Abstract
Chronic gastritis and intestinal metaplasia associated with naturally occurring colonization by Helicobacter aurati and two other microaerobic species were observed in Syrian hamsters. Thirty-five hamsters, between 7 and 12 months of age, were evaluated from two research and three commercial facilities. Microaerobic bacteria were cultured from the hamster stomachs. These bacteria included H. aurati, a fusiform, ureasepositive species; a second novel helical, urease-negative Helicobacter sp.; as well as a smaller, urease-negative Campylobacter sp. Southern blot analysis detected Helicobacter spp. DNA in the gastric tissues of all 35 hamsters; 15 hamsters also had Campylobacter sp. DNA in their gastric tissues. When examined by light microscopy, argyrophilic bacteria consistent with H. aurati or the second Helicobacter sp. were present in antral sections of 12 out of the 15 hamsters where bacteria were seen, while 9 out of the 15 hamsters had bacteria resembling the Campylobacter sp. The presence of Helicobacter spp. but not the presence of Campylobacter sp. was significantly correlated to gastritis severity (P < 0.0001 for Helicobacter spp., P = 0.6025 for Campylobacter sp.) and intestinal metaplasia, as measured by numbers of goblet cells (P = 0.0239 for Helicobacter spp., P = 0.5525 for Campylobacter sp.). Severely affected hamsters also had Giardia sp. within their metaplastic gastric pits. Hamsters with naturally occurring helicobacter-associated gastritis provide a model for studying the development of intestinal metaplasia and gastric giardiasis in H. pylori-infected humans.
Chronic gastritis caused by Helicobacter pylori is widespread in humans, even though clinical signs are often absent. In a proportion of patients, duodenal and gastric ulcers develop in conjunction with H. pylori colonization; persistent infection is also considered to be a risk factor in the development of gastric adenocarcinoma and lymphoma. Although the basic mechanisms for cancer development are not understood, stages in the process include chronic superficial gastritis, chronic atrophic gastritis, intestinal metaplasia, and dysplasia. To investigate the pathogenesis of these helicobacter-induced gastric lesions, experimental inoculation with gastric helicobacters has been performed in numerous laboratory animals, including mice, 24 34 36 rats, 11 Mongolian gerbils, 16 17 19 38 guinea pigs, 32 ferrets, 12 cats, 8 dogs, 27 pigs, 22 and macaques. 3 Of these experimental models, only Mongolian gerbils infected with H. pylori have evidence of intestinal metaplasia, a premalignant lesion, followed by gastric cancer in some animals. 18 31 37
Reports concerning natural infections of gastric helicobacter species and concomittant pathology are less common than inoculation studies. Subclinical gastritis has been documented in rhesus and cynomolgus macaques with naturally acquired H. pylori infection. 1 4 15 28 A correlation between H. mustelae and gastric cancer, MALT lymphoma as well as adenocarcinoma, has been demonstrated in ferrets. 6 9 10 Cheetahs colonized with H. acinonychis have gastritis with accompanying clinical signs of vomiting and wasting. 5 A closed cat colony has been shown to be chronically and naturally infected with H. pylori, 7 and the colony cats exhibited a diffuse lymphofollicular, atrophic gastritis with areas of mucosal dysplasia. Gastric inflammation was seen in certain strains of older mice in association with H. muridarum, a normal resident of the rodent lower intestine. 23 26 These natural helicobacter infections are of interest as animal models of gastritis and its sequelae for humans as well as for the clinical implications of such infections in the host species.
Helicobacter species identified previously in Syrian hamsters are H. cinaedi 14 and H. mesocricetorum, 33 reported to be normal components of the hamster gastrointestinal tract, and H. cholecystus, 13 recovered from hamster gallbladders. Recently we described the morphology, biochemical characteristics, and 16S rRNA sequence of H. aurati, a urease-positive, fusiform microaerobe found in the gastrointestinal tracts, including the stomachs, of adult Syrian hamsters. 25 Two other phenotypes, both negative for urease, were also recovered from the hamster stomach cultures; these were a long, slender, and helical phenotype that, by polymerase chain reaction (PCR), is a Helicobacter sp., and a short, curved Campylobacter sp. By 16S rRNA sequencing, these latter two species are also novel, but they have not been formally named. The morphologic lesions associated with these three species of bacteria in the hamster stomach are addressed in this report. In contrast to the complex enteritidies or “wet tail” syndromes that are well recognized in Syrian hamsters, gastric pathology in this species has not been described at length previously to our knowledge.
Materials and Methods
Thirty-five Syrian hamsters (Mesocricetus auratus) were examined from two research and three commercial facilities in the United States. There were 16 males and 19 females between 7 and 12 months of age. The research hamsters had been unmanipulated experimentally; previous sporadic mortalities in these two colonies had resulted in the submission of clinically normal animals to our diagnostic laboratory. The vendor animals were all healthy, retired breeders. Complete necropsies were performed following euthanasia with carbon dioxide and aseptic collection of stomach and cecal samples for culture and PCR. Gastrointestinal tissues were immersion-fixed in 10% neutral buffered formalin. Paraffin-embedded 5-µm sections were stained with hematoxylin and eosin. Gastric sections were also stained with a Warthin-Starry silver method to demonstrate argyrophilic bacteria, while the Alcian blue/periodic acid–Schiff (PAS) reaction, pH 2.5, technique was used to stain for mucopolysaccharides.
Gastric pathology was evaluated in a blinded fashion. Overall gastritis was rated as absent (0), mild (1), moderate (2), marked (3), or severe (4). Alcian blue/PAS sections of antrum 8 mm in length, beginning at the pyloric–duodenal junction as determined by the presence of villi, were used to assess intestinal metaplasia. The number of goblet cells in that area was counted and approximated when exceeding 50. Assigned scores were 0 (0–25 cells, normal), 1 (26–50 cells, slight increase), 2 (51–100, moderate increase), 3 (101–500, marked increase), or 4 (over 500, extreme increase).
Silver-staining of bacteria within antral gastric pits and glands was noted as present or absent for each hamster. Bacteria measuring over 6 µm long were consistent with H. aurati or the helical Helicobacter sp. Very short, slightly spiral bacteria were compatible with the Campylobacter sp. An analysis of variance (ANOVA) was used to determine the association of the Helicobacter spp. or Campylobacter sp. in antral sections with degree of gastritis and goblet cell score.
Antral stomach samples were cultured for microaerobic bacteria, and PCR with appropriate controls was performed on successful cultures and directly on tissue specimens, as described earlier. 25 Primer pairs specific for Helicobacter spp. and Campylobacter spp. were used that amplified 1,200 base-pair and 297 base-pair fragments, respectively. After stomach tissue samples were analyzed by PCR, Southern blots were performed to increase the sensitivity of detecting helicobacter and campylobacter DNA. The PCR products were run on a 1% agarose gel and then transferred to a nanomembrane (Hybond-N, Amersham Pharmacia Biotech, Buckinghamshire, England). A 1,200 base-pair product of H. hepaticus and a 297 base-pair product of C. jejuni were used as probes. The ECL Direct Nucleic Acid Labelling and Detection Systems (Amersham Pharmacia Biotech) protocol was followed for identification of Helicobacter and Campylobacter PCR products in the hamster stomach tissue samples.
To characterize the protozoa that were visualized in selected antral sections with light microscopy, DNA was extracted from paraffin-embedded sections using the Oncor Ex-Wax DNA Extraction Kit (Gaithersburg, MD). PCR was then performed with primers for Giardia sp. (reverse 5′-TTAGTGCTTTGTGACCATCGA-3′; forward 5′-AAGTGCGTCAACGAGCAGCT-3′) 21 and for Tritrichomonas sp. (reverse 5′-ATCACAAGTTACGCTCTAGC-3′; forward 5′-GGAAAGTCGGTCGACCCAATC-3′). A 171 base-pair fragment of the giardin gene was amplified with the Giardia primers, whereas the Tritrichomonas primers gave a 76 base-pair product of the rRNA gene. Ten microliters of DNA were added to 90 µl of the reaction mixture. After an initial denaturing step for 4 minutes at 94 C, amplification was performed using 35 cycles of denaturating for 1 minute at 94 C, annealing for 2 minutes at 58 C, and extension for 2 minutes at 72 C.
Results
Hamsters were normal except for two thin females obtained from one research group. No gross abnormalities were seen in the stomachs of the animals. Microaerobic bacteria were cultured from 10 out of the 35 hamster stomach samples; 25 many of these were mixed infections that contained at least two of the three species eventually isolated. Using PCR followed by Southern blot analysis, Helicobacter spp. DNA was detected in the gastric tissues of all 35 study hamsters, and 15 of the hamsters also had Campylobacter spp. DNA in their gastric tissues (data not shown).
Microscopically, gastric lesions were most severe in the distal antrum near the pyloric–duodenal junction, and consisted predominantly of multifocal to diffuse lymphoplasmacytic inflammation that had extended through the entire thickness of the mucosa (Fig. 1). In many cases, scattered heterophils and eosinophils were also present; rarely, mucosal lymphoid infiltrates formed distinct nodules in the deep mucosa. Intraepithelial lymphocytes were prominent in the superficial lining epithelium of severely affected animals. Varying degrees of mucosal epithelial hyperplasia and, occasionally, dysplasia and mucin depletion were also observed. Infrequently, dilated glands lined by low cuboidal epithelium were evident or complete loss of glands and mild fibrosis were present in the lamina propria of the antrum. Numerous goblet cells, indicative of intestinal metaplasia, were observed throughout the mucosa of inflamed regions of antrum (Fig. 2). The intestinal metaplasia was confirmed by Alcian blue pH 2.5/PAS staining (Fig. 3).
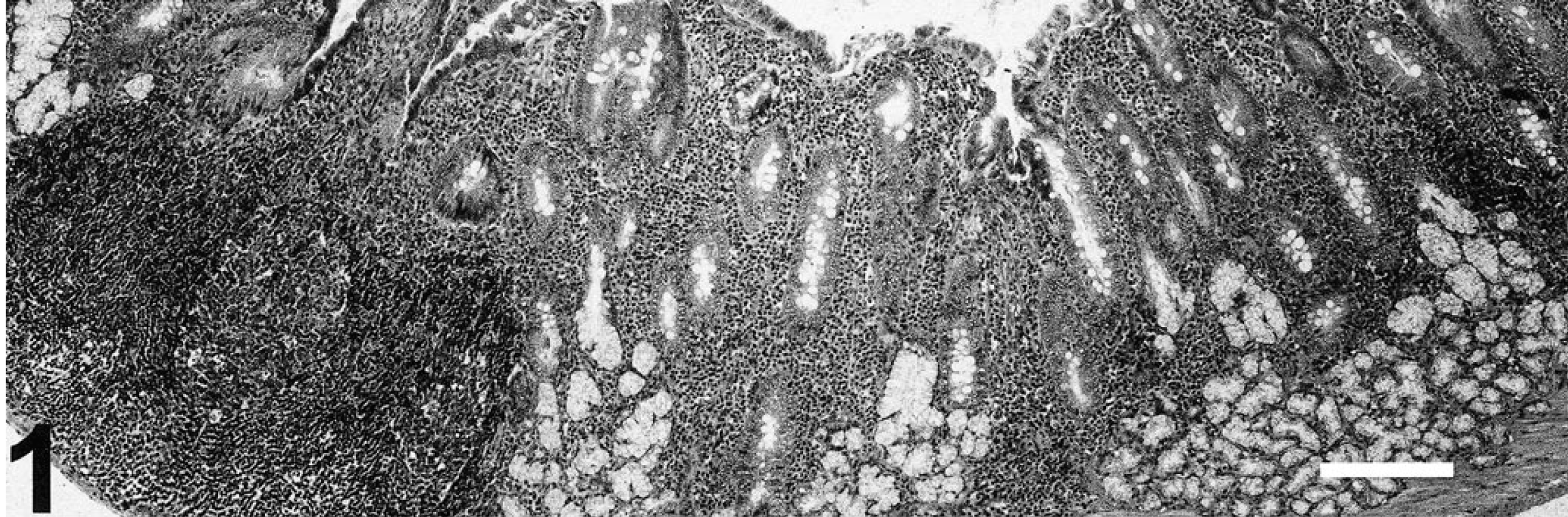
Gastric antrum of Syrian hamster infected with H. aurati and other two species of microaerobic bacteria. Severe inflammation is shown that extends to the muscularis mucosae. HE stain. Bar = 200 µm.
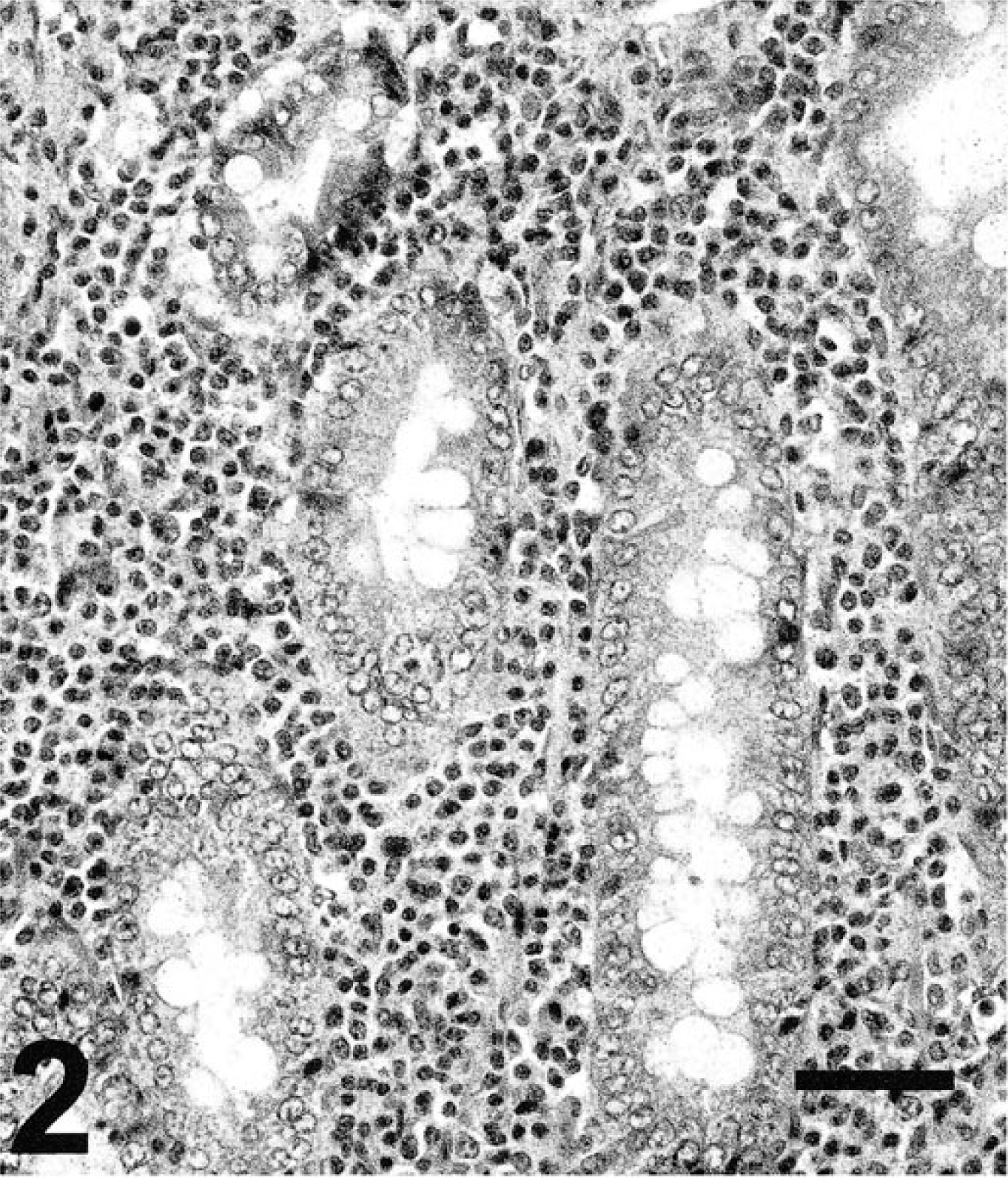
Higher power of antral area. Lymphoplasmacytic infiltrate and increased numbers of goblet cells are present; the latter is evidence for intestinal metaplasia. HE stain. Bar = 50 µm.
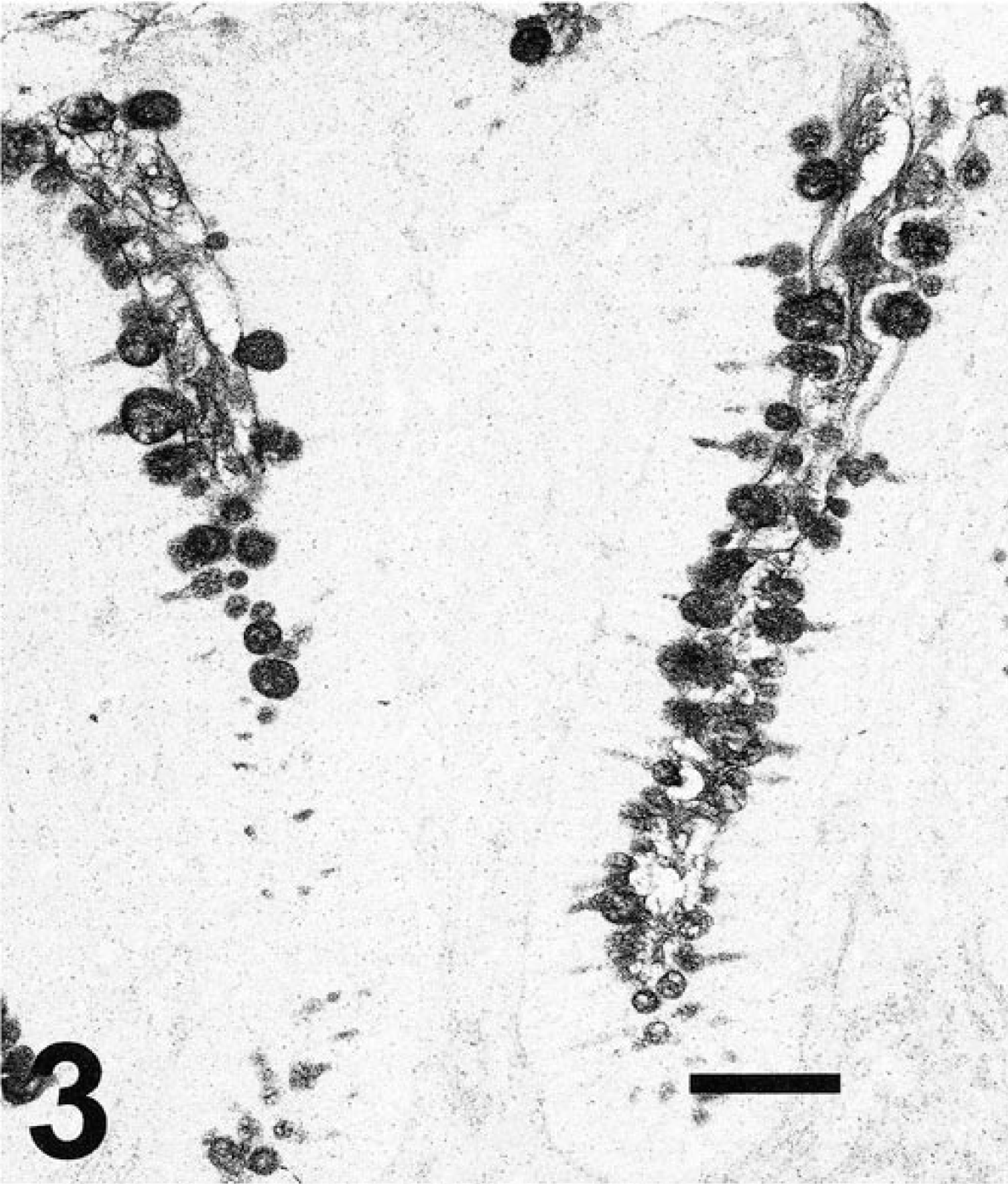
Inflamed area of hamster antrum. Goblet cells lining the gastric pits are demonstrated. Alcian blue pH 2.5/PAS stain. Bar = 50 µm.
In antral tissue sections from the study hamsters stained with the Warthin-Starry method, argyrophilic bacteria within gastric pits or glands were visible in 15 out of 33 hamsters; two animals were not available for this evaluation. Three morphological types of bacteria were seen: one was consistent with H. aurati, which is fusiform with a regular periodicity of darkly staining areas and tapered ends; the second was the unnamed helical Helicobacter sp.; and the remaining were the short Campylobacter sp. Due to variation in silver staining and section planes, H. aurati could not always be definitely distinguished from the helical Helicobacter sp.; as a result, these two helicobacters were considered together. When analyzing gastric lesions, bacteria resembling either H. aurati or the second Helicobacter sp. (Fig. 4) were the only gastric organisms observed in six hamsters, and three different animals had gastric mucosa containing only short bacteria consistent with the Campylobacter sp. (Fig. 5). The small campylobacter morphotype as well as the longer helicobacter were seen in the antral sections of another six hamsters.
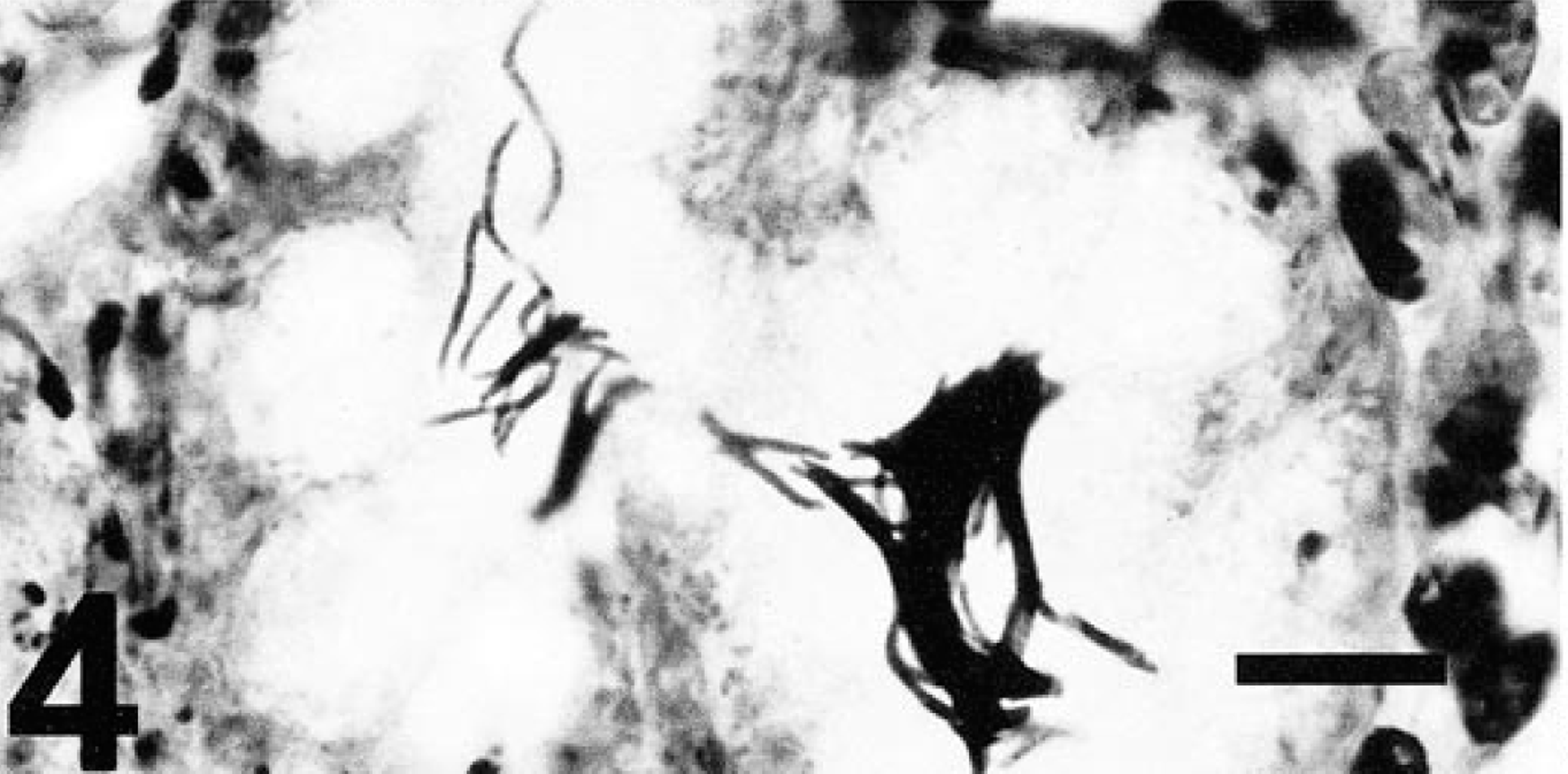
Gastric gland of inflamed area. Long argyrophilic bacteria are characteristic of H. aurati or the second Helicobacter sp. identified in Syrian hamsters. Warthin-Starry stain. Bar = 10 µm.
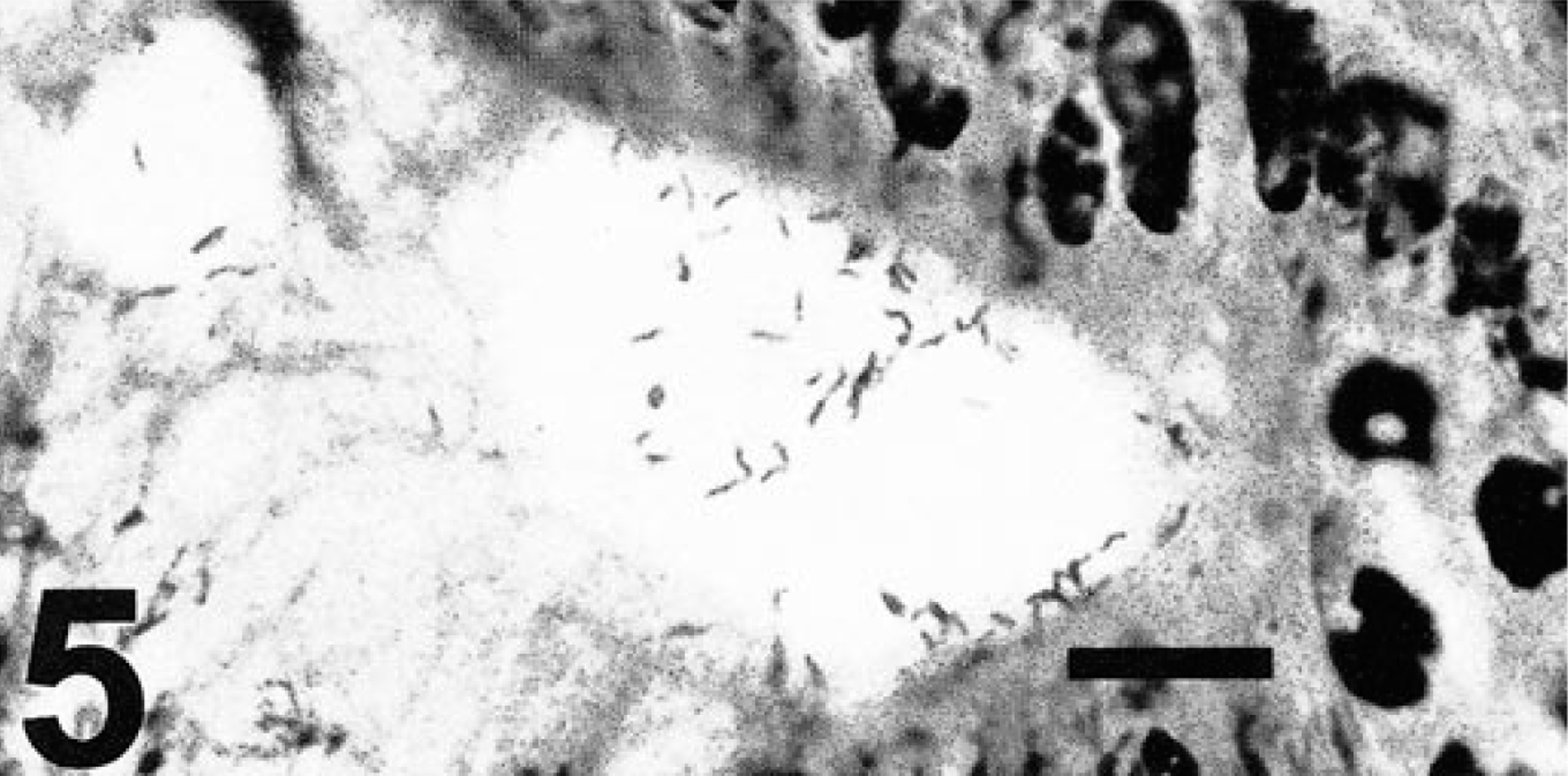
Gastric gland lumen. Short argyrophilic bacteria that appear like the Campylobacter sp. cultured from hamsters. Warthin-Starry stain. Bar = 10 µm.
The presence or absence of these types of bacteria in gastric tissue observed microscopically were compared to gastritis severity and numbers of goblet cells in Table 1. Among the 12 hamsters that had antral Helicobacter spp. observed microscopically, 6 had marked gastritis (score of 3) and 2 had severe gastritis (score of 4); the mean gastritis score for these 12 hamsters was 2.67. In contrast, gastritis was absent or mild (score of 0 or 1) in 18 of the 21 hamsters without Helicobacter spp. observed in gastric tissue, and their average gastritis score was 0.62. The nine hamsters that had small bacteria consistent with Campylobacter sp. had an average gastritis score of 2.11; marked gastritis scores (score of 3) occurred in four of these animals. Five of the 24 hamsters without campylobacter organisms present had marked or severe gastritis (score of 3 or 4), and the mean gastritis score for the 24 animals was 1.08. Using these data in an analysis of variance, gastritis severity was significantly correlated (P < 0.0001) with the presence of Helicobacter spp. in tissue sections and not (P = 0.6025) with the presence of Campylobacter spp.
Histological evidence of Helicobacter spp. or Campylobacter sp. in antral sections from Syrian hamsters compared to gastritis severity (0 = absent to 4 = severe) and goblet cell score (0 = normal to 4 = extreme increase).
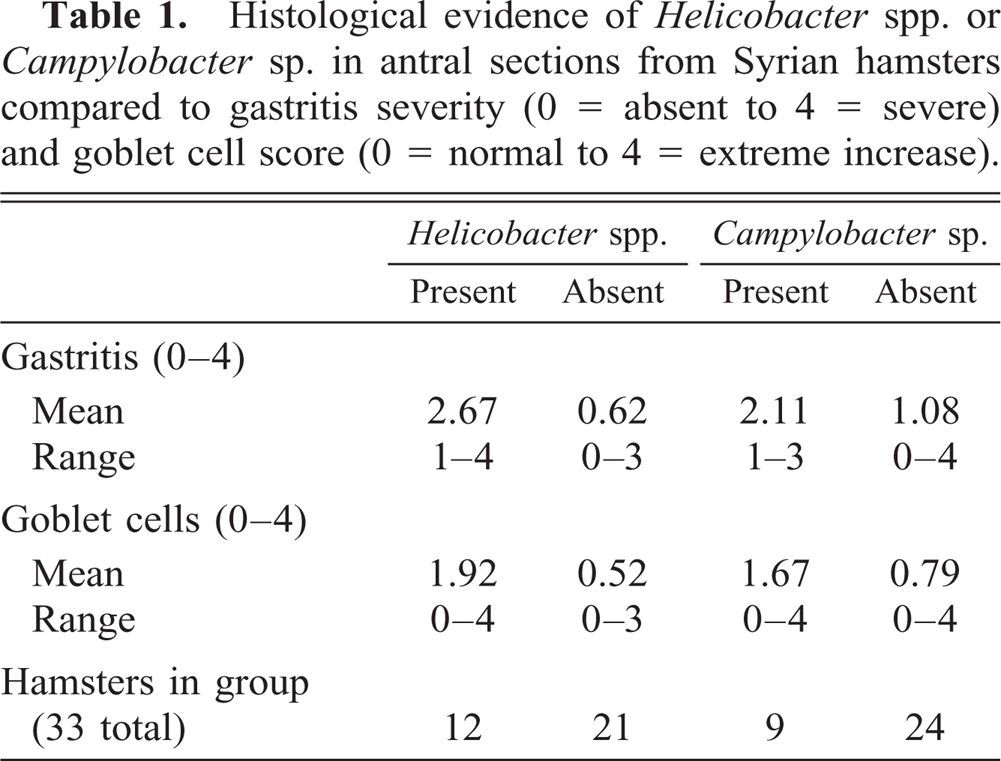
When the presence of these bacteria was compared with the finding of intestinal metaplasia as measured by relative numbers of goblet cells, hamsters with Helicobacter spp. averaged goblet cell scores of 1.92 compared with a mean of 0.52 in hamsters where Helicobacter spp. was not observed. Goblet cell scores averaged 1.67 in hamsters with Campylobacter sp. in gastric pits and 0.79 without Campylobacter sp. present. When an analysis of variance was applied to these data, goblet cell scores were significantly correlated (P = 0.0239) with the presence of Helicobacter spp. but not (P = 0.5525) with Campylobacter sp.
Argyrophilic bacteria were not obvious in the particular gastric pits that contained abundant goblet cells. Instead, these metaplastic sites were often accompanied by colonization of the gastric pits with numerous piriform or ovoid extracellular protozoa resembling Giardia or Tritrichomonas spp. (Fig. 6). PCR of two antral tissue samples identified the parasites in the gastric pits as Giardia and Tritrichomonas spp. (Fig. 7). One of the samples had only Giardia DNA present, while the other sample was positive by PCR for both Giardia and Tritrichomonas DNA. Intestinal metaplasia and protozoa were seen only in hamsters with marked or severe antral inflammation.
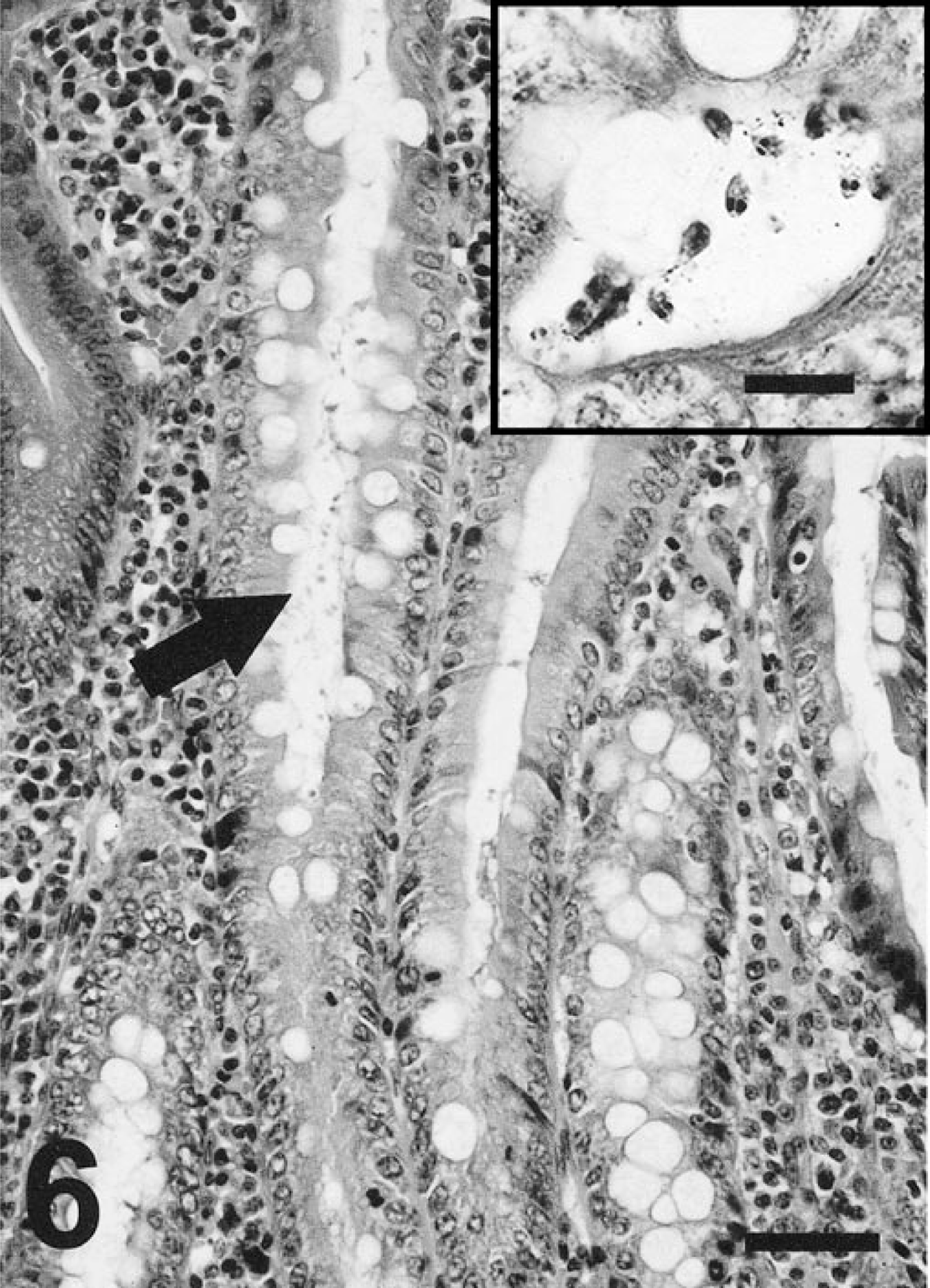
Area of intestinal metaplasia in hamster antrum. Lumen of gastric pit contains many protozoa (arrow). HE stain. Bar = 50 µm. Inset: Demonstrates ovoid shape and two nuclei consistent with Giardia. Warthin-Starry stain. Bar = 10 µm.
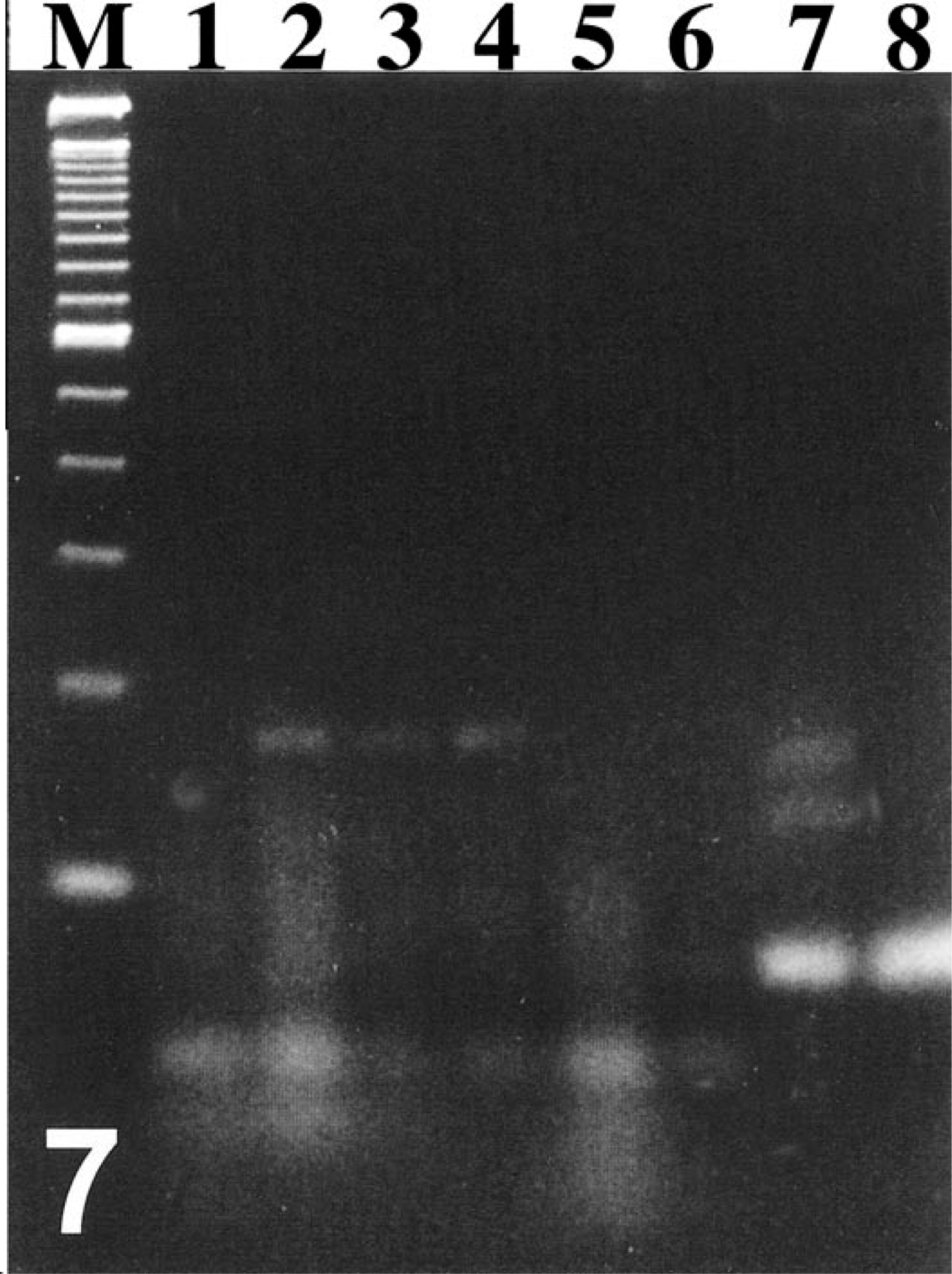
Ethidium bromide-stained agarose gel of PCR to identify protozoa in hamster antra. Lane M: 100 bp marker; 1: negative control Giardia; 2: hamster No. 6174, Giardia primers; 3: hamster No. 6172, Giardia primers; 4: positive control Giardia from hamster duodenum; 5: negative control Tritrichomonas; 6: hamster No. 6174, Tritrichomonas primers; 7: hamster No. 6172, Tritrichomonas primers; 8: positive control Tritrichomonas from mouse ileum.
Discussion
Chronic gastritis with intestinal metaplasia associated with naturally occurring gastric colonization by three species of microaerobic bacteria was documented in Syrian hamsters for the first time. Intestinal metaplasia is an important prognostic premalignant lesion for gastric cancer in stomach biopsies of people chronically infected with H. pylori, and the relationship between helicobacter species and metaplasia is being investigated. In some reports, Mongolian gerbils gavaged with H. pylori have intestinal metaplasia. 16–19, 31 37 However, a recent U.S. study of gerbils infected with different strains of H. pylori reported that intestinal metaplasia was not found. 38 The genetic background of the gerbils and strain of H. pylori, along with duration of infection, may in part explain these variable results. Recently it has also been shown that H. felis causes an aberrant metaplastic cell lineage to develop in C57BL/6 mice that is similar to goblet cells histochemically but not morphologically. 36 The mechanisms leading to premalignant intestinal metaplasia in humans with chronic H. pylori infection can now be studied in detail in a natural model of disease in hamsters. Whether gastric cancer develops in infected, aged hamsters requires additional studies.
Some hamsters with gastritis and intestinal metaplasia had extracellular protozoa within the gastric pits lined by intestinal-type epithelium. According to PCR that was done following microscopic examination of trophozoites, Giardia sp. were identified in both samples that were assayed and Tritrichomonas sp. in only one. Both of these protozoa can be common in laboratory hamsters. In general, giardia are considered opportunistic pathogens of the proximal small intestine; however, Tritrichomonas sp. are commensals found predominately in the cecum but can occur throughout the digestive tract. 35 It is of interest that humans with chronic atrophic gastritis, intestinal metaplasia, and H. pylori infection have been diagnosed with gastric giardiasis as well. 2 30 Treatment with proton-pump inhibitors also has been associated with human giardiasis in gastric biopsies. 20 29 Decreased gastric acidity with these syndromes and therapies are hypothesized to allow G. lamblia to extend proximally into the stomach from the duodenum. Though gastric pH was not measured, a hypochlorhydric environment may similarly explain aberrant giardia in the gastric mucosa of affected hamsters.
At this juncture, it is impossible to determine if the gastric pathology in the study hamsters is a result of one or more of the three microaerobes identified. Microaerobic cultures from the stomach samples often contained more than one species of bacteria. Also six hamsters had two types of bacteria in their gastric antra that were identified microscopically as consistent with the large helicobacter morphology and the smaller campylobacter. Specific PCR primers to amplify DNA from the three species in tissue sections are currently not available. Nevertheless, several factors support a potentially more important role in gastric inflammation for the two Helicobacter species, especially H. aurati. Most importantly, gastritis and goblet cell scores were significantly correlated with Helicobacter spp. in tissue sections but not with Campylobacter sp. Although the statistical analysis was not designed to distinguish between H. aurati and the second Helicobacter sp. because accurate identification was not always possible, the fusiform-shaped H. aurati seemed to be more common in histological sections. Pure cultures of H. aurati from hamster stomachs were also more successful than cultures of the other two species. 25 This may denote its preponderance in gastric tissue or simply reflect an ability of H. aurati to grow well in vitro. In any case, helicobacter DNA was found in the stomachs of all 35 study hamsters by Southern blot, compared with 15 of the hamsters that also had campylobacter DNA in their stomachs. Thus, the Helicobacter spp. were more common in hamster stomachs.
Finally, it can be speculated that H. aurati, which is the only urease-positive member of the hamster gastric microbiota, may colonize the gastric antrum more efficiently and earlier than the other two bacteria. The three microaerobic species were cultured from hamster cecal samples more often than from gastric tissues, 25 and hence, like H. muridarum in mice, 26 they may normally inhabit the hamster lower bowel and invade the stomach only under certain conditions. Colonization of the gastric mucosa has been thought to require the urease enzyme, which could serve a buffering capacity through the production of ammonia. An altered gastric microenvironment after infection with H. aurati could then allow subsequent colonization by the two urease-negative species. However, we have recently reported that C. fetus, a urease-negative campylobacter, can colonize scid mice and induce gastritis. 39 More studies are necessary to define the ecology and relative contributions of these three species to the inflammatory process in hamster stomachs. Because helicobacter DNA is widespread among commercial vendor animals and hamster pups cannot be foster raised to achieve germ-free status, transmission studies in hamsters are currently technically challenging. In our laboratory, we have thus far inoculated A/J and ICR scid mice with H. aurati, and colonization has been successful (unpublished data).
There was considerable variation in gastric lesion development and colonization by H. aurati and the other two microaerobic species among the different groups of hamsters studied. One important parameter that affected infection status and therefore gastric inflammation was the source of hamsters. The six animals from one vendor had no evidence of pathology or colonization by culture, even though helicobacter DNA was detected in their gastric tissue samples. Yeastlike cells were found in the stomachs of these hamsters. One possibility is that these hamsters are genetically resistant to microaerobic colonization or subsequent pathology, as previously noted in different mouse strains. Older hamsters, which generally came from the two research colonies sampled in this study, had higher gastritis scores than younger animals; helicobacter-induced lesions in humans also increase in severity with patient age or chronicity of infection. Additional stresses that contributed to helicobacter-associated pathology in animals maintained in a research environment cannot be ruled out.
Syrian hamsters are often used in biomedical research, especially in long-term toxicology studies. Hamsters with helicobacter and campylobacter-associated gastric inflammation may confound certain experimental studies. The differential expression of gastric pathology among infected hamsters is also important; those factors that modulate the pathologic changes seen are not well characterized but are undoubtedly important, similar to H. pylori infection in humans. In addition to the impact that infections with H. aurati and the other two microaerobic species may have on unrelated research involving Syrian hamsters, the development of intestinal metaplasia and gastric giardiasis in infected hamsters offers an attractive model for studying those aspects of H. pylori pathogenesis in humans.
Footnotes
Acknowledgements
We are grateful for the assistance of Drs. C. Dangler and M. Ihrig. This work was supported in part by grants R01-A137750 and T32-RR07036.
