Abstract
A 7-year-old neutered female English Setter presented with syncope, anemia, and weight loss. Clinical examination revealed a systolic murmur and echocardiography demonstrated a mass on the pulmonic valve. Postmortem examination confirmed the presence of a pulmonic valve mass that extended along the pulmonary trunk and into the left pulmonary artery. Multiple pale nodules were observed in the right lung. Microscopic examinations of the pulmonary artery mass and the lung nodules revealed a pleomorphic population of spindle cells often arranged in broad bands containing strap-like nuclei and eosinophilic cytoplasm devoid of cross striations. The neoplastic cells expressed vimentin and alpha-smooth muscle actin but did not express desmin, CD31, factor VIII, or S100. The presentation, histological features, immunocytochemical profiles, and behavior of this tumor were indicative of a primary pulmonary artery leiomyosarcoma with lung metastasis.
In animals, primary vascular neoplasms are derived more commonly from endothelium (hemangiomas, hemangiosarcomas, angioendotheliosis) or pericytes (hemangiopericytoma) and rarely from muscle (leiomyomas, leiomyosarcomas, glomangiomas). 5 11 15 While hemangiomas and hemangiosarcomas are regarded as the more commonly recognized neoplasms of vascular endothelium, angioendotheliosis, a multisystemic intravascular proliferation of endothelial cells, has also been described in the cat. 4 12 This neoplasm shares similarities with cutaneous angiomatosis in the bovine and pseudoangiosarcoma in man. Hemangiopericytomas are commonly observed in the dog and, while intimately associated with blood vessels, their true origin from pericytes continues to be controversial, leading to their classification in some instances as a subcategory of fibrosarcoma. 10 In many organs where leiomyomas and leiomyosarcomas develop, it is speculated that vascular smooth muscle may be the site of origin. 5 6 An uncommon benign subcutaneous vascular smooth muscle tumor, the glomangioma, has also been recorded in the dog. 14 This tumor is derived from the modified smooth muscle cells of the glomus body responsible for temperature regulation. In man, leiomyosarcomas of the pulmonary artery represent an infrequent manifestation of neoplasms of vascular smooth muscle, with the literature containing single case reports and reviews. 2 This report describes the clinical, echocardiographic, histologic, and immunocytochemical features of a primary pulmonary artery leiomyosarcoma in an adult dog. To the authors' knowledge, this case represents the first documentation of such a lesion in companion animals.
A 7-year-old neutered female English Setter weighing 18 kg was referred to the Veterinary Teaching Hospital at University College Dublin for evaluation of syncope, anemia, and weight loss. Three months before referral, the dog had had an ovariohysterectomy following a pyometra. A heart murmur was detected at this time but not investigated any further. Recovery from surgery was slow and her condition deteriorated. One month before referral, the dog developed syncopal episodes associated with exercise, which lasted 30 seconds and were flaccid in nature. On presentation, the dog was thin and had pale mucous membranes and a grade 5/6 systolic murmur audible on both sides of the thorax. Heart rate was 120 bpm and femoral pulses were strong. A CBC and biochemical profile revealed a normochromic, normocytic anemia (PCV 31%, MCV 66 fl, MCHC 34 g/dl) and elevated BUN (32.6 mmol/liter) and creatinine (256 µmol/liter). Urine specific gravity was 1.015.
Ultrasonography of the heart was performed using an Opus I ultrasound machine (Ausonics, Australia) and a 7.5 MHz sector transducer. Right-sided parasternal long and short axis views were obtained. Within the pulmonary artery outflow tract, a mixed echogenic 2-mm × 4-mm mass was identified on the pulmonic valve. With the views optimized for aortic outflow tract and aortic leaflets, the left coronary cusp was seen to be markedly thickened and hyperechoic. There was some thickening of the interventricular septum and left ventricular free wall. The right ventricular free wall was markedly thickened.
The owners were informed that the systolic murmur and syncopal episodes were likely to be related to the pulmonic and aortic valve lesions. Following discussion of the findings and in view of the severity of clinical signs, protracted course of illness, and poor prognosis, euthanasia was requested.
Postmortem examination confirmed the presence of a smoothly outlined, friable soft tissue mass with a broad base that was attached to the pulmonic valve. This mass had a polypoid projection (1 cm in length) that extended into the right ventricle (Fig. 1). The mass continued along the pulmonary trunk and extended into the left pulmonary artery. Growth within the wall and lumen was observed and both the pulmonary trunk and left pulmonary artery were almost totally occluded. There was focal nodular thickening of the left coronary aortic valve cusp, and hypertrophy of the right and left ventricular free walls was noted. The periphery of the right caudal lung lobe contained multiple grey, firm, noncystic nodules approximately 1 cm in diameter. Both kidneys were moderately reduced in size and nodular in outline. The left kidney contained a 1-cm cystic cavity in the caudal pole. The spleen had a 6-cm-diameter smooth-margined, blood-filled subcapsular mass in the body and similar 1–2-cm-diameter masses in the tail.
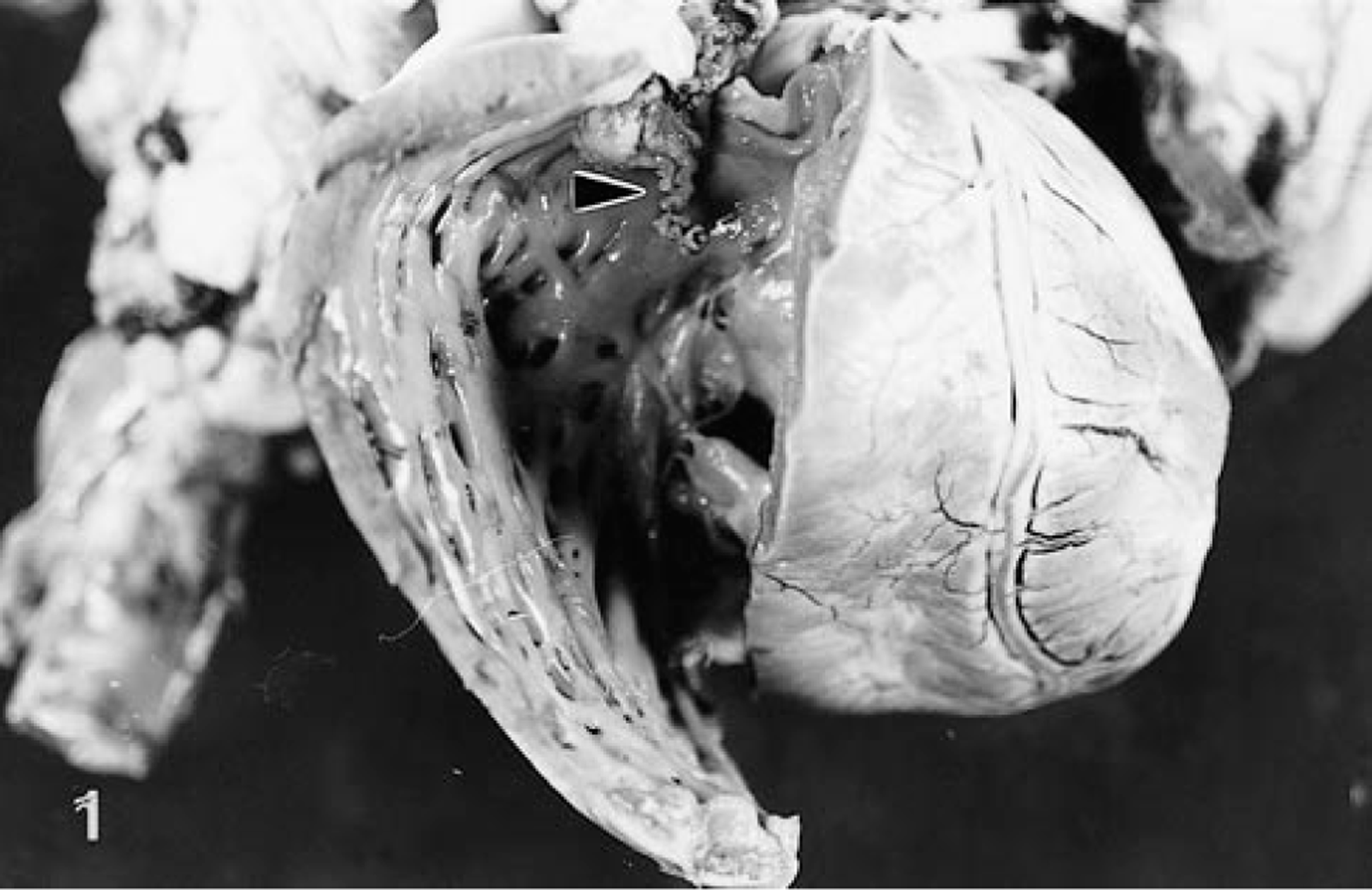
Heart. Right ventricle lumen exposed to reveal a smooth-outlined, polypoid mass (arrowhead) extending from the pulmonic valve.
Routine aerobic cultures of portions of the pulmonic valve mass failed to identify any bacteria.
Sections of pulmonic valve, pulmonary trunk, right and left pulmonary arteries, aortic valve, lung, liver, kidney, and spleen were fixed in neutral-buffered formalin and were paraffin embedded. Five-micrometer sections were cut and stained with hematoxylin and eosin. Selected sections were also stained with MSB, Massons trichrome and elastin. Sections of the mass within the pulmonary artery and in the lung were stained with a panel consisting of mouse monoclonal or rabbit polyclonal (factor VIII only) antibodies (Dako, Cambridge, UK) using commercial avidin-biotin streptavidine complex immunocytochemical techniques (StreptABComplex-HRP, Dako, Cambridge, UK). Antibody dilution were as follows: alpha-smooth muscle actin, 1:1,000; desmin, 1:50; vimentin, 1:40; factor VIII, 1:150; CD31, 1:50; and S100, 1:500. Tissue preparations included trypsinization (0.1% trypsin, 0.1% CaCl) for 30 minutes at 37 C for alpha-smooth muscle actin and desmin. For CD31 expression, antigen was unmasked with pressure cooker treatment for 90 seconds in 0.01 M sodium citrate. Antigen retrieval was not required for vimentin, factor VIII, or S100. Biotinylated rabbit antimouse or swine antirabbit antibodies were used as the secondary antibody (Dako, Cambridge, UK) and 3′3-diaminobenzidine tetrahydrochloride-hydrogen peroxide (Sigma, Missouri, USA) as chromagen.
The pulmonic valve mass consisted of a multinodular lesion blending into the connective tissue of the valve and into the endocardium of the right ventricle. This mass was composed of some tightly packed and some loosely arranged pleomorphic spindle cells. Nuclei were characterized by anisokaryosis, oval shape, polygonal or elongated shape, and a blunt end. The nuclear membrane was well demarcated and contained one or two small nucleoli (Fig. 2). Bizarre mitotic figures were common. Cell borders were indistinct and the cytoplasm was occasionally vacuolated. Both single cell necrosis and pockets of necrotic tissue with infiltrates of neutrophils were observed. Cells sometimes resembled smooth muscle tissue when arranged in broad bands containing strap-like nuclei and bands of eosinophilic cytoplasm without cross striations. Occasionally cells were separated by depositions of pale homogenous eosinophilic matrix. The surface of the pulmonic valve mass contained fibrin and necrotic debris.
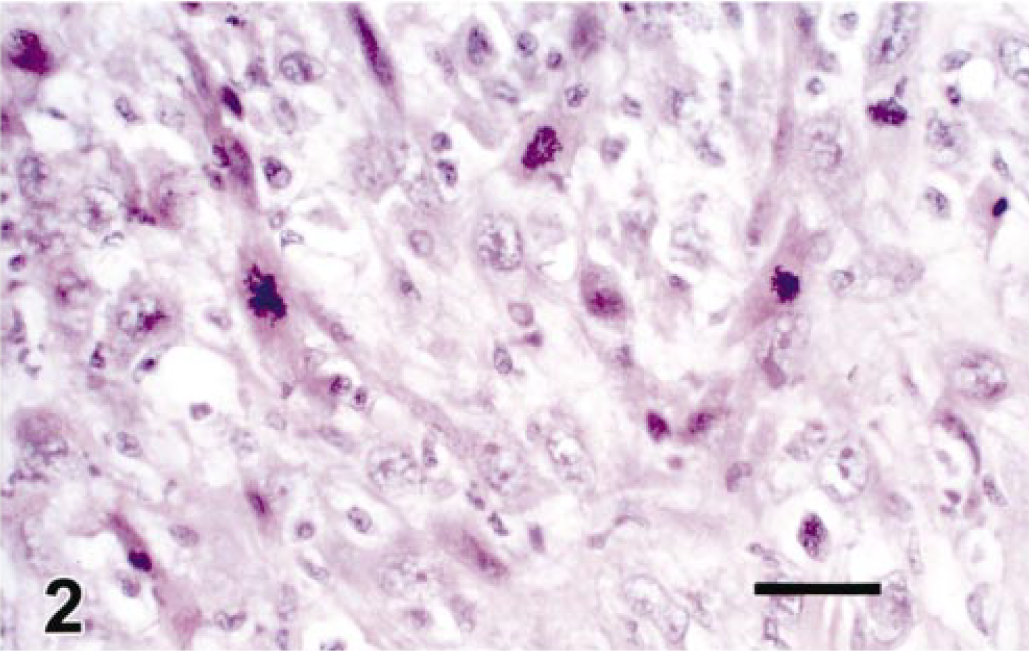
Pulmonic valve mass. Bands of pleomorphic spindle-shaped cells with small to large nuclei. Many mitotic figures noted. HE. Bar = 10 µm.
The neoplastic cell population extended along the pulmonary artery, partially replacing the tunica media. The luminal surface of the involved pulmonary artery had adherent fibrin adjacent to tumor cells.
The lung nodules observed on gross postmortem examination corresponded to multiple blood vessels filled with neoplastic cells, similar to those previously described in the pulmonary valve (Fig. 3). Vessels were enlarged and tortuous, with total obliteration of the vascular lumina and compression of blood vessel walls. In some situations, vessel walls could not be identified and neoplastic cells adjoined lung parenchyma.
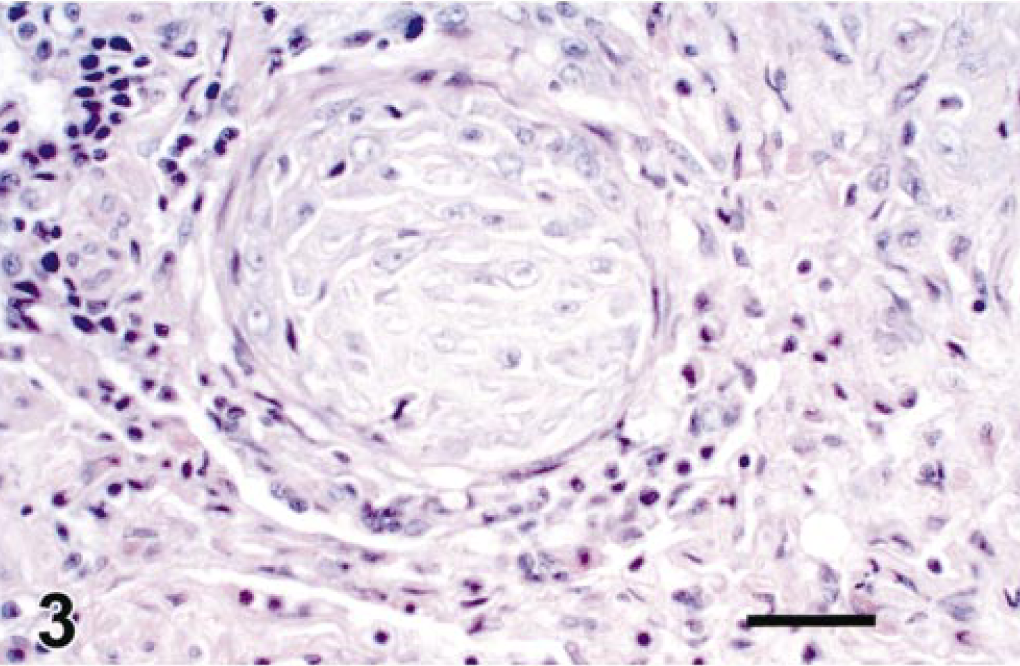
Lung. Blood vessels plugged with neoplastic cells and surrounded by mononuclear inflammatory cells. HE. Bar = 40 µm.
Immunocytochemistry revealed positive intracytoplasmic staining of neoplastic cells for alpha-smooth muscle actin (Fig. 4) and vimentin, but cells were negative for desmin, CD31, factor VIII, and S100. Morphological and immunocytochemical findings supported a diagnosis of an intrapulmonary artery leiomyosarcoma.
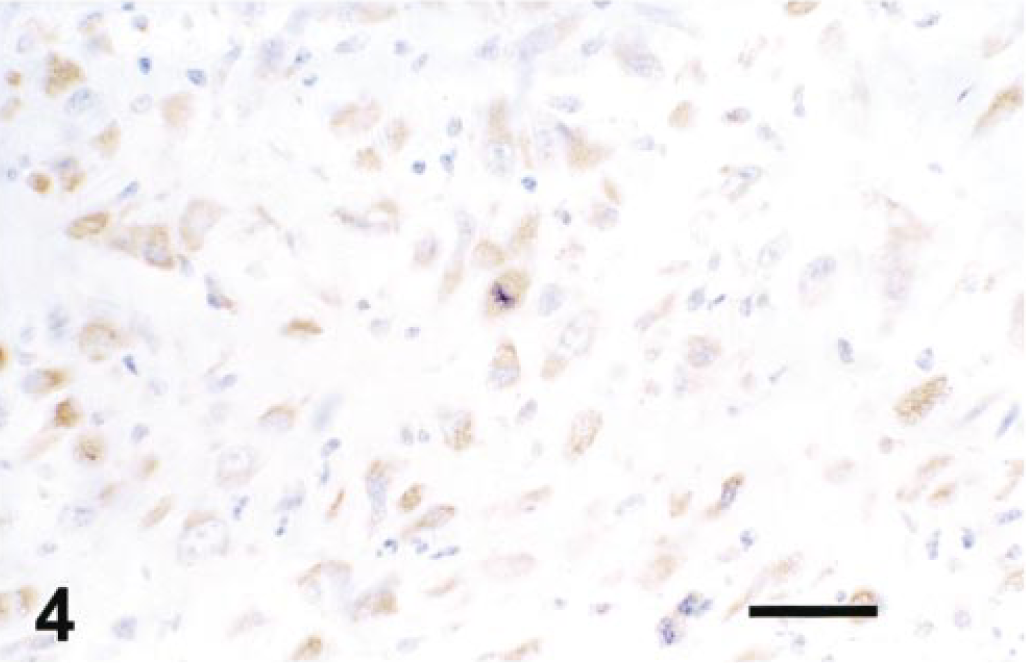
Lung. Alpha-smooth muscle actin expression in tumor cells. Avidin-biotin peroxidase complex method. Mayer's hematoxylin counterstain. Bar = 10 µm.
The ventricular surface of the aortic valve was thickened due to an irregular nodular proliferation of subendothelial fibroblasts. In this reaction, there were regions of hemorrhage and focal aggregations of hemosiderin-laden macrophages, lymphocytes, and plasma cells. A focal loss of endothelium and fibrin deposition was noted. There was evidence of organization of this thrombus, but accompanying surface inflammation and bacterial colonies were absent. It was speculated that such features were consistent with a nonbacterial thrombotic endocarditis. Other findings included splenic nodular hyperplasia, chronic interstitial nephritis, and a renal cystadenoma.
Leiomyosarcomas are rare in domestic farm and companion animals. 5 In the dog, they have been documented in the alimentary tract, urinary tract, spleen, and liver. 3 7 While leiomyosarcomas of the alimentary and urinary tract are generally accepted as originating from muscle layers within gastric, intestinal, and bladder walls, it is speculated that vascular smooth muscle may also be the site of origin in certain cases of leiomyosarcoma. 5 This is supported by a recent report of an intravascular leiomyosarcoma involving the skin of a cat. 6 To the authors' knowledge, this present case is the first report of a leiomyosarcoma of vascular smooth muscle of the pulmonary artery, and its presentation parallels many of the features observed in primary pulmonary artery sarcomas in man.
A review of 45 cases of intraluminal pulmonary artery sarcomas in man by Baker and Goodwin 2 documented sarcomas involving the pulmonary trunk, left and right pulmonary arteries, pulmonic valves, and right ventricular outflow tract. Lung involvement was noted in 25% of cases. The gross postmortem appearance of the present case was similar in many respects to those documented by Baker and Goodwin. 2 The tumor manifested as a large mass within the lumen of the pulmonary trunk and left pulmonary artery with long intraluminal protrusions to involve the pulmonic valve and distal portions of the left pulmonary artery. The intimate relationship of the wall of the pulmonary trunk to the mass coupled with the histological appearance supported an origin from vascular smooth muscle of the pulmonary trunk wall. The absence of tumor in the right pulmonary artery was interesting and paralleled findings of Baker and Goodwin, 2 who reported that, while intraluminal pulmonary sarcomas often involve the left pulmonary artery, no involvement of the right artery was noted. Another interesting feature was that metastatic spread was noted only in the right lung and may suggest that, while the right pulmonary artery did not provide an environment suitable for tumor growth, the lack of impediment of blood flow did facilitate tumor spread to the right lung fields.
In man, many pulmonary artery sarcomas have been reported as undifferentiated. 2 Leiomyosarcomas were documented in approximately 25% of cases and the remainder were either classified as rhabdomyosarcomas, chondrosarcomas, fibromyxosarcomas, fibrosarcomas, mesenchymomas, osteosarcomas, mxyosarcomas, or hemangiosarcomas. In general, the differentiation of leiomyosarcomas from fibrosarcomas and rhabdomyosarcomas may sometimes be difficult. 5 While the presence of myofibrils and centrally located strap-like nuclei is helpful, the absence of striated fibrils is not conclusive. In the present case, histological features of cell type and fascicular growth patterns, supported by the expression of vimentin and alpha-smooth muscle actin, indicate a mesenchymal tumor with smooth muscle differentiation. A nonsupporting feature was the absence of desmin expression. However, cases of leiomyosarcoma with desmin negativity are observed in man and are related to the fact that subpopulations of nonneoplastic smooth muscle cells may also fail to express desmin. 13 Absent or weak desmin expression has also been documented in canine and feline leiomyosarcomas. 1 6 While the expression of vimentin and alpha-smooth muscle actin in the absence of desmin expression would warrant the inclusion of hemangiopericytoma, undifferentiated sarcomas, and glomangiomas in the differential diagnosis, 8 9 14 the histological features, presentation, and behavior of this neoplasm would not be consistent with these tumors.
In the present case, malignancy was confirmed based on the invasive growth of the tumor, metastatic spread, and cellular morphology.
Footnotes
Acknowledgements
The authors acknowledge the technical assistance of Ms. A. Jenkins and Mr. I. McMillan, University of Glasgow, Ms. S. Worrell, Mr. C. King, Mr. B. Cloak, Ms. M. McCullagh, and Mr. M. Garrett, University College Dublin, and the advice and support of Dr. H. Larkin, Ms. C. Skelly, and Professors B. Sheahan, B. Jones, and C. Bellenger.
