Abstract
A 22-week-old 21-kg female Bernese Mountain Dog ingested a topical antipsoriatic preparation containing the synthetic vitamin D analog tacalcitol. The dog died after a history of lethargy, recumbency, paresis of the hindlimbs, increased rectal temperature, dyspnea, and hematemesis. Histologic examination revealed metastatic calcification in the kidneys, lungs, myocardium, brain, stomach, and tear glands. The appearance of soft tissue mineralization in multiple organs is consistent with hypercalcemia derived from excessive vitamin D uptake. Oral toxicity studies for tacalcitol in the dog are not available, but the present report emphasizes the extraordinary toxic risk of drugs containing this vitamin D analog to dogs.
The present report describes the disturbance of calcium homeostasis associated with tacalcitol ingestion in a dog. A 22-week-old, 21-kg, female Bernese Mountain Dog was presented to a private practitioner for investigation of lethargy, weakness, and anorexia. On physical examination, the dog was recumbent and appeared depressed. Also, the dog showed paresis of the hindlimbs, reddish mucosal membranes, an increased rectal temperature (39.9 C), and a slightly increased respiratory rate. Abdominal palpation did not elicit signs of pain. Therapy was initiated by administration of prednisolone and amoxicillin. During the subsequent night, the dog developed hematemesis, deteriorated rapidly, and died.
Gross necropsy findings were limited to the lungs, which appeared blood-filled and were of increased consistency. Subsequent histologic examination revealed widespread soft-tissue calcification in multiple organs including the kidneys, lungs, myocardium, brain, stomach, and tear glands. In particular, hematoxylin and eosin stains of kidney sections revealed mineralization of arteries as well as mineral deposition in the tubular basement membranes (Fig. 1). The Bowman's capsule and the basement membranes of glomeruli were also mineralized. Because of extensive postmortem degeneration, the lumen of the tubules contained mainly epithelial cell debris. Mineralized arteries and a few mineralized veins were found in the collapsed lungs. The pulmonary parenchyma also contained diffuse mineral deposits in the interstitium. Foci of mineralized myocytes were present in the myocardium. The basement membranes of ependymal cells in the brain and some arteries of the choroid plexus were also mineralized. Similarly, stomach sections revealed mineralization of the basement membrane of the mucosa. In addition, some parietal cells, the lamina muscularis, and some arteries were mineralized. In the tear glands of the third eyelid, the basement membrane was again the main target of mineralization. All these soft tissue calcifications were characterized by their purplish color upon hematoxylin and eosin staining. Using the von Kossa staining technique, on the other hand, these calcifications could be visualized as black salt deposits. For example, Fig. 2 illustrates the mineralization observed in stomach sections. A concomitant infection was suggested by the presence of inflammatory cells in the lungs and liver, whereas the accompanying lymph nodes, Peyer's patches, and the spleen were partially depleted. However, bacterial cultures failed to reveal a causal agent in either of the affected organs.
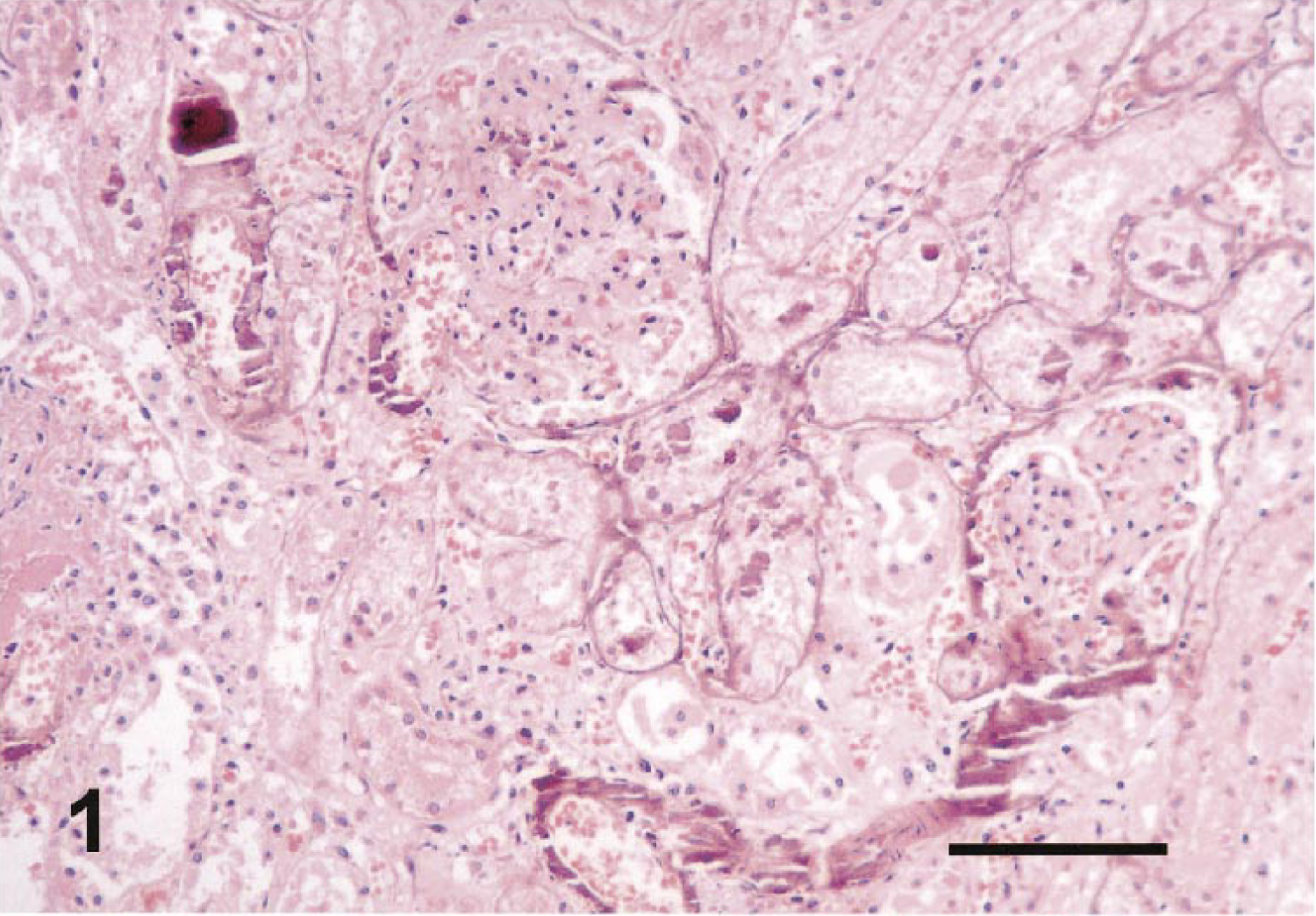
Histology of mineralized kidney. The section was stained with hematoxylin and eosin. Note the characteristic purplish color of calcium deposits in the blood vessel walls, in the basement membrane of renal tubules, and in the glomeruli. Bar = 150 μm.
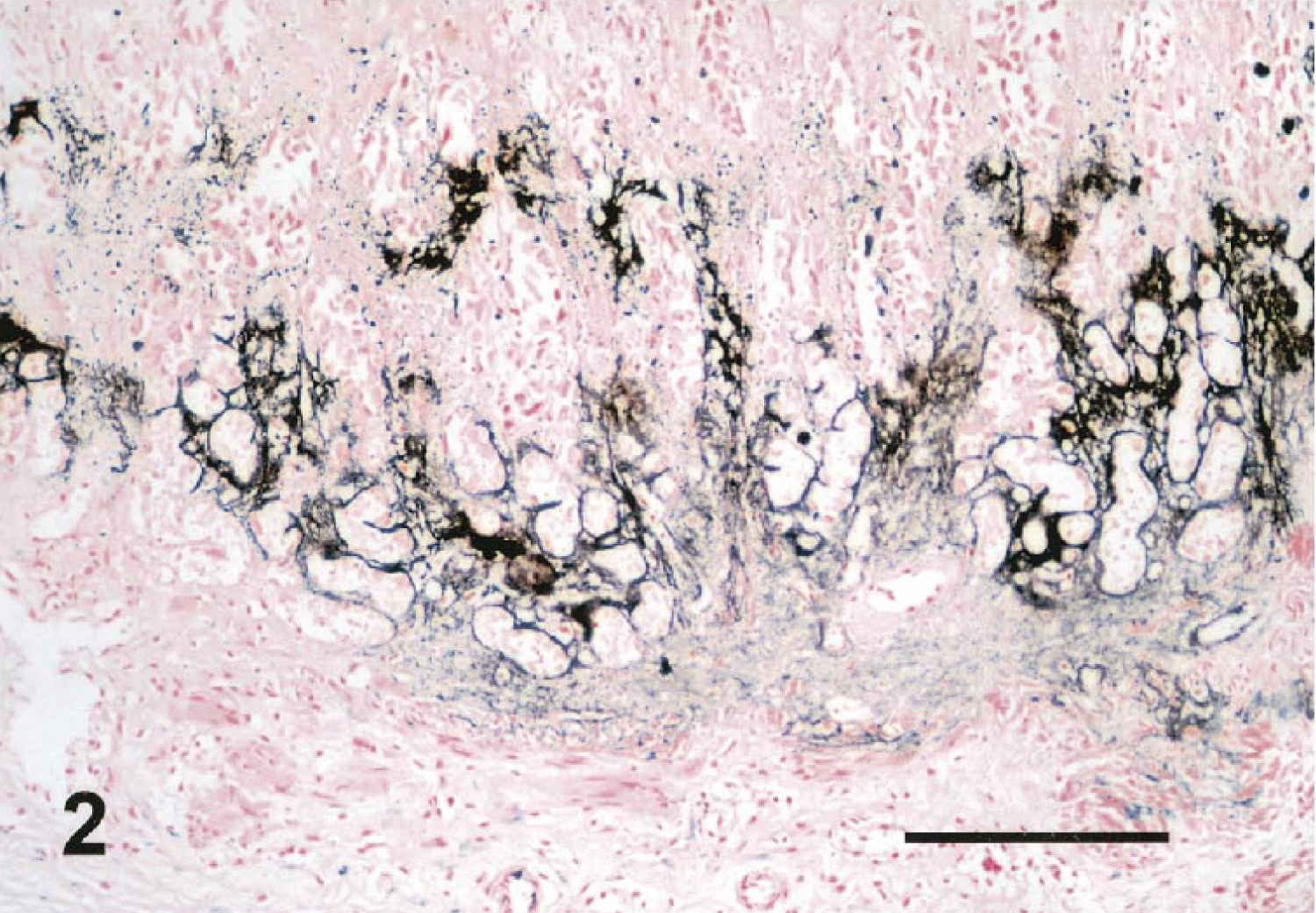
Von Kossa staining of a stomach section. Note the calcium deposits visualized as black staining upon treatment with silver nitrate. Bar = 300 μm.
The slight calcification observed in the lungs might have been dystrophic, associated with the inflammatory process in the pulmonary tissue. However, no plausible explanation could be found for the widespread metastatic calcification observed in all other organs. Renal failure is a possible cause of hypercalcemia, 3 but it is unlikely that the modest changes detected in the kidneys were responsible for such severe calcifications. Also, no macroscopic or microscopic evidence was found of neoplasia or changes in the pituitary or adrenal glands, ruling out paraneoplastic syndrome, hyperparathyroidism, and hypoadrenocorticism as the cause of the extensive calcification. On the other hand, the clinical and pathologic abnormalities in this case are identical to the known toxic effects of large doses of vitamin D, 2–5 prompting a tentative diagnosis of vitamin D poisoning. Upon further questioning, the owner remembered that he had found a damaged and emptied 20-g tube of a topical antipsoriatic preparation containing tacalcitol at 4 μg/g, indicating that the dog had ingested its entire contents 36–48 hours before the onset of clinical symptoms. Precise quantitation of the total amount of material ingested was not possible because the owner also mentioned possible previous exposures to the same drug. No evidence was found of other potential sources of excessive vitamin D uptake such as plants, dietary supplements, or rodenticide baits.
Vitamin D and its active derivatives are hormones that regulate serum calcium levels by stimulating calcium mobilization from bones, by increasing the intestinal absorption of calcium, and by reducing its renal excretion. 3 4 Vitamin D binds to cytosolic receptors within target cells, and the resulting receptor–hormone complex interacts with DNA either to enhance or to inhibit gene transcription. The hypercalcemia deriving from the excessive uptake of vitamin D causes precipitation of calcium salts preferentially between elastic or collagen fibers in blood vessels, kidneys, heart, lungs, and other organs. Such calcium depositions lead to massive cell injury and necrosis. In addition, hypercalcemia produces vasoconstriction leading to severe ischemic tissue damage. 3 4 Previous reports already illustrated the potential toxic risk that another synthetic vitamin D analog, calcipotriol, poses to dogs; 1 3 however, to the best of our knowledge, severe metastatic mineralization with fatal outcome following ingestion of tacalcitol has never been reported. Tacalcitol is a vitamin D analog that is widely used as an effective and apparently safe agent for the treatment of psoriasis because it inhibits keratinocyte proliferation and induces cell differentiation. Interestingly, the low tacalcitol concentration of the material ingested in the present case indicates that this compound is particularly toxic to dogs. Single-dose oral toxicity trials in the dog have not been reported, but the low median lethal dose (LD50; 10 μg/kg body weight) in single subcutaneous injections to Beagles confirms that dogs are indeed up to 40 times more susceptible to tacalcitol toxicity than mice and rats (Acute toxicity of TV-02 in subcutaneous administration to Beagles, Study No. S017A0D300; Teijin Institute for Bio-Medical Research, Tokyo, Japan, 1989 [Hilbe, unpublished]). The ingestion of products containing vitamin D or vitamin D analogs is particularly dangerous because of the late onset of serum electrolyte abnormalities and clinical signs of poisoning. As in the case reported here, this lag period may unfortunately delay the initiation of diagnostic and therapeutic measures. The recommended treatment after uptake of toxic doses of vitamin D or its analogs includes administration of fluids and electrolytes, diuresis with furosemide and administration of synthetic glucocorticoids, possibly in conjunction with calcitonin and a low-calcium diet. 1–3 This therapy must be sustained for a prolonged period of time (up to 2 weeks), while closely monitoring serum calcium concentrations as treatment is withdrawn. 5
