Abstract
The distribution pattern and associated tissue reactions with progressive changes in Besnoitia caprae cysts were investigated in 6 experimentally infected 16- to 20-month-old male goats. Each goat was subcutaneously inoculated with approximately 13 × 108 B caprae bradyzoites. The animals were examined daily for development of clinical besnoitiosis, and skin biopsies from distal parts of the limbs were taken at weekly intervals. At 15, 30, 60, 120, 180, and 365 days postinfection (DPI), 1 goat was euthanized. Samples were collected at autopsy from various organs for histologic and ultrastructural studies. No cysts were seen in tissue sections on 15, 30, and 365 DPI, but large numbers were present at 60, 120, and 180 DPI in the skin of the distal limbs, scrotum, and ears, with fewer in the tongue, palate, sclera, testicles, and spermatic cord. No cysts were seen in the lungs, liver, kidneys, spleen, central nervous system, or lymph nodes. Cyst numbers peaked at 60 DPI, then declined from 120 to 180 DPI. Degenerated cysts were relatively rare at 60 DPI but more numerous at 180 compared with 120. A granulomatous reaction—predominantly characterized by macrophages, lymphocytes, and plasma cells—surrounded each degenerated cyst. All goats showed testicular tubular degeneration with little or no spermatogenic activity. The sizes of cysts and their wall thickness, with the size of bradyzoites and some of their organelles, exhibited progressive chronologic changes.
Parasites of the genus Besnoitia are classified in the subfamily Toxoplasmatinae of the phylum Apicomplexa. 11 Natural besnoitiosis causes chronic disease in a range of livestock and wild animals.3,6,7,9,17
Except for B darlingi, B oryctofelisi, and B wallacei, whose definitive host is the domestic cat, the life cycles of other Besnoitia species are not fully understood.8,10,18 Some species can be mechanically transmitted from one intermediate host to other susceptible intermediate hosts through tachyzoites or bradyzoites. 17 Several species in the genus Besnoitia have been identified, but morphologic differences among them are poorly defined.9,10 B caprae has been recognized as a host-specific parasite of goats, but its definitive host is still unknown. The natural route of transmission of B caprae to the goat is not clearly understood, but tick or fly vectors may play a major role in mechanical transmission of the organism from infected to healthy goats. 18 No B caprae cysts have been reported in cattle, sheep, and donkeys grazing alongside infected goats.3,21,27
Although caprine besnoitiosis has been reported only in wild and domestic goats in Iran6,27 and in domestic goats in Kenya,3,4 it is possible that the infection is more widespread. Lack of reports of its presence may be simply due to the unfamiliarity of local veterinarians with the disease and its diagnosis. The presence of white sandlike cysts in the scleral conjunctiva, associated with typical subcutaneous cysts in distal parts of the limbs, is pathognomonic and crucial in diagnosis of caprine besnoitiosis.6,27 Thickening, alopecia, and hyperkeratosis of the skin in infected goats occur because of accumulation of Besnoitia cysts in the dermis and subcutaneous tissues, which cause chronic local inflammatory cell infiltration and tissue necrosis.26,27 Localization of Besnoitia cysts in the parenchyma of the testis and epididymis and their adverse effect on the spermatogenesis and fertility of male goats have been reported.13,23,25 The parasite causes heavy economic losses by reducing reproductive performance and damaging the skin with subsequent degradation of leather quality.22,24,25
To the best of our knowledge, there has been no experimental study of the distribution and chronology of development and degeneration of Besnoitia cysts in goats or other host species. Tissue reactions of the intermediate hosts to the developmental stages of Besnoitia cysts are also ill-defined. The present experiment was therefore designed to study the distribution of and changes in Besnoitia cysts, with the associated tissue reactions, in experimentally infected goats.
Materials and Methods
Isolation of Besnoitia caprae
Goats with chronic besnoitiosis were identified by observing Besnoitia cysts in their conjunctiva and by examining histologic sections of skin biopsies from the tarsal and carpal regions.3,6 Naturally infected goats exhibiting clinical features of caprine besnoitiosis were culled and euthanized according to National Veterinary Organization ethics rules, and the subcutaneous tissues were collected and stored at 4°C. To minimize surface contamination, tissue samples were individually washed in Dulbecco’s Modified Eagle Medium (DMEM), supplemented with 2% fetal calf serum, 1% antibiotic solution (10,000 IU penicillin and 10,000 mg streptomycin per milliliter) and 1% amphotericin B (250 mg/ml). The tissue samples were subsequently cut into 1- to 2-mm 3 pieces, which were placed in new DMEM solution for 10 minutes. The DMEM with liberated bradyzoites was collected and centrifuged at 770 × g for 15 minutes at 4°C. The pellets were resuspended in DMEM and the bradyzoites counted with a Neubauer chamber. They were then cryopreserved by resuspending in fetal calf serum containing 10% DMSO and by freezing and storing in liquid nitrogen.
Infection of the Goats
Six male goats, approximately 16 to 20 months old and from a breeding herd in a noninfected area, were determined to be free of Besnoitia infection based on the absence of grossly apparent cysts in the scleral conjunctiva and the absence of cysts in biopsies from the carpal and tarsal skin. No Besnoitia infection had been reported in goats from anywhere within 120 km of the area from which the goats were purchased. Each goat was subcutaneously inoculated in the dorsolateral thoracic area immediately caudal to the caudal angle of the scapula, with approximately 13 × 108 bradyzoites of B caprae. The infected goats were housed in separate boxes, each in a different room, and examined daily for development of clinical besnoitiosis.
Skin Biopsies
Based on an aseptic incisional technique, a rectangular biopsy (approximately 3 × 5 mm) from the skin of distal parts of the limbs was collected under local anesthesia from each goat at weekly intervals until 60 days postinfection (DPI) and then monthly from the surviving animals until the end of the experiment, at 365 DPI. Sections from 5 areas of each biopsy from the carpal skin of each animal were histologically examined for the presence of Besnoitia cysts.
Necropsy and Histopathologic Examination
For collection of material for histologic examination at autopsy, 1 goat was euthanized at each of 15, 30, 60, 120, 180, and 365 DPI, and tissue samples were collected from brain, spinal cord, sclera, lungs, heart, blood vessels, liver, kidneys, testicles, epididymis, tunica albuginea, tunica vaginalis, penile urethra, esophagus, rumen, omasum, abomasum, small and large intestine, and subcutaneous peritendinous soft tissue. Skin samples were taken from the tips of the pinnae, over the frontal sinuses, and from the carpus, tarsus, and scrotum. All samples were fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned at 5 μm, and stained with hematoxylin and eosin and Alcian blue / periodic acid–Schiff for light microscopic examination. The average number of cysts per microscopic field (each 3.14 mm 2 ) was scored as follows, based on a 10× objective lens and 10× eyepiece: up to 5, 10, 15, 20, 25 (+, ++,…, +++++, respectively). Biopsies (and autopsy material, when appropriate) were collected monthly from the same anatomic sites from 10 age-matched, clinically normal uninfected goats in the breeding herd. This herd, from which the experimental goats had been bought, was regularly examined clinically during the entire period of the experiment and showed no evidence of Besnoitia infection.
Electron Microscopy
Samples of dermis and subcutaneous tissue (1 × 1 × 1 mm) from the infected areas were prepared for electron microscopy as described previously. 28 Briefly, the tissue was fixed in 4% ice-cold glutaraldehyde for 2 hours, rinsed three times in buffered sodium cacodylate, postfixed in osmium tetroxide, and again rinsed in buffered sodium cacodylate. Dehydration in graded concentrations of acetone preceded embedding in epoxy resin. Sections of 60- to 90-nm thickness were cut with a diamond knife, mounted on copper grids, stained with uranyl acetate, counterstained with lead citrate, and examined with a transmission electron microscope (Philips CM10, Philips, Eindhoven, Netherlands).
Results
Clinical Signs and Gross Lesions
Infected goats were initially characterized by hyperthermia, depression, mild subcutaneous edema, and weight loss. Hyperthermia and edema disappeared by 30 and 60 DPI, respectively, whereas weight loss continued up to 210 DPI in the one surviving goat. Weight losses of 12 to 23% were seen in individual animals despite maintenance of normal appetites in all goats. Disappearance of edema was accompanied by progressive thickening, folding, or wrinkling of the skin with alopecia and hyperkeratosis.
Gross skin lesions showed multifocal to coalescing patchy alopecia and hypotrichosis, with thickened skin on the neck and around the muzzle, eyes, base of the ears, scrotum, carpus, elbow, stifle, and tarsus. Besnoitia cysts developed in the conjunctiva as early as 32 to 40 DPI. These were initially transparent minute blebs, but as they matured, they became translucent, resembling white sea-sand particles. The skin and eye lesions gradually diminished from 120 DPI and disappeared by 300 DPI.
At 60, 120, and 180 DPI, the subcutaneous tissues of the affected skin—with tendon sheaths and tendons, periosteal surfaces of limb bones, subcutaneous muscles, and scleral conjunctiva—exhibited variable numbers of spherical white granules clearly visible under a stereomicroscope at 25×. These were most numerous in skin covering the carpal and tarsal bones, the frontal bone, and the scrotum. Orchitis with enlargement of the testicles was another feature that started from 50 to 60 DPI. The testicles remained enlarged to 150 to 180 DPI, then gradually shrank to become hard and small from 270 DPI until the end of the experiment, at 365 DPI, in the surviving goat.
No clinical signs of caprine besnoitiosis were seen at any time during the experimental period, in animals in the primary herd from which the experimental goats had been purchased.
Histopathologic Findings
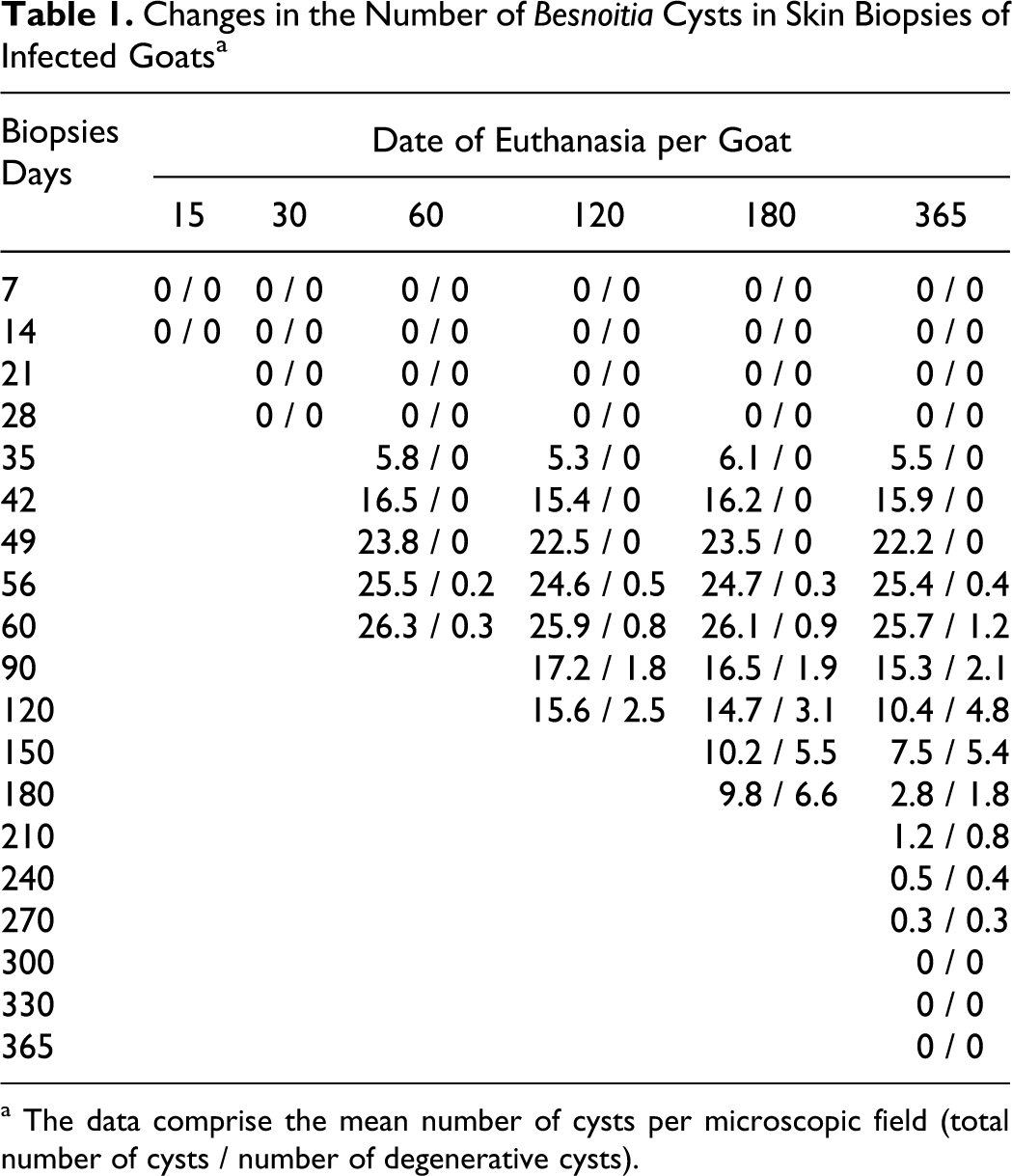
No cysts were found in biopsies from the goats in the noninfected breeding herd during the experimental period. By contrast, in the infected animals, Besnoitia cysts were seen for the first time in skin biopsies at 35 DPI (Table 1). Sections from all infected animals at 35, 42, 49, 56, 60, 90, 120, 150, and 180 DPI showed large numbers of cysts in the dermis and subcutis in skin biopsies from distal parts of the legs (Fig. 1).
Changes in the Number of Besnoitia Cysts in Skin Biopsies of Infected Goats a
a The data comprise the mean number of cysts per microscopic field (total number of cysts / number of degenerative cysts).
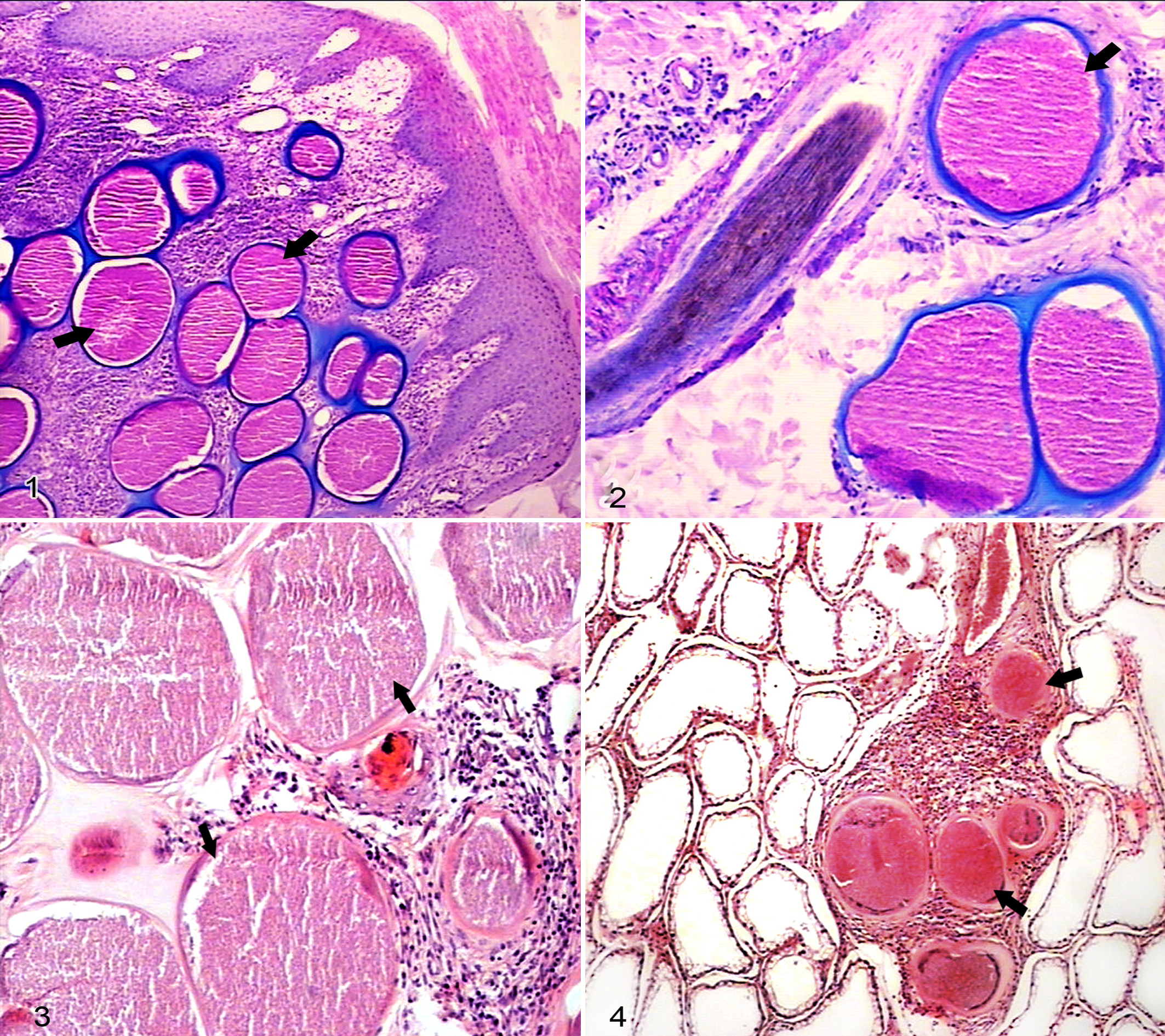
In the necropsy samples, cysts were numerous in skin of the distal limbs, scrotum, and pinna and over the frontal bone. Fewer cysts were present in the skin of neck, subcutaneous muscles, muscle fibers of tongue, submucosa of soft and hard palate, esophagus, testicles and epididymis, tunica albuginea, tunica vaginalis, spermatic cord, and glans penis of the animals euthanized at those times (Table 2). No cysts were found in the lungs, liver, kidneys, spleen, brain, spinal cord, digestive tract, and associated lymph nodes.
Chronology of Changes in the Number of Besnoitia capra e Cysts in Organs of Goats
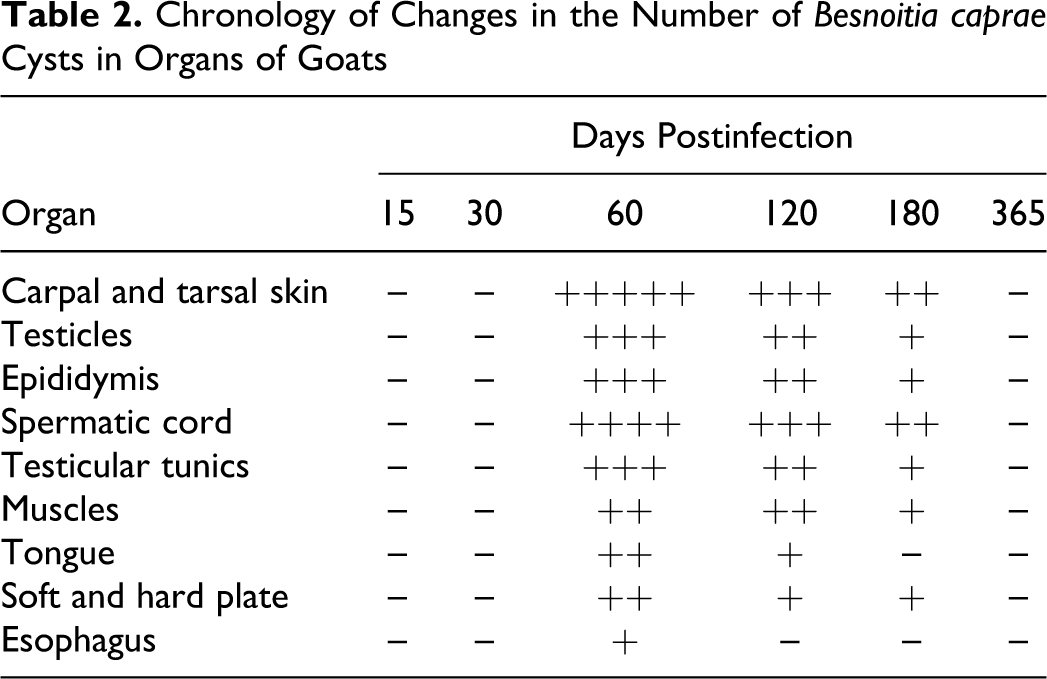
Fresh Besnoitia cysts appeared in the skin biopsies as early as 35 DPI, but there were no degenerating cysts before 56 DPI (Table 2). Cyst numbers were maximal at 60 DPI and declined thereafter. Only 3 degenerating cysts were found in the many skin biopsies examined on 270 DPI, and none were seen at 300 DPI or later. As shown in Table 2, the average number of cysts from five sections of carpal skin revealed a maximum infestation rate of 26.3 Besnoitia cysts per microscopic field (3.14 mm 2 ) at 60 DPI, with an average cyst diameter of 308.3 μm at 60 DPI (Table 3, Fig. 2). As cyst numbers declined, the proportion of degenerating cysts increased, as did their maximum diameter (average, 491.0 μm at 240 DPI) (Table 3). Cyst degeneration was accompanied by increasing granulomatous inflammation (Fig. 3).
Chronology of Changes in the Diameters of Cysts and Thickness of Cyst Walls a
a In micrometers. The dimensions shown are means ± SD of 100 cysts measured on days 60, 120, and 180 days postinfection and of 60 and 40 cysts on 210 and 240 days postinfection, respectively.
Skin cysts were found chiefly in the superficial dermis, with a few in the deep dermis and underlying muscles. Some cysts in the walls of skin blood vessels were associated with polypoid changes in the intima. The overlying epidermis showed varying degrees of hyperkeratosis and acanthosis. Each single cyst was usually encapsulated separately, but in rare instances, 2 to 8 cysts were located in the same capsule, as noted previously.
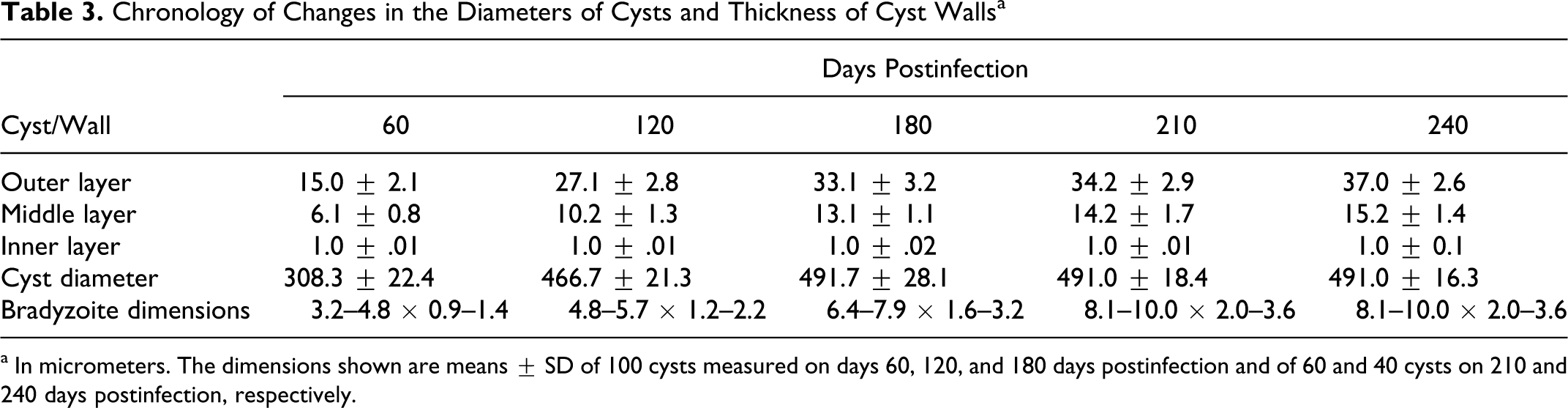
The testicles showed mild tubular degeneration, with infiltration of lymphocytes, macrophages, and eosinophils but no cysts, on 15 and 30 DPI. Most seminiferous tubules were hollow and lacked primordial cells and spermatogonia in the epithelial layer, which indicated little or no spermatogenic activity in these goats, compared with that in noninfected age-matched animals from the breeding herd. On 60, 120, and 180 DPI, Besnoitia cysts were found in the interstitial tissues of the testicles, epididymides, and spermatic cord and in the endothelium and intima of the blood vessels of these organs (Fig. 4). The cysts showed polypoid growth from the intima, which caused partial vascular occlusion and ischemic tissue necrosis, as well as ischemic atrophy of the seminiferous tubules. Cysts were occasionally located within and occluding the seminiferous tubules and epididymal ducts, with formation of spermatoceles. Degenerating cysts in these organs were most numerous at 180 DPI and were frequently surrounded by granulomatous reactions. Animals with such lesions showed severe tubular degeneration and atrophic seminiferous tubules. The epididymides of the animal euthanized at 60 DPI exhibited ductal atrophy, parenchymal fibrosis, and macrophage infiltration.
Host tissue reaction to cysts varied, being limited near young viable cysts in early stages of infection but more pronounced later around degenerating cysts. These latter cysts were infiltrated by mixtures of macrophages, lymphocytes, plasma cells, eosinophils, and fibroblasts, but no giant cells were detected (Fig. 3). In subcutaneous muscle, there was, in addition to cysts and muscle fiber necrosis, chronic myositis with macrophage and eosinophil infiltration around the cysts and diffusely between muscle fibers.
The walls of the Besnoitia cysts varied in thickness between earlier and later stages of infection (Table 3) but were always composed of three layers comprising (1) an external collagen-like capsule that stained deep blue with Alcian blue / periodic acid–Schiff, (2) a middle layer that enclosed host cell elements including nuclei, and (3) an innermost layer, a thin parasitophorous vacuole membrane that filled the entire cell cytoplasm and contained numerous closely packed bradyzoites. Histologic examination of lung and liver showed mononuclear cell infiltration in parasitized goats but no Besnoitia cysts and so could not be definitely identified as a reaction to the parasite.
Ultrastructural Studies
The three layers of the cyst wall seen under light microscopy were more conspicuous at the ultrastructural level (Table 3). The cysts were initially small and thin walled but became larger with thicker walls by 180 DPI. The outer layer was fibrous connective tissue, and it showed the most changes, becoming thick and collagenous in the later stages of infection. The middle layer contained host cell nuclei, endoplasmic reticulum, and other host cell organelles. Its thickness increased with time. Due to degeneration of the cysts and severe inflammatory cell infiltration, it proved impossible to measure wall thickness at 270 DPI. At all developmental stages, pseudopodia-like filamentous extensions consisting of loosely packed fibrillar material from the second layer formed interdigitations with the inner surface of the external layer of the capsule. The innermost layer was a thin (1 μm) parasitophorous vacuolar membrane lined by an amorphous granular layer within the cytoplasm of a hypertrophied fibroblast, containing numerous crescentic bradyzoites.
From 180 DPI onward, the size of the cysts remained almost constant, but there were clear changes in the shape and size of the bradyzoites and their organelles (Table 3; Fig. 5, left). In all developmental stages, each bradyzoite was bounded by a smooth three-layered pellicle, and each contained a conoid, several micronemes, and 6 rod-shape rhoptries (Fig. 5, right), which were moderately electron dense and extending from the anterior end to the level of the nucleus. Twenty-two regularly arranged anterior subpellicular microtubules originating from the polar ring extended posteriorly along the inner membrane of the bradyzoite to the posterior end. Amylopectin granules, rough endoplasmic reticulum, ribosomes, and mitochondria were more evident and numerous during earlier stages of infection, whereas lipid droplets were prominent in the cytoplasm of the bradyzoites during later stages. The amylopectin droplets were heavily osmiophilic, dense, homogenous bodies, more numerous at early stages of infection than the large, empty lipid droplet vacuoles later present. No enigmatic bodies were found in B caprae.
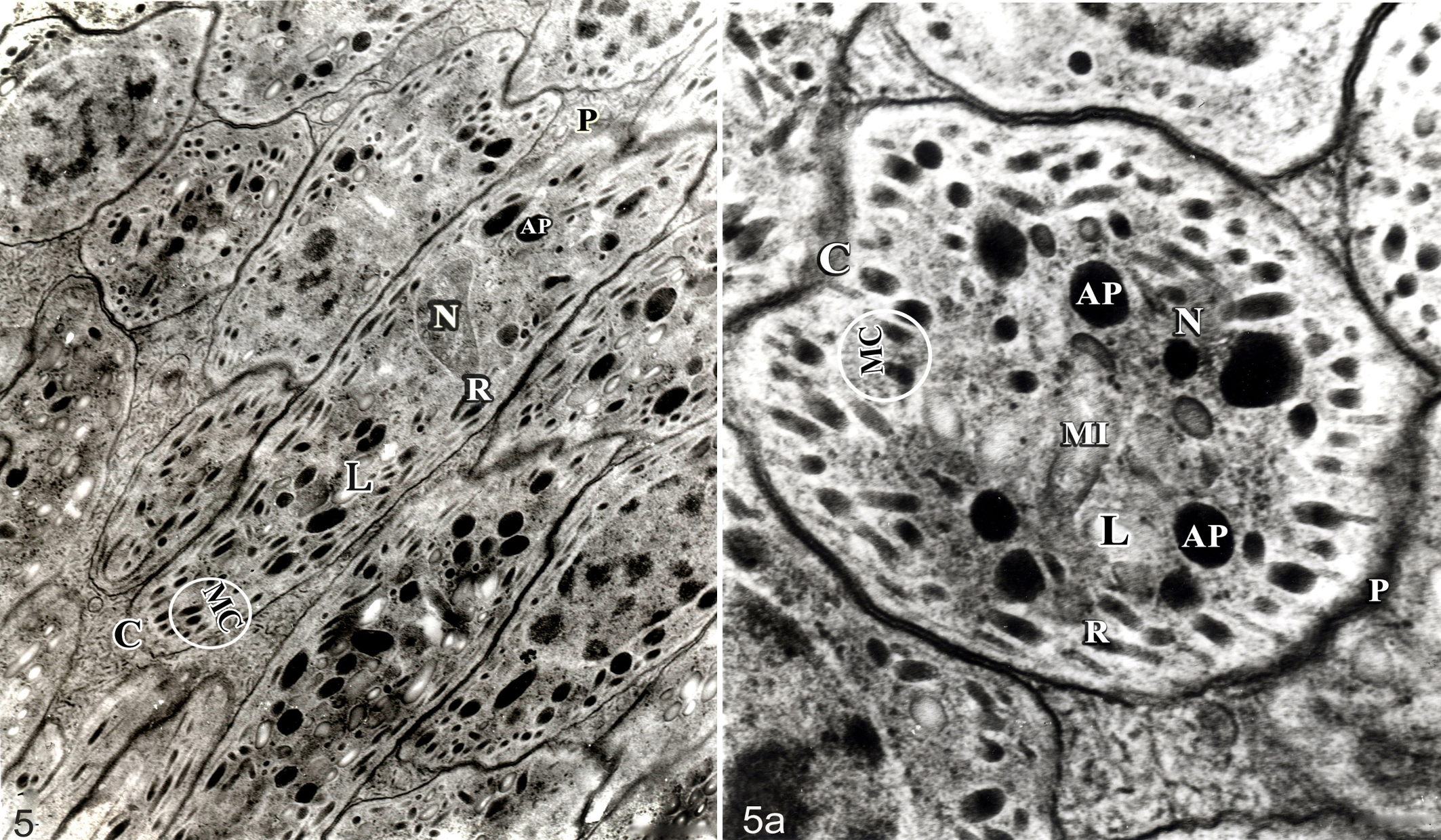
Besnoitia caprae bradyzoite; ultrastructure of longitudinal section (left) and cross section (right). Nucleus (N), conoid (C), rhoptries (R), amylopectin granules (AP), lipid granules (L), mitochondria (MI), micronemes (MC), pellicle (P).
Discussion
The development of B caprae cysts in goats in the present study, following subcutaneous inoculation of bradyzoites, shows that mechanical transmission may play a role in the spread of the parasite. This finding is consistent with the hypothesis that hematophagous insects may transmit some Besnoitia sp from an infected intermediate host to noninfected animals,1,18 which appears to be the case for B besnoiti in cattle where tsetse flies and tabanids have been implicated as vectors.2,5 The current experiment also confirms the predilection sites for this parasite described previously.24,27
The most serious limitations of this investigation were (1) the small number of animals available and (2) the noninfected animals were not exact controls for the experimental goats, given that it was impracticable to house them in precisely the same conditions. However, this shortcoming is mitigated to some extent by the large number of biopsy specimens that could be collected from each animal and the close correlation between the findings in the histology of the biopsies from several individuals in the earlier stages of the study. The availability of biopsy and autopsy material from the noninfected reservoir of clinically normal goats also provided a reasonable basis for confidence that findings from the experimental group give an accurate picture of the pathogenesis of experimental mechanical infection of goats with B caprae.
The acute stage of the disease is mainly correlated with proliferation of tachyzoites in blood vessel walls, whereas the chronic phase is characterized by cyst formation in subcutaneous tissues, testicles, epididymis, and scleroderma. The present experimental study found skin thickening and hair loss to be features of the chronic phase, as previously reported in relation to various naturally occurring besnoitioses,9,13,19 whereas resolution of the skin lesions was similar to that in some other Besnoitia infections. 16
The appearance of cysts at about 35 days after infection is somewhat earlier than that reported in native goats in Kenya, 21 with observation of Besnoitia cysts 56 and 60 days after bradyzoite inoculation. That experiment described cysts for a short period in some organs after infection but not the longer-term progressive changes in cysts and the associated tissue reactions during cyst degeneration, as shown in this study.
If the present findings in a limited number of goats are typical of caprine besnoitiosis, it seems that the mechanically transmitted parasite may not remain viable for more than about 270 days and that autoinfection does not occur. In mild infections, it seems possible that Besnoitia cysts may, paradoxically, survive longer because blood vessel occlusion is less likely and the resultant ischemic degenerative tissue changes may be less severe. Although this investigation found a gradual increase in the number of degenerating cysts, followed by their disappearance by 300 DPI in the one surviving goat, it would be premature to reach a conclusion on the mechanisms responsible for the spontaneous improvement of infected animals, given the few goats in the present study.
A long-term consequence of infection in clinical cases may be necrosis and mineralization of the testes, 13 with consequent temporary or permanent infertility.7,27 Similar mechanisms causing reduced fertility have been described in reindeer and cattle,1,14 although Njenga et al 22 reported that mild to severe chronic caprine besnoitiosis had an adverse effect on semen volume and sperm viability.
The progression of host tissue responses to Besnoitia infection in reindeer and rabbits (but not goats) has been described.12,15 It is not clear whether these reactions are triggered directly—by the presence of live cysts, their rupture, or their degeneration—or indirectly, by release of cytokines from damaged or necrotizing tissue in the vicinity of the cysts27,28 or by release of bradyzoites. The histologic findings in the present study are consistent with those of the natural besnoitiosis.3,6,27 There have been no previous morphometric studies on cysts and bradyzoites at histologic or ultrastructural levels, although Njenga et al 20 and Oryan and Azizi 24 described fine structural differences between Besnoitia isolated from goats and cattle.
The current study is the first investigation of the distribution pattern and chronology of formation and degeneration of B caprae cysts in goats and the morphologic changes in the cysts and bradyzoites up to 1 year after infection, with intermediate host tissue reactions to developmental stages of Besnoitia cysts. The results of the present experiment show that B caprae can be mechanically transmitted from an infected intermediate host to another healthy animal, thereby indicating that hematophagous arthropods or, possibly, epidermal contact may be involved in natural transmission. It also seems that the longevity of the infective parasite is less than 300 days and that the bradyzoites released from degenerating cysts are not biologically infective and are unable to develop new cysts in the predilection sites or other organs of the same animal.
Footnotes
Acknowledgment
We thank the authorities of Veterinary School, Shiraz University for their financial support and cooperation. We are grateful to Mr. L. Shirvani and Mr. G. Yousefi from the Department of Pathobiology, School of Veterinary Medicine, Shiraz University, Shiraz, Iran, for their technical assistance.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
