changes were observed in the lymphoid tissues. The follicular B-cell zone was replaced by histiocytic cells and multinucleated giant cells (Fig. 1). Moreover, cytoplasmic circoviral inclusion bodies appearing as grape-like clusters that were intensely basophilic were detected in the cells within germinal centers of the lymphoid tissues (Fig. 2). Typical inclusion bodies were found in 7 of 20 pigs examined. Inclusion bodies were found in the tonsils of 2 of 2 pigs (2/2), mesenteric lymph nodes of 6/7, Peyer's patches of 6/7, mediastinal lymph nodes of 5/5, pulmonary lymph nodes of 3/4, hepatic lymph nodes of 4/4, and mandibular lymph nodes of 2/2. Seven pigs had interstitial pneumonitis characterized by moderate thickening of the alveolar septa due to inflammatory cell infiltration by mononuclear cells and occasional type II pneumocyte hyperplasia. One had fibrinous necrotizing enteritis, two had fibrinous meningitis, and two had fibrinous polyserositis.
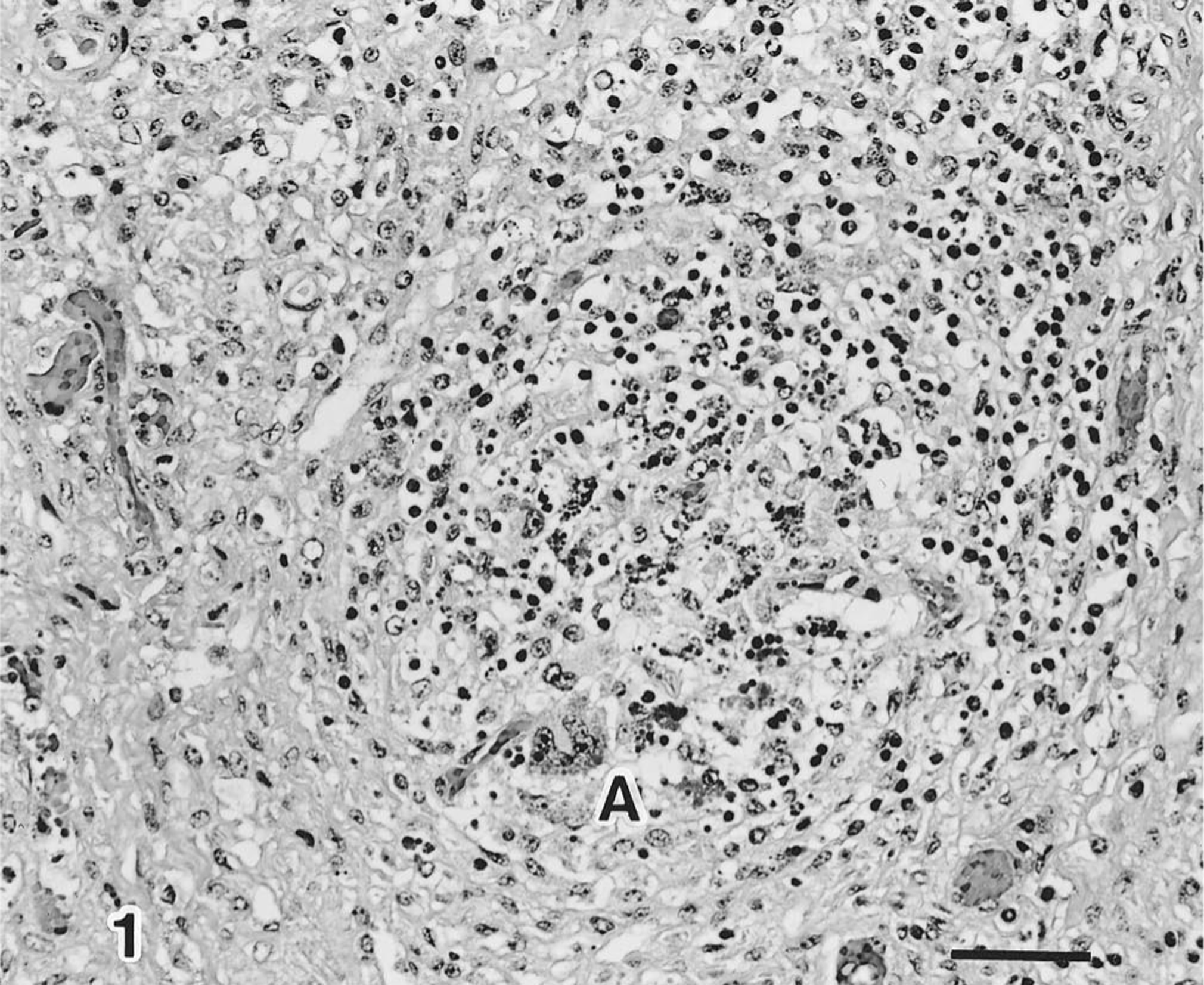
Tonsil; severe lymphoid depletion and giant cell in the lymphoid follicles (A). HE. Bar = 50 µm.
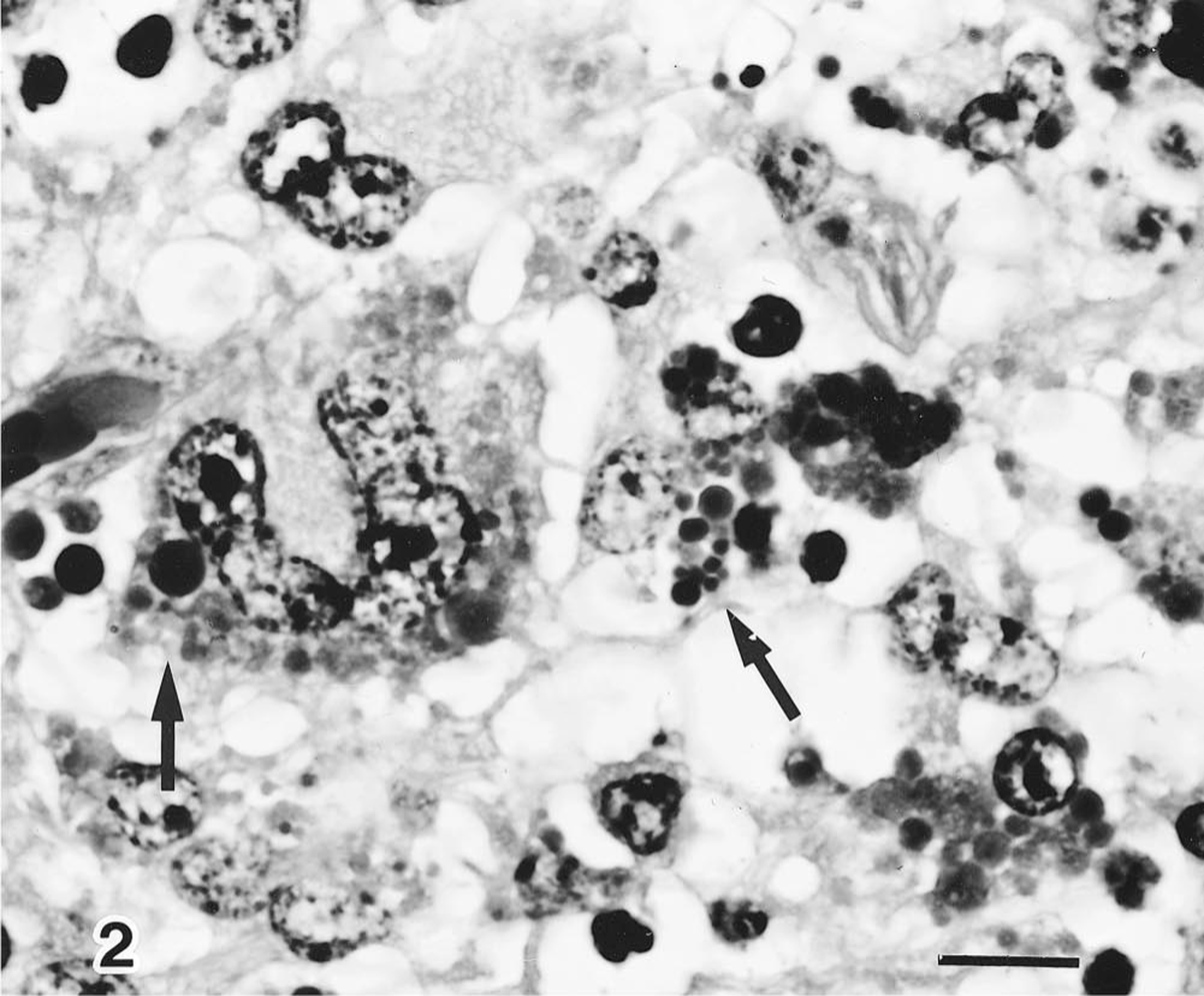
High magnification of A. Multiple intracytoplasmic inclusion bodies in the macrophages (arrows). HE. Bar = 10 µm.
Immunohistochemically, PCV-2 antigen was demonstrated in 7 of 20 pigs examined; antigen localized diffusely in the cytoplasm and to various sized, often multiple, and usually intracytoplasmic inclusions (Fig. 3). Antigen was detected in giant cells and macrophages distributed in the tonsil, mesenteric lymph nodes, ileal Peyer's patches, and lung, and its distribution was closely associated with circoviral inclusion bodies. PRRSV antigen was detected in one out of seven of the PCV-2 infected pigs. No PCV-1, HCV, ADV, or toxoplasma antigen was observed in any of the tissues or cases.
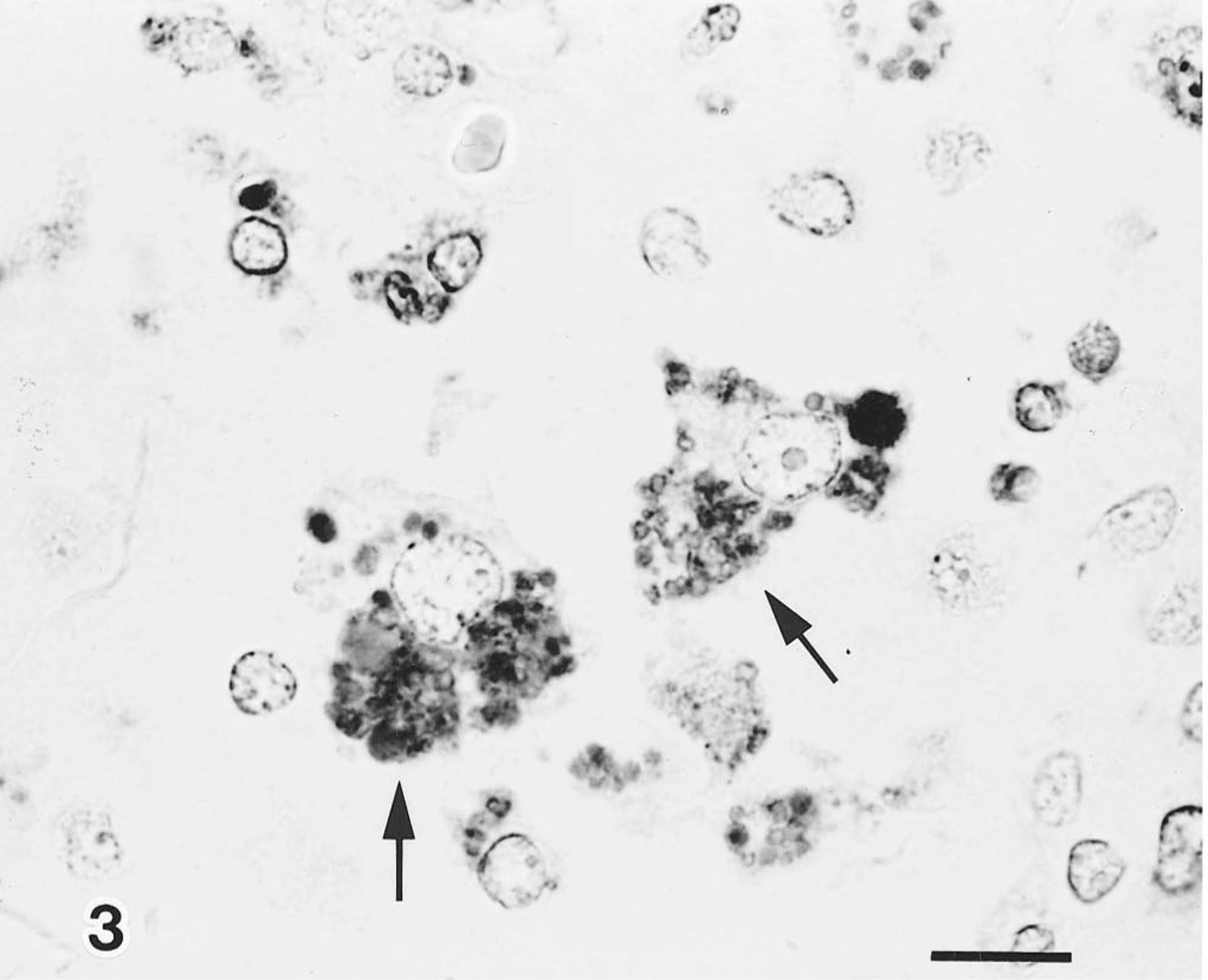
Mesenteric lymph node; PCV-2 antigen associated with the cytolasmic inclusion bodies (arrows). Anti-PCV-2 SAB. Bar = 10 µm.
Electron microscopically, cytoplasmic inclusions in macrophages in the lymphoid tissues were dense, non–membrane-bound paracrystalline arrays or semicircles of tightly packed icosahedral, 14–17-nm diameter, nonenveloped virus particles (Fig. 4). Particle size and morphology were consistent with previously described circoviruses. 5 13
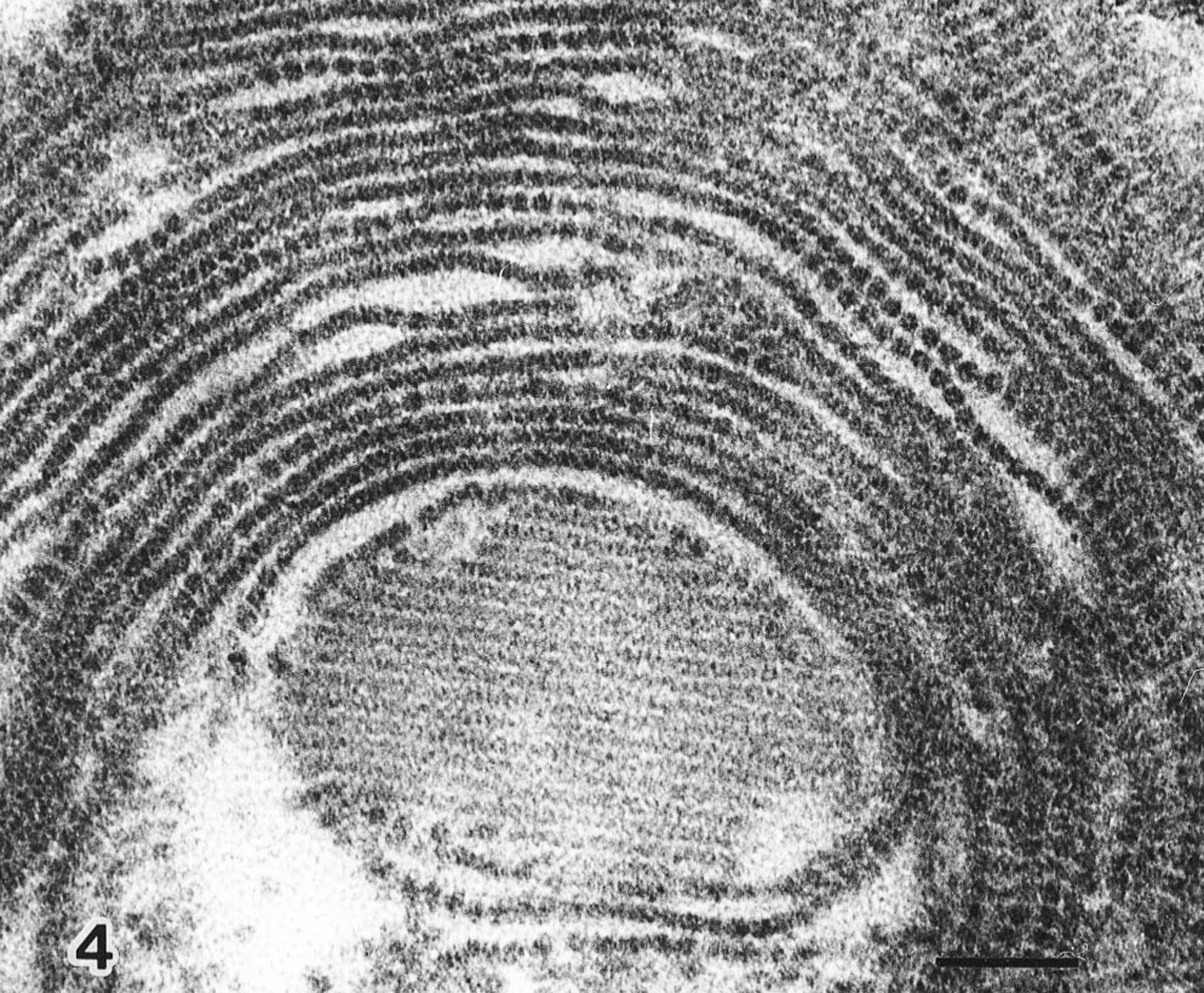
Mesenteric lymph node; virus particles in paracrystalline array and semicircles. Bar = 100 nm.
The diagnosis of PCV infection was made on the presence of characteristic macroscopic and microscopic lesions. 1–4, 6 12–14 In the present retrospective study, we demonstrated that seven pigs had characteristic lymphoid tissue lesions, consisting of severe lymphoid depletion, syncytial giant cell formation, and intracytoplasmic inclusion bodies in macrophages. Electron microscopy confirmed that the inclusion bodies consisted of circoviral particles. These pathologic lesions were similar to those of PCV-2 previously reported for this disease. 1 4 9 12–14
It is well known that PCV-1 is nonpathogenic but PCV-2 is pathogenic for pigs. 1–3, 6 In the present study, abundant PCV-2 antigen was demonstrated in giant cells and macrophages in lymphoid tissues. However, no PCV-1, HCV, or toxoplasma antigens were found in seven pigs, except one with PRRSV antigen. Thus, PCV-2 disease occurred in Japan as early as 1989.
Footnotes
Acknowledgements
We thank Mr. K. Kobayashi and Miss M. Shimada for preparing the histologic tissue sections and Dr. Y. Ando and Mr. T. Fujisawa for preparing the photographs.
