Abstract
We demonstrated that pigs are susceptible to acute infection by equine herpesvirus type 9 (EHV-9). Six 8-week-old SPF pigs were inoculated intranasally and four were inoculated orally with different doses of EHV-9, and observed for 6 days. Although neurological signs did not develop in any of the infected pigs, the six intranasally infected pigs and one of the orally infected pigs developed lesions of encephalitis consisting of neuronal necrosis, neuronophagia, and intranuclear inclusion bodies, distributed mainly in the rhinencephalon. EHV-9 antigen was localized in the necrotic neuronal cells and was closely associated with the presence of inclusion bodies. These findings clearly demonstrate that pigs are fully susceptible to EHV-9 infection following intranasal inoculation (but less so following oral inoculation), and that EHV-9 in pigs has a highly neurotropic nature.
Herpesviruses have been isolated from many different species. Infection in natural hosts is usually mild and can lead to persistent infection. Some herpesviruses may cross species barriers nd induce severe to fatal disease in other hosts. 9 Equine herpesvirus 9 (EHV-9), isolated from Thomson's gazelles in 1993, 2 is the causative agent of fatal meningoencephalitis in the gazelle. 10 Previously, it has been demonstrated that EHV-9 is virulent in suckling mice of the ICR strain by intracerebral inoculation and in 4-week-old mice by intranasal inoculation, causing both neurological symptoms and death. 2 Presently, the range of natural hosts of EHV-9 is unknown. The infectivity and pathogenicity of EHV-9 was also not clear in domestic animals, including pigs. In this study, 8-week-old pigs were inoculated with virus by different routes and doses to determine the susceptibility of pigs to EHV-9.
Twelve 8-week-old, specific-pathogen-free pigs with no serum antibodies against EHV-9, Aujeszky's disease virus (ADV), and porcine reproductive and respiratory syndrome virus (PRRSV) were used. The pigs were divided into three groups—orally infected pigs, nasally infected pigs, and uninfected controls—for the experiments, and the groups were housed separately in different blocks to prevent cross infection. EHV-9 isolated from the Thomson's gazelle 2 10 was used. The virus was passaged six times in Madin and Darby bovine kidney (MDBK) cells, and then twice in a pig kidney (PK) cell culture. The infected PK cell culture fluid was used for the inoculation of the pigs. Before inoculation with EHV-9, the pigs were anesthetized with ketamine hydrochloride (30 mg/kg intramuscularly) and xylazine (2.0 mg intramuscularly). Pig Nos. 1 and 2 were inoculated intranasally with a nebulizer with 3 ml of 107.0 tissue culture infectious dose (TCID)50/ml, pig Nos. 3 and 4 with 3 ml of 105.0 TCID50/ml, and pig Nos. 5 and 6 with 3 ml of 103.0 TCID50/ml of EHV-9, respectively. Pigs Nos. 7 and 8 were inoculated orally with a syringe with 3 ml of 107.0 TCID50/ml, and pig Nos. 9 and 10 with 3 ml of 105.0 TCID50/ml of EHV-9. Pig No. 11 was inoculated intranasally and pig no. 12 was inoculated orally with 3 ml of noninfected culture medium as controls. All pigs were observed clinically for 6 days and then euthanatized by an intravenous injection of pentobarbital sodium. For virus isolation, the nasal secretion of each pig, including the two controls, was swabbed before inoculation and at 2, 4, and 6 days after inoculation of the virus. At necropsy, the olfactory bulb, frontal pole of the brain, trigeminal ganglion, and palatine tonsils were collected. Each secretion and minced specimen was cultivated in rabbit kidney cell culture at 37 C for 7 days and was observed for cytopathic effect. Following gross examination at necropsy, tissue specimens from each pig, including the brain, trigeminal ganglia, palatine tonsil, and nasal mucosa, were fixed in a 10% neutral buffered formalin, and representative blocks of the central nervous system (cerebral cortex, frontal pole, motor area, occipital pole, corpus striatum, thalamus, colliculus caudalis, cerebral peduncles, pons, cerebellum, medulla oblongata, spinal cord, and trigeminal ganglia) and other tissues were embedded in paraffin, sectioned, and stained with hematoxylin and eosin (HE) for histology. Immunohistochemically, viral antigen was demonstrated by the avidin–biotin–complex (ABC) immunoperoxidase method with the Vectastain ABC kit. Anti–EHV-9, and anti-ADV, anti-PRRSV, and anti-hemoagglutinating encephalomyelitis virus (HEV) rabbit sera were all used as primary antibodies at dilutions of 1:1,024, 1:2,048, 1:8,192 and 1:2,048, respectively. Sections were counterstained with methyl green. Tissue sections from pig Nos. 11 and 12 and the serum from a nonimmunized rabbit were used as negative controls.
Clinically, all pigs (Nos. 1–6) inoculated intranasally and one (No. 8) inoculated orally showed a slight febrile reaction (over 39.5 C) over 2 to 3 days; the highest temperature of pig No. 3 was 40.0 C. No other clinical abnormality was observed in the infected pigs. The two control pigs (Nos. 11 and 12) remained healthy during the experimental period. Virologically, EHV-9 was recovered from the nasal secretions between 2 and 6 days postinoculation in pigs infected intranasally, but not orally (Table 1), with titers of 101.0 to 103.0 TCID50/ml. The virus was also isolated from the olfactory bulb of the six intranasally infected and one (pig No. 8) of the orally infected pigs, from the frontal pole of four (pig Nos. 2, 3, 4, and 6) of the intranasally, and one (pig No. 8) of the orally infected pigs, and from the trigeminal ganglion of one (pig No. 4) of the intranasally infected pigs. The highest titer at the olfactory bulb was 102.75 TCID50/0.1 ml in pig Nos. 5 and 6, at the frontal pole of the brain was 103.25 TCID50/0.1 ml in pig No. 4, and at the trigeminal ganglion was 100.75 TCID50/0.1 ml in pig No. 4. No virus was isolated from the two uninfected control pigs.
Isolation of EHV-9 from nasal secretions and various tissues of intranasally and orally infected pigs.∗
Values are expressed as log 10.
No macroscopic lesions were observed in any of the infected pigs. Microscopic lesions were confined to the central nervous system (Table 2) and consisted of a nonsuppurative encephalitis characterized by neuronal necrosis, neuronophagia, focal gliosis, perivascular infiltration of mononuclear cells, and intranuclear inclusion bodies. The distribution of the encephalitic changes was similar in the six intranasally infected and one (pig No. 8) orally infected pig. In the cerebrum, the most prominent lesion was found in the rhinencephalon and in the cerebral cortex. Neuronal necrosis was widespread in the mitral and tufted cell layers of the olfactory bulbs (Fig. 1) and was accompanied by infiltration of macrophages. The affected neurons were characterized by neuronal pyknosis and basophilia, diffuse to central chromatolysis, karyorrhexis, and multifocal cellular debris. Basophilic intranuclear inclusion bodies were present in many neurons and glial cells in this zone. Focal gliosis was found in the olfactory gyri. Severe neuronal necrosis and many intranuclear inclusion bodies were detected in the amygdaloid body, in the putamen, and in the caudate nuclei (Fig. 2). Encephalitic lesions were prominent in the pyramidal and large granular cell layers of the lobus frontalis and temporalis. Slight nonsuppurative meningitis, made up of macrophages and lymphocytes, was found in the lobus frontalis. No lesions were evident in the medulla oblongata, spinal cord, cerebellum, and trigeminal ganglia, nasal mucosa, palatine tonsils, and other tissues. No macroscopic or microscopic lesions were observed in the two noninfected controls.
Distribution of the histologic lesion and viral antigen in tissues of pigs infected intranasally or orally with EHV-9.∗
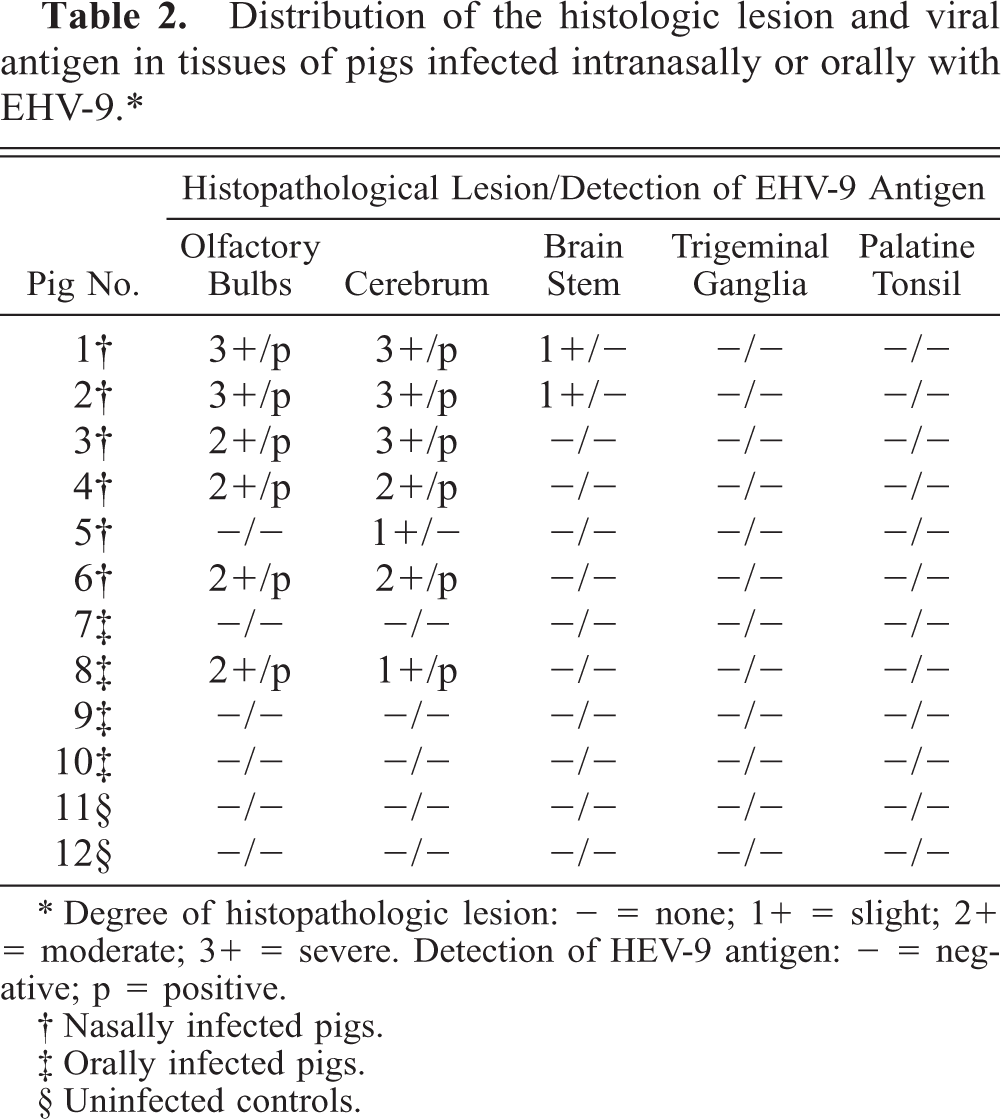
Degree of histopathologic lesion: − = none; 1+ = slight; 1+ = moderate; 3+ = severe. Detection of HEV-9 antigen: - = negative; p = positive.
Nasally infected pigs.
Orally infected pigs.
Uninfected controls.
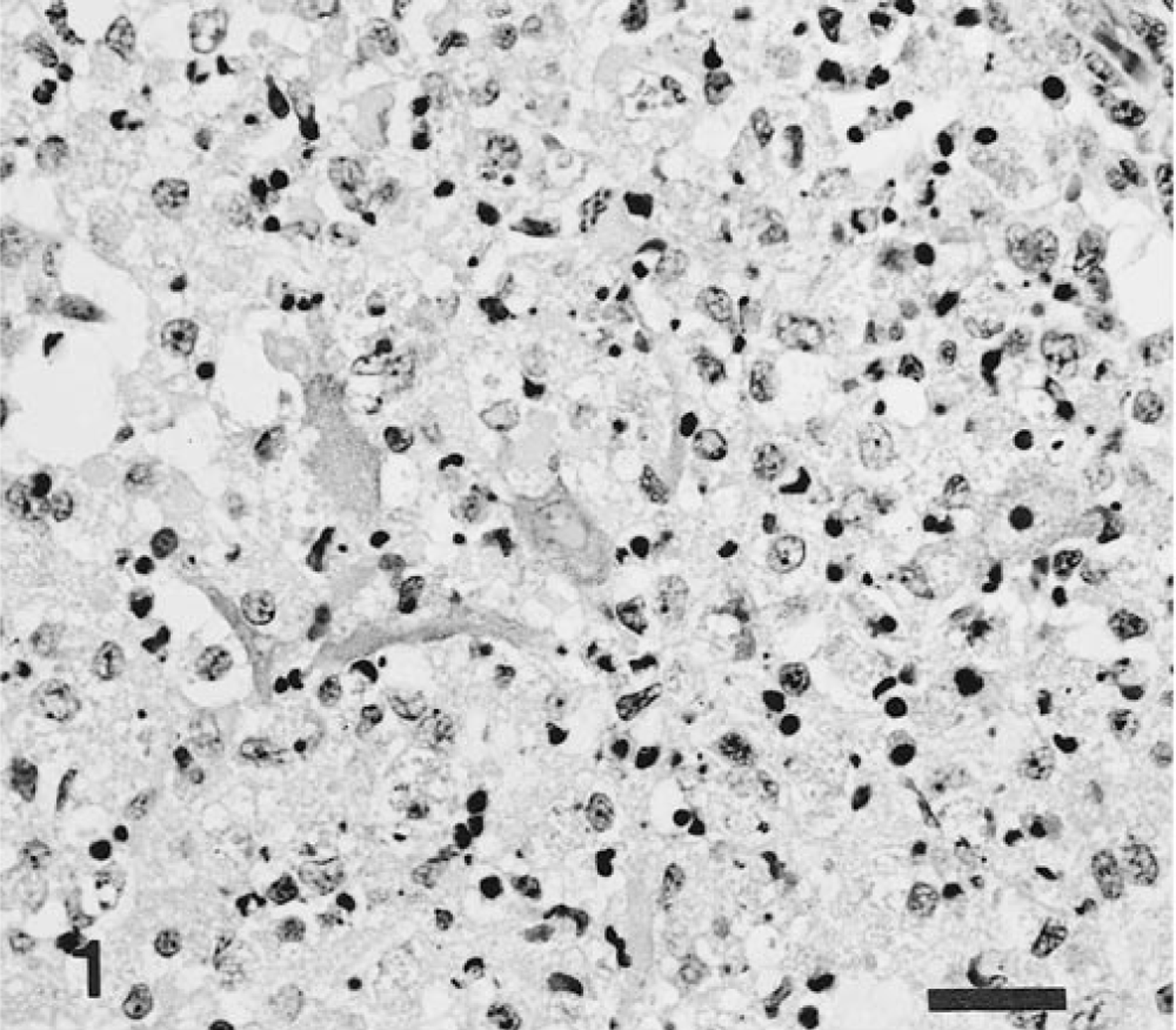
Brain, olfactory bulb; intranasal infection, pig No. 4. Neuronal necrosis is extensive in the inner granular and mitral layer or zone. HE. Bar = 25 μm.
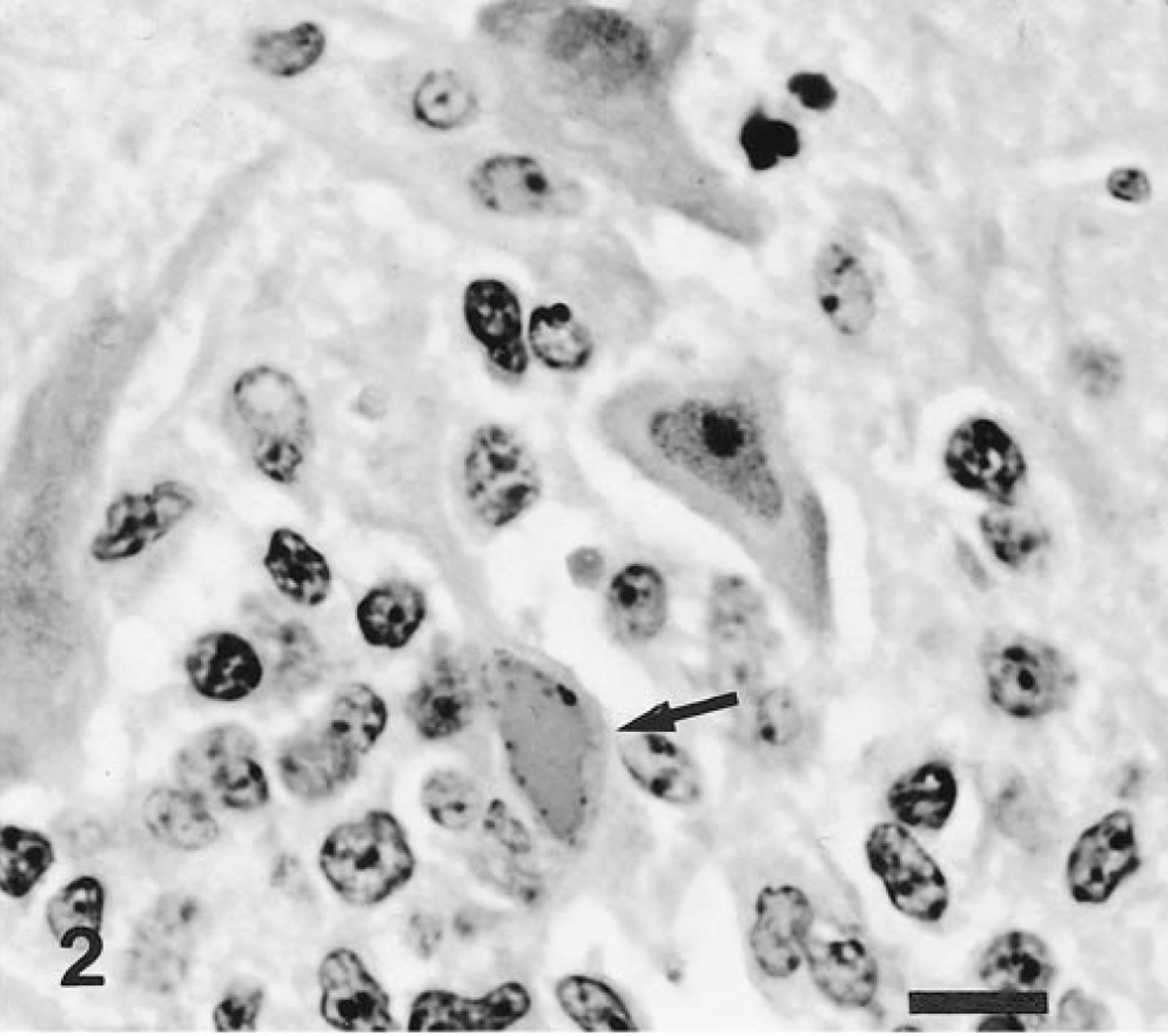
Brain, amygdaloid body; intranasal infection, pig No. 3. A basophilic intranuclear inclusion body (arrow) is present in a dead neuron. HE. Bar = 10 μm.
Immunohistochemically, EHV-9 antigen was detected in the nucleus and cytoplasm of the necrotic neurons. Antigen localization was closely associated with the presence of intranuclear inclusion bodies and neuronal cell necrosis (Table 2). In pig Nos. 6 and 8, a small amount of the EHV-9 antigen was also found in neuroglial cells in the mitral and tufted layer of the olfactory lobus (Fig. 3), as well as in the cortex of the lobus frontalis and temporalis. In four nasally infected pigs (Nos. 1–4), abundant EHV-9 antigen was widely distributed in the rhinencephalon (olfactory lobe, amygdaloid body, putamen, and caudate nuclei), and in the lobus frontalis, lobus temporalis, lobus parietalis, and lobus occipitalis (Fig. 4). No EHV-9 antigen was detected in the medulla oblongata, cerebellum, trigeminal ganglia, palatine tonsils, or other tissues in any of the infected pigs. No ADV, PRRSV or HEV antigens were found in any of the tissues of all the pigs.
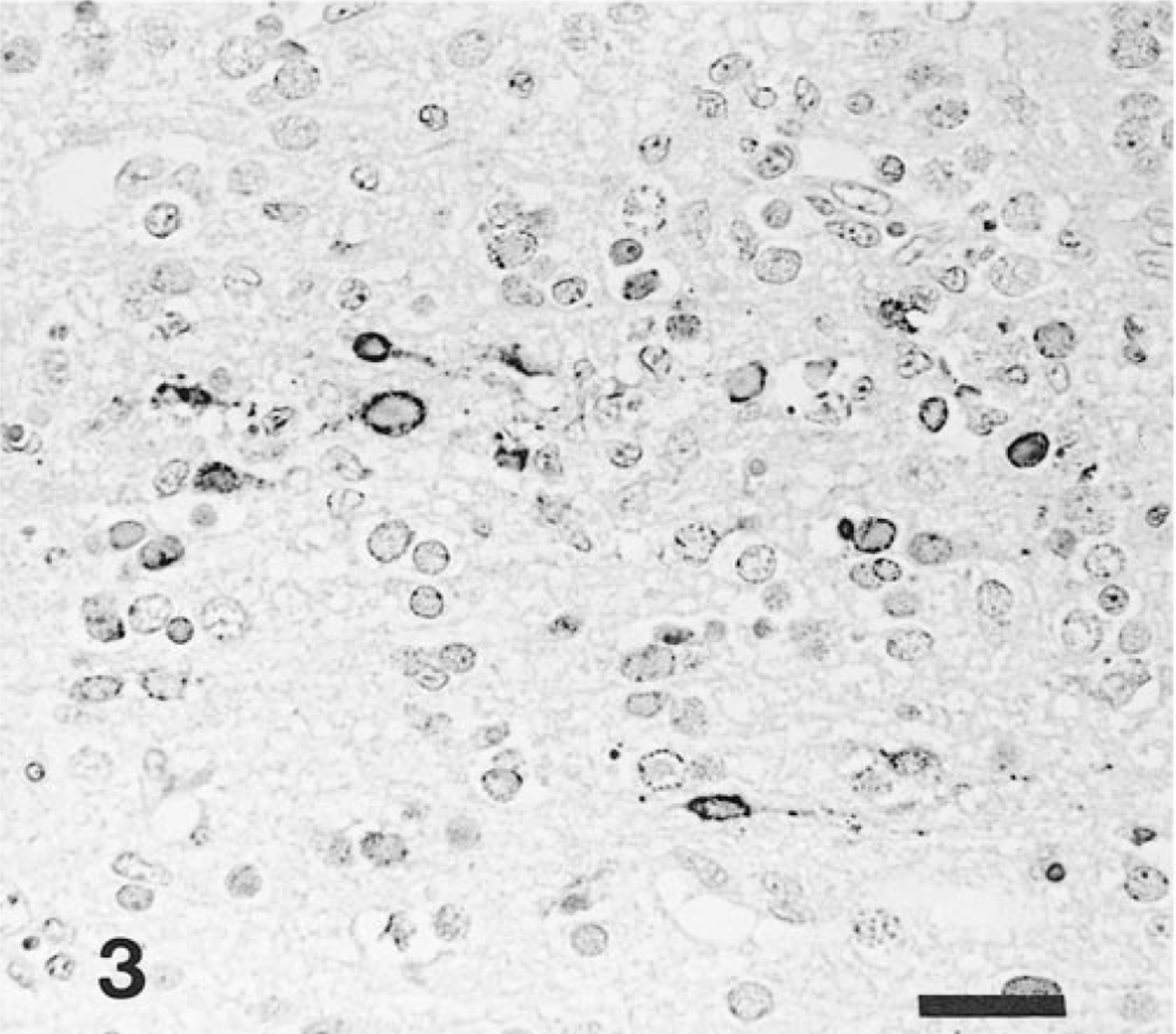
Brain, olfactory bulb; intranasal infection, pig No. 4. Abundant EHV-9 antigen is detected in the inner granular neurons. Anti–EHV-9 ABC. Bar = 25 μm.
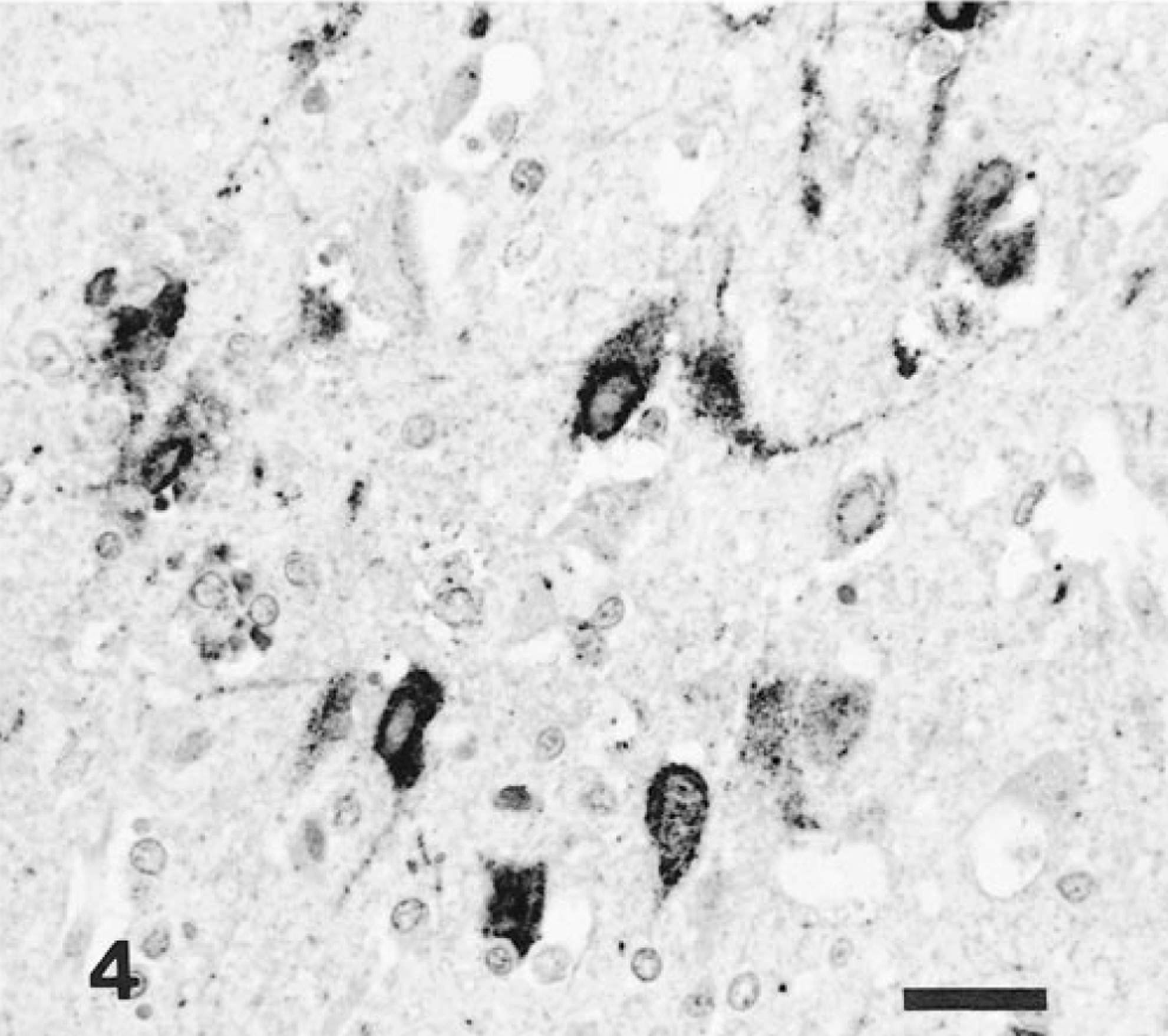
Brain, amygdaloid body; intranasal infection, pig No. 3. EHV-9 antigen is present in necrotic neurons. Anti–EHV-9 ABC. Bar = 25 μm.
Although neurological signs did not develop in any of the infected pigs, histologically, encephalitis consisting of neuronal necrosis, neuronophagia, intranuclear inclusion bodies, and perivascular cuffing was observed in all six intranasally infected pigs and in one orally infected pig; lesions were localized mainly in the rhinencephalon and in the cerebral cortex and were not observed in the cerebellum or spinal cord. These findings closely resemble those of a previous report 10 of gazelles that were naturally infected with EHV-9, and pigs and cattle infected experimentally and naturally with the other herpesviruses. 3–5
Immunohistolochemically, EHV-9 antigen was demonstrated intralesionally in the neuroglial cells and in necrotic neurons. The close association of antigen with neuronal necrosis and intranuclear inclusion bodies suggests that EHV-9 replication led to the neuronal cell death. Moreover, presence of EHV-9 antigen and the severity of the encephalitis corresponded closely with the intranasal route of infection and the challenge dose.
Herpesvirus 1 in different species replicates well in the naso-oral mucosa and produces necrotizing rhinitis 8 and tonsillitis. 6 In the present study, EHV-9 was recovered from nasal secretions of the six nasally infected pigs, although no histologic lesions were detected in the nasal mucosa. Tonsillitis was not present in the palatine tonsils of the four orally infected pigs. These results may indicate that EHV-9 replicates only at a low level in the nasal mucosa and even less in the oral mucosa of pigs.
The pathway of the virus to the central nervous system from the naso-oral mucosa has been investigated with many viruses, such as herpesviruses 1 3 5 and HEV virus. 7 These viruses travel centripetally in the nerve fibers from the nasal mucosa to the trigeminal ganglion, olfactory lobes, or both, and also from the oral mucosa to the trigeminal ganglion. 1 3–5 In the present study, the six nasally infected pigs and one orally infected pig developed the typical induced encephalitis but did not develop epithelial necrosis and peripheral neuritis in the nasal cavity; nor did they develop tonsillitis or trigeminal ganglionitis. Pigs are therefore susceptible to the EHV-9 infection in the central nervous system following intranasal inoculation. Further study is needed to clarify the neuropathogenesis in pigs infected with EHV-9.
Footnotes
Acknowledgements
We thank Mr. M. Kobayashi and Miss M. Shimada for preparing the histological sections and Dr. Y. Ando and Mr. T. Fujisawa for preparing the photographs.
