Abstract
A 17-y-old Rocky Mountain gelding was presented to the Virginia-Maryland Veterinary Teaching Hospital because of a 4-wk history of anorexia, weight loss, lethargy, and fever of unknown origin. Abdominal ultrasound revealed lymphadenomegaly of the abdominal and colonic lymph nodes, thickening of the wall of the large colon, and a mass associated with the large colon. The horse was euthanized given a poor prognosis. On autopsy, an ~20-cm diameter mass was found within the mesocolon between the right ventral and right dorsal colon. The mass had invaded through the colonic walls and formed a fistula between the 2 involved lumina. On histologic evaluation, the mass consisted of small numbers of large neoplastic lymphocytes, numerous small lymphocytes, and many foamy macrophages. A diagnosis of T-cell–rich, large B-cell lymphoma was made based on immunohistochemical staining for CD79a, CD3, and Iba1; concurrent infection with equid herpesvirus 5 was confirmed with in-situ hybridization (ISH). To our knowledge, neither a trans-colonic fistula resulting from alimentary lymphoma in a horse nor detection of intralesional equid herpesvirus 5 in equine alimentary lymphoma by ISH has been reported previously.
A 17-y-old Rocky Mountain gelding was referred to the Virginia-Maryland Veterinary Teaching Hospital (VTH; Virginia Tech, Blacksburg, VA, USA) with a 4-wk history of weight loss, lethargy, and hyporexia. Intermittent fever and diarrhea were noted ~1 wk before presentation at the VTH. Prior to referral, the gelding was examined on the farm, and a sample of blood was taken for a CBC and sent to the Virginia Tech Animal Laboratory Services (ViTALS). Treatment on the farm included flunixin meglumine (1.1 mg/kg; 10 mL) and minocycline (4 mg/kg PO q12h). Physical examination findings upon presentation to the VTH included normal temperature (37.5°C), heart rate (40 bpm), and respiratory rate (12 bpm), normal gut sounds, and soft manure (cowpie consistency). The CBC was repeated on presentation; results were similar to those on the farm (Table 1) and included evidence of systemic inflammation with probable underlying stress response, titrational metabolic acidosis (anion gap: 10.5, range: 1.0–9.0 mEq/L; CO2: 27, RI: 28–36 mEq/L), and mild hypoalbuminemia (27 g/L; RI: 32–40 g/L). Transabdominal ultrasound was performed and revealed a large mass in the large colon with up to 2.0-cm thickening of the adjacent colonic wall and enlarged, up to 4.0-cm diameter, colonic lymph nodes within the right abdomen. The horse was euthanized given a poor prognosis and was submitted for postmortem examination.
CBC abnormalities in a horse with intestinal lymphoma on day 0 at the farm of origin and 8 d later (day 8) when presented to Virginia-Maryland Veterinary Teaching Hospital.

On postmortem examination, an ~20-cm diameter, firm, white-to-tan, irregularly shaped mass was found within the mesocolon between the right ventral and right dorsal colon, closely adjacent to the diaphragmatic flexure. On cut section, the mass contained soft, granular, blackened necrotic areas and pockets of pus. The walls of colonic segments adjacent to the mass were expanded by edema (Fig. 1A). A transmural mucosal ulcer, connected to a trans-colonic fistula, formed a continuous open track from the lumen of the right ventral colon through the mass and into the lumen of the right dorsal colon (Fig. 1B). The regional abdominal lymph nodes were enlarged and tan-to-brown with a loss of cortical and medullary architecture. The abdominal cavity contained increased amounts of yellow transparent ascitic fluid. All other thoracic and abdominal organs were unremarkable. Aerobic bacteriologic culture of the purulent exudate within the mass yielded no growth.
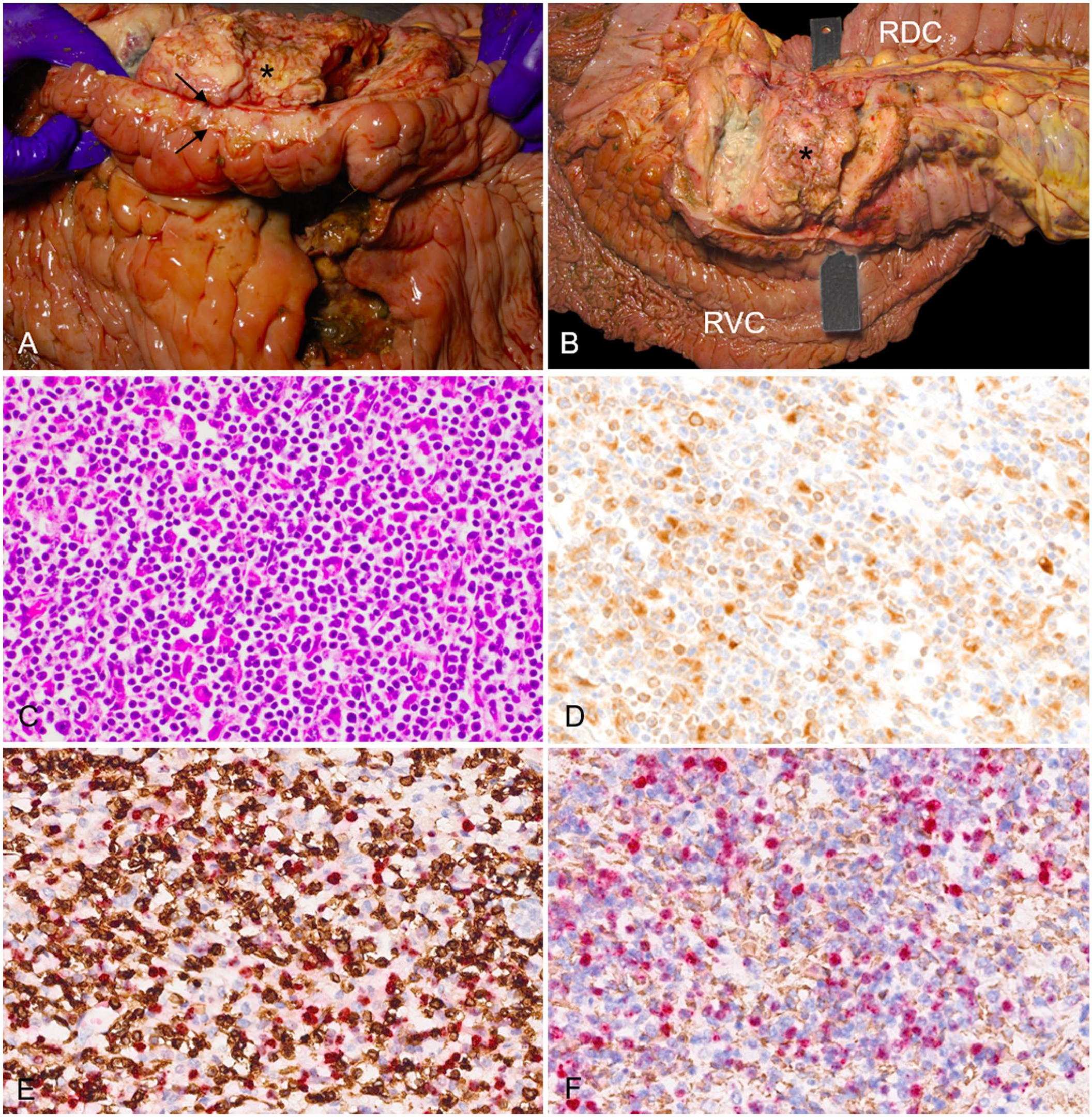
Large colon of a Rocky Mountain gelding with colonic T-cell–rich, large B-cell lymphoma.
Sections of the mesocolonic mass, large colon, and associated lymph nodes were examined with routine H&E stain, and representative samples of the mass were further assessed by immunohistochemical (IHC) staining for detection of CD79a, CD20, Pax5, CD3, and ionized calcium-binding adaptor molecule 1 (Iba1) leukocyte markers as well as equid herpesvirus 5 (EHV5; Equid gammaherpesvirus 5) nucleic acid by in-situ hybridization (ISH). Formalin-fixed, paraffin-embedded (FFPE) tissue sections were processed at the New York Animal Health Diagnostic Center (NYAHDC; Cornell University, Ithaca, NY, USA) using standard protocols and an automated IHC stainer (Bond-Max automated IHC staining system; Leica). Briefly, serial 4-µm tissue sections mounted on charged slides were deparaffinized (AR9222, Bond dewax solution; Leica), and after heat epitope retrieval (AR9640, Bond epitope retrieval solution 2; Leica), the slides were incubated with the respective primary antibody for 15 min followed by a polymeric horseradish peroxidase (DS9800; Bond polymer refine detection; Leica) linker antibody conjugate detection system, and hematoxylin counterstain (DS9390; Leica) for 5 min. For T-cell detection, the ready-to-use mouse monoclonal IgG2a anti-human T-cell antigen receptor complex (PA0553, Ready-to-use primary antibody CD3, clone LN10; Leica) was used. For B-cell detection, ready-to-use mouse monoclonal IgG1 anti-human CD79a (PA0599, clone JCB117; Leica), rabbit polyclonal IgG anti-human CD20 (PA5-16701; Thermo Fischer) diluted 1:400, mouse monoclonal IgG1 (clone 1EW, PA0552; Leica), and anti-human pro-, pre-, and mature B-cell Pax5 encoded nuclear transcription factors were used. After a 5-min peroxide block (DS9800; Leica), macrophages–dendritic cells (Iba1) were detected by using a rabbit polyclonal antibody raised against a conserved human, rat, and mouse synthetic peptide of the Iba1 C-terminal sequence (019-19741; Fujifilm Wako) diluted 1:3,000. Normal equine lymph node served as a positive antibody and method control for T and B cells, and for macrophage–dendritic cells; omission of the primary antibody on serial sections served as a negative method control. Positive immunolabeling was identified as brown staining with diaminobenzidine (DAB).
For localization of EHV5 nucleic acid in FFPE tissue sections, a highly sensitive and previously validated ISH method (RNAscope; Advanced Cell Diagnostics) 14 was performed at the NYAHDC using a standard protocol and an automated ISH research platform (Discovery Ultra; Roche). After deparaffinization and target retrieval (760-248, mRNA sample prep kit; Roche), 4-μm tissue sections mounted on charged slides were hybridized with the 200 μL of a 20ZZ ISH probe targeting a 1,478-nucleotide region of the EHV5 gB gene encoding a highly expressed viral envelope glycoprotein (GenBank AF050671.1; 463701, V-EHV5-gB, Advanced Cell Diagnostics). This was followed by signal amplification (760-236, mRNA RED probe amplification kit; Roche), detection (760-234, mRNA RED detection kit; Roche), and counterstaining with hematoxylin. A section of a horse lung with equine multinodular pulmonary fibrosis that was EHV5 PCR assay positive served as a positive control. Positive signal was identified as red staining within infected cells.
For co-localization of EHV5 nucleic acid in T and B cells, and in macrophage–dendritic cells, serial 4-µm FFPE tissue sections mounted on charged slides were processed for ISH followed by the IHC procedure without deparaffinization and heat epitope retrieval.
The abdominal mass consisted of an unencapsulated, poorly demarcated, and densely cellular population of round cells that infiltrated and effaced all layers of the right ventral and right dorsal colonic wall and the mesocolonic lymph nodes. The mass was composed of sheets of 3 distinct round-cell populations separated by a moderately dense fibrovascular stroma (Fig. 1C). 1) The large neoplastic lymphocytes had moderate amounts of lightly eosinophilic cytoplasm, round-to-oval nuclei with coarsely stippled chromatin, and single, prominent nucleoli. This cell population had a mitotic count of 1 in 2.37 mm2, exhibited cytoplasmic immunoreactivity for CD79a antigen, and accounted for 30–40% of the cell population by IHC (Fig.1D). 2) The small round cells had scant lightly basophilic cytoplasm, small nuclei, and a condensed chromatin pattern. This cell population had cytoplasmic immunoreactivity for CD3 and accounted for 50–70% of the cell population by IHC (Fig. 1E). 3) The large round cells had abundant, brightly eosinophilic cytoplasm containing needle-like eosinophilic inclusions and large, round, vesicular nuclei. These cells had cytoplasmic immunoreactivity for Iba1 antigen and accounted for <15% of the cell population by IHC (Fig. 1F). Neoplastic cells did not express CD20 or Pax5 antigens by IHC (data not shown). Similar cell populations were identified replacing and effacing the architecture of mesocolonic lymph nodes.
Throughout the mass, the lymph nodes and the colon were random, locally extensive necrotic regions bordered by granulomatous inflammation consisting of many large foamy macrophages and multinucleate giant cells. Small caliber blood vessels within the mass and colon contained fibrin thrombi. Detection of EHV5 nucleic acid by ISH in a representative sample of the mass revealed strong nuclear signal (Fig. 2). Double IHC-ISH revealed a lack of co-localization between EHV5 signal and both CD3 and Iba1 cells (Fig. 1E, 1F). Unfortunately, the CD79a antibody–detection method was incompatible with the ISH protocol. However, given the lack of co-localization of EHV5 with CD3 and Iba1, the EHV5 signal was most likely detected in B cells (diagnosis by exclusion).
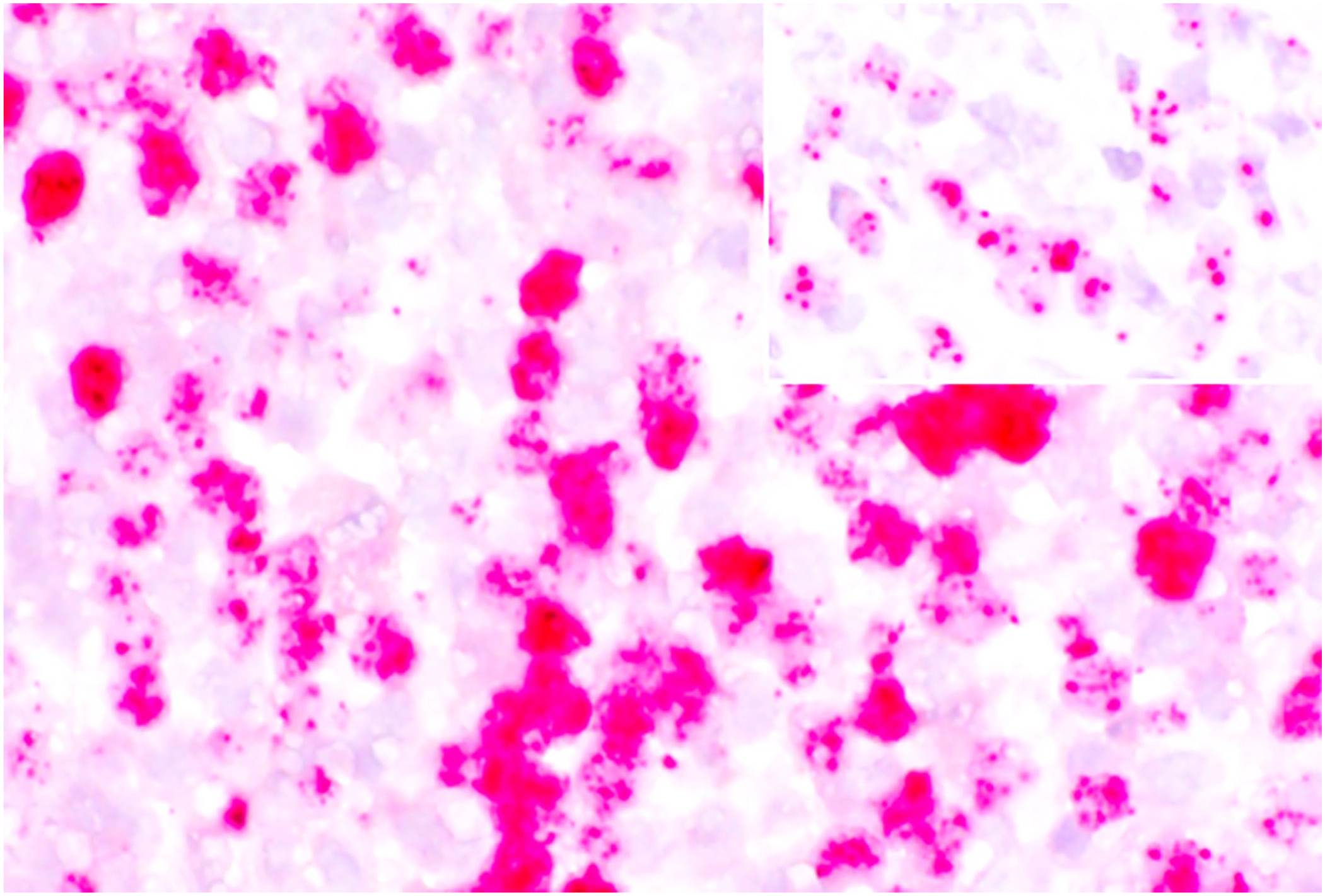
Photomicrographs of an in-situ hybridization assay to detect equid herpesvirus 5 (EHV5) nucleic acid. Strong nuclear EHV5 signal. Inset: positive control (horse lung with equine multinodular pulmonary fibrosis, confirmed with PCR).
A diagnosis of a T-cell–rich, large B-cell lymphoma (TCRBCL) with formation of a trans-colonic fistula and concurrent EHV5 infection was made based on the clinical presentation, gross autopsy findings, cell morphology on routine H&E stain, and results of CD79a, CD3, and Iba1 IHCs and ISH assays. We found no cases of intralesional detection of EHV5 in equine alimentary lymphoma nor trans-colonic fistula resulting from equine alimentary lymphoma in a search of Google, PubMed, CAB Direct, Web of Science, and Scopus, suggesting that no descriptions of these conditions have been reported in horses.
Equine intestinal neoplasia is rare; the most commonly reported type is lymphoma, followed by gastrointestinal stromal tumor, adenocarcinoma, and leiomyosarcoma.18,20 Other less commonly reported alimentary neoplasms include leiomyoma, squamous cell carcinoma, myxosarcoma, ganglioneuroma, peripheral nerve sheath tumor, and carcinoid.16,18 Alimentary lymphoma is the third most commonly reported equine lymphoma, behind multicentric and cutaneous lymphoma.2,5,18 There is no reported breed or sex predisposition for alimentary lymphoma, which is typically seen in adult horses.5,16,18 This neoplasm has been reported in both the small and large intestines, with higher prevalence in the small intestine. When concurrent infiltration of both the small and large intestinal segments occurs, alimentary lymphoma may be difficult to differentiate from multicentric lymphoma.2,5,16–18 Primary alimentary lymphoma has been suggested to develop from the gut-associated lymphoid tissue (GALT). 18 The presence of tumor cells in multiple locations in our case makes determination of the origin of the lymphoma more challenging; however, potential primary sites may include the GALT, one or more mesenteric lymph nodes, or a multicentric lymphoma originating in the abdominal lymph nodes, although this seems less likely given that no extra-alimentary organ systems or distant lymph nodes were affected.
Historically, TCRBCL is the most commonly reported subtype of alimentary lymphoma. 18 However, a 2020 analysis reported that enteropathy-associated T-cell lymphoma (EATL1; 8 of 20 cases) and monomorphic epitheliotropic intestinal lymphoma (MEITL/EATL2; 9 of 20 cases) are more common than TCRBCL (3 of 20 cases). 2 EATL1 cases consist of large, CD3+ T cells; EATL2 tumors are composed of small, CD3+ T cells; epitheliotropism is a prominent feature in both. 2 In contrast, TCRBCL consists of a mixed population of cells and epitheliotropism is absent, features that are consistent with our report. 2 Metastasis to both regional lymph nodes and lungs is commonly reported with equine alimentary lymphoma; EATL1 may metastasize at a slower rate compared to TCRBCL.2,18 In our case, all abdominal lymph nodes were enlarged, and histology confirmed the presence of neoplastic cells similar to those found in the mesocolonic mass. We noted no metastases in the lungs or other distant organ systems.
An important finding in our case was the detection of concurrent EHV5 nucleic acid signal within neoplastic cells (likely B cells) of the mesenteric mass. EHV5 infection has been associated with cutaneous and multicentric equine lymphoma, as well as gastric ulcer syndrome, and equine multinodular pulmonary fibrosis (EMPF).10,11,14,20–22 To our knowledge, the demonstration of EHV5 within lesions other than EMPF has not been reported previously. A nearly 5-fold higher prevalence of EHV5 infection is found in horses with lymphoma (67% PCR assay positive) compared to lymphoid tissue obtained from healthy control horses (14% PCR assay positive). 22 A single report of remission of lymphoma in a submandibular lymph node upon successful treatment of EHV5 with acyclovir is suggestive of an etiologic association. 21 The association between EHV5 infection and the development of EMPF is not completely understood. Studies propose that, after inhalation, EHV5 viral particles travel to the tonsillar crypts via the mucociliary apparatus, using glycoprotein B directly to infect lymphocytes within the lymphoid follicles.8,20 Infected lymphocytes then enter the bloodstream and/or lymphatics, infect other lymphocytes via cell–cell transfer, and travel to other sites, including the lungs. 20 Pulmonary fibrosis has been reproduced by experimental infection of horses with EHV5, and it has been proposed that fibrosis is a consequence of EHV5 viral latency within the lungs.6,23
There is worldwide genetic diversity of EHV5 strains, which are carried with imported horses, in some cases with more than one strain. 1 Although we did not directly demonstrate a causative link between EHV5 and lymphoma, the in-situ detection of the virus could indicate viral involvement. Hypothetically, the gB gene sequence variation reported in the EHV5 strains, or the reactivation and combination of strains, can be implied in the development of lymphoma or EMPF in horses 1 ; however, this requires further investigation.
Other herpesviruses have also been associated with lymphoproliferative disorders in both veterinary and human medicine, including Epstein-Barr virus (Human gammaherpesvirus 4), 13 Kaposi sarcoma-associated herpesvirus (Human gammaherpesvirus 8), 7 and Marek disease virus (Gallid alphaherpesvirus 2), that infect various cell types, including B cells, natural killer cells, T cells, and epithelial cells, resulting in latency within T cells, and neoplastic transformation of CD4+ T cells. 3 The mechanisms of neoplastic transformation in these viral infections are multifactorial but may inform future research into the contribution of EHV5 in the development of equine lymphoma.
Consistent with our case, clinical signs associated with intestinal neoplasia in horses are typically nonspecific and include anorexia, chronic weight loss, intermittent colic, diarrhea, depression or lethargy, and pyrexia.11,16,18 Common clinical pathology findings include anemia, hyperfibrinogenemia, hyperproteinemia, hyperglobulinemia, and hypoalbuminemia. In lymphoma cases, anemia can be attributed to various causes, including inadequate production of erythrocytes as a result of myelophthisis of the bone marrow, and blood loss attributed to gastrointestinal ulceration.17,18 These explanations may have contributed to the anemia seen in our case, although bone marrow was not examined. Several hematologic and biochemical changes seen in lymphoma have been attributed to the inflammatory stimulus by neoplastic cells, including hyperfibrinogenemia caused by increased production of cytokines, such as interleukin-6 (IL-6), and hyperproteinemia characterized by increased serum globulins in response to chronic antigenic stimulation.12,17,18 In addition to the inflammatory stimulus of the neoplastic cells, monocytosis and neutrophilia may be a response to the mucosal ulceration and tumor necrosis noted in our report. 12 Lymphopenia has been reported in horses with lymphoma, and hypoalbuminemia is commonly seen and attributed to both protein-losing enteropathy associated with mucosal damage and decreased production as a negative acute-phase protein.9,12,16,18
Intestinal fistulas are rare, abnormal communications between gastrointestinal lumina in different segments. 24 Common acquired causes of intestinal fistulas include post-surgical complications, trauma, presence of a foreign body, inflammatory bowel disease, diverticulitis, neoplasia, and chemotherapy or radiation treatment.15,19 Fistula formation occurs after perforation when adjacent structures are also affected by infiltration by the inciting cause; this can occur because of pressure necrosis caused by a mass or feed impaction, regional inflammation, and/or secondary infection.19,24 In horses, there are rare reports of enterocutaneous fistulae as a result of a congenital umbilical hernia or penetrating trauma to the equine patient’s abdomen. 4 In our case, fistula formation is attributed to several factors, including pressure necrosis by the large mesocolonic mass, transmural invasion of the colonic wall by neoplastic cells, and regional inflammation secondary to the mesenteric mass and necrosis therein. Bacterial colonies were not seen histologically in areas of necrosis and inflammation.
Footnotes
Acknowledgements
We thank Dr. Thomas Cecere for his gross photography assistance, Jinhua Zhang and Katherine Trainque for the histotechnology, and Robin T. Pasiak at the Animal Health Diagnostic Center histology laboratory at Cornell University for performing the IHC and ISH assays.
Declaration of conflicting interests
The authors declare no potential conflict of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
