Abstract
The golden Syrian hamster (Mesocricetus auratus) is frequently used as a model to study virulence for several Leptospira species. Onset of an acute lethal infection following inoculation with several pathogenic Leptospira species has been widely adopted for pathogenesis studies. An important exception is the outcome following inoculation of hamsters with live L. borgpetersenii serovar Hardjo, the primary cause of bovine leptospirosis and a cause of human infections. Typically, inoculation of hamsters with L. borgpetersenii serovar Hardjo fails to induce clinical signs of infection. In this study, the authors defined LD50 and ID50 for 2 strains of L. borgpetersenii serovar Hardjo: JB197 and 203. Both strains infected hamsters with ID50 values of approximately 1.5 × 102 bacteria yet differed in tissue invasion and interaction with leukocytes, resulting in widely divergent clinical outcomes. Hamsters infected with strain 203 established renal colonization within 4 days postinfection and remained asymptomatic with chronic renal infections similar to cattle infected with serovar Hardjo. In contrast, hamsters infected with strain JB197 developed a rapidly debilitating disease typical of acute leptospirosis common in accidental hosts (eg, humans) with an LD50 of 3.6 × 104 bacteria. Evidence that strain JB197 resides in both extracellular and intracellular environments during hamster infection was obtained. Development of models that result in chronic and acute forms of leptospirosis provides a platform to study L. borgpetersenii pathogenesis and to test vaccines for the prevention of leptospirosis.
Leptospirosis, caused by pathogenic Leptospira species, is one of the most widespread zoonoses known. Although there is considerable genetic diversity among pathogenic Leptospira, the clinical manifestation of disease caused by these bacteria is similar and ranges from a mild, largely asymptomatic chronic infection to an acute, potentially lethal infection. 14 At least 0.5 million human cases occur per year, with mortality rates ranging from 5% to 15%. 19 Chronically infected maintenance host animals are sources of infection, with viable bacteria being transmitted either by direct contact with infected bodily fluids or by exposure to urine-contaminated water. 20 Bacteria gain entry by passage across mucous membranes or abraded skin, disseminate hematogenously, and then localize primarily in liver and kidney. In acute infections, virtually all organs may become infected. Bacterial passage from infected kidneys to the bladder and release of bacteria during urination can result in exposure of new hosts. 14
Bovine leptospirosis is primarily caused by serovar Hardjo. 35 Cattle infected with serovar Hardjo rarely present clinical signs of infection except during pregnancy, where infection may cause abortion, stillbirth, or birth of weak offspring. Because of the influence on herd health and productivity, infections caused by serovar Hardjo probably have a greater economic impact in beef-producing countries than any other leptospiral serovar. 31 Characterization of serovar Hardjo pathogenesis has been difficult due to the finding that this serovar occurs in at least 2 distinct species—L. borgpetersenii and L. interrogans 30 —and to the lack of a small animal model for experimental serovar Hardjo infection. Although golden Syrian hamsters are susceptible to acute lethal infection with many pathogenic Leptospira species, 15,25 only 3 reports have described successful acute infection of hamsters with serovar Hardjo, 17,31,33 and these all preceded the discovery that different species share the same serologic classification, so it is not possible to know which species was used. In the present work, we tested 2 well-defined strains of L. borgpetersenii serovar Hardjo in the hamster model, with the discovery that inoculation with different strains of the same species results in vastly different clinical outcomes: either an acute or chronic infection.
Materials and Methods
Bacterial Culture
L. borgpetersenii serovar Hardjo strains 203 and JB197 were isolated from beef steers during slaughter 24 and propagated in a modified EMJH 12,16 semisolid medium, T80/40/LH, containing 5-fluorouracil (100 μg/ml) as described previously. 13,36 Animal infections used cultures from liver homogenates and were either in their first or second in vitro passage. Both serovar Hardjo strains have been demonstrated to infect cattle. 6 In addition, strain 203 has been used extensively as infectious challenge for cattle studies, 2,7,8 whereas the genome sequence of strain JB197 was recently reported. 10
Animals
Female golden Syrian hamsters (Mesocricetus auratus) were purchased from Harlan Laboratories (Madison, WI) and were approximately 50 to 80 g in weight at the time of inoculation. Animals were acclimated to the facility for at least 1 week before initiating studies. All animal experiments were conducted with approval of the Institutional Animal Care and Use Committee at the National Animal Disease Center.
Animal Infections
LD50 and ID50 calculations were done using the method of Reed and Muench, 28 using 8 groups of animals (n = 5) per strain. All members of a group were housed together but kept separate from all other groups to prevent cross-infection. All animals within a group received the same dose and strain of bacteria, administered by intraperitoneal injection. Intraperitoneal injection was chosen as the route of infection for these studies based on published international standards for evaluating leptospiral vaccine potency. 27 Prior to infection, bacteria were quantified by direct counting under dark field using a Petroff-Hauser chamber, and 10-fold dilutions ranging from 1 × 102 to 1 × 109 bacteria per animal were prepared for inoculation. Animals were observed twice daily until the first appearance of clinical signs within an infection group, then at least thrice daily. The onset of clinical signs of acute infection was used as an endpoint, rather than death, in calculating the LD50 to minimize animal distress. Clinical signs included huddling, lack of appetite, ruffled coat, and external hemorrhage. Previous observations indicated that animals exhibiting these signs following infection were unlikely to survive. Animals exhibiting clinical signs of leptospirosis were anesthetized by intramuscular injection of xylazine (50–200 mg/kg) and ketamine (100 mg/kg) and euthanized by decapitation. For calculation purposes, these animals were considered dead by infection. In addition, because strain 203–infected animals showed no outward clinical signs of infection, 1 animal per group infected with the 3 highest concentrations of bacteria was sacrificed at weekly intervals for preparation of tissue to assess infection status. All animals that survived to 30 days postinfection (DPI) were sacrificed, and tissue sections were collected for analysis. To test and validate infectious dose results for each serovar Hardjo strain and determine the progression of bacterial dissemination during infection, subsequent studies used groups of animals infected with selected bacterial concentrations, and when needed, animals were sacrificed before exhibiting clinical signs of infection to follow the progression of bacterial dissemination into tissue. Hamsters that received no bacteria were used as negative controls throughout these studies.
Detection of Bacteria
Liver and kidney samples were prepared for bacteriologic culture by gently homogenizing tissue and inoculating as described above. 36 Cultures were incubated for 6 months and periodically examined for the presence of bacteria using dark field microscopy before being designated as negative. Infected animals were also identified through the use of dark field microscopy and direct immunofluorescence microscopy of tissue homogenates following methods described previously. In addition, microscopic examination of formalin-fixed paraffin-embedded (FFPE) tissue sections stained by a modified Steiner-Steiner silver stain (Sigma Aldrich, St Louis, MO) was used to detect the presence of spirochetes in tissue.
Microscopic and Electron Microscopic Analysis
Tissue samples were collected at necropsy and fixed in 10% neutral buffered formalin for histopathology using standard techniques. Blood smears were prepared during necropsy and fixed with acetone before processing for immunofluorescence microscopy. FFPE tissue sections were stained either with hematoxylin and eosin or modified Steiner-Steiner silver stain. Microscopic examination was done using a Nikon Eclipse E800 microscope.
Immunofluorescence microscopic analysis of samples was done as described previously. 22 Rabbit antisera used for localization of Leptospira in tissue included anti-LipL32 (a gift from Dr David Haake) or a high-titer rabbit anti-Leptospira serum conjugated to FITC (prepared in house). Images were captured with a RT3 Slider CCD camera (Diagnostic Instruments, Inc., Sterling Heights, MI) mounted on a Nikon Eclipse E800 microscope.
Samples were prepared for transmission electron microscopy by placing approximately 1-mm square cubes of tissue in 2.5% glutaraldehyde in 0.1M sodium cacodylate. Samples were washed with 0.1M cacodylate buffer, stained with 2% OsO4 in 0.4M cacodylate buffer for 1 hour, followed by 2 washes with 0.4M cacodylate buffer. Stained samples were dehydrated gradually with increasing concentrations of ethanol before placing in epoxy propane resin. Epoxy resin–embedded tissue samples were cut to 100-nm thickness using an ultramicrotome and placed on 200 mesh copper grids. Samples were viewed using a Tecnai G2 Biotwin transmission electron microscope (FEI, Hillsboro, OR).
Results
Infection and Lethality of L. borgpetersenii Serovar Hardjo Strains in Golden Syrian Hamsters
To test the ability of 2 strains of L. borgpetersenii serovar Hardjo to establish infections in golden Syrian hamsters, animals were inoculated with live cultures by intraperitoneal injection to ensure that these studies were consistent with OIE standards for leptospiral vaccine potency testing. 27 Both L. borgpetersenii serovar Hardjo strains tested in this study infect cattle, 6 belong to the same species, and are genetically similar. However, strains 203 and JB197 can be differentiated on the basis of restriction endonuclease polymorphisms into subtypes A and B, respectively. 30 Serovar Hardjo strains 203 and JB197 have similar capacities to infect hamsters but vary in hamster virulence (Supplemental Table 1, available at http://vet.sagepub.com/supplemental). Both strains infected hamsters with ID50 values of approximately 1.5 × 102 bacteria. It was not possible to determine the LD50 for L. borgpetersenii serovar Hardjo strain 203; all animals survived, including those that received 1 × 109 bacteria, and none exhibited clinical signs of infection throughout the 30-day duration of the study at any of the tested infectious doses. In contrast, L. borgpetersenii serovar Hardjo strain JB197 is hamster lethal with a LD50 of 3.6 × 104 bacteria. The onset of clinical signs of strain JB197 infection ranged between 4 and 25 DPI, with most animals receiving 1 × 107 or more bacteria showing signs of infection by 5 DPI. Animals that received lower bacterial loads either survived until the end of the study (eg, most animals infected with 1 × 106 bacteria) or had delayed onset of clinical signs of infection starting at 10 DPI and continuing until 25 DPI (Supplemental Table 1).
Microscopic Analysis of Steiner-Steiner-Stained Sections: Tissue Distribution of Serovar Hardjo Strains

a Tissue distribution is based on evaluation of all infectious doses (1 × 102–109).
b Tissue distribution is based on evaluation of 1 × 103 bacterial dose except for intestine, which was done with 1 × 102 bacteria per animal.
Bacterial Dissemination
The progression from intraperitoneal inoculation through the first week of infection was analyzed and compared for both strains 203 and JB197 by histopathology. Hamsters exhibited peritonitis at 1 DPI; spirochetes were found localized to a mixed inflammatory infiltrate containing lymphocytes and macrophages. Perivascular accumulations of lymphocytes were common by 2 DPI (Figs. 1, 2), and in hamsters infected with strain JB197, spirochetes were often found around vessels and within vessel walls (Fig. 2, inset); this was not a feature seen in strain 203–infected hamsters. By 3 DPI, mesenteric perivascular lymphocytic infiltrates were more pronounced, and this pattern continued into 4 DPI for both strain 203 and strain JB197, which also induced hemorrhage in the mesentery (data not shown).
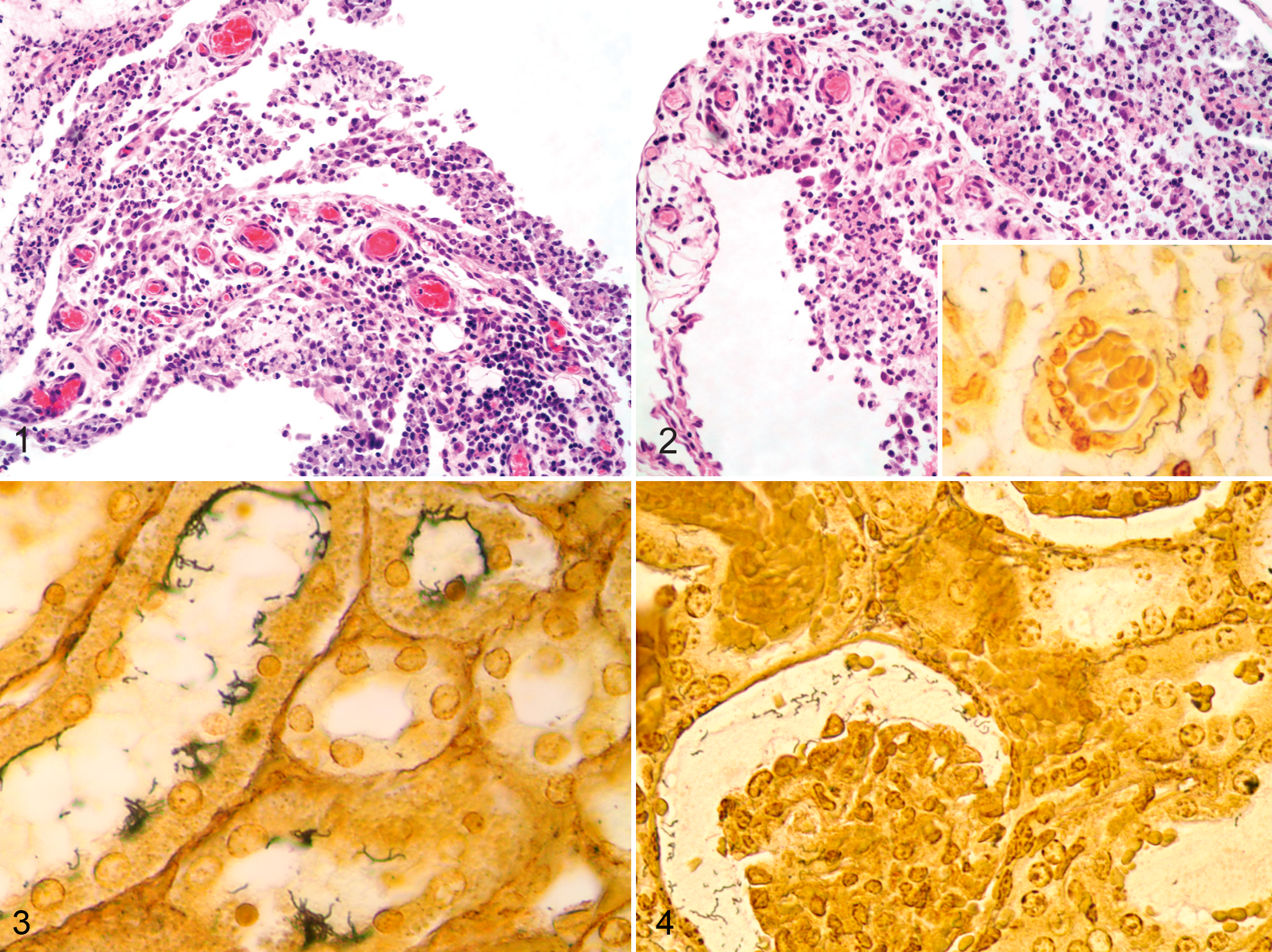
Mesentery, hamster infected intraperitoneally with Leptospira borgpetersenii serovar Hardjo strain 203, 2 days postinfection (DPI). Note lymphocyte infiltration with perivascular accumulations of lymphocytes. HE.
Olfactory bulb, hamster infected intraperitoneally with Leptospira borgpetersenii serovar Hardjo strain JB197, 10 days postinfection (DPI). Note absence of inflammatory cell infiltration. HE. Inset: Spirochete in a serial section of tissue. Steiner-Steiner Silver.
The first organ in which bacteria were detected beyond the injection site was the pancreas of strain JB197–infected hamsters. Bacteria were apparent by 3 DPI, with hemorrhage in the interstitium and pancreatic islets. By 4 DPI, pancreatic hemorrhage was more extensive in JB197-infected hamsters (data not shown). Spirochetes were rarely detected in the pancreas of strain 203-infected hamsters.
Bacterial colonization of the kidney was compared for both serovar Hardjo strains tested. Bacteria were detected by microscopic examination of silver-stained FFPE tissue sections in the kidney at 4 DPI in strain 203–infected hamsters and by 3 DPI in hamsters infected with strain JB197. By day 4 DPI, large numbers of bacteria were detected in silver-stained renal tissue sections. Bacteria were also detected in kidney tissue by dark field examination of kidney homogenates. Kidney tissue collected either at the end of the study (30 DPI) following infection with strain 203 (Fig. 3) or on 10 DPI following infection with strain JB197 (Fig. 4) was heavily colonized. Strain 203 appeared restricted to renal tubules, whereas strain JB197 was also seen in the urinary space of Bowman capsules and the interstitium.
Liver, hamster infected intraperitoneally with L. borgpetersenii serovar Hardjo strain JB197, 4 DPI. Transmission electron microscope image showing tightly packed spirochetes in the intracellular space. Note tight junction adjacent to spirochetes (arrows). Osmium tetroxide. Bar = 500 nm.
Tissue Distribution of L. borgpetersenii Strains in Infected Hamsters
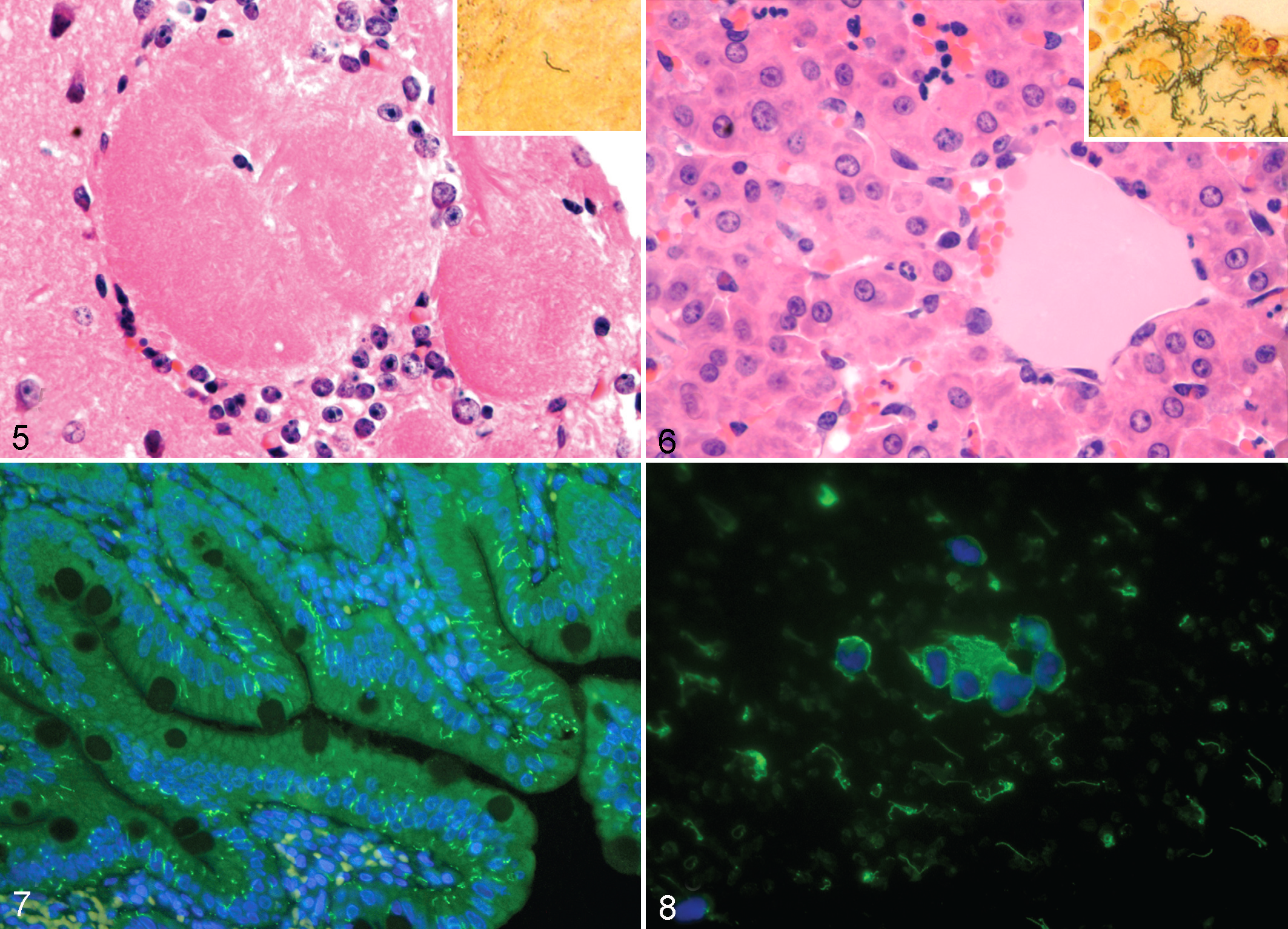
Tissue samples were processed for histopathology from hamsters infected with strains 203 and JB197 to assess the extent of bacterial dissemination during infection, and these data are summarized in Table 1. A detailed comparison of tissue distribution, hemorrhage, and bacterial distribution is provided in Supplemental Table 2. Samples of tissue from animals in all strain 203 infection groups were analyzed together since this strain failed to produce a lethal infection. Analysis of the tissue distribution of strain JB197 was limited to animals that received 1 × 102 to 1 × 103 bacteria; these 2 infection groups were selected because the infectious dose was below the LD50, and animals either survived to the end of the study or underwent a protracted infection typically lasting 12 DPI before clinical signs of infection were observed. Strain 203 was found in kidney and spleen of all infected animals and rarely in brain, liver, or pancreas. In contrast, strain JB197 was consistently detected in all organs analyzed (Table 1, Figs. 4–8), except uterus, where it was found in some but not all animals examined.
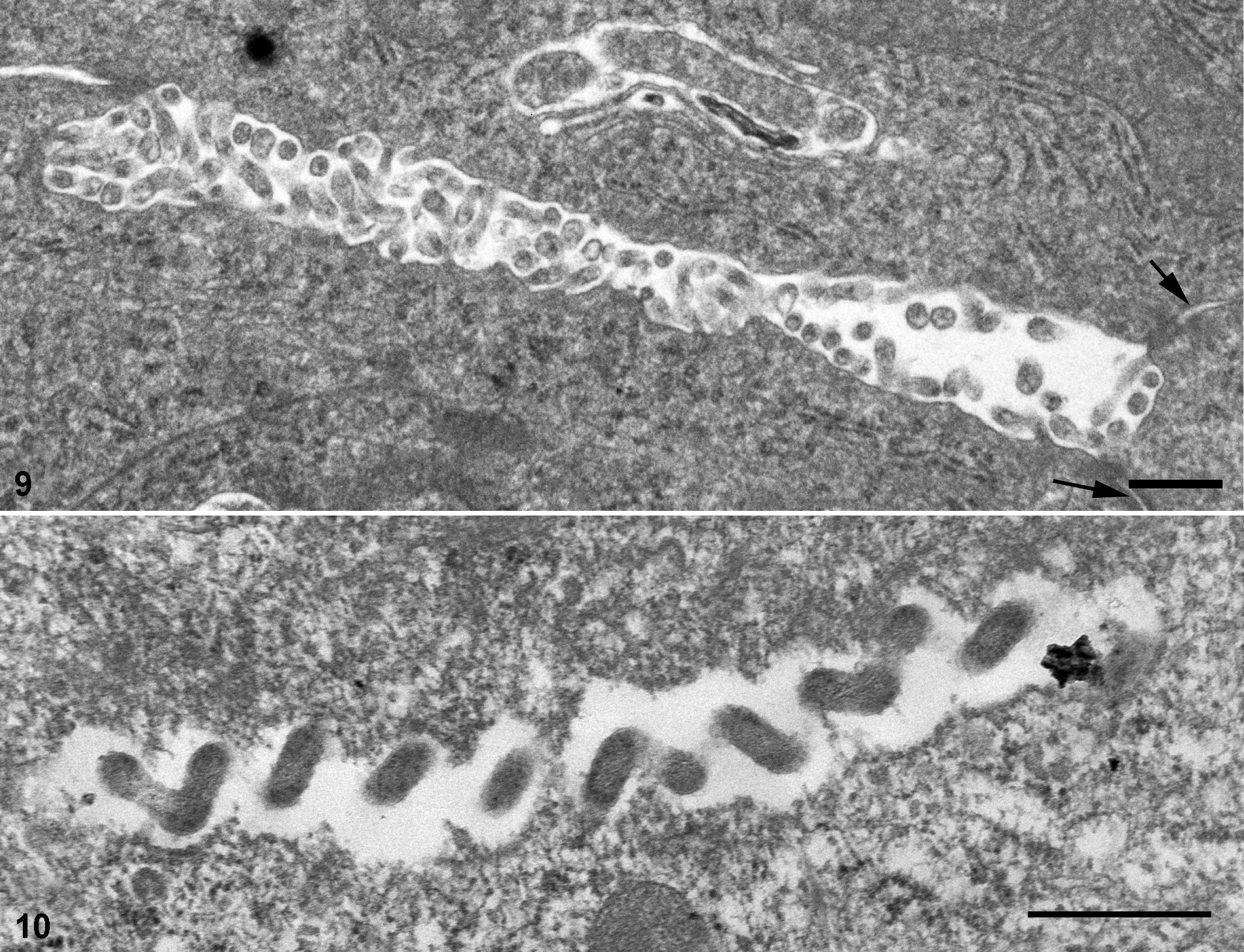
Within the central nervous system of hamsters infected with strain JB197, spirochetes were detected in the cerebellum, cerebrum, meninges, brain stem, and olfactory bulbs (Fig. 5). Lungs of acutely infected hamsters were often heavily colonized, and hemorrhage was often detected in the alveolar spaces (data not shown). In the livers of animals infected with strain JB197, Leptospira were predominantly found in tight junctions within the intercellular space (Fig. 6). Except where the space was filled by bacteria, hepatocytes remained in tight association, without evidence of dissociation (Figs. 6, 9). In each of these tissues, bacterial colonization was predominantly extracellular (Fig. 9), although intracellular bacteria were occasionally detected (Fig. 10). Inflammatory cells were found within blood vessels and in areas of overt hemorrhage but were not generally observed to extravasate into spirochete-infected tissue (Figs. 5, 6; data not shown). Leptospira were widely distributed throughout tissue of the small intestine (Fig. 7), and typically, bacteria were found as intracellular forms (Fig. 10).
Strain JB197 forms aggregates within blood. Aggregations of spirochetes in close association with white blood cells were frequently detected in blood smears from hamsters infected with JB197 (Fig. 8) and in blood vessels found throughout FFPE tissue sections (data not shown). Preliminary studies using electron microscopy of blood samples suggest Leptospira aggregate predominantly with neutrophils (data not shown).
Discussion
In this study, we describe development of a golden Syrian hamster model to study L. borgpetersenii serovar Hardjo pathogenesis. While cattle may be a normal host for serovar Hardjo, experimental design is often limited due to availability of appropriate facilities and related costs. In addition, cattle usually do not exhibit clinical signs of infection, thus limiting evaluation of disease progression. Mice have limited susceptibility to Leptospira infection and therefore do not provide a useful model for leptospirosis. In contrast, golden Syrian hamsters are susceptible to lethal infection with several species of Leptospira 15,25 and provide an excellent model for leptospirosis. However, hamster infection studies using serovar Hardjo have yielded inconsistent results; strains often varied in virulence or clinical outcome, 17,31,33,34 or immunosuppression was needed to achieve lethal infection. 1 Contributing to the confusion, previous reports that described lethal serovar Hardjo infections in hamsters were done before the discovery that serovar Hardjo is included in more than 1 species 9 ; it is unclear if the strains analyzed were L. borgpetersenii or L. interrogans. This is a critical point; genetic differences between L. borgpetersenii and L. interrogans result in substantial differences in phenotype between these 2 species. 10 Therefore, development of chronic and lethal infection models should help identify virulence traits that differ between well-defined strains of L. borgpetersenii serovar Hardjo and provide a way to directly compare virulence attributes among various pathogenic Leptospira species.
Lack of an infection model using small animals has contributed to the slow development of effective serovar Hardjo vaccines and detection of potential virulence traits for L. borgpetersenii. Although the intraperitoneal injection of live bacteria used here does not replicate a normal route of Leptospira infection, this route is routinely used to evaluate vaccine potency. 27 The route of infection used in this study is important to consider when evaluating some aspects of tissue invasion (eg, peritonitis) that are most likely due to the route of inoculation. However, it is important to document these events to better evaluate the development of disease in this model.
Hamsters inoculated with L. borgpetersenii serovar Hardjo strain 203 did not induce clinical signs of infection at any dose tested; therefore, an LD50 could not be determined for this strain. In contrast, the low ID50 (100 cells) for this strain and its ability to quickly colonize the kidneys indicate strain 203 is an efficient pathogen that can establish an asymptomatic, chronic infection. Thus, hamster infection using strain 203 may be a useful model for understanding features of host–pathogen interactions that occur in maintenance hosts, such as cattle. In hamsters infected with strain 203, bacteria localized primarily in the kidneys and spleen, with kidney infection being detected within 4 DPI. Within kidneys, Leptospira were found exclusively in the renal tubules. Other tissues rarely contained spirochetes following infection with strain 203, a finding in sharp contrast to the widespread proliferation of strain JB197. Strain 203, like strain JB197, was found primarily in an extracellular state. However, intracellular forms of JB197 in the liver, brain, and small intestine were detected. Although intracellular forms of L. interrogans have been reported using tissue culture systems, 3,23 to our knowledge detection of intracellular forms of L. borgpetersenii in animal tissue has not been previously reported.
Infection with strain JB197 established renal infection by 3 DPI and rapidly disseminated throughout the body. This rapid spread from the inoculation site may help account for the observed virulence; most hamsters infected with strain JB197 underwent an acute, lethal infection (LD50 = 3.6 × 104 bacteria). Strain JB197 virulence may be rapidly lost; we found empirically during the course of this study that consistent initiation of an acute infection required use of bacterial cultures that had undergone < 2 rounds of in vitro passage (data not shown). Generally, the delay between inoculation and onset of clinical signs of acute infection (4 to 25 DPI) was dependent on dose, with animals that received fewer bacteria having a longer delay before clinical signs appeared, yet the change from asymptomatic infection to clinical disease, including the appearance of external hemorrhage, was rapid and usually occurred over a 24-hour period. Animals infected with > 1 × 107 bacteria developed clinical signs of disease early (< 5 DPI) as compared with animals that were inoculated with < 1 × 105 bacteria, where clinical signs of infection were not seen until at least 10 DPI. Animals that received 106 bacteria developed chronic infections, but the kidneys exhibited gross pathologic evidence of infection, and animals were often reduced in size at the termination of the study as compared with normal controls. To help explain these results, we base our current model on findings with another spirochete, Borrelia burgdorferi. Hamsters infected with B. burgdorferi produce significant levels of bactericidal IgG within 1 week after infection, 21 and sera from infected mice confer passive protection. 4,5 Additionally, the infectious dose directly affects recognition of B. burgdorferi antigens; sera from animals infected with < 104 bacteria do not recognize OspA, a protein downregulated during infection, while animals infected with > 104 bacteria produce antibodies that react with antigens expressed during in vitro culture, including OspA. 4 In our model, animals initially develop an acquired immune response to antigens expressed by the inoculum—that is, antigens preferentially expressed by the bacteria during in vitro culture. However, animals that received > 107 bacteria (strain JB197) likely have sufficient bacterial load before immune mediated clearance is possible, resulting in clinical infection. Animals inoculated with fewer bacteria (< 105) should develop a primary immune response to in vitro expressed proteins, and because of the smaller infectious dose, the host achieves partial immune-mediated bacterial clearance. Bacteria in the population that survive the initial clearance are expected to express a different (in vivo) protein profile; subsequent bacterial replication should result in bacterial loads capable of inducing clinical disease before adequate development of immunity to in vivo–expressed antigens. Animals receiving inocula at the threshold between rapid and delayed onset of clinical disease may have sufficient bacterial loads containing bacteria expressing both in vivo and in vitro antigenic profiles and develop an immune response capable of restricting dissemination of bacteria expressing either protein profile. To test this hypothesis, future studies will focus on characterizing the specificity of antibodies developed during hamster infection using different infectious doses.
There are several differences in the pathology induced by L. borgpetersenii serovar Hardjo strain JB197 infection as compared with descriptions of L. interrogans serovar Icterohaemorrhagiae infection. 26,29 Hamsters infected with strain JB197 had multifocal small areas of centrilobular hepatocellular degeneration and necrosis, a result of heavy bacterial colonization. However, generalized dissociation of hepatocytes was not observed, and hepatocytes retained tight junctions adjacent to where bacteria reside despite bacteria being packed within intercellular spaces. In contrast, liver tissue from guinea pigs 26 or hamsters 29 infected with L. interrogans exhibit cellular dissociation with a smaller bacterial load. Surprisingly, inflammatory cells were primarily restricted to capillaries and areas of hemorrhage despite bacterial dissemination into brain, liver, and lung tissue in hamsters infected with strain JB197. In contrast, inflammatory cell infiltration was reported to occur in guinea pigs infected with L. interrogans. 26 These findings suggest that there may be differences in how L. borgpetersenii and L. interrogans strains interact with components of the host innate immune system.
A striking feature of the late stages of infection with strain JB197 was the appearance of spirochetes in the blood, either swimming free or in large bacterial aggregates with one or more leukocytes. This is the first known report showing that Leptospira form aggregates with leukocytes during infection and may be a unique feature of acute L. borgpetersenii serovar Hardjo infection; preliminary hamster infection studies with L. interrogans serovar Pomona led to detection of bacteria in blood but not in association with leukocytes. It is unclear if these aggregates develop as a result of an active bacterial process involving adherence to leukocytes, possibly via the CR3 receptor; 11 are a possible consequence of antibody development; or are the result of extracellular traps (NETs) 32 released by neutrophils. If these aggregates involve neutrophils, this may help to explain the clinical outcome of infection with strain JB197. Neutrophil activation and release of toxic cellular constituents is implicated in acute lung injury, 18 and widespread neutrophil activation following in vivo replication of serovar Hardjo may contribute to organ damage, leading to a rapid shift from asymptomatic infection to acute onset of clinical disease.
The chronic and acute models of serovar Hardjo infection described here provide a basis for ongoing pathogenesis studies. When comparing tissue distribution and pathology of serovar Hardjo infection by these 2 strains, we selected infectious doses below the LD50 for strain JB197 to mimic a more normal course of disease rather than simply provide an overwhelming infection resulting from inoculation with high numbers of bacteria. Future experiments that use sub-LD50 infectious doses of strain JB197 should make it possible to replicate a natural course of infection to help understand the progression of acute leptospirosis. In addition, development of a lethal infection model for serovar Hardjo should enable potency testing of Leptospira vaccines following accepted international standards, whereas development of a chronic infection model may enable testing of vaccines suitable for reducing infection in maintenance hosts.
Footnotes
Notes
Acknowledgements
We thank Richard Hornsby and Ami Frank for exceptional technical support; Judith Stasko and Virginia Montgomery for histopathology support; and the animal caretakers at the National Animal Disease Center. We thank Dr David Haake for his generous gift of LipL32 antiserum.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
