Abstract
Expression of interferon regulatory factor 4 (IRF4)/multiple myeloma oncogene-1 (MUM1) in peripheral T-cell lymphoma (PTCL) in people is associated with a poorer survival outcome compared to cases of PTCL lacking MUM1 expression. The aim of this study was to determine whether MUM1 is expressed in canine peripheral T-cell lymphoma not otherwise specified (PTCL-NOS). For comparison, the presence of MUM1 antigen was also investigated in canine diffuse large B cell lymphoma (DLBCL). Nine cases of PTCL-NOS and 9 cases of DLBCL diagnosed by a commercial veterinary diagnostic laboratory were selected. Positive immunohistochemical labeling for MUM1 was observed in PTCL-NOS (2 out of 9 cases) and DLBCL (3 out of 9 cases). These findings suggest that a subset of neoplastic T and B lymphocytes can express MUM1. The role of MUM1 in the biological behavior and outcome of canine lymphoma (CL) requires further investigation on a larger number of cases.
Keywords
Canine lymphoma (CL) is a common neoplasm with an estimated incidence rate of 20 to 100 cases per 100,000 dogs and is comparable to non-Hodgkin lymphoma in humans. 20 According to the World Health Organization (WHO) classification criteria, 17 CL has different phenotypic subtypes. The subtype is known to have an impact on prognosis and is used to inform treatment protocols.9,20 Approximately 60%–70% of CL are B-cell, 30%–40% T-cell, and less than 1% null (non-B non-T cell).11,16 Diffuse large B-cell lymphoma (DLBCL) is the most common subtype of CL. Peripheral T-cell lymphoma not otherwise specified (PTCL-NOS) is the second most common subtype of CL and comprises a heterogenous group of T-cell lymphomas.11,16 High-grade T-cell lymphoma is an aggressive disease, and when treated with cyclophosphamide, hydroxydaunorubicin, vincristine, and prednisone (CHOP)-based protocols, dogs have a complete remission rate as low as 40%, relapse earlier and have shorter survival time than dogs with a comparable stage, high-grade B-cell lymphoma. 9
Interferon regulatory factor 4 (IRF4)/multiple myeloma oncogene-1 (MUM1) is a transcription factor associated with cell growth, particularly B-cell maturation. Multiple myeloma oncogene-1 immunohistochemistry is routinely used for diagnostic confirmation of plasma cell tumor in dogs. 12 In addition, expression of MUM1 has been demonstrated in some CLs of B-cell origin. 12 Expression of MUM1 within PTCL-NOS in humans is associated with a poorer survival outcome compared to tumors which do not express this transcription factor.7,15 The purpose of this retrospective study is to determine whether canine PTCLs-NOS express MUM1, and if the degree of expression differs between DLBCLs and PTCLs-NOS.
A retrospective search was performed at IDEXX Diagnostic Laboratories Ltd (Wetherby, UK) on cases submitted between 2018 and 2020. Only cases with a diagnosis of PTCL-NOS and DLBCL and the immunophenotype already confirmed by immunohistochemistry for B and T cell antigens were included in this study. A total of 9 cases of PTCL-NOS and 9 cases of DLBCL were collected. The samples consisted of whole node extirpation (17 samples) and Tru-cut biopsies (1 case of PTCL-NOS). Haematoxylin and eosin (H&E) stained slides and immunohistochemical slides of all cases were reviewed by 2 board certified pathologists (E. R., S. L. C.) and classified according to the WHO criteria. 17 The following features were recorded for each case: nodular versus diffuse growth pattern, nuclear size, and the number of mitoses per high-power field. Nuclear size was determined as small (<1.5x the size of a red blood cell), intermediate (1.5–2x the size of a red blood cell), or large (>2x the size of a red blood cell). The number of mitotic figures was counted in 1 40X digital high-power field (HPF) in the area of highest mitotic activity and multiplied by 1.2 to calculate the mitotic count (MC) in 1 standard HPF (0.237 mm2) according to the Meuten et al. 8 Lymphomas with 0 to 5 mitoses in 0.237 mm2 were graded as low grade, those with 6 to 10 mitoses/0.237 mm2 were graded as medium grade, and those with greater than 10 mitoses/0.237 mm2 were graded as high grade. 17
To demonstrate the presence of MUM1 antigen in the T-cell or B-cell population, a double immunohistochemical procedure with the T cell marker CD3 or B cell marker CD20 and MUM1 was performed according to the following method. 10 CD20 was selected as B-cell marker for the double immunohistochemical procedure instead of PAX5 because CD20 labeling is membranous/cytoplasmic (instead of nuclear) and thus did not interfere with the MUM1 labeling.
Briefly, 3 micrometers thick sections were cut and mounted on KP-PLUS positive charged slides (Klinipath, The Netherlands), incubated overnight at 37°C, deparaffinized in 3 changes of Histo-Clear Histology Clearing Agent (National Diagnostics, Atlanta, Georgia, USA) and re-hydrated in ethanol solutions of decreasing concentration.
For heat-induced epitope retrieval (HIER), sections were treated at full pressure with Access Retrieval Unit (Menarini, Italy) in ethylenediaminetetraacetic acid (EDTA) buffer (pH 9) for 90 seconds at 125°C. The sections were then rinsed in Tris Tween buffer (pH 7.5).
Endogenous peroxidase activity was quenched at room temperature with 3% hydrogen peroxide in phosphate buffered saline (PBS). The sections were incubated for 30 minutes at room temperature with the primary antibody MUM1 (MUM1p clone, Dako) in a 1:50 dilution. The sections were then washed with Tris Tween buffer (pH 7.5) to remove the excess primary antibody.
For primary antibody detection, the sections were incubated with EnVision+ System HRP labeled polymer anti-mouse secondary antibody (Dako, Carpinteria, California, USA) for 30 minutes at room temperature. The sections were washed with Tris Tween buffer (pH 7.5) to remove any excess labeled polymer and incubated with Envision FLEX HRP Magenta Substrate Chromogen System (Dako, Carpinteria, USA) for 5 minutes. Excess Magenta was removed by washing the section with distilled water followed by Tris Tween buffer (pH 7.5). Sections were then treated with 200 mM sulphuric acid to remove residual HRP activity without impacting upon the intensity of the HRP Magenta stain.
The sections were then treated for 5 minutes at room temperature with 3% hydrogen peroxide in PBS to quench endogenous peroxidase activity. The sections were then incubated for 30 minutes with either CD3 antibody (polyclonal, Dako, Carpinteria, USA) in a 1:300 dilution or CD20 antibody (polyclonal, Fisher, Waltham, MA, USA), in a 1:700 dilution. For primary antibody detection, the sections were incubated with EnVision+ System HRP labeled polymer anti-rabbit secondary antibody (Dako, Carpinteria, USA) for 30 minutes at room temperature.
Tris Tween buffer was used to remove excess labeled polymer from the sections. This was followed by two 5-minute incubations with 3,3’-diaminobenzidine (DAB) substrate-chromogen (EnVision+ System, Dako, Carpinteria, USA). Sections where rinsed twice with distilled water for 5 minutes each time. Tissues were counterstained using Gill’s hematoxylin and mounted using clear resin and coverslips for long-term storage. A normal canine lymph node was used as positive control; negative control was obtained replacing the primary antibody with Dako REAL Antibody Diluent.
A semi-quantitative evaluation of the double immunolabeled slides was performed according to the criteria described by Heo et al: 7 cases with less than 5% neoplastic cells showing positive immunolabelling for MUM1 antibody were considered negative, while cases with more than 5% of neoplastic cells labeling positive were considered positive.
To calculate the number of MUM1 positive neoplastic cells within each group of lymphomas, digital image analysis was performed using an Aperio Leica CS2 scanner with QuPath-Software (v.0.2.3). The complete area of the tissue was inspected, starting from the left upper corner of the tissue, until 100 MUM1 positive cells were counted. The ratio of MUM1 positive and CD3 (or CD20) positive and negative cells was determined. A cell was considered double positive when it had both brown cytoplasmic and red nuclear labeling.
The results of histological examination of PTCL-NOS cases are summarized in Table 1. Peripheral T-cell lymphoma not otherwise specified cases showed a diffuse growth pattern. Five out of 9 cases were composed of intermediate-sized cells, 3 were composed of large cells, and 1 of small cells. The histological grade was low in 4 cases, intermediate in 2, and high in 3 cases. None of the PTCL-NOS showed plasmacytoid features.
PTCL-NOS cases. Summary of the results of the histological analysis and evaluation of the double immunohistochemical labeling for CD3 and MUM1.
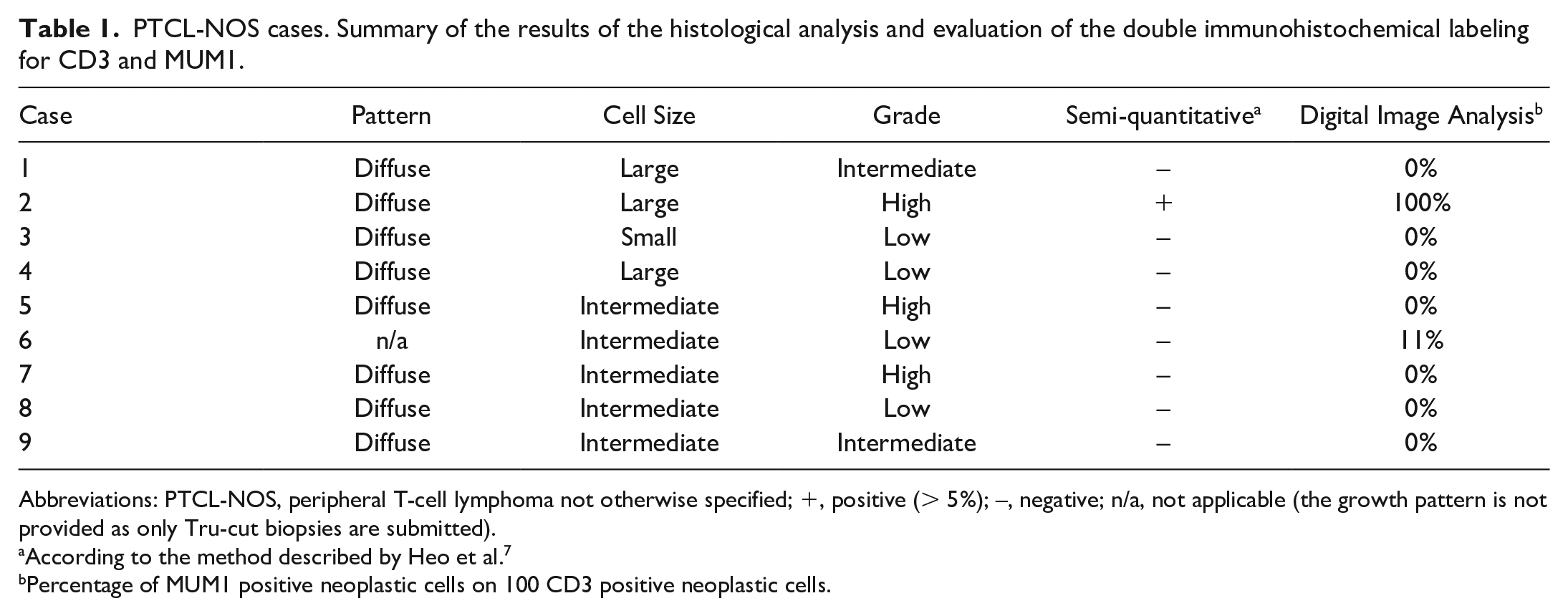
Abbreviations: PTCL-NOS, peripheral T-cell lymphoma not otherwise specified; +, positive (> 5%); –, negative; n/a, not applicable (the growth pattern is not provided as only Tru-cut biopsies are submitted).
According to the method described by Heo et al.7
Percentage of MUM1 positive neoplastic cells on 100 CD3 positive neoplastic cells.
DLBCL showed a diffuse growth pattern and large (6 out of 9 cases) or intermediate to large (3 cases) cell size. The histological grade was intermediate in 5 cases and high in 4 cases. None of the DLBCL were subtyped as plasmablastic lymphoma. MUM1 labeling was nuclear in both PTCLs-NOS and DLBCLs.
Semi-quantitative evaluation of the double immunolabelled slides detected the presence of double positive cells in 1 out of 9 cases of PTCL-NOS (Fig. 1, a and b) and in 2 out of 9 cases of DLBCL. With digital image analysis, this increased to 2 out of 9 cases of PTCL-NOS and 3 out of 9 cases of DLBCL showing double positive cells. No difference in the pattern and degree of MUM1-positive labeling was noted between PTCLs-NOS and DLBCLs.
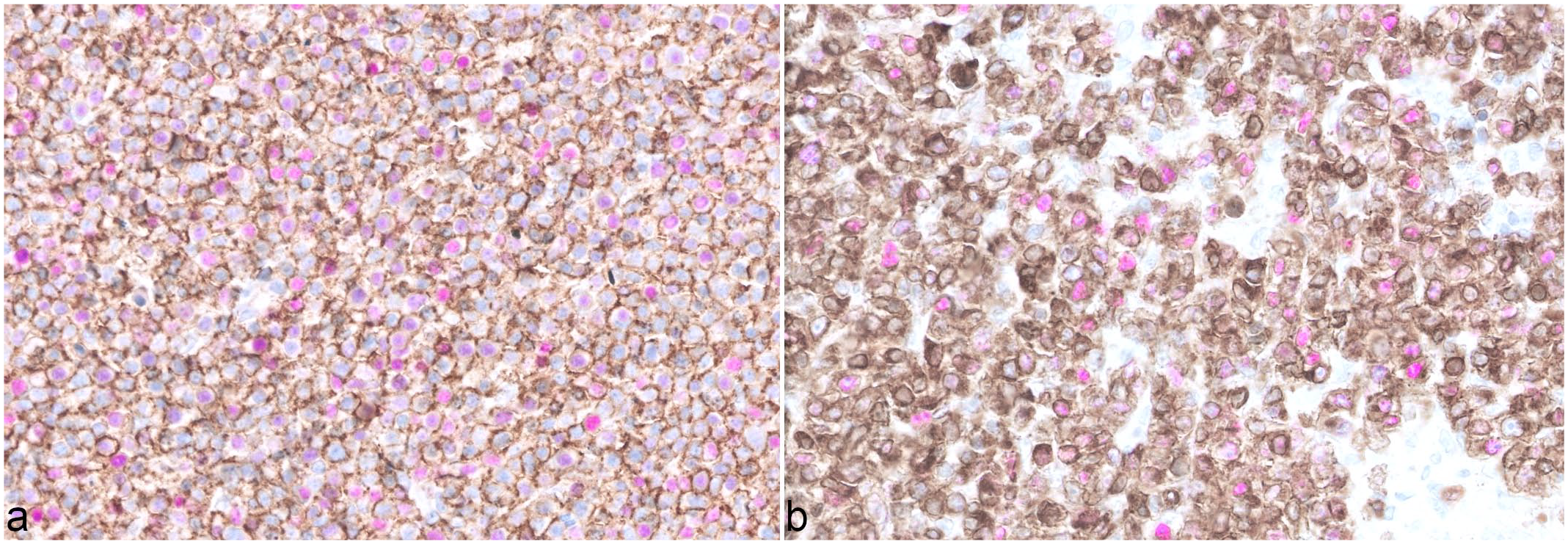
(a) Peripheral t-cell lymphoma not otherwise specified (PTCL-NOS), case no. 2, lymph node, dog. The neoplastic cells have strong membranous immunolabelling for CD3. Nuclear immunolabelling for MUM1 is also noted, diffusely, in the same neoplastic cells. (b) PTCL-NOS, case no. 6, lymph node, dog. The neoplastic cells have strong membranous immunolabelling for CD3. Nuclear immunolabelling for MUM1 is also noted in 11% of the CD3-positive neoplastic cells. Double immunohistochemistry for CD3 (DAB chromogen) and MUM1 (HRP Magenta chromogen).
In humans, PTCLs are aggressive non-Hodgkin lymphomas characterized by frequent relapse and poor survival outcome.2,18 Most patients with PTCL are treated mainly with anthracycline-based chemotherapies, such as CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisolone); however, their response to this therapy is not as good as patients who have B-cell lymphomas. 3 MUM1 has been suggested as a potential prognostic factor and therapeutic target in PTCL in humans.5,7,15 In veterinary oncology, the expression of MUM1 in CL has previously been reported in a small number of cases of B-cell lymphoma and anaplastic lymphoma. 12 To our knowledge, the expression of MUM1 has not been previously investigated in canine PTCL-NOS.
In the present study, positive immunolabelling for MUM1 was demonstrated in canine PTCL-NOS cells expressing CD3, using a double immunohistochemical assay. One of the positive cases was a high-grade, large cell lymphoma. In human pathology, MUM1 is reported to be frequently expressed in anaplastic lymphoma of large cell type.7,15,19 A correlation between the cell size, grade, and MUM1 expression can be thus hypothesized also in canine species; however, a study with a larger number of cases is needed to evaluate this hypothesis.
Based on the high variability in grade, cell size, and MUM1 expression noted in PTCL-NOS in this study series, it is likely that different subsets of neoplastic T lymphocytes are grouped under the general definition of PTCL-NOS. Human DLBCL is also a heterogeneous disease with a variable clinical course. The subclassification of DLBCL according to the stages of B-cell differentiation during germinal center maturation, has prognostic significance.1,13,14 For this reason, in human pathology, immunohistochemical detection of MUM1 is used in conjunction with other markers to determine the cell of origin of DLBCL.4,6 A subset of canine B cell lymphomas also express MUM1, as demonstrated by the results of this study and previously shown by Ramos-Vara et al. 12
In conclusion, a better characterization of the expression profile of neoplastic cells in canine PTCL-NOS and DLBCL, with the help of immunohistochemistry and/or gene expression profiling, might allow an accurate subclassification and might also represent an important part of the diagnostic work-up, helping with the development of new therapeutic strategies. In addition, it might allow the identification of subtypes of lymphoma that display a more favorable outcome and a better response to therapy. Finally, prospective studies on a larger number of cases with complete follow-up data are required to definitively establish a correlation among tumor grade, cell size, and MUM1 expression and to determine any potential prognostic significance of MUM1 in canine PTCL-NOS and DLBCL.
Footnotes
Acknowledgements
The author(s) thank Mr Stefano Gandolfo for the technical assistance in the photographic plate design.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
