Abstract
A commercial diet fed to a colony of inbred strain 13 guinea pigs for approximately 6 weeks was subsequently recalled for excessive levels of vitamin D. Twenty-one of 62 animals exhibited clinical signs, including anorexia, lethargy, and poor body condition. Nine affected and 4 clinically normal animals were euthanized for further evaluation, including serum chemistry, urinalysis, and gross and/or histopathology. Macroscopic findings included white discoloration in multiple organs in 8 animals, and microscopic evaluation confirmed multiorgan mineralization in tissues from 7 animals. Serum 25-hydroxyvitamin D levels were elevated in 10 animals. Serum inorganic phosphorus and alkaline phosphatase levels were increased in all exposed animals; however, total calcium and ionized calcium levels were not significantly higher in exposed animals than in control strain 13 guinea pigs from a different institution. The data support a diagnosis of hypervitaminosis D with metastatic calcification. Following the diet recall, the remaining guinea pigs increased their food intake and regained body condition. Diagnostic testing of 8 animals euthanized approximately 3 months after returning to a normal diet demonstrated that serum parathyroid hormone remained significantly lower, and ionized calcium and ionized magnesium were significantly higher, in recovered animals compared to controls and exposed animals. These results indicate that diagnostic tests other than serum calcium are necessary for a diagnosis of hypervitaminosis D in guinea pigs.
Vitamin D is a fat-soluble steroid hormone necessary for calcium (Ca) and phosphorus (P) homeostasis and bone mineralization. 4,18 Recent studies demonstrate a broader role for vitamin D in maintaining health, including protection against autoimmunity, infection, and cancer. 1,23 Synthesis of the active form, 1,25 dihydroxyvitamin D (1,25(OH)2D; calcitriol), is a multistep process that begins either with synthesis of vitamin D3 (cholecalciferol) in the skin following exposure to UV irradiation or from dietary intake of vitamin D2 (ergocalciferol) or D3. 18 Ingested vitamin D is transported to the liver, where it is converted to 25-hydroxyvitamin D (25(OH)D), the major circulating form of the vitamin. 25(OH)D is then transported to the kidney, where it is converted to the active form, 1,25(OH)2D, in the proximal tubules. Vitamin D exerts its effects through binding of the vitamin D receptor (VDR), a nuclear protein moderately to highly expressed in most tissues, and by regulating gene transcription in a number of cell types.
Hypervitaminosis D typically occurs following ingestion of high doses of vitamin D, generally through excess supplementation or from ingestion of toxic plants or contaminated sources. 2,11,21 In hypervitaminosis D, circulating levels of 25(OH)D as well as multiple metabolites are increased while 1,25(OH)2D typically remains within the normal range, indicating a tight regulation at this level. 12 It has been suggested that when vitamin D is present in toxic doses, inactive metabolites including 25(OH)D may overcome the regulatory mechanisms and enter the cells to bind the VDR and stimulate gene transcription. Reported effects of hypervitaminosis D include an increase in intestinal absorption of calcium and increased bone resorption with resultant hypercalcemia and hypercalciuria. 12 Intestinal absorption of P is also increased and, along with high Ca levels, leads to soft tissue calcification. Clinical signs associated with hypervitaminosis D result from perturbations in body mineral levels and tissue mineralization and include anorexia and depression, stiffness, and lameness. 18
Soft tissue calcification, or metastatic calcification, has been documented in guinea pigs since the 1940s and is still reported as a disorder that primarily affects males older than 1 year. 8,19 The majority of reported spontaneous and experimentally induced cases resulted from altering dietary P, magnesium (Mg), and/or Ca, with one group determining that “unsuitable amounts, or possibly ratios, of calcium and phosphorous are the principal cause of calcification.” 6 Metastatic calcification has also been reported to occur subsequent to hypervitaminosis D in several species, including dogs, cattle, and humans. 9,16 –18,24 In this article, we describe hypervitaminosis D in a closed colony of inbred strain 13 guinea pigs fed a commercial laboratory animal diet that was subsequently recalled for elevated levels of vitamin D.
Materials and Methods
Animals
Exposed animal colony
A colony of 62 inbred strain 13 guinea pigs consisting of breeding pairs and experimental animals was housed in standard guinea pig caging in an Association for Assessment and Accreditation of Laboratory Animal Care International (AAALAC)–approved facility at Massachusetts General Hospital (MGH, Boston, MA). Animals ranged in age from neonate to 33 months. The colony was established in November 2009 from 3 breeding pairs received from the US Army Medical Research Institute of Infectious Diseases (USAMRIID, Frederick, MD) and was negative for simian virus 5, Sendai virus, pneumonia virus of mice, reovirus type 3, lymphocytic choriomeningitis virus,
Control animals
Control serum was obtained from strain 13 guinea pigs at USAMRIID. Animals were housed in standard guinea pig caging (LabProducts, Seward, DE). The room was maintained with a light cycle of 12:12 hours and with no fewer than 12 air changes per hour. Colony animals were negative for pathogens indicated above; they were not tested for GpAV. Animals were fed ad libitum Teklad2040 S global sterilizable guinea pig diet (Harlan, Madison, WI) containing 1.5 IU/g vitamin D3, 1.1% Ca, 0.8% P, 0.5% nonphytate P, 0.3% Mg, and 1.3% K. Regular dietary supplement included spinach offered a minimum of twice weekly. All research was conducted under an IACUC–approved protocol in compliance with the Animal Welfare Act, Public Health Service (PHS) policy, and other federal statutes and regulations relating to animals and experiments involving animals.
Bones for comparative histology were harvested from 1 male Hartley guinea pig (case No. 28), obtained from Elm Hill Labs (Chelmsford, MA) and used in a reproductive study at Massachusetts Institute of Technology (MIT, Cambridge, MA). The guinea pig was housed in standard guinea pig caging (Allentown, Allentown, NJ). The room was maintained with a light cycle of 12:12 hours and with 10 to 12 air changes per hour. Animals were fed ad libitum Purina 5025 global sterilizable guinea pig diet (Harlan, Madison, WI) containing 3.1 IU/g vitamin D3, 1.1% Ca, 0.6% P, 0.37% nonphytate P, 0.35% Mg, and 1.57% K. All research was conducted under an IACUC-approved protocol in compliance with state, local, and federal regulations.
Animal number designation
Individual animals undergoing diagnostic tests are indicated in Supplemental Table S1. Thirteen animals (case Nos. 1–13) that were exposed to the contaminated diet, their sex, and the age at which they were euthanized for blood and/or tissue collection are indicated in the columns on the left (“Exposed” animals). The exposed animal group consisted of 7 males ranging from 4 months of age (case No. 1) to 33 months (case No. 7) and 6 females ranging from 5 months of age (case No. 8) to 28 months (case No. 13). Serum was obtained from 6 guinea pigs at USAMRIID as controls and was analyzed using the same instrumentation as that used for exposed animals for serum chemistries (Suppl. Table S1, middle columns, “Control”). The control group consisted of 3 males (case Nos. 14–16) ranging from 14 to 15 months old and 3 females (case Nos. 17–19) ranging from 12-15 months of age. Blood and/or tissues were obtained from 8 animals from the exposed colony 3 months after they were returned to normal chow (Suppl. Table S1, right columns, “Recovered”). The recovered group of animals consisted of 1 male that was 7 months old (case No. 20) and 7 females between 4 and 17 months of age (case Nos. 21–27). Clinical signs exhibited by exposed and recovered animals while on the high vitamin D diet are also shown.
Clinical Chemistry and Urinalysis
Exposed and recovered animals were euthanized by intraperitoneal injection with Euthasol euthanasia solution (Butler Schein Animal Health, Dublin, OH) containing 390 mg pentobarbital sodium and 50 mg phenytoin sodium. The control Hartley guinea pig (case No. 28) was euthanized by intraperitoneal injection with Fatal Plus (Vortech Pharmaceuticals, Dearborn, MI). Blood samples were collected via cardiac puncture at necropsy into serum separator tubes. Control strain 13 guinea pigs were anesthetized by intramuscular (IM) injection with ketamine (60.6%; Vedco, St Joseph, MO), xylazine (33.3%; AnaSed, Vedco), and acepromazine (6.0%; Boehringer Ingelheim, St Joseph, MO) at 0.2 ml/400 g of body weight. Blood was collected from the cranial vena cava. Serum chemistries were analyzed on a Dri-Chem 7000 (Heska Corporation, Loveland, CO). Urine was collected from exposed animals by cystocentesis at necropsy and held at room temperature for immediate analysis. The color and clarity were recorded, and a direct wet mount microscopic examination was performed. Urine dipstick (Chemstrip; IDEXX, North Grafton, MA) was performed, and refractometer readings for urine-specific gravity (USG) were taken. Urine sediment was analyzed microscopically with unstained and Wright-Giemsa–stained slides. Urine and cytology preparations were reviewed by a board-certified veterinary pathologist (D.E.B.). Serum 25(OH)D, ionized calcium (iCa), ionized magnesium (iMg), parathyroid hormone (PTH), and calcitriol were measured at the Michigan State University Diagnostic Center for Population and Animal Health. Ionized Ca and iMg levels were adjusted for pH per standard methods.
Histopathology
Tissue sections were fixed in 10% neutral-buffered formalin and were routinely processed and embedded in paraffin according to an accepted histologic technique. Bones were decalcified in 20% formic acid. Then, 5-μm-thick sections of all tissues were stained with hematoxylin and eosin (HE), and some were also stained by von Kossa’s technique with HE counterstain for histopathological evaluation by 2 board-certified veterinary pathologists (N.M.P. or T.M.A.).
Statistics
Serum chemistry values were compared between groups of animals using the Mann-Whitney nonparametric test. Statistical analysis was performed using GraphPad Prism 4.0 (GraphPad Software, La Jolla, CA), with
Results
Clinical History
A colony of inbred strain 13 guinea pigs consisting of breeding and experimental animals was maintained at MGH from the fall of 2009 until the fall of 2012. Over a 6-week period in the summer of 2012, twenty-one of the 62 colony animals began exhibiting nonspecific clinical signs, including anorexia, lethargy, and poor body condition (PBC). No prior medical problems other than occasional complications associated with breeding had been observed in the colony. Based on the presenting signs and widespread distribution of affected animals, differential diagnoses focused on infectious and metabolic diseases. During the period in which in-house diagnostics were being performed, the manufacturer of the guinea pig feed used in the facility announced a recall of several lots because of high levels of vitamin D in the formulation. Comparison with manufacturing dates indicated that our facility had received 3 lots of potentially tainted feed. One lot was determined by the manufacturer to contain 565 IU/g, more than 350 times the 1.5 IU/g typically found in the diet. To the best of our estimation, the first animal presented with anorexia and reluctance to move 2 weeks after receiving the contaminated diet, with an additional 3 animals presenting with PBC during the following 12 days. In total, approximately 30% of the animals, both males and females, exhibited nonspecific clinical signs as listed in Supplemental Table S1. Severity of presenting signs in exposed males was not obviously age dependent, while signs tended to be more advanced in sows older than 9 months than in younger females.
Gross and Microscopic Findings
Nine of the 21 animals showing clinical signs and 4 clinically normal animals were euthanized for blood collection and/or necropsy and tissue collection. Gross lesions were observed in most animals exhibiting clinical signs and in 1 clinically normal 4-month-old male (case No. 1) (Suppl. Table S2). Lesions included white discoloration of the cardiac muscle, gastrointestinal (GI) tract, skeletal muscles, and/or kidneys; cystic kidneys; and thickening of the urinary bladder wall (Suppl. Table S2, Fig. 1). Histological evaluation was performed on the tissues indicated in Supplemental Table S2 from 4 males exhibiting clinical signs (case Nos. 2, 4, 6, and 7) and from 3 clinically normal animals (case Nos. 1, 5, and 9). Microscopic lesions were observed in tissues from all 7 animals and tended to be more extensive than those observed grossly. Microscopic lesions included mild to marked mineralization of heart, lung, stomach, kidneys, and/or skeletal muscle. Fibrosis and inflammation composed predominately of macrophages were associated with the mineralization. Lesions were less severe in clinically normal animals, with mild hepatocellular vacuolization and mild (case Nos. 5 and 9) to moderate (case No. 1) mineralization of the indicated organs (Suppl. Table S2).
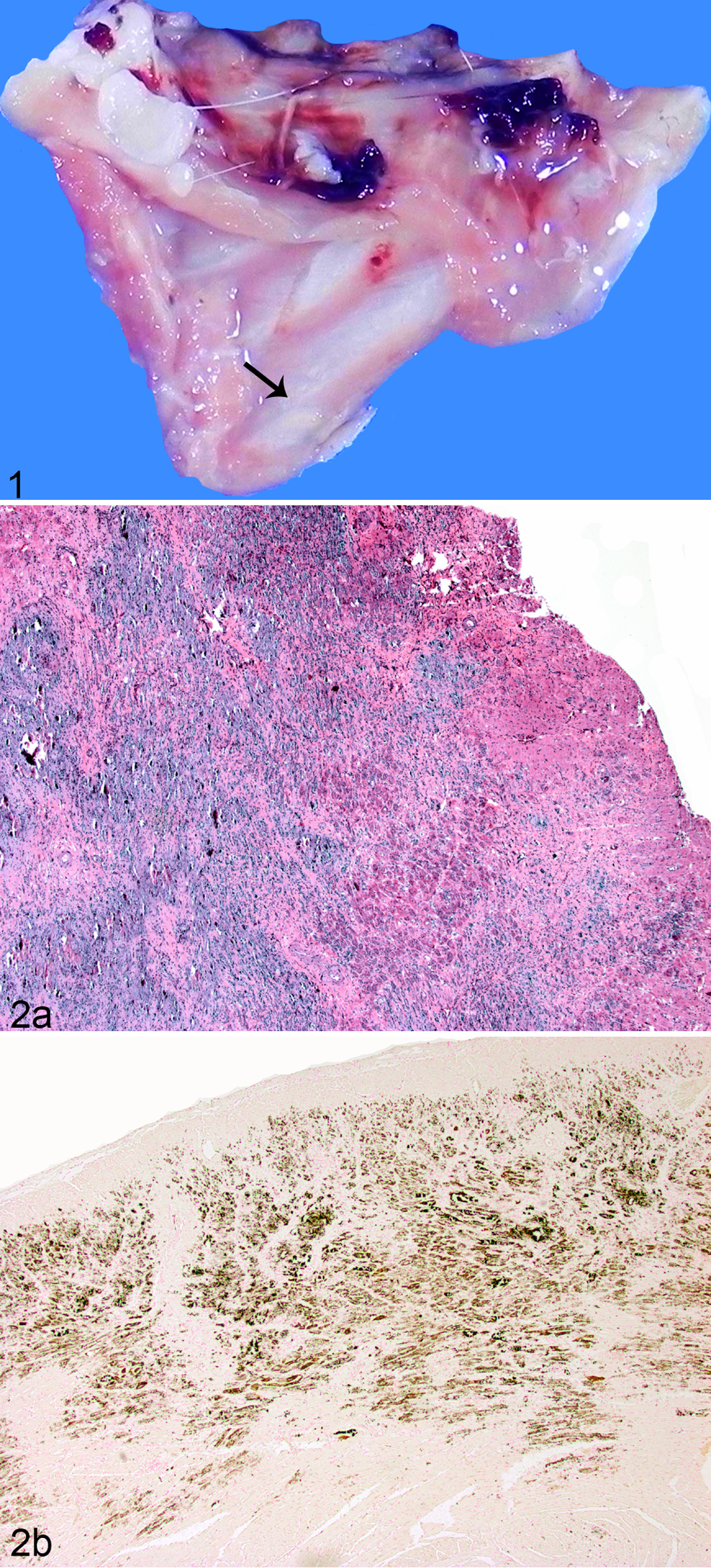
Hypervitaminosis D, mineralization, intercostal muscle, guinea pig, case No. 4. White discoloration on intercostal muscle (arrow).
Figures 2 and 3 show representative pathological changes observed in a 27-month-old male guinea pig (case No. 6). Marked multifocal to coalescing myocardial mineralization with myofiber necrosis was found throughout the cardiac tissue (Fig. 2a). Vascular basement membrane mineralization and focal arteriolar subintimal fibrosis were seen at higher magnification (not shown). Von Kossa staining confirmed the presence of mineral deposition in these tissues (Fig. 2b). Prominent mineralization was also present in cortical and medullary renal tubules and glomerular basement membranes (Fig. 3).
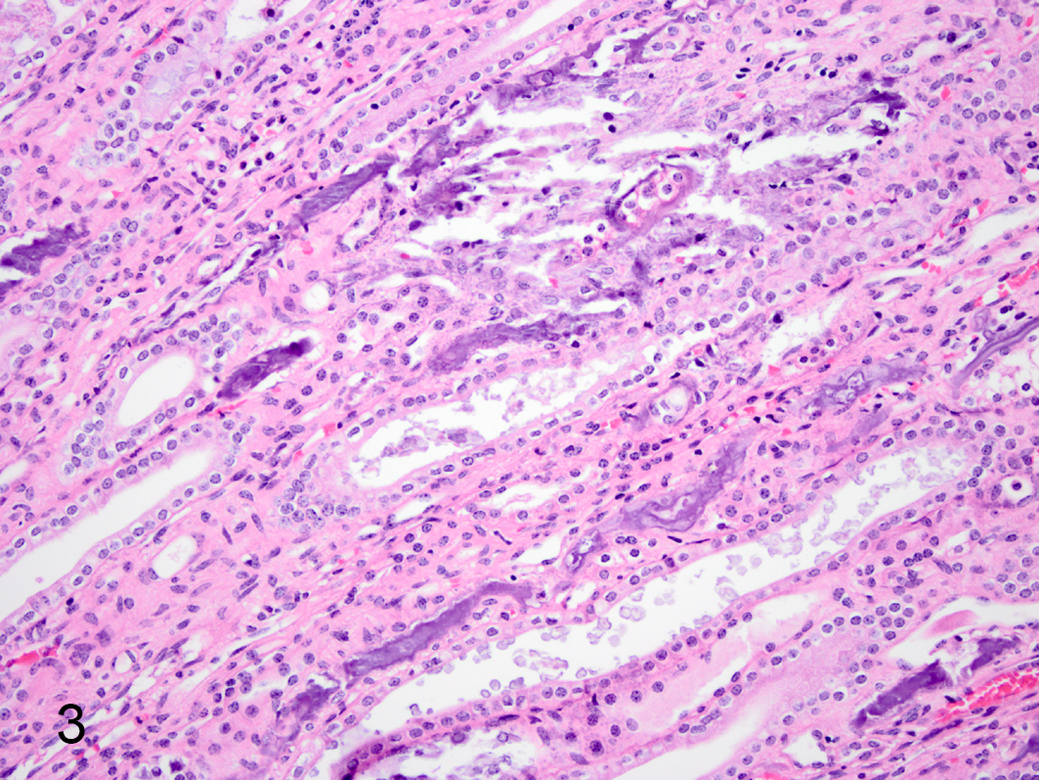
Hypervitaminosis D, renal tubular mineralization, kidney, guinea pig, case No. 6. Foci of mineralization within medullary tubules. Hematoxylin and eosin.
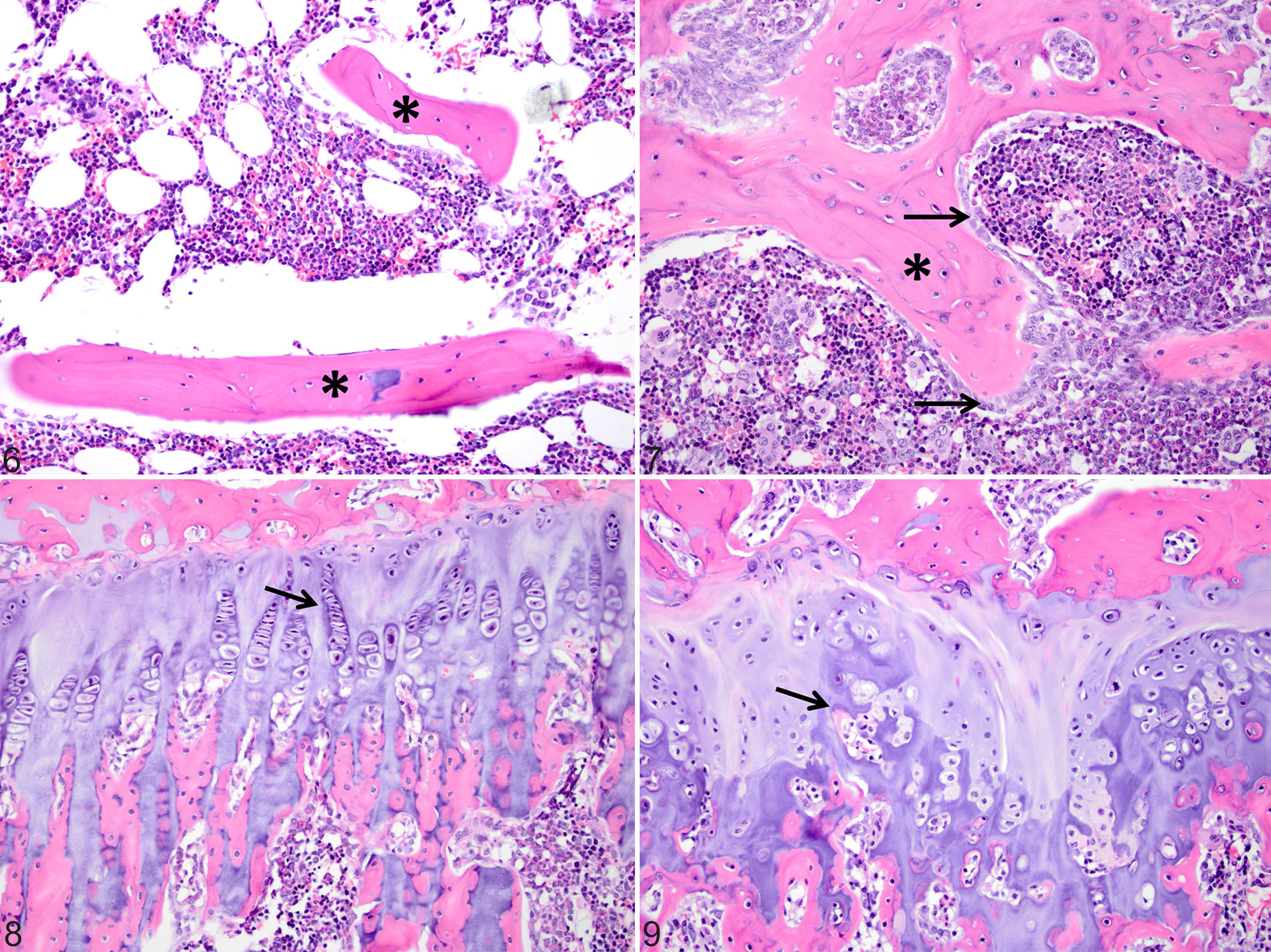
HE-stained sections of bone from the tibia, femur, and rib, from an exposed 6-month-old male (case No. 4) were examined (Figs. 5, 7, and 9) and compared with sections from an age- and sex-matched outbred Hartley guinea pig as a control (case No. 28) (Figs. 4, 6, and 8). Histopathologic changes of osteosclerosis were seen in the affected guinea pig. Lesions were most pronounced in the tibia, where the medullary trabeculae were markedly thickened by wide osteoid seams with increased numbers of osteoblasts, although osteoclasts were also present (Figs. 4 and 6, control; Figs. 5 and 7, exposed). The physis was slightly thinner, with regions of increased eosinophilia of the physeal and articular cartilage due to loss of matrix proteoglycans. Within the physis, chondrocytes were somewhat disorganized, with some lateral dispersion of normally palisaded cells and poor delineation of chondrocytes in the proliferative zone (Fig. 8, control; Fig. 9, exposed). Within the articular cartilage, there was reduced basophilia, consistent with proteoglycan loss, particularly in the regions of territorial matrix immediately surrounding chondrocytes, and reduced delineation of chondrocyte palisades. The articular cartilage was thinned in some areas, and its surface was multifocally roughened and irregular. The periosteum was also thickened and contained foci of mineralization, which were also seen in the ligaments and muscles of the stifle joint, as well as in regions of the articular cartilage of the tibia (not shown).
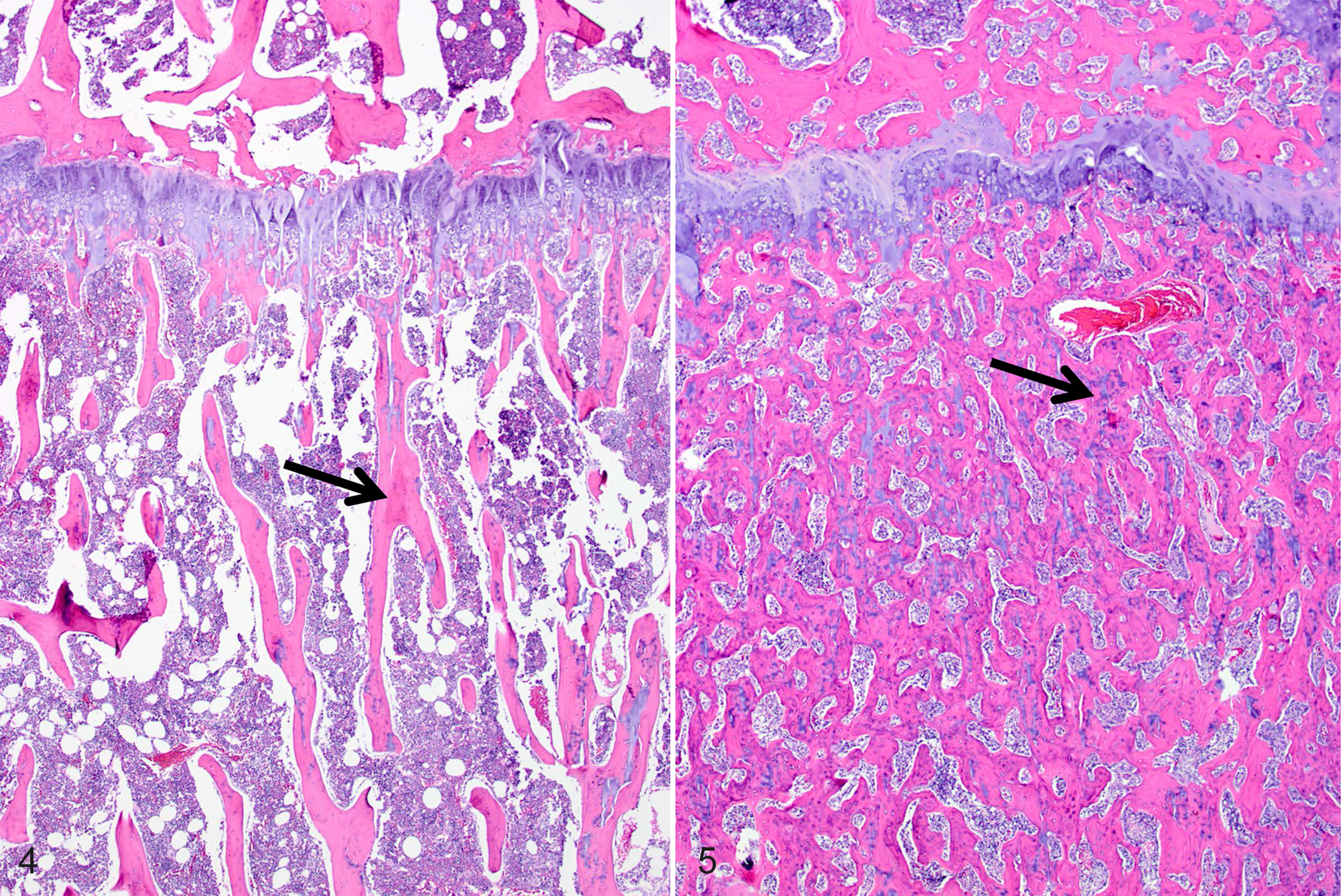
Normal tibia, guinea pig, case No. 28. Trabecular bone (arrow) from a control animal. Hematoxylin and eosin (HE).
Normal tibia, guinea pig, case no. 28. Trabecular bone (asterisks) from a control animal. Hematoxylin and eosin (HE).
Additional lesions of note in animals exhibiting clinical signs included moderate chronic interstitial nephritis and diffuse centrilobular hepatocellular vacuolation consistent with fatty change (not shown). Blood vessel changes consistent with hyaline arteriolar sclerosis were seen in the urinary bladder of a 33-month-old male guinea pig (case No. 7) and the heart of a 5-month-old male (case No. 2). Colony animals were serologically positive for GpAV but did not show any gross or histological evidence of clinical disease attributable to GpAV (data not shown).
Clinical Pathology and Urinalysis
Twelve of the exposed animals had blood collected for serum chemistries; 10 samples were submitted for vitamin D analyses as well as comprehensive metabolic chemistries. Because normal values had not been established for the exposed colony, serum samples were obtained from the originating colony at USAMRIID to use as controls (Suppl. Table S1, middle columns).
Results from the vitamin D panel are shown in Figs. 10 to 14 and Supplemental Table S3. The average serum concentration of 25(OH)D in exposed animals was 954.4 ± 35.30 nmol/l (Fig. 10). This was significantly higher (
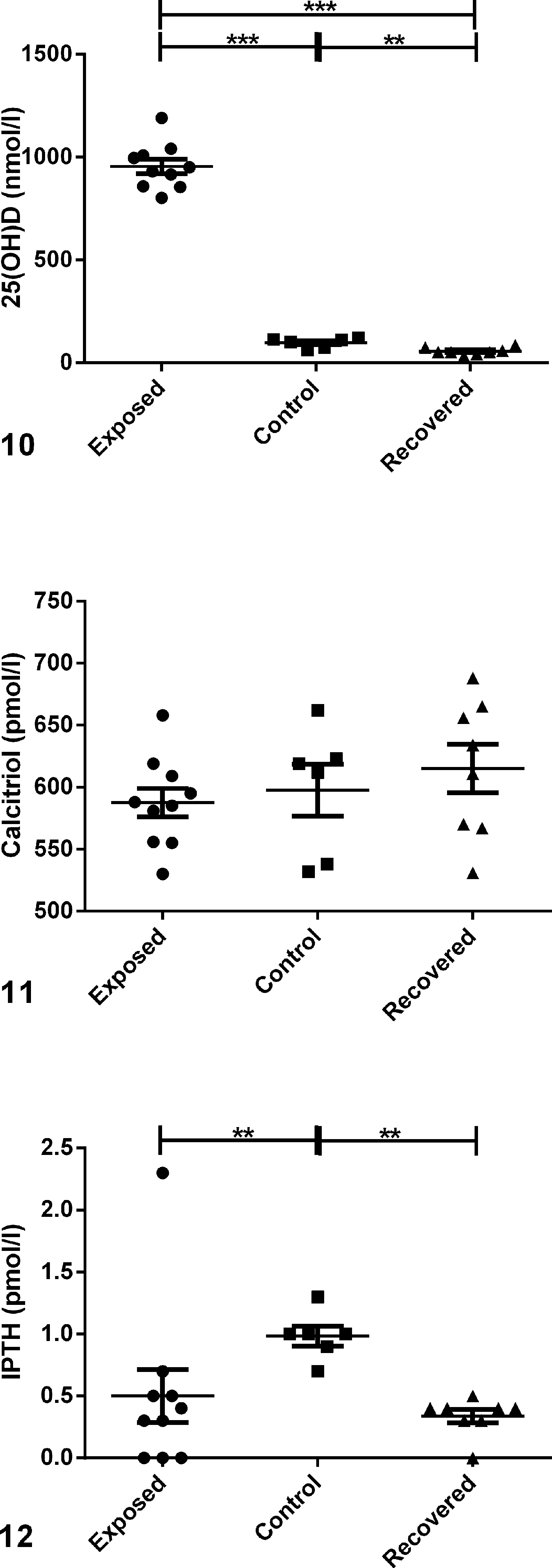
Vitamin D panel comparing serum levels in guinea pigs exposed to excessive dietary levels of vitamin D (“exposed”), nonexposed control animals (“control”), and animals exposed to excessive dietary vitamin D and returned to normal food for approximately 3 months (“recovered”); scatter plots.
iCa and iMg were measured as part of the vitamin D panel in this study. iCa measures only Ca that is free in the blood, whereas serum total calcium measures free or ionized Ca; complexed calcium that is bound to phosphate, sulfate, citrate, and lactate; and Ca bound to calcium binding protein (CBP). 10,22 Therefore, iCa is a better indicator than serum total Ca of Ca homeostasis. No significant difference in serum iCa levels was observed between exposed and control animals (Fig. 13). In fact, the trend indicated slightly lower levels in exposed animals than in control animals. Although not statistically significant, serum iMg also tended to be lower in exposed animals than in control animals (Fig. 14).
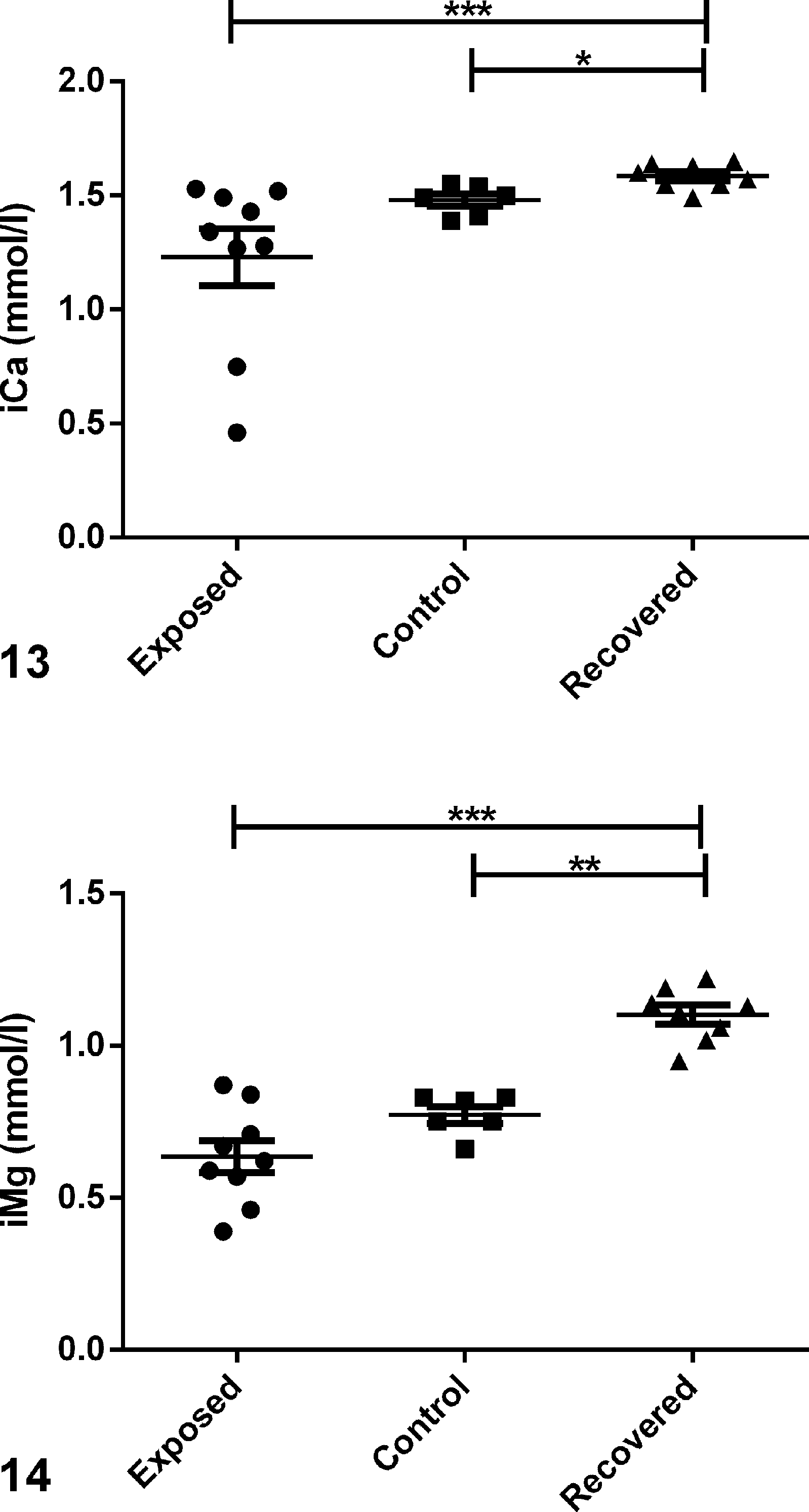
Vitamin D panel comparing serum levels in exposed, control, and recovered guinea pigs; scatter plots.
Serum chemistry values are shown in Figs. 15 to 19 and Supplemental Table S4. Consistent with hypervitaminosis D, exposed animals had significantly (
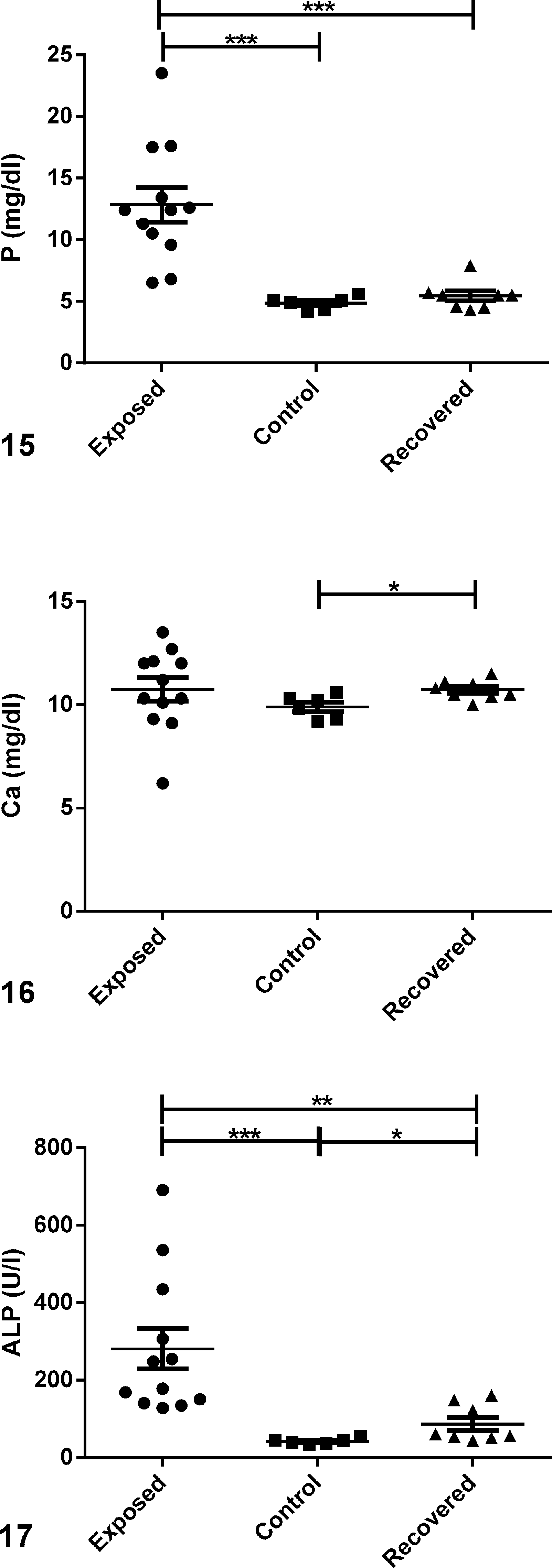
Serum chemistries comparing serum levels in exposed, control, and recovered guinea pigs; scatter plots.
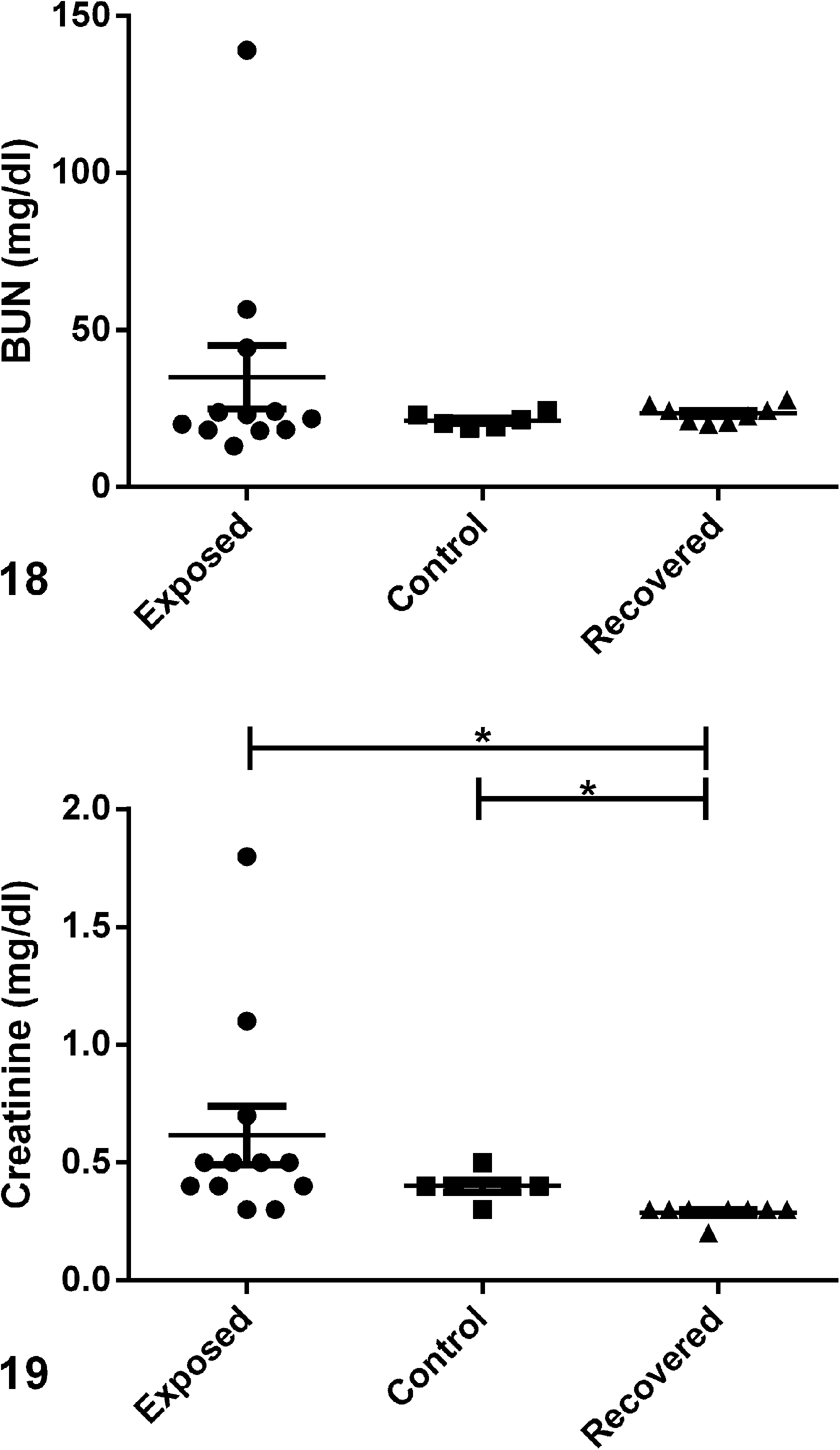
Serum chemistries comparing serum levels in exposed, control, and recovered guinea pigs; scatter plots.
Alkaline phosphatase (ALP) was markedly higher in serum from exposed animals than from controls (281.2 ± 52.49 U/l vs 42.50 ± 3.00 U/l,
Urine was collected from 4 males (case Nos. 1, 2, 6, and 7) and 3 females (case Nos. 9, 12, and 13). Microscopic examination of urine sediment from all 7 animals revealed crystalluria. Crystal morphology was most consistent with calcium carbonate, amorphous phosphate, and calcium oxalate; however, without biochemical urolith analysis, the composition cannot be specifically defined. All urine samples were positive for blood, ranging from 2+ to 4+, and 6 of the 7 samples were positive for protein, ranging from trace to 4+ (Suppl. Table S5). Interestingly, the 3 males with increased BUN (case Nos. 2, 6, and 7) also had low urine pH between 5 and 6, while the remaining 4 animals had normal urine pH between 8 and 9 (Suppl. Table S5). The normal alkaline urine was too cloudy to ascertain an accurate USG, while USG of the 3 males with acidic urine ranged from 1.020 to 1.026.
Three-Month Follow-up
Recalled guinea pig diet was discarded and replaced with normal feed in mid-July 2012. Guinea pigs remained mildly anorexic for several weeks but gradually resumed normal food intake over 4 to 5 weeks. To determine if the colony had any chronic effects from prolonged intake of the tainted diet, 1 male and 7 female guinea pigs ranging in age from 4 to 17 months were euthanized for serum and tissue collection 3 months after the diet recall and placement of a newly formulated diet. Five animals (case Nos. 20–24) did not exhibit signs of clinical disease while on the tainted diet (Suppl. Table S1, right columns). The 3 older animals (case Nos. 25–27) had mild PBC. Gross lesions were not recorded for any of the 8 animals at the time of necropsy.
Sections of liver, lung, heart, and kidney were submitted from 3 females ranging in age from 4 to 15 months (case Nos. 22, 24, and 26). Kidney sections from all 3 animals exhibited chronic nephropathy with mineralization of the cortical and medullary tubules and glomerular basement membrane (not shown). Bronchiolar and/or alveolar mineralization was observed in lung sections from 2 guinea pigs (case Nos. 22 and 26), and mineralization of the myocardium was evident in sections from 1 animal (case No. 26, not shown). All animals had mild to moderate vacuolar changes in the liver (not shown).
Serum 25(OH)D had decreased in all animals to 55.50 ± 6.10 nmol/l (Fig. 10). Levels were significantly lower in recovered animals compared with either exposed (
Consistent with the lower serum vitamin D levels, serum P in recovered animals was comparable to levels observed in control animals (5.44 ± 0.40 mg/dl vs 4.87 ± 0.22 mg/dl; Fig. 15). Serum total Ca, like iCa concentrations, was slightly higher in recovered animals than in controls (10.73 ± 0.17 mg/dl vs 9.98 ± 0.23 mg/dl;
Discussion
We describe the clinical, serum biochemical, gross, and histopathological findings associated with diet-induced hypervitaminosis D and metastatic calcification in an inbred guinea pig colony. Vitamin D toxicity has been rarely reported in guinea pigs, but metastatic calcification subsequent to improper mineral ratios has been extensively studied in this species since the 1940s.
6,7,14,15
Soft tissue calcification has also been observed in guinea pigs fed the toxic plant,
In the current case report, animals were fed a commercial diet containing high levels of vitamin D for approximately 6 weeks before it was recalled. Vitamin D levels were more than 350 times the normal formulation in at least 1 of the 3 bags fed to guinea pigs during that time frame. Approximately 30% of the animals presented with nonspecific signs. Gross and microscopic lesions were present in both clinically normal animals and those exhibiting clinical signs of disease, although lesions tended to be more severe in clinically ill animals. Microscopic analyses revealed more extensive tissue mineralization than was observed grossly. Animals regained appetite and body condition and appeared clinically normal within several weeks after the recalled diet was removed and they were returned to normal feed. However, microscopic evaluation of kidneys from 3 animals in the recovered colony demonstrated chronic lesions that could potentially affect the long-term health of the animals.
Hypervitaminosis D with soft tissue calcification was confirmed following extensive diagnostic testing. Soft tissue mineralization was suspected based on gross lesions observed in multiple organs in most exposed animals, including one that was clinically normal. Microscopic examination confirmed multifocal mineralization, including in organs that appeared grossly normal, in both exposed and recovered animals. In addition, osteosclerosis was seen in the tibia of 1 exposed guinea pig evaluated. The bone changes were consistent with those associated with hypervitaminosis D.
In most species, hypercalcemia is considered the hallmark of vitamin D toxicity, 31 but we and others have found that serum calcium levels are a poor indicator of vitamin D status in the guinea pig. 11 In our study, there was no significant difference in total serum Ca or iCa concentrations between exposed and control animals. In fact, the trend indicated slightly lower iCa levels in exposed animals. This could be due to the high serum P, which pulls Ca from the ionized pool, leading to lower iCa concentrations in connection with hyperphosphatemia. Our findings contrast slightly to those reported by Jenson et al 11 for a colony of guinea pigs carrying a genetic mutation for α-mannosidase that were also affected by the diet recall. Similar to data obtained with the strain 13 guinea pigs, serum total Ca concentrations were normal in guinea pigs with α-mannosidosis. However, iCa levels were determined to be “slightly higher than expected.” 11
The propensity for guinea pigs to develop metastatic calcification in the absence of overt hypercalcemia may be related to the physiologic mechanisms governing Ca homeostasis in this species. O’Dell et al 20 performed studies comparing intake vs excretion of various minerals, including Ca, Mg, K, sodium (Na), and P, in guinea pigs fed various compositions of diet. They concluded that the absorption of Ca is much higher in guinea pigs than in most other species and that the kidneys excrete a relatively high proportion of the excess in the urine. In the face of the excessive dietary vitamin D in this study, it was not surprising that animals exhibited crystalluria and multifocal mineralization in the kidneys. The ability of guinea pigs to excrete high levels of Ca in the urine under normal circumstances may explain why BUN and creatinine were not significantly increased in most of the exposed animals despite tissue mineralization and crystalluria. Azotemia was apparent only in the 3 male guinea pigs exhibiting the most severe clinical disease while on the tainted diet (case Nos. 2, 6, and 7). All 3 animals had clear urine with an acidic pH ranging from 5 to 6 rather than the normal cloudy urine with a pH of 8 to 9. BUN and creatinine were modestly to markedly increased in these animals. Acidic urine was previously reported in guinea pigs fed a high-phosphorus, acidic diet and was also accompanied by clearing of the normally cloudy urine. 20
While high serum 25(OH)D is the most conclusive method for diagnosing hypervitaminosis D, it is not routinely performed in laboratory animals and requires relatively large volumes of serum or plasma. Other biochemical markers that may be useful to diagnose hypervitaminosis D in guinea pigs include serum P, ALP, and PTH. Consistent with hypervitaminosis D in other species, serum levels of 25(OH)D and P were higher in exposed animals than in controls, and serum PTH levels were lower. High serum ALP in exposed animals was most likely multifactorial, due to altered bone metabolism secondary to vitamin D toxicity, and also affected by anorexia and subsequent hepatic lipidosis, a conclusion supported by histopathology in the exposed guinea pigs. The highest concentrations of ALP were observed in animals <6 months of age. Young animals tend to have higher serum concentrations of ALP due to bone growth, 13 and the extremely high concentrations seen in serum collected from case Nos. 1, 3, 5, and 8 most likely resulted from a combination of altered bone growth and liver disease.
Ca*P is frequently measured in humans and other animals, with a high product considered a contributing factor in soft tissue mineralization. 5,28 Prolonged hyperphosphatemia potentiates soft tissue calcification when Ca*P is high. 25,28 A Ca*P of >70 has been used as a predictor of metastatic calcification in horses with systemic calcinosis and in dogs with renal failure. 3,27 Although measurement of the Ca*P product is not routinely used to predict metastatic calcification in guinea pigs, we found that all but 1 exposed animal (case No. 10) had a Ca*P >70. Ca*P was below 70 in all control animals and in all but one of the recovered animals. Additional studies are needed to determine if Ca*P is a reliable prognosticator of tissue calcification in guinea pigs.
Previous reports in humans and other animals demonstrate recovery of biochemical parameters, including serum Ca, P, and PTH levels, following treatment or cessation of high levels of vitamin D administration. By contrast, we found that serum PTH concentrations remained low while iCa and iMg were significantly higher in recovered animals than in exposed animals actively consuming high dietary vitamin D or in control animals. Based on the knowledge that complete recovery and normalization of biochemical parameters can take more than 6 months due to the storage of vitamin D in fat, one possibility is that PTH levels remained low due to high levels of vitamin D stored in fat with prolonged release. The biological significance of the changes in either iMg or iCa observed is unknown given that the normal ranges of these elements have not been determined for the guinea pig. Moreover, differences in the control colony from USAMRIID vs exposed or recovered animals from MGH could be due to dietary and/or environmental differences between the colonies. Thus, the relevance of the differences observed in iCa and iMg to the diagnosis or prognosis of hypervitaminosis D in guinea pigs is unclear but is an area that warrants further study. We predict that the decrease in serum P following return to a normal diet allows for the increases in serum iCa (and possibly iMg) because the P is no longer pulling minerals from the ionized pools.
In conclusion, we report a case study of hypervitaminosis D and metastatic calcification in a colony of inbred strain 13 guinea pigs. Our data confirm the recently reported finding that serum Ca levels do not appear to be an accurate indicator of vitamin D status in guinea pigs. 11 Hyperphosphatemia was consistently observed in exposed animals and is likely a better diagnostic indicator of hypervitaminosis D in this species. Serum PTH and possibly iMg appear to be decreased in guinea pigs with hypervitaminosis D, and Ca*P product is increased (>70) and thus may also be valuable markers for hypervitaminosis D. Data obtained in this case study indicate that diagnostic tests other than serum Ca are necessary for a diagnosis of hypervitaminosis D in guinea pigs.
Footnotes
Acknowledgement
We thank Dr J. G. Fox and Dr R. P. Marini for review of the manuscript, E. Robbins and A. Terestre for assistance with manuscript preparation, and the MGH-CCM Clinical Pathology Laboratory and MIT-DCM Histopathology Laboratory for assistance with diagnostic testing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This report was supported in part by R21 OD011193 (H.H.).
