Abstract
An 8-month-old Ayrshire heifer had a rapidly growing mass in the axillary region of the left thoracic limb. The mass surrounded the distal humerus and entrapped nerves of the brachial plexus, causing an abnormal gait. Histologically, the mass was composed of clusters and cords of round to polygonal cells with scattered, spindle-shaped cells. The neoplastic cells stained positively for vimentin and cytokeratin. No staining was found with S-100 protein, kappa and lambda light chains, or T-cell markers by immunohistochemistry. On electron microscopic evaluation, the cytoplasm of the neoplastic cells contained few organelles, principally rough and smooth endoplasmic reticulum and mitochondria. This synovial sarcoma has histologic and ultrastructural features characteristic of the poorly differentiated subtype of synovial sarcoma in the human classification system.
Keywords
Synovial sarcomas are malignant tumors of undetermined origin. They are rarely encountered in animals but have been reported in dogs, cats, cattle, horses, and ferrets. 4–6 Affected animals are usually adults. Lymph node and pulmonary metastases commonly occur. 4 The tumor is highly destructive locally and progression is usually slow, but it can pursue a fulminating course ending in loss of organ function within a few months requiring euthanasia. We report a rapidly growing synovial sarcoma in a heifer with pulmonary and lymphatic metastases.
An 8-month-old purebred Ayrshire heifer was presented at the Veterinary Teaching Hospital of the University of Montreal for a rapidly growing mass in the left forelimb. On clinical examination, there was a firm 20-cm-diameter ulcerated and mobile expansive mass localized in the subcutaneous tissue of the craniolateral aspect of the distal end of the humerus. The left prescapular lymph node was severely enlarged. The animal had an abnormal gait resulting from mechanical impairment and from a neurological deficit probably caused by nerve compression. A bilateral increase of respiratory sounds and cardiac arrythmia were also noted. No other abnormality was present on clinical examination. Biochemistry and hematology values were within the reference range, and serum selenium level was marginally low. The animal was seronegative for bovine leukemia virus.
Ultrasound examination of the mass revealed hyperechoic tissue of homogeneous density with abundant vascularization. Fine-needle aspirations of the mass and of the prescapular lymph node revealed a highly cellular population composed mainly of round neoplastic cells with some mature lymphocytes, plasma cells, and macrophages. Marked anisocytosis and anisokaryosis were evident. Neoplastic cells contained a round or pleomorphic cleaved nucleus with clumped chromatin and irregular nucleoli. Atypical mitotic figures were occasionally seen. The morphological appearance of the cells (lymphoblast-like) was suggestive of lymphoma. In view of the poor prognosis, the animal was euthanatized.
On postmortem examination, there was a 30 × 10–cm mass surrounding the distal part of the humerus, close to the radiohumeral joint. No bone or articular invasion was noted. Although the joint capsule was not involved, the mass was intimately attached to the tendon sheet of the biceps brachii. The mass was poorly delineated, and it compressed and infiltrated the surrounding muscles of the forelimb and entrapped nerves of the brachial plexus. It was white, firm, and fibrous, and on cut section, it contained numerous randomly distributed 5–10-cm-diameter foci of necrosis, sometimes coalescent. The prescapular lymph node was greatly enlarged and was similar to the mass on cut section. Fifteen to 20 round, firm, white, well-circumscribed nodules 1–3 cm in diameter infiltrated the lung parenchyma. On cut section, they were white, uniform, and well delineated. Tracheobronchial lymph nodes were moderately enlarged. A mild suppurative bronchopneumonia affected the apical region of the cranial lobes.
Several samples of the principal mass, prescapular and tracheobronchial lymph nodes, and pulmonary nodules were fixed in 10% neutral buffered formalin, embedded in paraffin, and stained with hematoxylin-phloxin-saffron for light microscopic examination. Immunohistochemical staining was performed using commercially available antibodies for identification of S-100 protein (Immunon, Pittsburg, PA), vimentin, cytokeratin, kappa and lambda light chains (Biogenex Laboratories, San Roman, CA), and CD3 (Dako Laboratories, Glostrup, Denmark).
The avidin-biotin-peroxidase method (Vectastain ABC kit, Vector Laboratories, Burlingam, CA) was used with 3-amino-9-ethylcarbomazole as the peroxidase substrate and Mayer's hematoxylin for counterstaining. Positive control was present on each slide. Formalin-fixed samples of the mass and pulmonary nodules were fixed in 5% gluteraldehyde, postfixed in osmium tetraoxide, and routinely processed for electron microscopic examination.
Histologically, the mass was poorly circumscribed, and it infiltrated the adjacent muscle and entrapped and slightly infiltrated the nerves of the brachial plexus (Fig. 1). It consisted of clusters and cords of round cells separated by wide strands of loose fibrovascular tissue. The cells were polygonal with scant, sometimes vacuolated, dark eosinophilic cytoplasm with poorly defined borders. Their variably sized and variably shaped nuclei were centrally located and occasionally contained one or two prominent dark nucleoli and irregularly clumped chromatin. Randomly distributed spindle-shaped neoplastic cells arranged in thin bundles were sometimes mixed with round cells. The mitotic rate was moderate, varying from one to three mitotic figures per high-power field. Scattered, variably sized coalescent foci of coagulative necrosis were randomly distributed throughout the mass. Individual cell necrosis and hemorrhage were also observed. The subcapsular sinus of the tracheobronchial lymph node was widened by neoplastic cells, causing partial destruction of the normal node architecture. Pulmonary nodules were encapsulated and compressed surrounding parenchyma, producing small foci of atelectasis. They were composed of neoplastic cells similar to those of the main mass.
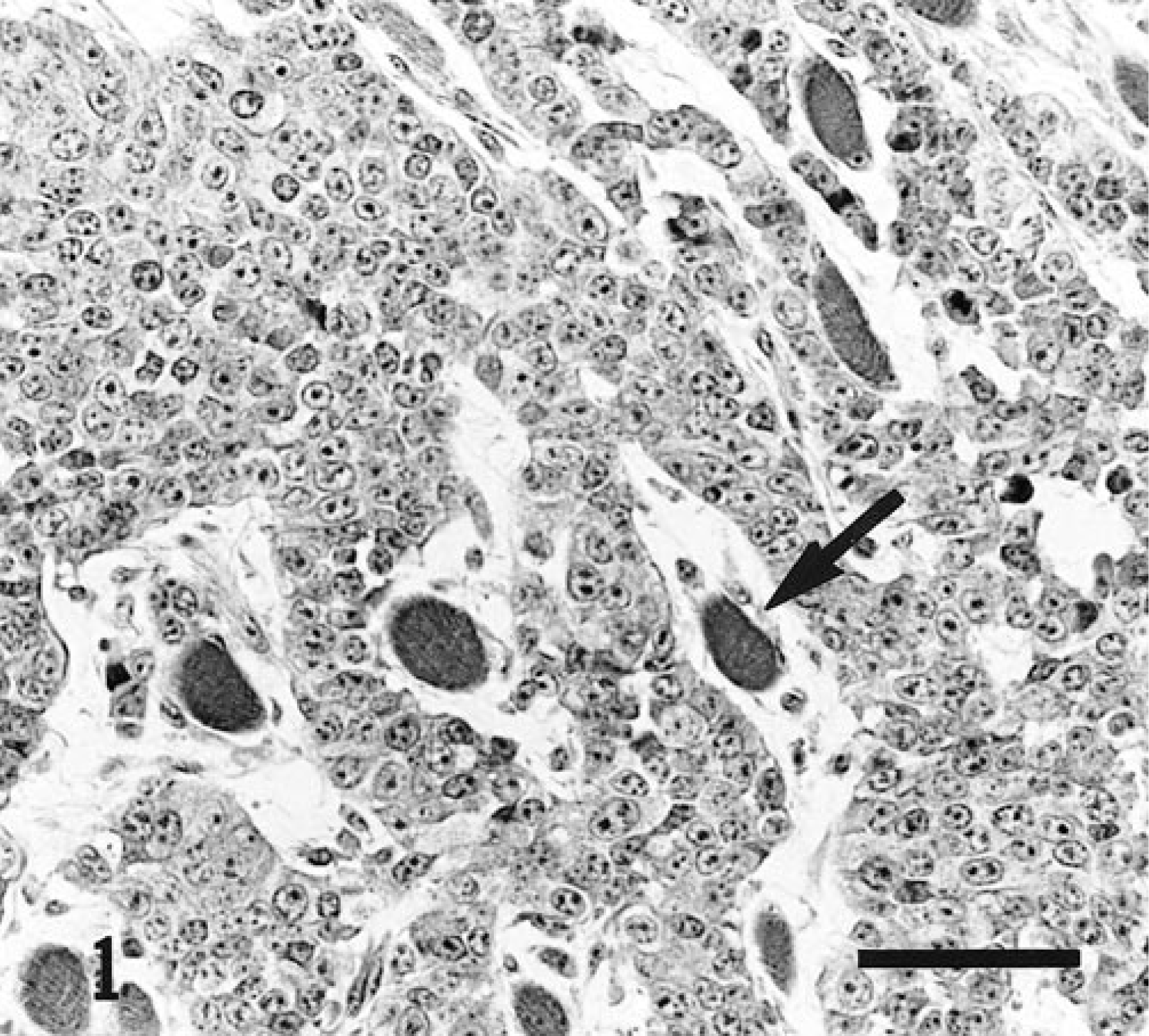
Mass; 8-month-old Ayrshire heifer. Clusters and cords of round and poorly differentiated cells separated by wide strands of loose fibrous tissue. Atrophic or slightly degenerated myocytes are entrapped in the mass (arrow), showing the invasive behavior of the tumor. Hematoxylin-phloxin-saffron. Bar = 50 µm.
The neoplastic cells stained positively for vimentin and cytokeratin (Fig. 2). S-100 protein, kappa and lambda light chains, and CD3 staining were negative.
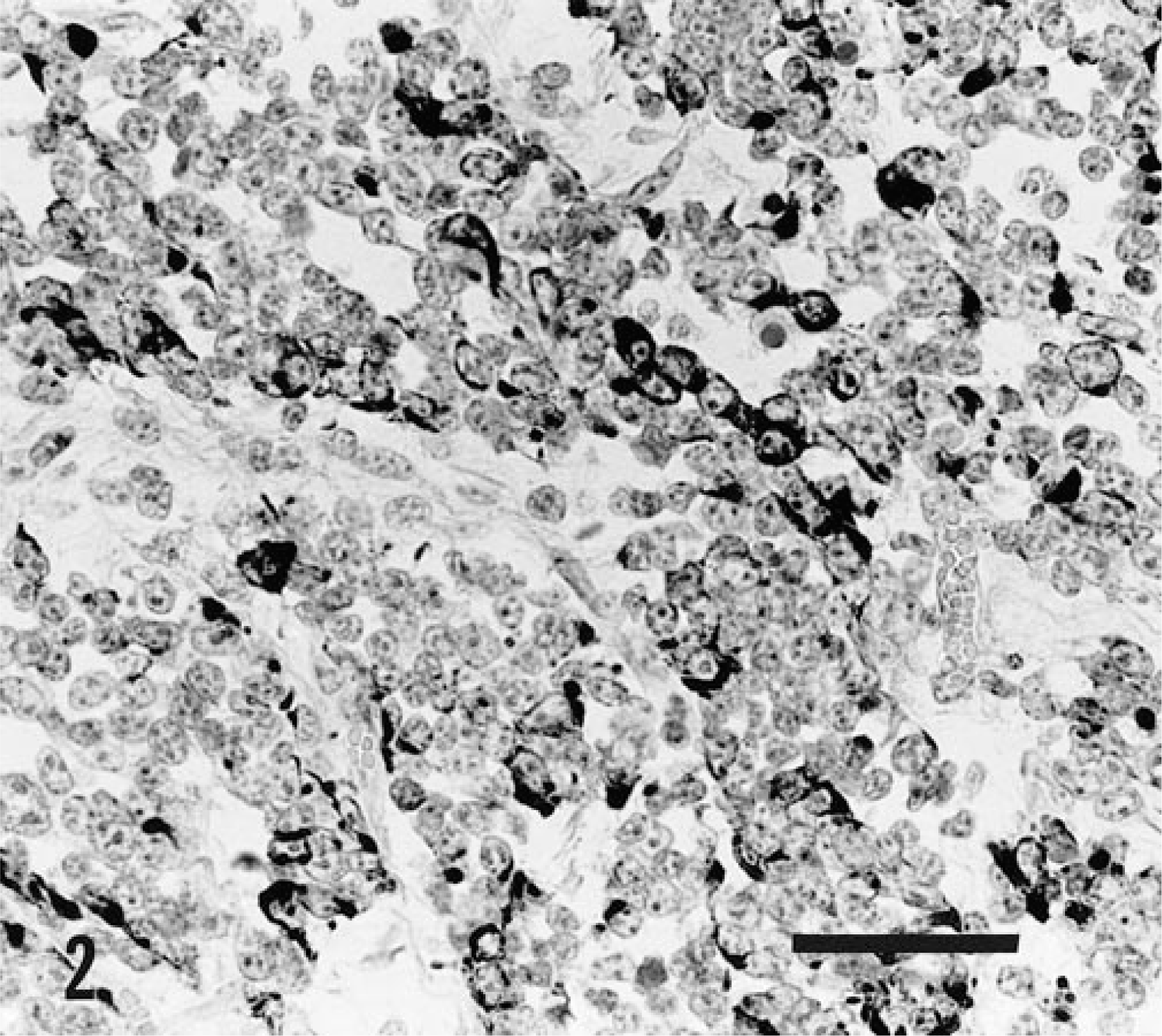
Mass; 8-month-old Ayrshire heifer. The neoplastic cells reacted positively on immunohistochemical staining with the cytokeratin antibody. Avidin-biotin-peroxidase method, Mayer's hematoxylin counterstain. Bar = 50 µm.
On electron microscopic examination, the neoplastic cells were mostly polygonal, forming small clusters and showed marked anisokaryosis (Fig. 3). A few scattered, spindle-shaped cells were sometimes surrounded by a flocculent matrix or a few collagen fibrils. The cytoplasm of the cells contained few organelles, principally rough and smooth endoplasmic reticulum and mitochondrias. Occasionally, short and thin microvillous projections of the cytoplasmic membrane were observed.
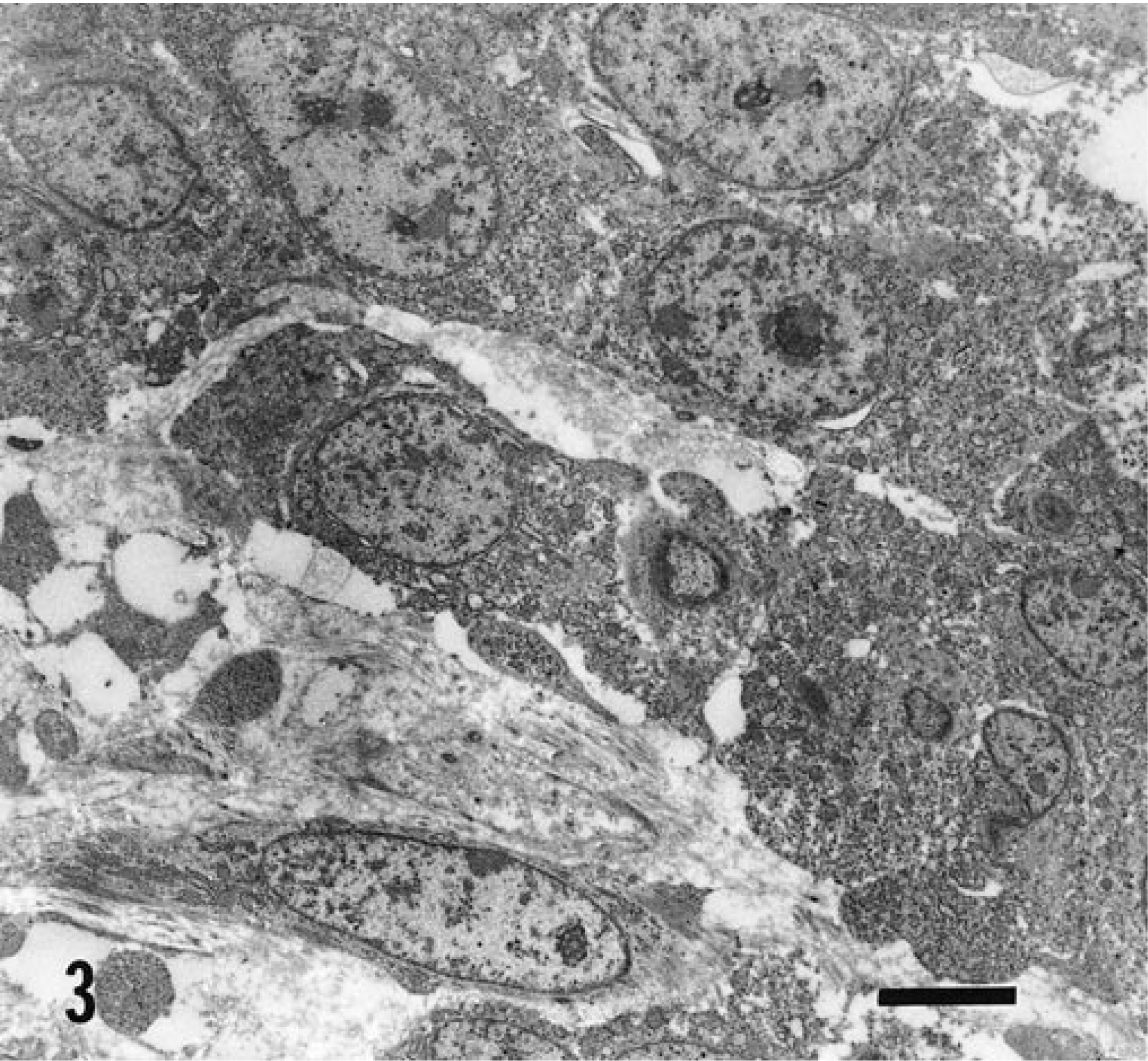
Mass; 8-month-old Ayrshire heifer. Clusters of polygonal neoplastic cells with marked anisokaryosis; some scattered spindle-shaped cells (lower left) are present. Uranyl acetate and lead citrate stain. Bar = 4 µm.
In view of the microscopic and electron microscopic examination and of the immunoperoxidase results, a synovial sarcoma was diagnosed. Synovial sarcoma can take on various morphologic appearance on light microscopic examination, which can occasionally make it challenging to identify. It can be confused with other soft tissue sarcomas, or, as is this case, the poor differentiation of the neoplastic cells can make it histologically indistinguishable from other small, blue, round cell tumors, including primitive neuroectodermal tumor, malignant nerve sheath tumor, and lymphoma. 2 In these cases, immunohistochemistry and electron microscopy can be useful to determine the tumor cells origin and phenotype. 1–2
Despite cytologic features suggesting the high possibility of a lymphoma, this diagnosis was rapidly ruled out in view of the negative results of immunohistochemistry for both T- and B-cell markers. The strong positivity for cytokeratin and vimentin was suggestive of synovial sarcoma but did not completely exclude other tumors. Immunophenotypic overlap due to partial epithelial differentiation is often shown by neoplastic cells of primitive origin. Malignant peripheral nerve sheath tumor (MPNST) and primitive neuroectodermal tumor (PNET) were excluded by the negative results of immunohistochemistry with antibodies to S-100 protein. Indeed, in a report of eight human cases, immunohistochemistry on poorly differentiated synovial sarcoma has shown that more than 87% of these are negative for S-100 protein. 2 MPNSTs and PNETs generally do not express high molecular weight cytokeratin antigen, whereas S-100 protein expression has been reported in 40–80% and in up to 68% of these tumors, respectively. 2
Malignant mesothelioma can have a similar immunophenotype. However, the mass and the pulmonary metastases did not involve serosal surfaces. Furthermore, these two tumors may be differentiated ultrastructurally by the presence of numerous prominent microvilli on the surface of mesothelial cells, which were absent in this case. 5
Synovial sarcomas are more frequent in people than in animals. They are subdivided into four main histologic types: the biphasic (or classical) type, characterized by the presence of well-differentiated epithelial and mesenchymal components, which are more or less equally represented; the monophasic spindle cell type; the monophasic epithelial cell type; and the poorly differentiated synovial sarcoma. 5
In animals, as in people, the classic bimorphic synovial sarcoma infrequently poses a diagnostic problem. 1–2 In this case, the lack of differentiation of the neoplastic cells, the presence of both epithelial and mesenchymal characteristics on immunohistochemistry staining, and electron microscopic examination are features of the poorly differentiated synovial sarcoma subtype of humans. Local invasion, metastasis, and anaplasia of the tumor cells reflect the malignant nature of the neoplastic process.
The age of the heifer seems unusual for this type of tumor in an animal. In people, however, adolescents and young adults are more frequently affected. 5
In general, synovial sarcomas are located in the deep soft tissues, usually in the vicinity of joints. 3–5 They are believed to arise from a pluripotent primitive mesenchymal precursor cell outside the synovial membrane rather than from synovioblastic cells lining a synovial cavity. 5 , 6
Footnotes
Acknowledgements
We thank Jules Deslandes for technical assistance and Dr. Kelly Rewbotham, who referred the case.
