Abstract
Sixty male BALB/c mice were inoculated intraperitoneally with either a sublethal or a lethal dose of Burkholderia mallei China 7 strain, then killed at multiple time points postinoculation. Histopathologic changes were qualitatively similar in both groups and consisted of pyogranulomatous inflammation. In sublethal study mice, changes were first seen at 6 hours in mediastinal lymph nodes, then in spleen, liver, peripheral lymph nodes, and bone marrow at day 3. These changes generally reached maximal incidence and severity by day 4 but decreased by comparison in all tissues except the liver. Changes were first seen in lethal study mice also at 6 hours in mediastinal lymph nodes and in spleens. At day 1, changes were present in liver, peripheral lymph nodes, and bone marrow. The incidence and severity of these changes were maximal at day 2. In contrast to sublethal study mice, the incidence and severity of the changes did not decrease through the remainder of the study. The most significant difference between the two groups was the rapid involvement of the spleen in the lethal study mice. Changes indicative of impaired vascular perfusion were more frequently seen in the sublethal study mice. Our findings indicate that mice are susceptible to B. mallei infection and may serve as an appropriate model for glanders infection in a resistant host such as human beings. Additionally, by immunoelectron microscopy, we showed the presence of type I O-antigenic polysaccharide (capsular) antigen surrounding B. mallei.
Glanders is a zoonotic disease caused by Burkholderia (formerly Pseudomonas) mallei, a gram-negative, aerobic, non–spore-forming bacillus. Earlier reports 13 stated that B. mallei lacks a capsule; however, there is now evidence to the contrary. 19 This organism is thought to be an obligate mammalian pathogen 22 with solipeds serving as the reservoir for infection. 1 5 While glanders is still endemic in areas of Asia, the Middle East, Northern Africa, as well as various Mediterranean regions, 2 13 the Western Hemisphere is currently free of the disease. 2
Natural glanders infections occur primarily in horses, donkeys, and mules, 15 but most mammals have some degree of susceptibility. 11 Natural disease is occasionally seen in goats, sheep, dogs, and cats, 5 17 and it has been rarely reported in carnivores that have eaten infected horse meat. 5 17 Cattle, pigs, 5 and birds 11 are very resistant to natural disease. Experimentally, most domesticated animals can be infected, with the exception of cattle, pigs, and rats. 11 Guinea pigs and hamsters are the laboratory rodents most susceptible to glanders infection. 11 Miller et al. found that, while there appears to be a variance in susceptibility among individual guinea pigs, hamsters are more uniformly susceptible to infection with B. mallei. 15 Vyshelesskii 24 stated his studies showed that cats were most susceptible to the species tested, followed by guinea pigs, rabbits, field mice, and moles. Laboratory mice are only slightly susceptible to infection unless the organisms are given in very high doses. 18
Even in times when the worldwide prevalence of glanders in horses was high, the occurrence of this disease in human beings was rare. 17 There have been no documented epidemics of glanders in humans. 10 However, the finding of glanders-associated nodules at autopsy in large numbers of people with equine contact suggests that the prevalence of mild subclinical infections may be higher than previously suspected. 21 Although human susceptibility to B. mallei has not been studied in depth, the organism has proven to be highly infectious in laboratory settings. 17 22 It has been stated that there are few organisms as dangerous to work with as the glanders bacillus. 17 21
We previously reported a time-course study of glanders pathogenesis in Syrian hamsters inoculated intraperitoneally (IP) with B. mallei. 8 Because of their uniformly high susceptibility, hamsters have been used extensively as models for glanders infection. 15 However, as models for infection in human beings, hamsters fall short because people are moderately resistant to glanders. 10 24 To study glanders pathogenesis in an animal model with resistance/susceptibility similar to human beings, we chose laboratory mice, specifically the BALB/c strain. To elucidate glanders pathogenesis in mice, we performed a time-course study of histopathologic, ultrastructural, and immunohistologic findings in BALB/c mice inoculated IP with either a sublethal or lethal dose of the organism. To our knowledge, this is the first time-course study of glanders in mice.
Materials and Methods
Inoculum
Burkholderia mallei 23344 (China 7 strain) was obtained from the American Type Culture Collection (ATCC, Rockville, MD). It was cultivated on 4% glycerol tryptone agar medium (Difco Laboratories, Inc., Detroit, MI) at 35–37C, as recommended by ATCC. This strain was serially passaged three times in hamsters and designated GB15.1–3. The optical density and a standard curve were used to determine the colony-forming units (CFU) per milliliter of a 25-ml overnight culture of B. mallei. Cultures were suspended in Hank's balanced salt solution to achieve a concentration of 106 CFU/ml. The inoculum was administered IP in the left lower abdominal quadrant with a 25-gauge needle. We previously determined the LD50 of B. mallei GB15.1–3 in BALB/c mice to be 7 × 105 organisms (D. Waag and D. Brown, unpublished data). In the sublethal study, a challenge dose of 106 CFU was used, while in the lethal study, the dose was 107 CFU.
Bacterial isolation
Bacterial loads per milliliter of blood and per gram of spleen, liver, and lung were determined by plating known volumes on 4% glycerol tryptone agar (Difco Laboratories, Inc.) plates and counting colonies.
Animals
Sixty male, specific pathogen-free, BALB/cAnNCr mice, weighing between 20 and 25 g and approximately 6–8 weeks old, were randomly divided into two dosage groups. In the first (sublethal) group, 40 mice were injected IP with 0.5 ml of an inoculum containing 106 CFU of B. mallei. In the second group (lethal), 20 mice were injected IP with 0.5 ml of an inoculum containing 107 CFU of B. mallei. To serve as age- and sex-matched controls, 12 mice were injected IP with 0.5 ml of Hank's balanced salt solution alone.
In our LD50 trials, BALB/c mice proved to be similarly susceptibility to B. mallei as other inbred strains, such as C3H/HeN, DBA/1, and A/J, while being much more susceptible than C57BL/6 (D. Waag and D. Brown, unpublished data). We chose the BALB/c mouse for this study because of the wide availability of genetic variants (such as knockouts) in this strain. Some of these variants may prove to be valuable in future glanders pathogenesis studies.
Preparation of antisera
Rabbit anti-B. mallei serum was prepared as previously described. 8
Necropsy
Beginning at 6 hours postinoculation (PI), five sublethal study mice were killed in a CO2 chamber. This procedure was repeated daily through day 7. In the lethal study, five mice were killed at 6 hours and day 1 PI, six mice at day 2, and four mice (one killed, three found dead) at day 3, the final time point of the study. At each time point in each study, one control mouse was killed. Necropsy procedures were conducted as previously described. 8
Histopathology
Fixed tissue samples of all major organs/tissues from each mouse were routinely processed as previously described. 8
Immunohistochemistry
Immunohistochemical procedures for both light microscopic and electron microscopic examination were performed as previously described. 8 On paraffin-embedded tissues, an antibody dilution of 1:15,000 was used, while for immunoelectron microscopy, either a 1:10,000 or 1:15,000 dilution was employed.
Electron microscopy
Tissue processing for transmission electron microscopy (TEM) was performed as previously described. 8
Immunoelectron microscopy on cultured bacteria
Burkholderia mallei, strains China 7 and Budapest (not animal passaged), were prepared as previously described 3 for electron microscopic study of capsule. Briefly, the bacilli were grown overnight at 37 C in Luria-Bertani (LB) broth containing 4% glycerol. The LB broth consisted of 10 g of tryptone (Difco Laboratories, Inc.), 5 g of yeast extract (Difco Laboratories, Inc.), 5 g of NaCl (Sigma Chemical Co., St. Louis, MO), and 40 ml of glycerol (Sigma Chemical Co.). Distilled water was added to make a volume of 1 liter, and the pH was adjusted to 6.8. One hundred microliters of the saturated culture was inoculated into 3 ml of fresh medium, and the culture was grown until it reached middle logarithmic phase, approximately 5 hours. Culture was placed upon 400-mesh nickel grids for 2 minutes, blocked for 30 minutes with 0.5% BSA in PBS, and incubated with a 1:100 dilution of the primary antibody for 30 minutes. The primary antibody consisted of polyclonal rabbit antisera produced against an unbranched homopolymer of B. pseudomallei with the structure −3)-2-O-acetyl-6-deoxy-B-D-manno-heptopyranose-(1-, also known as B. pseudomallei type I O-antigenic polysaccharide (O-PS) (kindly provided by Dr. Don Woods, University of Calgary Health Sciences Centre, Calgary, AB, Canada). Grids were washed three times with PBS/BSA and incubated for 30 minutes in a 1: 50 dilution of goat anti-rabbit IgG conjugated with 5 nm colloidal gold (Sigma Chemical Co.). Grids were washed again three times with PBS/BSA, three times with distilled water, and examined by TEM as described above.
Results
Bacterial cell counts in tissue
Bacterial counts in blood, spleen, liver, and lung for mice in the sublethal and lethal studies are presented in Figs. 1 and 2, respectively. At most time points, the greatest number of bacilli were present in the spleen, then in the liver. For sublethal study mice, at 6 hours and day 1 PI, B. mallei was cultured from the spleens and livers of all mice and from the blood and lung from only one of four (different) animals. On days 2 and 3, the blood from two animals was culture positive, but after day 3, blood cultures were negative. B. mallei was not cultured from any liver samples at day 4 but was cultured from the livers of one mouse each at days 5 and 6 and two mice at day 7. Glanders bacilli were cultured from all spleens from 6 hours to day 2 and also at day 6. On days 3–5, bacilli were only found in two of four mice and at day 7 in three of four. The lung was culture positive in one mouse each at 6 hours and days 1, 2, and 6.
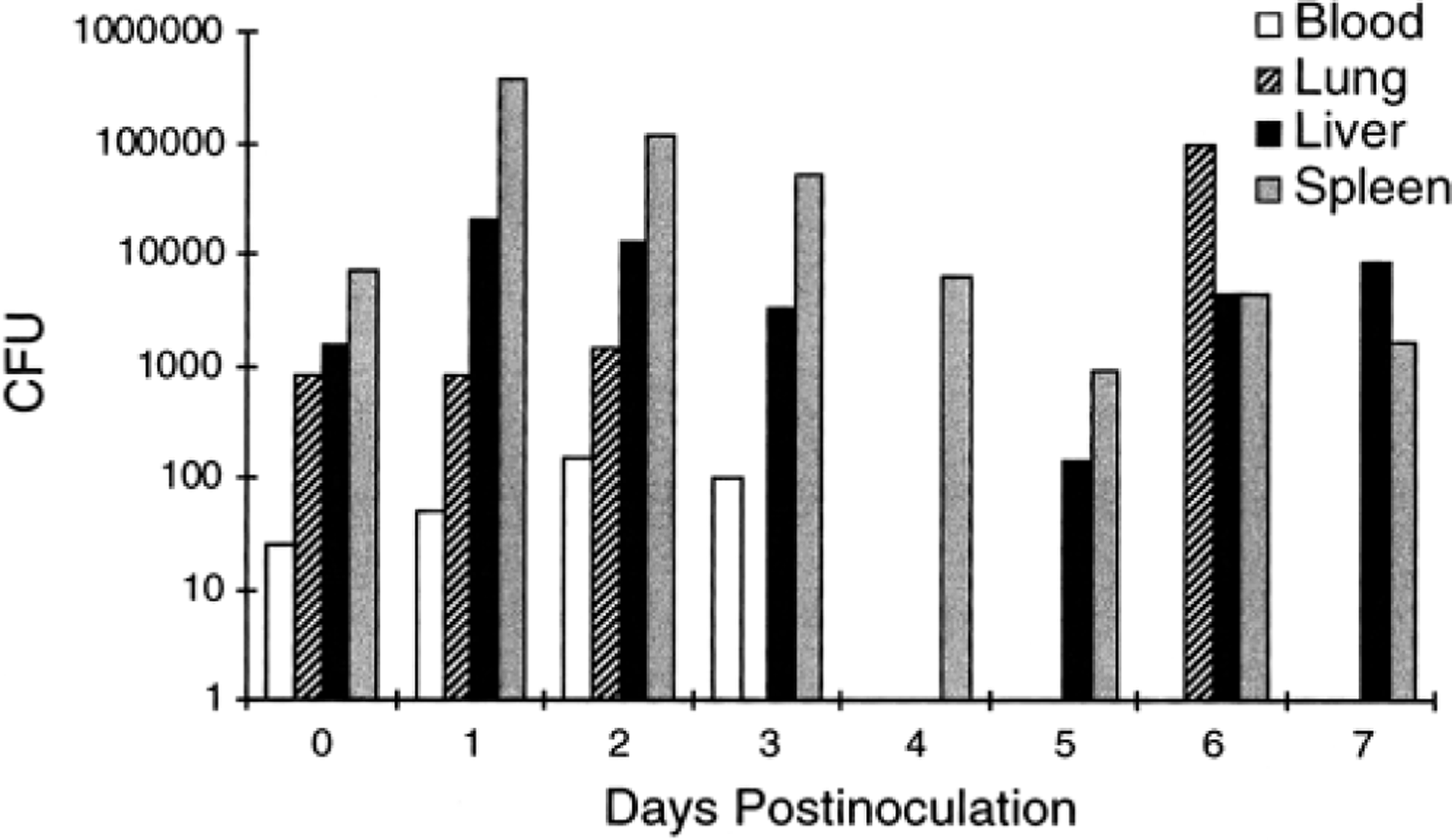
Time course effects on bacteria counts in mouse blood (CFU/ml), lung (CFU/g), liver (CFU/g), and spleen (CFU/g) after IP inoculation with 106 CFU of B. mallei. Glanders bacilli were cultured from all organs at 6 hours PI, but histopathologic changes in the spleen and liver were not found until day 1. Glanders-related changes were absent from lung throughout the study. Cultured blood bacteria were not present after day 3. Mice exhibited signs of clinical recovery beginning at day 5, although cultured bacteria in the spleen and liver were still high. Vertical axis indicates mean values; absence of a bar indicates no organism cultured. For days 1–7, n = 5.
Time course effects on bacteria counts in mouse blood (CFU/ml), lung (CFU/g), liver (CFU/g), and spleen (CFU/g) after IP inoculation with 107 CFU of B. mallei. Glanders bacilli were cultured from all organs at 6 hours PI. Although histopathologic changes were present in the spleen at 6 hours PI, they were not present in the liver until day 1 and were never seen in the lung (Table 2). Note that, with the exception of blood, cultured bacteria increased in lung, liver, and spleen until the end of the study. Cultured blood bacteria were not present after day 2. Vertical axis indicates mean values. For 6 hours PI and day 1, n = 5; for day 2, n = 6; for day 3, n = 4.
For lethal study mice, B. mallei was cultured from most of the sampled tissues. Exceptions include negative culture in the liver and lung of one of four mice (different animals) at day 2. Bacilli were cultured from lung, liver, and spleen in the one surviving day 3 animal, but the blood was culture negative.
Clinical observations
In sublethal study mice, clinical signs of infection were first observed at day 1 and consisted of slight ruffling of fur. At day 2 PI, mice also began huddling behavior, which increased in severity through day 4 PI. At day 5, two of the five mice in the group appeared clinically normal and active, and at day 7, all mice were clinically normal except for slightly ruffled fur. In lethal study mice, clinical signs of infection were first observed at day 1 and consisted of moderately ruffled fur and slight huddling behavior. At day 2, all mice were lethargic, somnolescent, and ataxic when stimulated to move. By day 3, three of the four mice in the group were dead; the fourth was moderately active but ataxic. All control mice were clinically normal throughout the study.
Gross pathologic findings
In sublethal study mice, the first gross pathologic changes were mild fibrinous adhesions of spleen to the greater curvature of the stomach, with frequent entrapment of the pancreas seen at day 1. Adhesions became progressively more severe with increasing time in the study. At day 2, mild splenomegaly was present in all mice and increased in severity at the later time points to as much as three times normal size in some mice. Also at day 2, moderate thymic atrophy was present in all mice and remained a consistent finding in all mice throughout the study. At day 5 miliary, pinpoint white foci were seen beneath Glisson's capsule and on the cut surface of the liver of one mouse. In lethal study mice, the first gross pathologic change was mild splenomegaly seen in one mouse at day 1. Mild splenomegaly as well as mild peritonitis involving the spleen and stomach were found in all mice from day 2 on. Also at day 2, the livers of three mice were pale, with a diffuse mottled appearance in one of these.
Histopathologic findings
The incidence of glanders-induced histopathologic changes in sublethal and lethal study mice at the various time points is summarized in Tables 1 and 2, respectively. In general, the most prevalent change in affected tissues in both dosage groups consisted of infiltration of polymorphonuclear (PMN) leukocytes in multiple tissues in the very early (6 hours PI) stages of infection, followed by the infiltration of equal or greater numbers of macrophages. This infiltrate was variably dense and well demarcated and was only rarely organized into discrete pyogranulomas.
Histopathic changes in mice serially killed after intraperitoneal inoculation with 106 CFU of Burkholderia mallei.

Number of mice with histopathologic changes at that time point.
n = 5 at all time points.
Abbreviations: MEDLN = mediastinal lymph node; MESLN = mesenteric lymph node; SMLN = submandibular lymph node; INLN = superficial inguinal lymph node.
Histopathologic changes in mice serially killed after intraperitoneal inoculation with 107 CFU of Burkholderia mallei.
Number of mice with histopathologic changes at that time point.
Abbreviations: MEDLN = mediastinal lymph node; MESLN = mesenteric lymph node; SMLN = submandibular lymph node; POPLN = popliteal lymph node; INLN = inguinal lymph node.
Sublethal study mice
(i) Spleen. Splenic changes were first seen at day 1 PI and consisted of infiltration of PMN leukocytes primarily in the marginal zone of the white pulp and a miliary distribution of pyogranulomatous inflammation in the red and white pulp. After day 1, the inflammation was purely pyogranulomatous with occasional foci of leukocyte necrosis. This infiltrate was multifocal, moderately well localized (Fig. 3), and the severity of this lesion was most intense at days 2 and 3 and then gradually diminished. Lymphoid depletion of the white pulp was present in the spleens of all mice at days 3 and 4 and in four of five mice at days 5–7. Pyogranulomatous capsulitis and/or steatitis were present in all mice at day 1, in three mice at day 2, and in only one mouse at day 3. Thrombosis and occasional vasculitis of splenic veins were present in one mouse each at days 3 and 6, two mice at day 5, and three mice at day 7.
Spleen; BALB/c mouse, day 3 after IP inoculation with 106 CFU of B. mallei. Note that this pyogranulomatous inflammatory cell infiltrate is moderately well demarcated from cells in the surrounding red pulp, a typical finding in spleens from sublethal study mice. In contrast, similar infiltrates tended to coalesce and were less discrete in lethal study mice. HE. Bar = 27 µm.
(ii) Liver. Liver changes were first seen at day 1 and consisted of a miliary distribution of a pyogranulomatous inflammation that was most severe at day 3 and did not diminish with time. This lesion is shown in Fig. 4 in our previous report. 8 Generally, necrosis of infiltrated leukocytes was rare except at day 2, when extensive leukocyte necrosis was present in four of five mice. At day 2, acute to subacute portal hepatitis was present in two mice, with the incidence increasing to include all mice on days 6 and 7. Hepatocellular coagulative necrosis, without cellular inflammatory infiltrate, was present in four mice at day 2, decreasing in incidence to none from day 6 on. Thrombosis, with or without vasculitis, was found in from one to two animals each from day 3 on (Fig. 4). In three mice, from days 3 to 7, there was vasculitis alone. At all time points, half of the mice with areas of hepatocellular coagulative necrosis had obvious venous thrombosis.
Liver; BALB/c mouse, day 3 after IP inoculation with 106 CFU of B. mallei. There is a thrombus within a portal vein. The general boundaries of the thrombus are indicated with arrowheads while the residual (or possibly recanalized) lumen of the vein is marked with an L. We believe the pale eosinophilic hepatocytes present in the lower half of this field to be injured as the result of ischemia. They are contrasted by more normal hepatocytes in the top half of the field. Attending the injured hepatocytes is a mild infiltrate of PMN leukocytes and macrophages. The changes present in this micrograph are typical of those found in sublethal study mice in the later stages of the infection. HE. Bar = 30 µm.
(iii) Mediastinal lymph node/mediastinum. Changes in the mediastinal lymph nodes at 6 hours consisted of single to multiple foci of acute inflammation, usually in the subcapsular cortex (Fig. 5) but also in medullary cords. At days 1 and 2, the character of the infiltrate was pyogranulomatous and was seen in the subcapsular cortex of most mice. The severity of the infiltrate diminished at the later time points. Other less common lesions present in a few animals included cortical lymphoid depletion, thrombosis, and pyogranulomatous mediastinitis.
Intrathoracic lymph node; BALB/c mouse, 6 hours after IP inoculation with 106 CFU of B. mallei. In the subcapsular cortex (C), note the focally extensive infiltrate of PMN leukocytes with some admixed macrophages between arrowheads. This infiltrate extends down from the surface of the node (arrow), perhaps down an axial sinus. A few infiltrated leukocytes are also scattered in the subcapsular sinus. The subcapsular sinus is marked in several places with asterisks. HE. Bar = 33 µm.
(iv) Mesenteric/other lymph nodes/thymus. A pyogranulomatous infiltrate was present in the paracortex of mesenteric lymph nodes in one mouse each at days 1, 3, 4, and 6, in two mice at day 5, and in three mice at day 2. Cortical lymphoid depletion was common at days 2 and 3 but was rare thereafter. Other lesions noted less often in submandibular and superficial inguinal lymph nodes included cortical lymphoid depletion, acute to pyogranulomatous inflammation, and venous thrombosis. In the thymus, cortical lymphoid depletion was frequently present in mice from days 2 through 5, with a lower incidence thereafter. There were no glanders-related lesions found in popliteal lymph nodes.
(v) Bone/bone marrow. Changes in the bone marrow of the femur/tibia consisted of multiple foci of acute myelitis in the metaphyseal and, to a lesser degree, epiphyseal marrow of femur/tibia in four mice at day 2. From day 3 on, the infiltrate was also present within the diaphyseal marrow and was mostly pyogranulomatous (Fig. 6). Unlike other tissues, leukocyte necrosis was a frequent finding at all time points (Fig. 6). Mild pyogranulomatous myelitis was also found in the marrow pockets of turbinate bone and calvarium of one mouse each at days 3 and 5.
Bone marrow of long bone, hindlimb; BALB/c mouse, day 3 after IP inoculation with 106 CFU of B. mallei. Note the extensive focus of pyogranulomatous inflammatory cell infiltrate, which is surrounded by more normal medullary cellular components. Within the inflammatory infiltrate, there is scattered necrosis of leukocytes and presumably also medullary tissue. HE. Bar = 27 µm.
(vi) Male reproductive. Involvement of the testis, epididymis, and accessory sexual organs generally consisted of a pyogranulomatous inflammatory infiltrate within the tunica vaginalis and only rarely involved the parenchyma of the tissue. The incidence was highest in accessory sex organs/epididymis at days 1 and 3.
Lethal study mice
(i) Spleen. Splenic changes were first seen at 6 hours and consisted of minimal to mild acute inflammation in the marginal zone of the white pulp and rarely the red pulp. At day 2, the character of the infiltrate was pyogranulomatous and, with time, the involvement of the red pulp increased but the infiltrates were never well localized. Mild to moderate lymphoid depletion of the white pulp was present in two mice each at days 1 and 2, but at day 3, was present in four of five mice and was moderate to marked in severity.
(ii) Liver. Liver changes were first seen at day 1 and were similar to those seen in sublethal study mice, peaked in severity at day 2 and were not changed with time thereafter. Inflammatory infiltrates were often large enough to result in considerable hepatocyte loss. Multifocal zones of hepatocellular coagulative necrosis were a very frequent finding at all time points. Mineralization of degenerated and necrotic hepatocytes (Fig. 7), occasionally evident at the early time points, was observed in all mice at days 2 and 3. At day 1, three of five mice had thrombi in hepatic veins. Mild granulomatous portal hepatitis was seen in two of five mice at day 3.
Liver; BALB/c mouse, day 3 after IP inoculation with 107 CFU of B. mallei. There is a cluster of necrotic hepatocytes with mineralization of the peripheral cells. This lesion was a common finding in day 2 and 3 lethal study mice. HE. Bar = 27 µm.
(iii) Mediastinal lymph node/mediastinum. Changes in the mediastinal lymph nodes were first seen at 6 hours and consisted of minimal to mild infiltrates of PMN leukocytes within the subcapsular sinuses. The infiltrates were more severe and deeper within the node at day 1 but were pyogranulomatous from day 2 on. Cortical lymphocyte depletion was seen only occasionally in mice from day 1 on. Necrotizing pyogranulomatous mediastinitis was present in one mouse at day 1.
(iv) Mesenteric/other lymph nodes/thymus. Pyogranulomatous inflammation was present in the cortex of mesenteric lymph nodes of one mouse at day 1 and all mice thereafter. Cortical lymphoid depletion was an infrequent finding. Likewise, acute to pyogranulomatous lymphadenitis of submandibular lymph nodes was found in one mouse at day 1 and all mice thereafter, but lymphoid depletion was infrequent. Lymphadenitis of popliteal lymph nodes was seen in 3 mice at days 1 and 3 and all mice at day 2. In superficial inguinal lymph nodes, lymphadenitis was present in all mice from day 1 on. Cortical lymphoid depletion was rare in these nodes. Acute thymitis was seen in two mice each at days 2 and 3. Lymphoid depletion of the thymic cortex was also consistently seen from day 1 on and was of marked severity at days 2 and 3.
(v) Bone/bone marrow. Changes in bone marrow were similar to those in sublethal study mice but were first seen as early as day 1. These lesions peaked in severity at day 2 and did not diminish. By day 2, the infiltrate and necrosis were extensive and little residual myeloid tissue remained. Giemsa stain revealed abundant bacilli in the marrow from several day 2 mice (Fig. 8). Bacilli were usually scattered in short chains of 2–4 cells but were occasionally clustered in sheaves. Most bacteria appeared to be intracellular (Fig. 8) within macrophages and/or reticuloendothelial (RE) cells.
Bone marrow of long bone, hindlimb; BALB/c mouse, day 2 after IP inoculation with 107 CFU of B. mallei. The arrow points out a cluster of intracellular glanders bacilli, presumably within a macrophage. The light-staining cells at the top of the field did not appear to be viable, while viable, residual marrow cells at the bottom of the field stain more intensely. Giemsa stain. Bar = 14 µm.
(vi) Other. Mild pyogranulomatous steatitis was seen in the adipose tissue surrounding the testis and epididymis in two mice at day 2.
Immunohistochemistry
In general by light microscopy, B. mallei antigen was present most often in the cytoplasm of macrophages and/or RE cells, although several exceptions are noted below. Immunolabeled cells were always most numerous in areas of pyogranulomatous inflammation. Typically there was diffuse cytoplasmic staining of labeled cells. The profiles of individual bacteria could be identified only rarely and with great difficulty.
Sublethal study mice
By light microscopy, immunolabel was first noted at 6 hours PI in mediastinal lymph nodes within the subcapsular sinuses and in the subcapsular cortex. The distribution of immunopositive cells increased at day 1 throughout the cortex and into the medullary sinuses. In the spleen and liver at day 1, immunopositive cells were present in areas of inflammation. In the bone marrow at day 2, scattered immunopositive RE cells were found in the metaphysis, within macrophages in areas of inflammation in the diaphysis, and only rarely in the epiphysis.
Lethal study mice
In mediastinal lymph nodes at 6 hours PI, immunolabel was similar to that seen in sublethal study mice, with distribution increasing at the later time points. In spleens at 6 hours PI, macrophages and/or RE cells containing antigen were occasionally found in the marginal zone but at day 1 PI were widely scattered throughout the red pulp. In livers at 6 hours PI, antigen was occasionally seen in Kupffer cells, and at day 1 PI, it was frequently present in inflamed foci. In hepatic veins of one mouse, immunolabeled bacilli were observed within intravascular macrophages, and several appeared to be extracellular. In the bone marrow at day 1, there were loosely aggregated immunopositive cells throughout the marrow, and at day 2, the immunolabeling was very intense. Loosely aggregated immunopositive cells were scattered in inflamed areas of popliteal lymph nodes at both days 1 and 2.
Electron microscopy
In both sublethal and lethal study mice, bacilli were only rarely found in spleens and livers and were usually intracellular PMN leukocytes or macrophages that appeared undamaged. Intracytoplasmic bacilli were surrounded by a thin, uniform, electron-lucent halo presumed to have been the capsule lost during processing. This halo is illustrated in Fig. 14 of our previous report. 8 By immunoelectron microscopy (IEM), colloidal gold particles were distributed evenly around the periphery of bacteria, never within the protoplasm of the bacillus. Typical distribution of colloidal gold is shown in Fig. 15 of our previous report. 8 Occasionally, IEM was useful in identifying a distorted, degenerate bacillus that would have otherwise not been recognized.
Immunoelectron microscopy on cultured bacteria
Immunolabeling of glanders bacilli with the rabbit anti-B. pseudomallei type I O-PS revealed the O-PS to be arranged in a thick, uniform zone surrounding each bacillus (Fig. 9). The immunolabeled zone was consistent with the capsule and was generally thicker in Budapest strain bacilli (Fig. 9) than in China 7 (not shown). The density of colloidal gold particles was similar with both strains.

Transmission electron photomicrograph. Cultured B. mallei Budapest strain. Note the wide, uniform zone of colloidal gold particles around the bacterial cell indicating immunolabeling of the capsule. Rabbit anti-B. pseudomallei type I O-PS serum, goat anti-rabbit IgG conjugated to 5 nm gold. Bar = 500 nm.
Discussion
In this study, we present the pathologic changes induced in mice inoculated IP with either a sublethal or a lethal dose of B. mallei. The cardinal lesion at the gross level in both study groups was splenomegaly. Histologically, the principal lesions consisted of pyogranulomatous inflammatory cell infiltrates in multiple organs. Necrosis of both leukocytes and somatic tissue was common in bone marrow but not in other tissues.
In mice from both the sublethal and lethal studies, bacterial cultures revealed glanders bacilli in the blood, lung, liver, and spleen by 6 hours PI. The blood was culture-positive in one to two sublethal study mice through day 3 and through day 2 in all lethal study mice. It is likely that glanders bacilli injected IP were carried to intrathoracic lymph nodes by way of transdiaphragmatic lymphatics. 8 14 From intrathoracic nodes, bacilli entered the venous blood via the thoracic duct. The cessation of positive blood cultures in our mice may have signaled the completion of transfer of bacteria from the peritoneal cavity to the blood. It is also possible that bacteria spilled into the blood from the intrathoracic lymph nodes seeded early in the infection. However, the presence of low numbers of glanders bacilli found in histologic sections of these nodes argues against this mechanism.
In sublethal study mice, we were surprised at the inconsistent isolation of bacteria from blood, lung, and liver. For example, no bacteria were detectable in livers of any day 4 PI mice, but cultures were once again positive in livers of some mice from day 5 on. Clearly in mice from this study, the spleen was the major target tissue for glanders infection. Spleens were uniformly infected by 6 hours PI. The numbers of cultured bacteria peaked at day 1 and gradually diminished through day 4, at which point the numbers remained fairly consistent through the remainder of the study.
Sublethal study mice began to show evidence of clinical recovery at day 5, but our culture data presented in Fig. 1 clearly show that high numbers of bacteria remained in the liver and spleen of these animals at the end of the study. Although we have not yet studied chronic glanders infection, one of us (DMW) has cultured B. mallei from vaccine survivors (both mice and hamsters) at 60 days PI. This suggests that B. mallei may have the propensity for latency, as does the very closely related organism B. pseudomallei, 9 the causative agent of melioidosis.
The differences in histopathologic changes observed in mice with sublethal infections were largely quantitative rather than qualitative. In the spleen, e.g., the character, distribution, and severity of the pyogranulomatous inflammatory cell infiltrate was similar in mice from both study groups. However, in sublethal infections, the infiltrates were generally more discrete. In lethal infections, the infiltrates tended to coalesce.
The hallmark liver lesion seen in all sublethal study mice and at the early time points in lethal study mice was pyogranulomatous inflammation. Interestingly, the liver lesions in both groups did not diminish with time, as tended to be the case in other tissues. Earlier studies in mice killed about 40 days PI revealed liver lesions comparable in incidence/severity to the ones reported here (D. Fritz, D. Waag, and R. Byrne, unpublished observations). In time, miliary pyogranulomas were accompanied by sharply demarcated, variable-size zones of hepatocellular degeneration and necrosis (presumed to be an ischemic change). In lethal study mice from day 2 on, there was frequently mineralization of the peripheral hepatocytes in these ischemic zones. We were unable to determine why hepatocellular mineralization was not seen in livers of sublethal study mice. Finally, venous thrombosis and, to a lesser degree, vasculitis of hepatic veins were more commonly found in sublethal study mice from day 3 on. Perhaps the longer disease course in the sublethal infection allowed these vascular changes to develop.
The results of previous studies suggested that RE-rich tissues are especially susceptible to localization of B. mallei and genesis of lesions. 7 8 19 Our findings in this study corroborate the importance of RE tissues, particularly in a glanders-resistant species such as the mouse. In this species, organs without significant RE tissue were only occasionally affected. Intrathoracic lymph node lesions in lethal study mice were somewhat more severe and extensive than in sublethal study mice. However, involvement of extrathoracic lymph nodes, bone marrow, and even the spleen was consistently greater in lethal study mice. This was most likely due to the higher infectious dose received by the former, which resulted in a heavier localization of glanders bacilli in the RE-rich tissues. The severity and incidence of lesions in intrathoracic lymph nodes and spleen actually decreased in sublethal study mice killed near the end of the study. We believe that many of the lesions (with the exception of those in liver) seen in sublethal study mice at the later stages of the study were resolving. This is consistent with the apparent clinical recovery and culture data from these mice.
The present study in mice has highlighted some significant similarities and differences between glanders infection in mice and hamsters. Similarities included pyogranulomatous inflammatory response induced by the glanders bacillus in both species and a tropism for infection in RE-rich tissues (such as spleen, lymph nodes, bone marrow, and liver). Also in livers of subacute study mice, vascular thrombosis (Fig. 4) frequently resulted in discrete areas of hepatocellular degeneration and necrosis. In hamsters, we noted evidence of impaired vascular perfusion, especially in the later stages of the glanders disease process. 8 Infarcts of the spleen and bone marrow were present in a number of these animals.
Differences between mice and hamsters included the general limitation of glanders in mice to the RE-rich tissues, whereas in hamsters, it is widely disseminated. 8 The characteristic histopathologic change in mice included pyogranulomatous inflammation, which generally lacked discrete organization. Also, necrosis was never extensive and hemorrhage was rarely present. In hamsters, however, discrete pyogranulomas with central necrosis and persistence of leukocytic karyorrhectic debris were typical, and the inflammation was frequently accompanied by hemorrhage. In hamsters, we frequently saw septic thrombi in many tissues and postulated that each served as the nidus of areas of inflammation in hamsters. However, in mice, septic thrombi were never observed. Finally, by TEM, there were far fewer bacteria in the infected tissues of mice and they were always single. In hamsters, occasional phagocytes would contain three or more organisms, as is illustrated in Fig. 15 of our previous report. 8 Also, phagocytic cells in infected mice showed no evidence of injury, whereas in hamsters, phagocytic cells containing glanders bacilli were frequently degenerate or necrotic.
There is little information about pathologic findings in human beings infected with the glanders bacillus. The organisms tend to localize at the site of entry (skin, nasal or laryngeal mucosa) and induce formation of nodules that ultimately ulcerate, resulting in lymphangitis and lymphadenopathy. When the organisms are spread systemically, many tissues can be seeded. These tissues include skin, 1 skeletal muscle, 23 bone, 23 joints, 10 less commonly brain/meninges, 23 and rarely nasal 10 or ocular. 23 The lungs, liver, and spleen are often affected. 1 Histologically, the lesions are described as being granulomatous 13 and occasionally as being abscesses, 12 23 implying a significant component of PMN leukocytes and necrosis within the lesion.
With the exception of intralesional necrosis, the character of the inflammatory cell infiltrate is the same in mice as in human beings. As in people, the lesions are often seen in liver and spleen. But a major difference is that, in our mice, lung lesions were never present. Lung involvement is common in many animal species in natural infections with B. mallei. 1 23 Undoubtedly, many of these infections followed aerosol exposure, unlike in the mice in this study. Future pathogenesis studies of mice following aerosol exposure to the glanders bacillus should provide a more relevant exposure route for the mouse as a model.
For this study in mice, we used B. mallei GB15.1–3, a hamster-passaged China 7 strain, whereas in our IP hamster study, we used B. mallei GB-18, a hamster-passaged Budapest strain. 8 Although earlier studies in our laboratory found the China 7 strain to be significantly more virulent than the Budapest strain in both mice and hamsters, we found the difference in virulence to primarily affect the LD50 and the character of the disease (D. Waag and D. Brown, unpublished data). To produce glanders infection in mice inoculated IP, a very high dose of a very virulent strain of the bacillus was necessary. This again underscores the resistance of mice to glanders infection.
Earlier reports 13 stated that B. mallei lacks a capsule, but through the use of ruthenium red processing of tissue, Popov et al. 19 20 showed that the glanders bacillus does indeed have a capsule. We have demonstrated the capsule with immunolabel against B. pseudomallei type I O-PS (Fig. 9). In studies of B. pseudomallei, a pathogenic bacillus closely related to B. mallei, it was previously thought that type I O-PS was truly an O-antigen of lipopolysaccharide (LPS). 3 It is now known to be capsule, not LPS (D. DeShazer, unpublished data). B. pseudomallei mutants that lack this capsule have dramatically attenuated virulence in hamsters (D. E. Woods, personal communication). Unlike B. pseudomallei, B. mallei appears to lose the capsule after passage in the laboratory (not animal passage) (D. DeShazer, unpublished data). Preliminary findings by one of us (DD) indicate there is a corresponding decrease in virulence after laboratory passage, suggesting that capsule is an important virulence factor for B. mallei, as it is for other pathogenic bacteria. 16 Studies to clarify these interesting findings are in progress.
Several authors report finding L-forms (cell-wall deficient) of B. mallei within RE cells in infection of white mice 7 and white rats. 6 Ferster and Kurilov 7 postulated that persistence of bacteria in these cells resulted in the proliferation of RE cells and ultimately granuloma formation. It has been suggested by others 4 that many bacteria, when exposed to a hostile environment in their host, can change into a cell-wall-deficient form that can survive/persist in a latent state and cause chronic disease. Regardless, we were unable to identify any L-form glanders bacteria in the tissues of our mice.
We previously reviewed pathologic findings in glanders infection from a variety of laboratory animals as reported by other authors. 8 Of these reports, only Ferster and Kurilov 7 presented pathologic data in mice (Balb/c) that were injected subcutaneously with various doses of both virulent and avirulent (for mice) strains of glanders bacilli. In general, the histopathologic changes they noted in the spleens and extrathoracic lymph nodes are similar to those in our sublethal and lethal study mice. However, there are several major differences between the findings in their study and ours. They described pneumonic foci (presumably pyogranulomatous) in the lungs of their mice at day 5 PI. They also described an abundance of giant cells in the peribronchiolar connective tissue of their mice. In contrast, we found no glanders-related changes in the lungs of any mice and no multinucleated giant cells in any tissues from our mice. Ferster and Kurilov 7 also described liver lesions consisting of perivascular lymphohistiocytic infiltrates and multifocal Kupffer cell proliferations, which contained multinucleated giant cells. In contrast, the predominant lesions in the livers of our mice were miliary pyogranulomas and multiple foci of hepatocellular ischemic change. Subacute portal inflammatory cell infiltrates were often present but never contained giant cells.
In reporting their study, which compared glanders infection of both mice and hamsters, Ferster and Kurilov 7 suggested that significant differences exist in glanders pathogenesis between the susceptible (hamster) and nonsusceptible (mouse) host. Comparing our findings in this mouse study with our previous study in hamsters, 8 we conclude that glanders pathogenesis is similar in these two hosts following IP inoculation. Although the severity and distribution of lesions differ between mouse and hamster, the overall character of the lesions is essentially the same.
Historically, the incidence of glanders infection in human beings has been low, suggesting they have a moderate resistance to infection by B. mallei. Because of the high infectivity of human beings involved in laboratory work with the glanders bacillus, 17 22 we believe this organism has potential for emerging as a human pathogen. In light of the resistance of the organism to many antibiotics and past failures in the development of vaccines against B. mallei, continued study is needed on appropriate animal models. Hamsters are believed to be the most uniformly infected and highly susceptible rodents for glanders infection. 15 At the time of earlier glanders pathogenesis studies, inbred mouse strains were not readily available. Mice have similar resistance to glanders infection as human beings, which prompted us to use the inbred mouse as a model for glanders infection in a nonpermissive host. In the course of this study, we developed appropriate infective doses for the BALB/c mouse that produced uniform sublethal and lethal infections.
Although more difficult to infect with B. mallei than hamsters and some other potential animal models, the inbred mouse has significant advantages. First, with inbred mice, immunological assays to examine cell-mediated immunity (such as lymphocyte proliferation assays) can be performed on immune cells pooled from different animals. Second, the varieties of reagents against cell surface markers (such as cell phenotype and activation markers) are much more readily available for the mouse than the hamster. Third, with inbred mice (particularly the BALB/c), there is a wide availability of genetic variants (such as knockouts), and we anticipate that some of these variants will be of value in future glanders pathogenesis studies. Finally, from a convenience standpoint, mice are smaller than most other laboratory animals so more can be housed in a limited space. There is no denying the utility of animals other than mice for studying some aspects of glanders infection. For example, if vaccine and antibiotic therapies can be developed to protect hamsters against B. mallei challenge, they certainly should protect other animal species as well. But to develop these therapies and to study glanders pathogenesis and infection in the nonpermissive host, the mouse is clearly a superior model.
Footnotes
Acknowledgements
We thank Lynda Miller for necropsy support; Lynda Miller, Jeff Brubaker, and Quang Vu for histologic preparations; Kathy Kuehl and D'Angelo Austin for electron microscopic and immunohistochemical preparations; and Steve Ferendo, Lorraine Farinick, and Larry Ostby for visual information support. We are grateful to Dr. Keith Steele for sharing his expertise in the immunohistochemical procedures and to Dr. Don Woods for supplying the rabbit anti-B. pseudomallei type I O-PS serum. Finally, we thank Dr. Gary Zaucha, Dr. Keith Steele, and Katheryn Kenyon for their critical reviews of this manuscript. In conducting research using animals, the investigators adhered to the Guide for the Care and Use of Laboratory Animals (Committee on Care and Use of Laboratory Animal Resources, National Research Council, NIH Publication No. 86–23, revised 1996). The U.S. Army Medical Research Institute of Infectious Diseases animal facilities and animal care and use program are accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International. The views, opinions, and/or findings contained in this report are those of the authors and should not be construed as an official Department of the Army position, policy, or decision unless so designated by other documentation.
