Abstract
Spontaneous hepatic neoplasms were identified in two adolescent (<5 years of age) male cynomolgus monkeys (
Keywords
Spontaneous hepatocellular tumors are rare in nonhuman primates (Table 1). In a survey of necropsy records from 1,065 monkeys (32 species), a single case of hepatocellular neoplasia (hepatocholangiocellular carcinoma) was described in an adult African green monkey (
Spontaneous hepatic neoplasms reported for nonhuman primates.

The affected monkeys were part of larger shipments from Covance Research Products, Texas Primate Center (Alice, TX). These monkeys were born and raised within primate colonies in Vietnam. Birth dates were included with animal order information. Both monkeys were serologically negative for simian retrovirus, simian immunodeficiency virus, herpes B virus, herpes simian virus 1, and simian T-lymphotrophic virus 1.
Each monkey was enrolled in a separate drug safety study involving dissimilar drug candidates. These drug candidates were not recognized carcinogens or hepatotoxins in rodents or monkeys, based on findings of studies employing higher or similar drug doses or in studies of longer duration. Pretest physical and ophthalmic exams, electrocardiograms, blood pressures, and clinical chemistry and hematology parameters were within normal ranges for both animals. Comprehensive necropsies and histopathologic evaluation of all major tissues were performed for both animals. Pathologic alterations relevant to hepatic tumors were not identified in extrahepatic tissues. Representative sections from each major liver lobe of each animal were fixed in 10% neutral buffered formalin and processed routinely for histopathology.
Liver sections were stained with hematoxylin and eosin (HE). Additional stains applied to representative tissue sections from each animal were Masson's trichrome stain, periodic acid–Schiff (PAS) stain with and without diastase treatment, Wilder's reticulin stain, and Turnbull's blue (TB) and Prussian blue (PB) stains for iron. Representative tissue sections were stained immunocytochemically by the immunoperoxidase method with mouse anti-human monoclonal antibodies directed against low molecular weight cytokeratin (LMWCK [reacts with cytokeratins 7 and 8]; Becton Dickinson, San Jose, CA), cytokeratin (CK) 7 (Dako Corp., Carpinteria, CA), CK 8 (Dako Corp.), CK 18 (Dako Corp.), CK 19 (Dako Corp.), fibronectin (Novocastra, Vector Labs, Burlingame, CA), Ki-67 (MIB-1 antibody, Immunotech, Westbrook, ME), and carcinoembryonic antigen (CEA; Cell Marque, Austin, TX) and with rabbit anti-human polyclonal antibodies directed against high-molecular-weight CK (HMWCK [reacts with CK 6 and CK 9]; Dako Corp.), broad-spectrum CK (BSCK [reacts with CKs 3 or 4, CK 5, CK 6, CK 8, CKs 14 or 15, and CK 16]; Dako Corp.), glutathione S-transferase-
Summary of results∗ of selected staining procedures on nonneoplastic liver and hepatocellular tumors in cynomolgus monkeys.

− = negative; + = faintly positive; + + = moderately postive; + + + = strongly positive.
Staining of periportal hepatocytes.
Staining of cells of primitive ducts.
Staining of scattered foci of hepatocytes.
At termination, monkey No. 1 was 58 months old and had been treated for 4 weeks with an experimental metalloproteinase inhibitor (low dose). Grossly, a round raised tan mass 18 mm in diameter was embedded in the diaphragmatic surface of the left medial liver lobe. The remaining hepatic parenchyma was grossly normal.
Microscopically, the mass consisted of solid sheets and trabeculae (often multilayered) of neoplastic hepatocytes (NHs) partially surrounded by a fibrous capsule. Tumor cells were larger (3–6-fold) than nonneoplastic hepatocytes (NNHs) in adjacent tissue, but NHs maintained many characteristic hepatocellular features, including abundant eosinophilic cytoplasm and well-demarcated cell borders (Fig. 1). Cell nuclei were round to oval, of various sizes, enlarged, and vesicular, with prominent single or multiple nucleoli. Cellular pleomorphism was pronounced in many tumor areas and was typified by cells with irregular shape and multilobulated and/or multiple nuclei. Mitoses were numerous (up to 8/20× field) and many were atypical. At tumor borders, NHs interrupted the continuity of the fibrous capsule and were present in adjacent hepatic parenchyma. Parenchyma surrounding the neoplasm was multifocally compressed and atrophic and contained mixed inflammatory cell infiltrates, including large and small mononuclear cells and neutrophils. The solitary tumor was classified as an HCC. Metastases were not evident in other tissues.
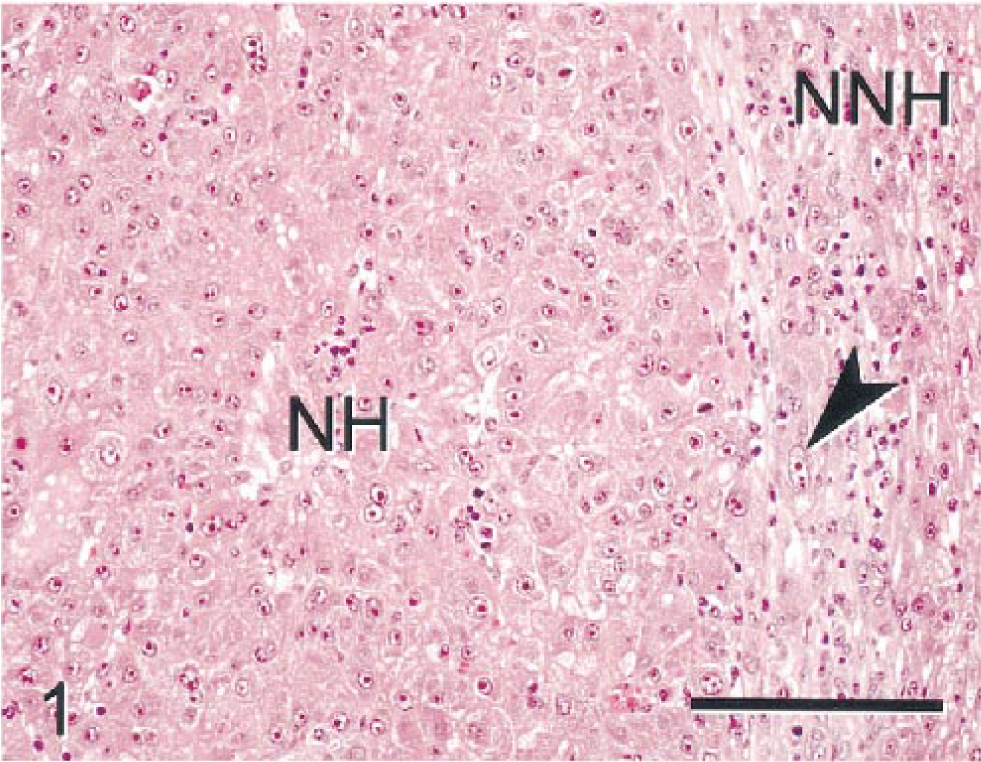
Hepatocellular carcinoma; monkey No. 1. Increased cell size and cellular atypia are evident in neoplastic hepatocytes (NHs). Compressed and atrophic nonneoplastic hepatocytes (NNHs) are adjacent to tumor. Atypical neoplastic cells were present within the tumor capsule/nonneoplastic tissue in many regions of the tumor boundary (arrowhead). HE. Bar = 150 µm.
PAS stains with and without diastase demonstrated depletion of PAS-positive material (glycogen) in NHs relative to NNHs. LMWCK and CK 18 stained most NHs, although staining intensity varied from none to moderate. Bile duct epithelium and hepatocytes in some periportal regions also stained in nonneoplastic parenchyma. Although most NHs were negative for CK 8, GST, CEA, and AFP, scattered collections of NHs stained intensely with these markers (Figs. 2, 3). CK 8, CK 18, and GST also stained bile duct epithelia in nonneoplastic parenchyma. BSCK, CK 7, and CK 19 did not react with NHs but reacted strongly with nonneoplastic bile duct epithelia. HMWCK weakly stained nonneoplastic bile ducts but did not react with neoplastic parenchyma. Ki-67 (MIB-1) reacted with large numbers of NHs (Fig. 4), whereas staining of NNHs was not observed. Staining was largely limited to the nucleus in most NHs, although many hepatocytes in mitoses had staining of chromatin and cytoplasm. Lesser numbers of stromal and inflammatory cells in neoplastic and nonneoplastic parenchyma and bile duct epithelial cells in nonneoplastic tissue showed nuclear staining with Ki-67. A fine meshwork of fibronectin staining occurred around NHs and NNHs. Pericellular staining for fibronectin within the tumor was more prominent than in nonneoplastic parenchyma, perhaps because of increased amounts of pericellular stroma. The fibrous capsule surrounding the tumor and normal hepatic stroma also stained intensely for fibronectin. The cytoplasm of NHs did not stain with fibronectin, but scattered aggregates of NNHs had intense diffuse cytoplasmic staining (Fig. 5). NHs and NNHs did not stain significantly for iron with TB or PB stains. Thus, staining pattern for iron was not useful in differentiating neoplastic from nonneoplastic cells.
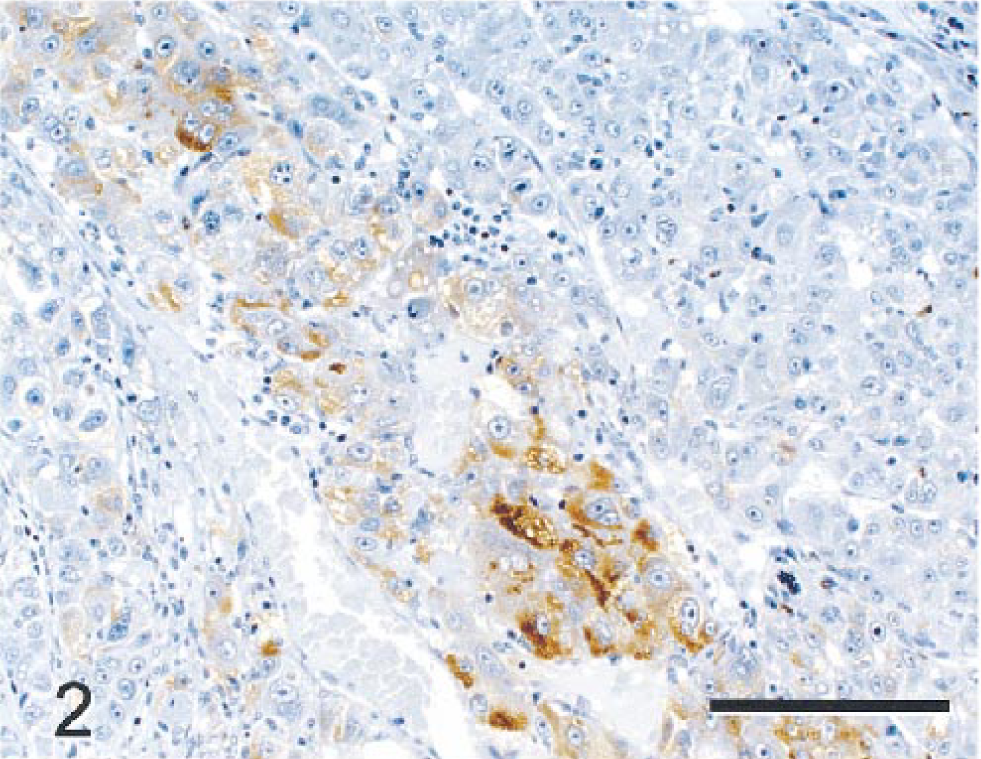
Hepatocellular carcinoma; monkey No. 1. Carcinoembryonic antigen (CEA)-positive tumor cells are mixed with tumor cells not staining with this immunocytochemical marker. Immunocytochemical stain for CEA, diaminobenzidine chromogen, counterstained with hematoxylin. Bar = 150 µm.
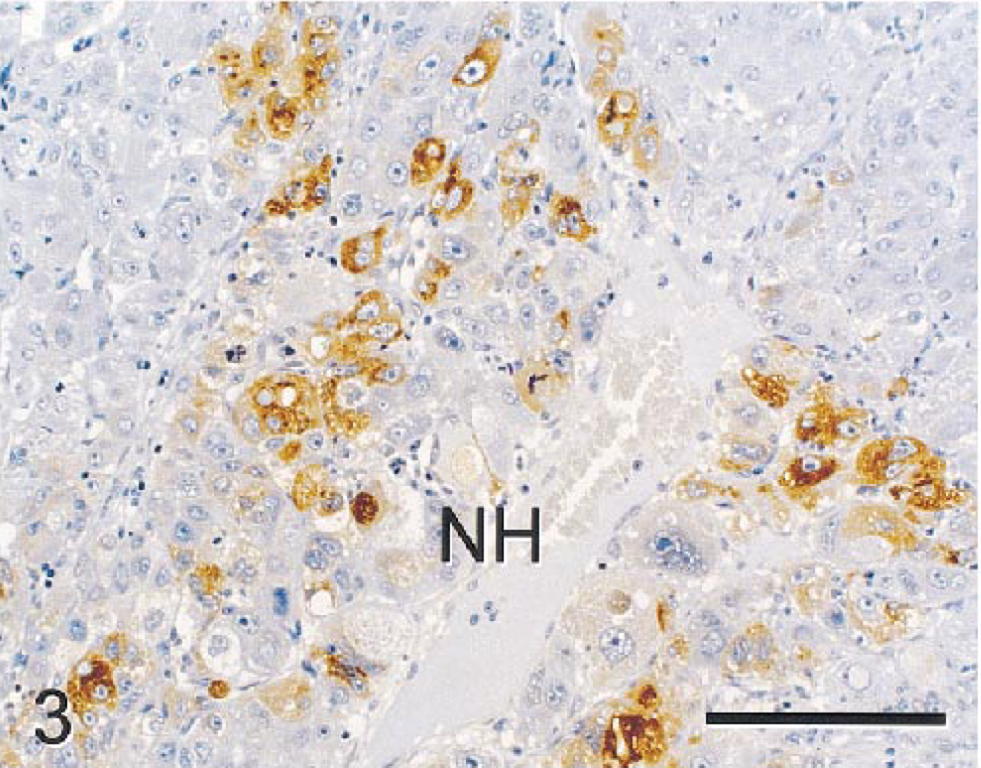
Hepatocellular carcinoma; monkey No. 1. α-Fetoprotein (AFP)-positive tumor cells are mixed with tumor cells negative for this immunocytochemical marker. Notice prominent pleomorphic features of some tumor cells. Immunocytochemical stain for AFP, diaminobenzidine chromogen, counterstained with hematoxylin. Bar = 150 µm.
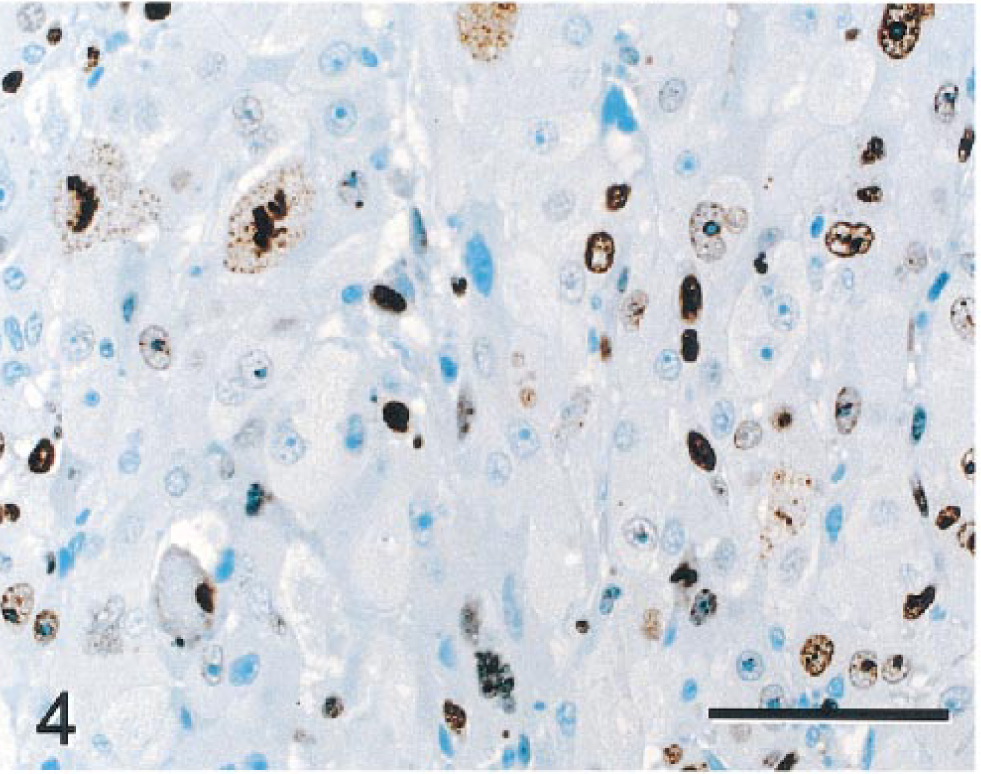
Hepatocellular carcinoma; monkey No. 1. Numerous neoplastic hepatocytes have nuclear staining with Ki-67 (MIB-1), reflecting cell proliferation. Cells in mitosis have staining of cytoplasm and chromatin. Nonneoplastic hepatocytes did not stain with this marker (not demonstrated). Immunocytochemical stain for Ki-67(MIB-1), diaminobenzidine chromagen, and counterstained with hematoxylin. Bar = 70 µm.
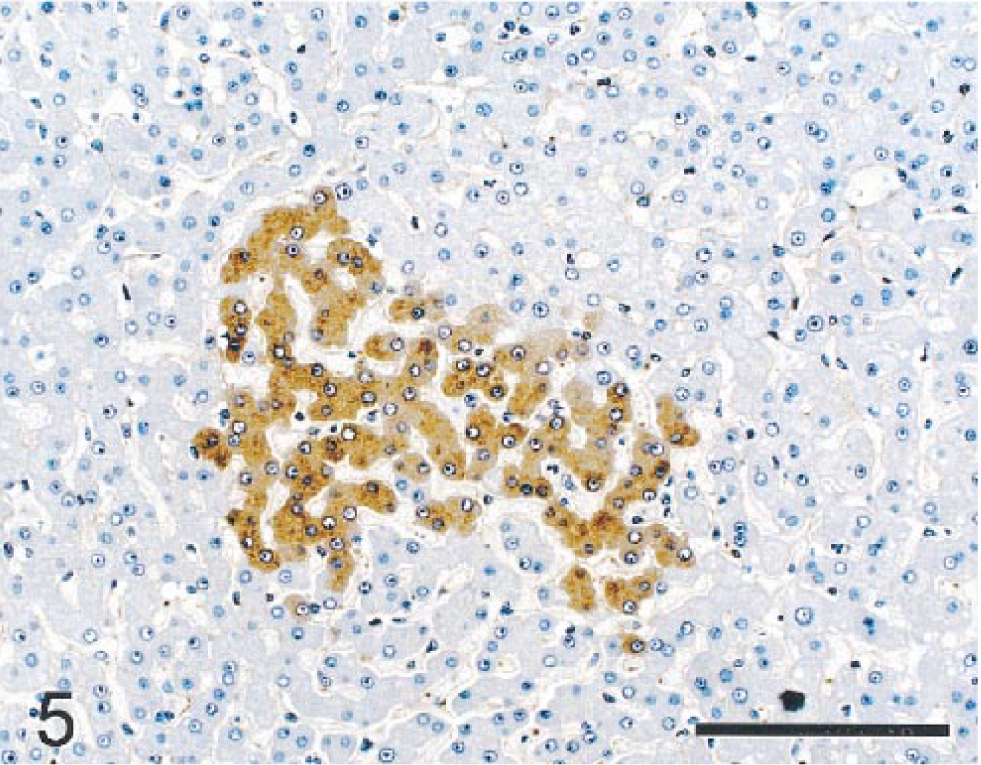
Nonneoplastic hepatic parenchyma; monkey No. 1. Aggregates of nonneoplastic hepatocytes had intensely positive cytoplasmic staining for fibronectin. A fine meshwork of fibronectin-positive material is also present lining sinusoids. Immunocytochemical stain for fibronectin, diaminobenzidine chromagen, counterstained with hematoxylin. Bar = 150 µm.
Other liver lobe sections contained small aggregates of morphologically altered hepatocytes (hepatocellular foci or foci of cellular alteration) within normal appearing parenchyma (Fig. 6). Foci were of various sizes but were smaller than hepatic lobules. Cells within these foci were of uniform character and differed from surrounding hepatocytes by size (smaller or larger) and/or tinctorial properties (vacuolated, clear, or eosinophilic). Cellular pleomorphism/atypia was not evident in foci, and adjacent parenchyma was not compressed. Scattered portal regions had minimal to mild fibrosis and bile duct hyperplasia confined to expanded portal regions.
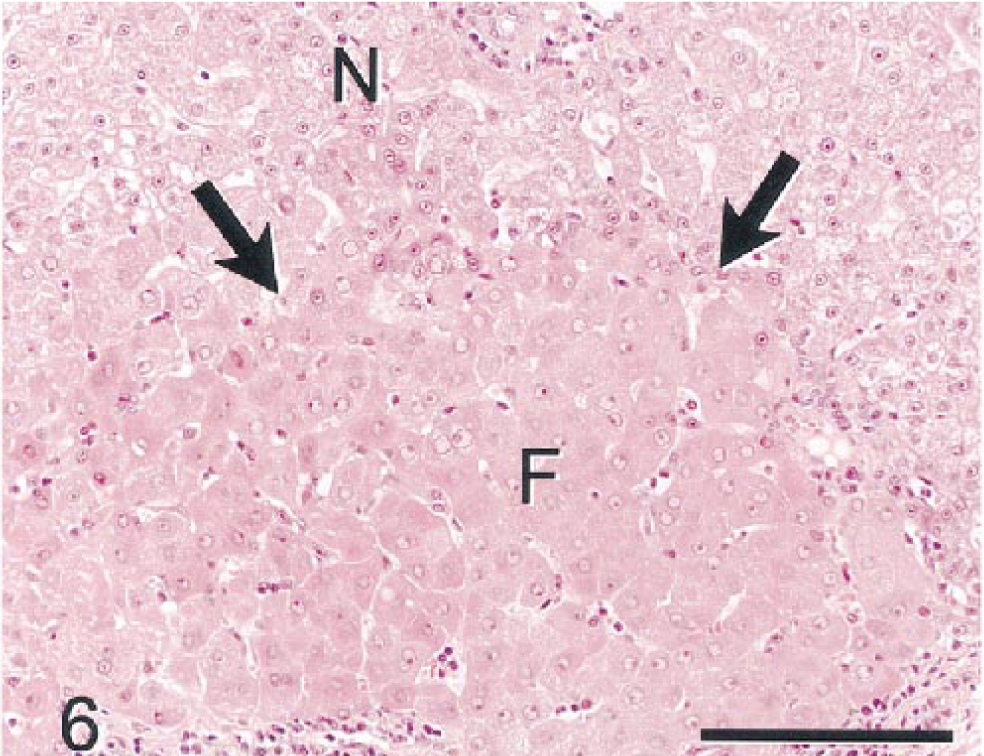
Nonneoplastic hepatic parenchyma; monkey No. 1. Focus of hypertrophic eosinophilic hepatocytes (F) with enlarged vesicular nuclei is surrounded by hepatocytes of normal size and tinctorial properties (N). There is no compression of parenchyma surrounding the focus of altered hepatocytes (arrows). HE. Bar = 150 µm.
At termination, monkey No. 2 was 59 months old and had been treated for 2 weeks with a test compound being developed as a cytostatic agent for inhibition of cell proliferation. Grossly, the liver was abnormally firm with an irregular rough surface. The cut surface showed irregular pale discoloration and lobulation in fibrotic parenchyma. Discrete neoplastic nodules or masses were not apparent grossly.
Microscopically, several sizes of poorly delineated to partially circumscribed nodules of atypical to bizarre NHs were evident in multiple liver lobes. NHs within nodules formed solid sheets lacking normal trabeculae or sinusoidal spaces (Fig. 7). These cells were enlarged (3–6-fold), had homogenous or vacuolated eosinophilic cytoplasm, and were highly pleomorphic in size and shape. Cell nuclei were enlarged, round to irregularly shaped, and vesicular with prominent nucleoli and marginated chromatin. Multinucleated cells and cells with multilobulated nuclei were common in all nodules. Only a single mitotic figure was evident in the tumor nodules. One tumor nodule had irregularly shaped primitive ducts and cell clusters formed by neoplastic cells that were smaller and more basophilic than most NHs. Many of the cells forming these primitive ducts and cell clusters stained with antibodies that stained nonneoplastic bile duct epithelium (BSCK, GST, CK 7, CK 8, and CK 18), indicating that the tumor cells were potentially of bile duct origin or differentiation (Fig. 8). Nonneoplastic parenchyma was compressed at margins of most nodules. Metastases were not evident in extrahepatic tissues. All but one of the tumor nodules were classified as HCCs. The tumor with cells forming primitive ductlike structures was classified a mixed hepatocholangiocellular carcinoma (MHC).
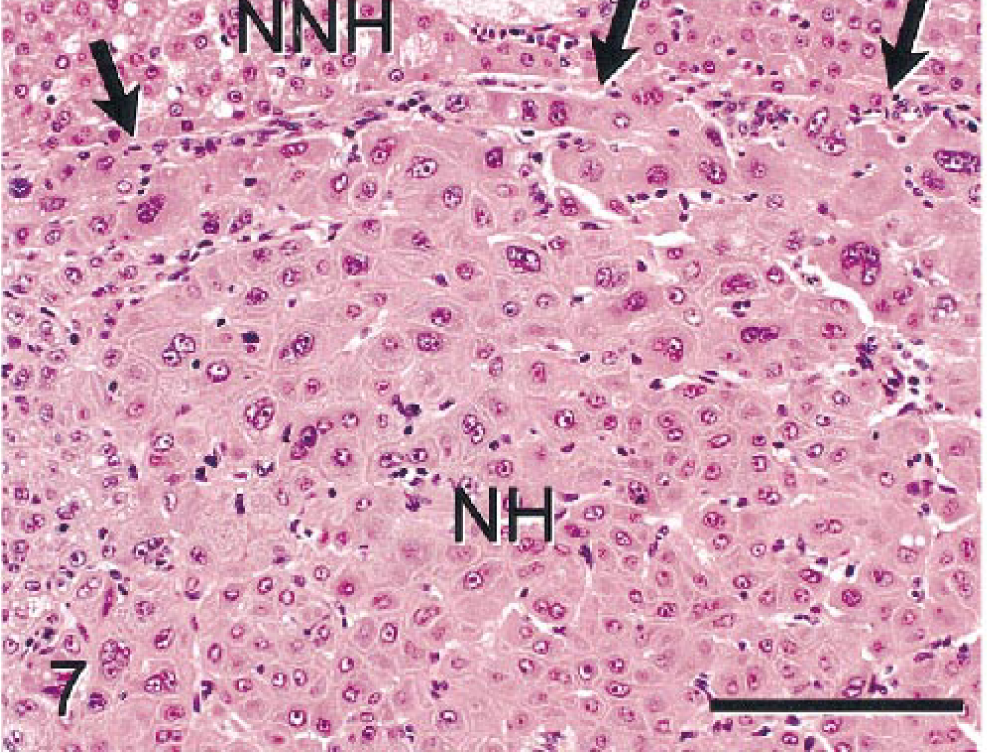
Mixed hepatocholangiocellular carcinoma; monkey No. 2. Solid sheets of atypical neoplastic hepatocytes (NHs) compress adjacent nonneoplastic hepatocytes (NNHs). Tumor cells had eosinophilic to vacuolated cytoplasm and enlarged pleomorphic nuclei. Arrows indicate border between NHs and NNHs. HE. Bar = 150 µm.
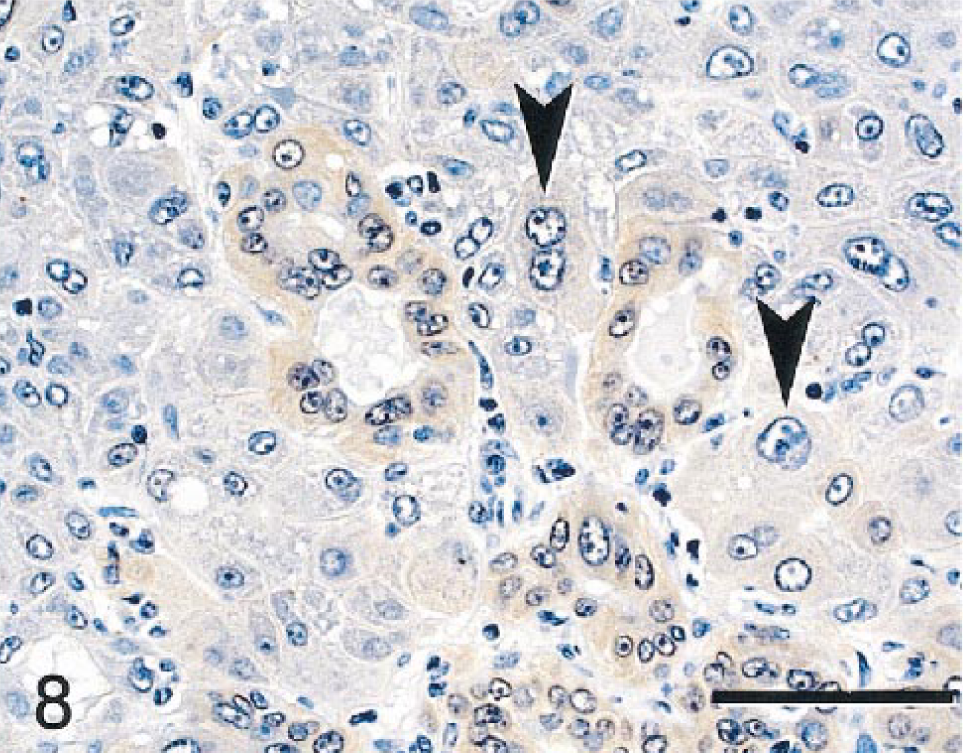
Mixed hepatocholangiocellular carcinoma; monkey No. 2. Primitive ductlike structures within the tumor had modestly postitive staining with glutathione S-transferase-
NHs and (regenerative) NNHs stained poorly for glycogen with PAS and did not stain with TB, HMWCK, BSCK, CK 19, CEA, or AFP. NHs of the MHC stained prominently with LMWCK (Fig. 9A,B), CK 7, CK 8, CK 18 (Fig 10), and GST, but intensity of staining of individual tumor cells within the mass varied from none to marked. In nonneoplastic parenchyma, LMWCK and CK 18 staining was enhanced in periportal hepatocytes. Bile duct epithelium and basophilic tumor cells forming primitive ducts stained with BSCK, LMWCK, CK 7, CK 8, CK 18, and GST (Fig. 8). Fibronectin staining of pericellular stroma within neoplastic nodules was slightly more pronounced than in regenerative hepatic lobules. As in monkey No. 1, scattered aggregates of hepatocytes within the nonneoplastic parenchyma had moderately intensive staining with fibronectin. Nonneoplastic bile ducts stained intensely with CK 19, whereas primitive bile ductlike structures in the MHC did not. Ki-67 staining was not apparent in neoplastic cells or in regenerative hepatocytes and bile duct epithelium in nonneoplastic tissues. Few stromal cells and mononuclear inflammatory cells had nuclear staining. Tumor nodules and foci of atypical cells did not stain for iron with PB in contrast to much of the nonneoplastic parenchyma, which was slightly to moderately siderotic. Staining with TB was uniformly negative in neoplastic and nonneoplastic tissue.
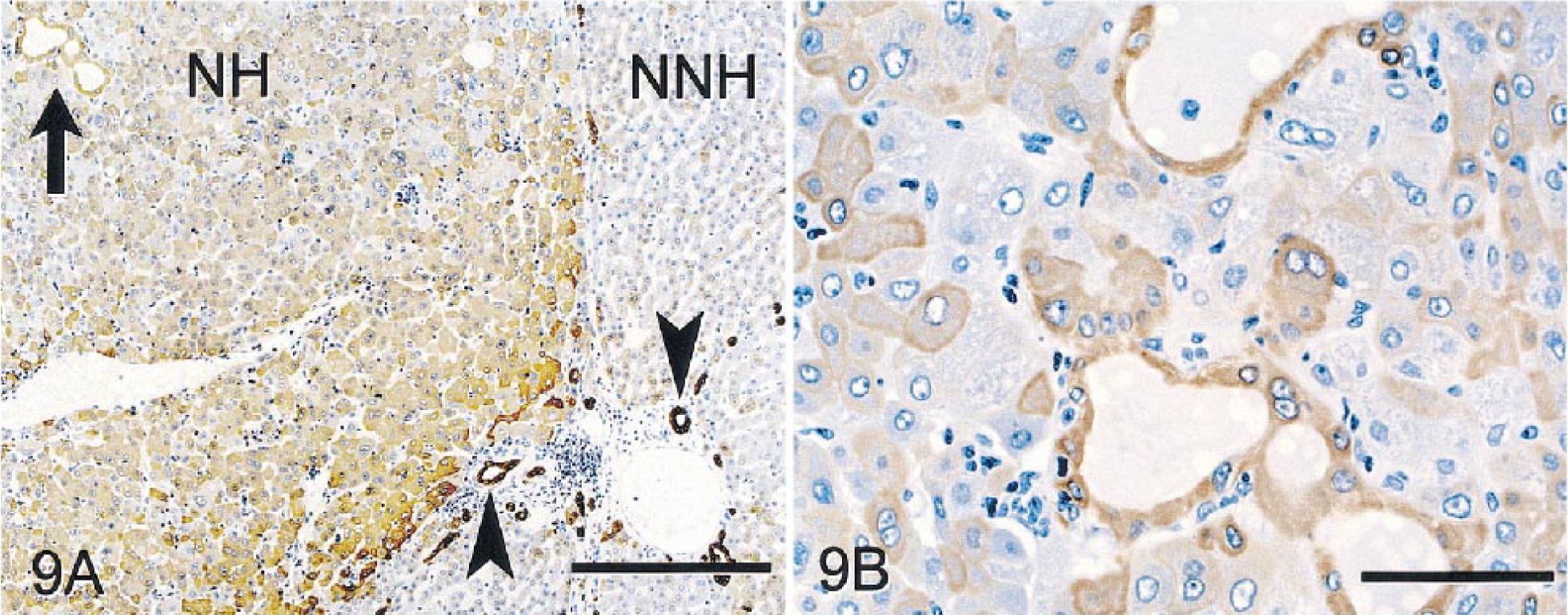
Mixed hepatocholangiocellular carcinoma; monkey No. 2. Immunocytochemical stain for LMWCK, diaminobenzidine chromagen, counterstained with hematoxylin.
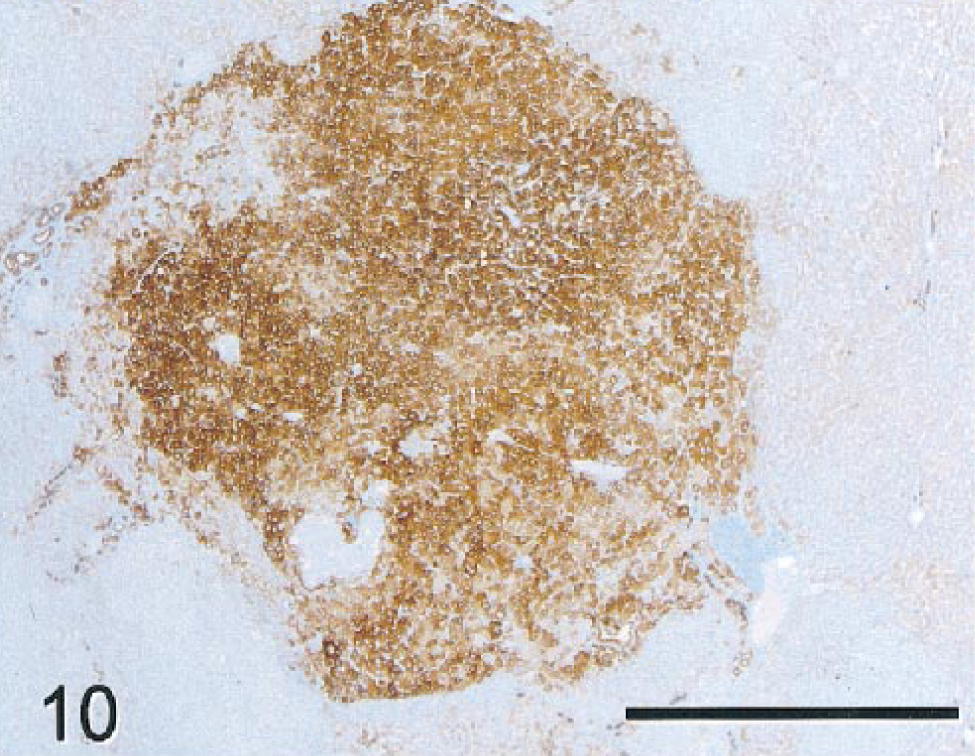
Mixed hepatocholangiocellular carcinoma; monkey No. 2. Neoplastic nodule stained intensely positive for CK 18. Nonneoplastic hepatocytes in periportal regions stained less intensely than neoplastic cells with this marker. Bile ducts also had strong staining comparable to that in the neoplastic nodule (not shown). Immunocytochemical stain for CK 18, diaminobenzidine chromagen, counterstained with hematoxylin. Bar = 1,000 µm.
The nonneoplastic hepatic parenchyma was diffusely cirrhotic, with prominent regenerative hepatocellular hyperplasia (Fig. 11). Regenerative hepatocytes were normal to slightly reduced in size and were arranged as single-layered trabeculae separated by well-defined sinusoidal spaces. Although architecture of trabeculae and sinusoids was preserved, lobular architecture was distorted by bands of fibrosis extending from portal regions and by the irregular length and angles of hepatic cords. Bile duct hyperplasia was prominent in fibrotic regions, and small bile ducts were evident in periportal parenchyma. Discrete altered hepatocellular foci (foci of cellular alteration) were also present in the regenerative parenchyma.
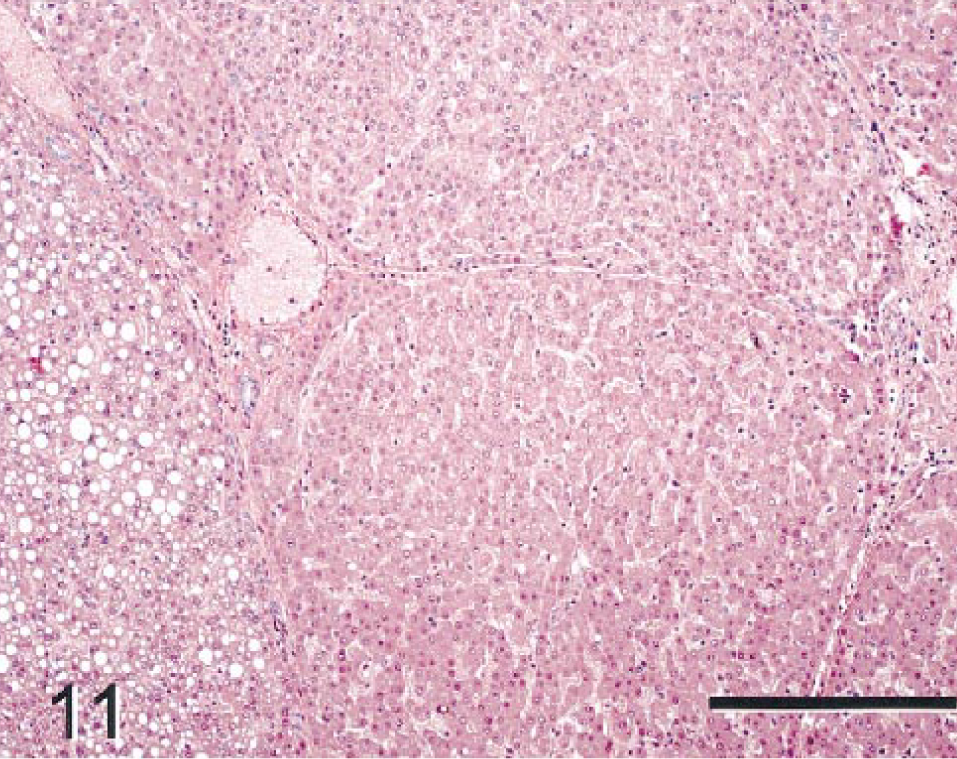
Nonneoplastic hepatic parenchyma; monkey No. 2. Regenerative hepatocellular hyperplasia and portal fibrosis are shown. Regenerative hepatocytes in some areas are vacuolated but do not have conspicuous cellular atypia. The trabecular/sinusoidal architecture was preserved throughout the section, although lobular architecture was distorted by fibrous septa. HE. Bar = 300 µm.
Spontaneous hepatic neoplasia has not been described in cynomolgus monkeys and most reports of hepatic neoplasia in nonhuman primates are of tumors in aged animals. Neoplasms in this report were in comparatively young cynomolgus monkeys. Cynomolgus monkeys are considered sexually mature, defined histologically by complete maturation of the male reproductive tract, by 4–4.5 years of age. Both of these monkeys were <5 years of age at necropsy and were considered adolescent to young adult relative to traditional landmarks of human development. No other cases of hepatic neoplasia were identified in nearly 2,000 wild-caught or captive-bred cynomolgus monkeys used at our facility, supporting the low occurrence of hepatic neoplasia in this species.
Histologic features of the neoplasm in these cases are consistent with morphologic characteristics defining HCC and MHC in other species. In monkey No. 2, neoplastic nodules were multicentric involving several liver lobes. Spontaneous multicentric hepatic carcinomas within a background of chronic cirrhosis have not been reported in monkeys. Widespread cirrhosis with regenerative hepatocellular hyperplasia in this animal was probably related to chronic or chronic-active injury. Although minimal and multifocal bile duct hyperplasia and portal fibrosis have been seen at low incidence in monkeys at our facility, the diffuse and moderate degenerative response observed in monkey No. 2 has not been observed. The original insult to the liver of this monkey could not be determined. Certain hepatotoxins, such as aflatoxin B1 and diethylnitrosoamine (DEN), have been shown experimentally to cause widespread chronic liver disease and neoplasia in monkeys. 14 These monkeys had not been treated with experimental compounds prior to initiation of our studies. We could not rule out exposure of this monkey to natural hepatic carcinogens such as certain aflatoxins or hepatitis C virus (nonA-nonB hepatitis virus) in the country of origin.
Ki-67 is expressed in cells throughout the cell cycle but is not expressed in resting cells. Thus, this immunocytochemical marker can serve as a useful index of cell proliferation. The paucity of mitoses and Ki-67 (MIB-1) labeling in regenerative tissue and the neoplastic nodules of monkey No. 2 are consistent with the cytostatic properties of the test compound given this animal. This situation contrasts with the florid Ki-67 labeling and numerous mitoses occurring in tumor cells of monkey No. 1, in which the drug candidate was not cytostatic in nature.
Limited immunocytochemical procedures have been conducted for experimentally induced liver tumors of monkeys, and were conducted here to further characterize changes in these spontaneous neoplasms. GST reactivity, normally found in biliary epithelium, has been used as a marker for preneoplastic hepatic lesions and primary hepatic tumors in rodents. 13 The utility of this marker in nonhuman primates has not previously been explored. In these monkeys, GST reactivity was evident in normal biliary epithelia, in relatively small regions of NHs in monkey No. 1, and in primitive ductlike structures and NHs in monkey No. 2. The vast majority of NHs in both cases did not stain with this marker.
CKs represent a family of intermediate filaments forming a substantial part of the cellular cytoskeleton in epithelium and epithelial tumors. 6 Certain anti-CK antibodies, including CKs 7, 8, 18, and 19, have been used to characterize HCCs induced by DEN in monkeys. 6 These investigators indicated that most HCCs displayed one of three CKs proported to be found in hepatocytes (CK 8, CK 18, or CK 19), but seldom did tumors express all three. Some DEN-treated animals had heterogenous (negative and positive) CK staining within the same tumor. Also, CKs that are normally expressed by bile ducts only (CK 7 and 19) were expressed by NHs in a significant number of DEN-induced tumors. We also found that certain CKs normally displayed by biliary epithelium (but not hepatocytes) were displayed in regions of NHs. LMWCK, CK 7, CK 8, and CK 18 stained NHs in at least some tumor nodules. Intensity of LMWCK, CK 7, CK 8, and CK 18 staining varied among tumor nodules, and positive and negative regions occurred within individual tumors. AFP and CEA are commonly considered markers of undifferentiated cells. 10 AFP and CEA staining were not evident in normal or regenerative hepatocytes of these animals but were multifocally positive in NHs of monkey No. 1 only. Staining patterns for LWMCK, CK 7, CK 8, CK 18, AFP, and CEA demonstrated altered immunophenotypic expression by tumor cells. This immunophenotypic expression is heterogeneous within tumor nodules and differs among cases.
Fibronectin is produced by hepatocytes and is a normal component of hepatic stroma. Fibronectin production is increased in proliferating hepatocytes 5 and has been demonstrated in HCCs in humans. 8 Staining of pericellular matrix around NHs was increased in both monkeys. Cytoplasmic staining with fibronectin did not occur in NHs, but aggregates of NNHs stained intensely. These cells were not associated with recognizable increases in pericellular matrix. The significance of such cytoplasmic staining in NNHs is uncertain. These cell clusters may represent a novel type of altered hepatocellular foci not previously recognized by routine histochemical staining.
Altered hepatocellular foci (foci of cellular alteration) histologically reminiscent of those seen in rodents were present within the nonneoplastic parenchyma in both monkeys. These foci have not been reported as a spontaneous finding in monkeys, but we have observed these foci sporadically in treated and control cynomolgus monkeys at our institution. The etiology of these foci and their relationship to the development of neoplasia in primates is unknown. In rodents, these types of morphologically altered hepatic foci are considered potentially preneoplastic lesions in the process of chemical carcinogenesis. 7
These cases of hepatic neoplasia add to the limited number of reports of spontaneously occurring hepatic tumors in nonhuman primates and document the spontaneous occurrence of this condition in an additional primate species. Immunocytochemical characterization of these tumors demonstrated variability in phenotypic expression of NHs within and among tumors and provides the first evaluation of such markers in spontaneously occurring hepatic tumors of nonhuman primates. The description of these hepatocellular neoplasms and their extensive phenotypic, histochemical, and immunohistochemical characterization should serve as a useful reference for researchers who may encounter similar lesions in nonhuman primate species.
