Abstract
Sarcoma samples from 33 dogs, 25 subcutaneous and 8 articular, were submitted for cytokeratin immunohistochemistry. Eight of the 25 subcutaneous sarcomas (32%) expressed cytokeratin in 1% to 50% of the neoplastic cells. Of the 7 articular sarcomas evaluated, 1 (14%) expressed cytokeratin in 10% of neoplastic cells. The Kaplan-Meier survival analysis showed that the mean overall survival of dogs with subcutaneous sarcomas (28.1 months [confidence interval [CI]:17.8, 38.4]) did not significantly differ from those with articular sarcomas (24.8 months [CI = 0.5, 29.0]). Overall survival of dogs with sarcomas (both locations combined) immunoreactive for cytokeratin (31.2 months [CI = 17.8, 44.6]) did not differ from those not immunoreactive for cytokeratin (22.0 months [CI = 8.4, 35.6]). Therefore, cytokeratin expression does not indicate synovial origin (P = .64) and neither sarcoma location (P = .76) nor cytokeratin expression (P = .53) affects patient overall survival in this small study. The use of cytokeratin immunohistochemistry is not helpful to determine synovial origin of sarcomas in dogs.
In many veterinary publications, the term synovial sarcoma has been used to denote a tumor arising from synoviocytes;2,4,7,8,13,15 however, this name is a misnomer 10 adopted from human medicine where it is used to denote a rare soft tissue sarcoma with partial epithelial differentiation that does not arise from synovium.12,14 This tumor in humans can occur in many locations, including viscera, and is diagnosed by identifying a specific chromosomal translocation; the cell of origin is unknown. 14 No similar tumor has been identified in animals.
Because the tumor called synovial sarcoma in humans is sometimes biphasic and the epithelial component can express cytokeratin, previous publications have erroneously suggested using cytokeratin to identify tumors of synovial origin in animals.4,8,10,13 Our contention is that cytokeratin expression by sarcomas does not indicate synovial or articular origin.
To collect cases of subcutaneous soft tissue sarcomas, the surgical pathology archives of the University of Tennessee College of Veterinary Medicine were searched for the terms “skin” and “soft tissue sarcoma.” Only cases from 2017 were examined to maximize follow-up information retrieval, by allowing enough time to evaluate overall survival while staying within 5 years of diagnosis, so that medical records would still be available. Cases without skin in the section were excluded in order to include internal positive control tissue for the cytokeratin immunoreactivity. The same surgical pathology archive was searched for the words “synovial” and “sarcoma.” As these cases are much less common, this search included the years 2016 through 2023. For this study, articular sarcomas were defined as mesenchymal tumors that were primarily within the joint capsule; histiocytic sarcomas and sarcomas with myxoid matrix were excluded. Submission forms were reviewed for age, breed, weight, sex, location, and overall survival time. Additional case history was obtained by calling the referring clinics. Treatment history and cause of death were not able to be retrieved.
Immunohistochemistry (IHC) was performed at the Kansas State University Veterinary Diagnostic Laboratory using a Leica Bond Rxm stainer. Pretreatment was 20 minutes in ethylenediaminetetraacetic acid (EDTA) pH 9.0 at 100°C. The primary antibody (mouse monoclonal AE1/AE3, Leica) was diluted 1:1 with Bone Primary Antibody Diluent (Leica) and incubated with the tissue for 15 minutes at ambient temperature. The slides were then treated with PowerVision Poly-AP Mouse IgG (Leica) for 25 minutes at ambient temperature. Positive reaction was detected with Refine Red Detection (Leica), and slides were counterstained with hematoxylin. For the subcutaneous sarcomas, slides with skin attached were used to provide internal cytokeratin positive control tissue. A small piece of haired skin was added to the articular sample slides by the laboratory performing the IHC to serve as a positive control for those samples. Surrounding mesenchymal tissues served as the negative control. Hematoxylin and eosin-stained archived slides and IHC slides were reviewed by the same board-certified pathologist (LEC) to ensure consistency. The percentage of neoplastic cells immunoreactive to cytokeratin antibodies was estimated by scanning the entire glass slide. The number of mitotic figures in 10 contiguous high-power fields (2.37 mm2) was counted, starting in the most mitotically active area. Histologic tumor-free distance was measured.
Fisher’s exact test was used to compare tumor location and cytokeratin presence. Overall survival outcomes were analyzed for tumor location, cytokeratin presence, and entirety of excision using Kaplan-Meier survival analyses with a log-rank (Mantel-Cox) test. The significance of cytokeratin presence was tested for cases with subcutaneous sarcomas, as well as with the full cohort, regardless of location. Cox regression was used to test for a relationship between mean overall survival times and number of mitoses. All data analyses were performed using SPSS 29.0 (IBM Corp, Armonk, New York) using an alpha of .05.
The 25 subcutaneous sarcomas were located on the limb (12/25), trunk (10/25), and head (2/25); the location of one was not provided. The histopathologic findings of the subcutaneous sarcomas were consistent with perivascular wall origin: interlacing bundles of spindle cells and whorls around vessels. 1 All sarcomas either extended to the surgical margins (incomplete excision) or were within 2 mm (complete but close excision). The articular sarcomas looked very similar but were within the joint capsule and were less likely to contain whorls (Fig. 1). The 8 articular sarcomas were located in the stifle (3/8), shoulder (2/8), elbow (1/8), tarsus (1/8), and interphalangeal joint (1/8). One of the 8 articular sarcomas was unable to be processed for IHC, but signalment and survival data were available, so that case was only included in those analyses. Of the 7 articular sarcomas immunoreactive for cytokeratin, 1 case was positive in approximately 10% of cells (Fig. 1e).
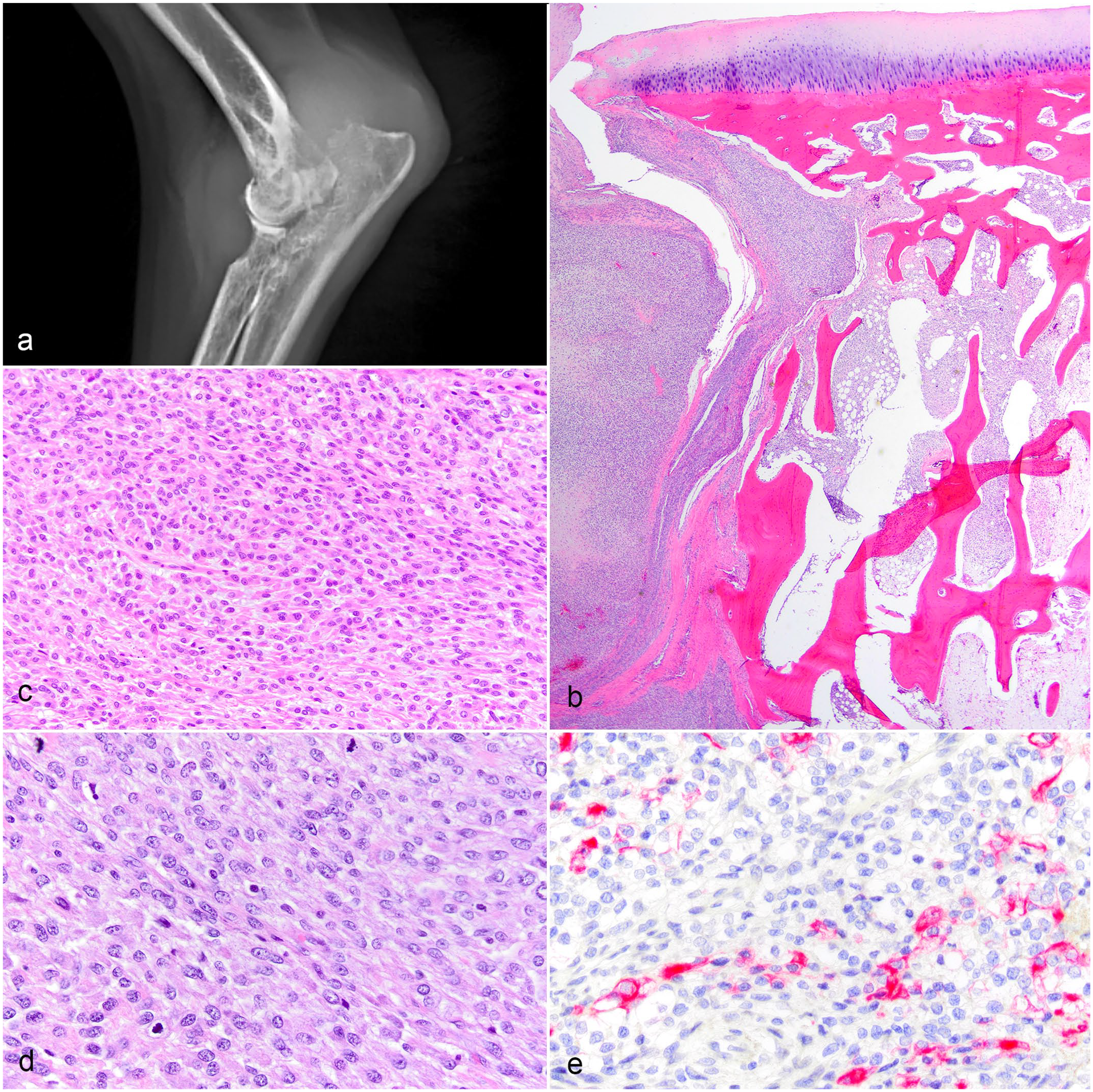
Articular sarcomas, dogs. (a) Radiograph of elbow joint with a soft tissue mass surrounding the joint and causing lysis in multiple bones. (b, c) Proximal radius, same case as (a). (b) Spindle cell mass inside the joint capsule and invading into bone. Hematoxylin and eosin (HE). (c) The mass is composed of spindle cells that are not histiocytic or myxomatous. HE. (d, e) Shoulder joint sarcoma. (d) The sarcoma is composed of spindle cells that are not histiocytic or myxomatous. HE. (e) Cytokeratin expression is present in 10% of cells. Pancytokeratin AE1/AE3 immunohistochemistry.
Eight of the 25 subcutaneous soft tissue sarcomas (32%) expressed cytokeratin in approximately 1% to 50% of the neoplastic cells (Fig. 2). The cytokeratin-expressing spindle cells were scattered to patchy in distribution. The cells that expressed cytokeratin were identical to those that did not; spindle shaped with no epithelioid features. The expression was diffusely cytoplasmic. The overlying epidermis and adnexal epithelium expressed cytokeratin in all subcutaneous sarcoma samples (internal positive control). There was no statistical difference (P = .64) between the percentage of subcutaneous sarcomas that were immunoreactive for cytokeratin (8/25, 32%) and the percentage of articular sarcomas that were immunoreactive for cytokeratin (1/7, 14%).
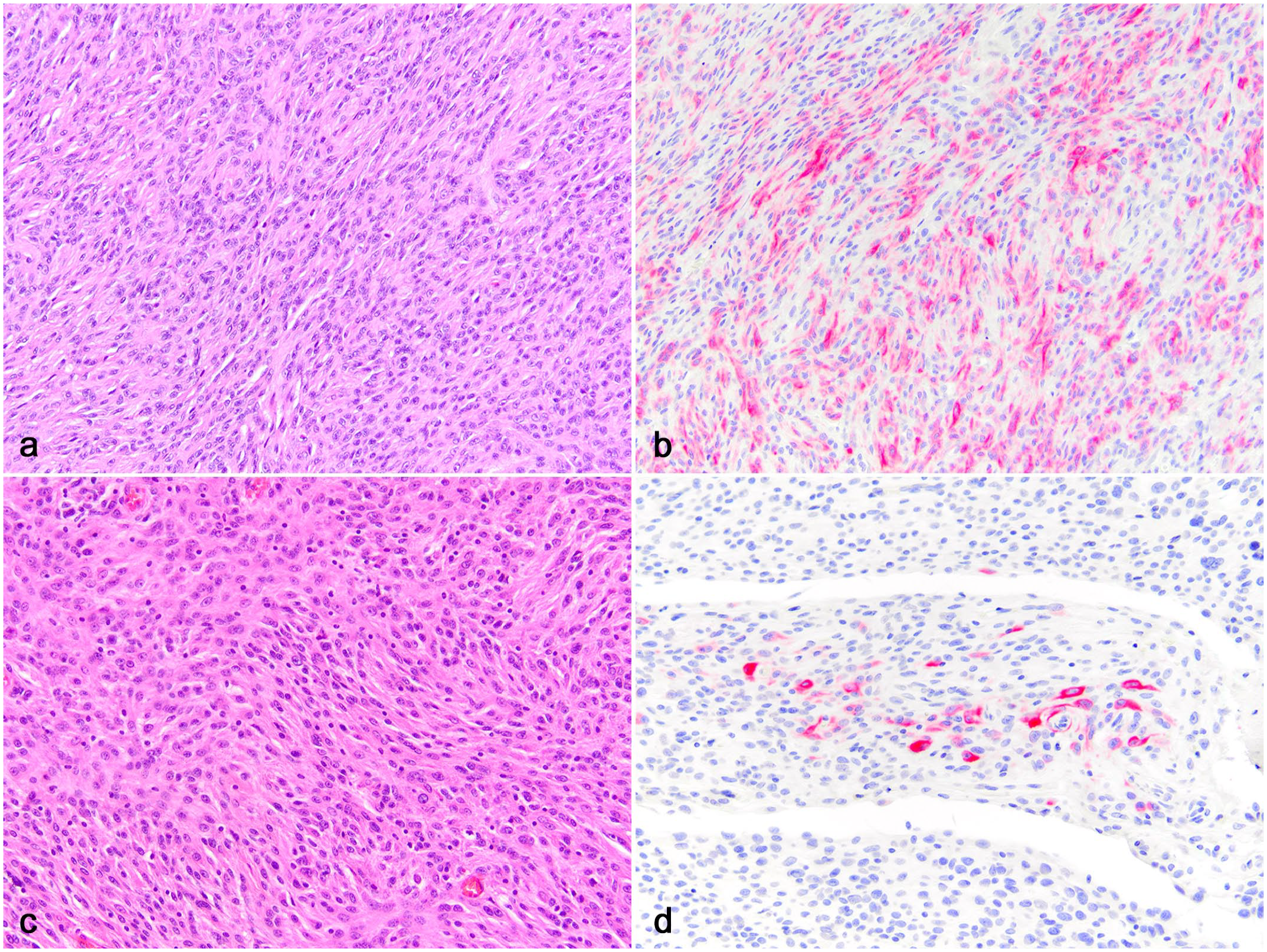
Subcutaneous sarcomas, dogs. (a, b) Soft tissue sarcoma. (a) The sarcoma is composed of spindle cells. Hematoxylin and eosin (HE). (b) 50% of the spindle cells express cytokeratin. Pancytokeratin AE1/AE3 immunohistochemistry (IHC).(c, d) Soft tissue sarcoma. (c) The sarcoma is composed of spindle cells. HE. (d) 1% of spindle cells express cytokeratin. Pancytokeratin AE1/AE3 IHC.
Signalment and overall survival data were obtained on 19 of the 25 subcutaneous sarcomas and all 8 of the articular sarcomas. There was no statistical difference (P = .70) between the mean overall survival time of the cases with subcutaneous sarcomas (28.6 months [confidence interval [CI]:18.4, 38.7]) and cases with articular sarcomas (24.8 months [CI = 0.5, 49.0]). Similarly, there was no statistical difference (P = .63) between the mean overall survival time of the cases of subcutaneous sarcomas that were immunoreactive for cytokeratin (31.2 months [CI = 17.8, 44.6]), and those that were not (23.4 months [CI = 10.7, 36.0]). There was no statistical difference (P = .49) in mean overall survival time between the cases (both locations combined) that were immunoreactive for cytokeratin (20.9 months [CI = 9.1, 32.8]) and those that were not (29.0 months [CI = 17.1, 40.8]). There was no statistical difference (P = .14) in mean overall survival time between the cases with incomplete surgical excision (34.8 months [CI = 23.1, 46.6]) and those with close (less than 2 mm) margins (15.3 months [CI = 7.8, 22.9]). The soft tissue sarcomas had 0 to 18 (average 5.9) mitotic figures per 2.37 mm2. The articular sarcomas had 0 to 48 (average 17.5) mitotic figures per 2.37 mm2. There was no significant relationship between the number of mitoses and overall survival time (P = .71, odds ratio = 1.0 [CI = 0.96, 1.1]).
There were no breed or sex predispositions in either the subcutaneous or articular sarcoma categories. The size of dogs with subcutaneous sarcomas averaged 24.6 kg (range = 4.8-53.2 kg) and the size of dogs with articular sarcomas averaged 15.9 kg (range = 2.6-28.5 kg). No breed was overrepresented in the expression of cytokeratin in their sarcomas.
Overall survival times were over 2 years for both sarcoma locations. Not enough information regarding local recurrence or metastases was available to be evaluated statistically, but metastasis does not always affect survival time in subcutaneous sarcomas. 5 All excisions were either incomplete or close, but margin status did not affect overall survival in this study. The number of mitotic figures in 2.37 mm2 also did not affect overall survival in this study, although it has in other studies. 5 The sarcomas in this study were not graded, because only 1 slide was examined, making the percentage of tumor necrosis unreliable.
One limitation of this study was that treatment history was not available. Therefore, we were unable to determine how different treatments may have affected the results.
Cytokeratin and vimentin are common markers used to identify cells of epithelial and mesenchymal origin, respectively. 9 Most neoplasms express one or the other, but some may express both, such as renal cell tumors, mesotheliomas, chordomas, and meningiomas. 9 Scattered cytokeratin expression in 5% to 50% of neoplastic cells has been reported in many types of human mesenchymal tumors, including fibrosarcomas, rhabdomyosarcomas, leiomyomas, leiomyosarcomas, gastrointestinal stromal tumors, liposarcomas, malignant peripheral nerve sheath tumors, and primitive neuroectodermal tumors.9,11 Cytokeratin expression was also previously documented in 2 of 17 canine soft tissue sarcomas. 3
The cell of origin of the subcutaneous soft tissue sarcomas with whorls is thought to be nonendothelial perivascular wall cells. 1 The cell of origin of articular (nonhistiocytic and nonmyxomatous) sarcomas is unknown, and they likely represent a heterogeneous group of tumors. The pathogenesis of cytokeratin expression by mesenchymal tumors is unknown.
In a review of 35 synovial tumors in dogs, 11 were neither histiocytic nor myxomatous. Of those 11, 5 (45%) expressed cytokeratin, 4 which is a higher percentage than in this study. The low overall number of cases in each study, especially this one, may explain the discrepancy. In that review, 4 cytokeratin expression was erroneously used to identify sarcomas as synovial origin. Human synovial sarcomas are sometimes biphasic and the epithelioid component can express cytokeratin, but those tumors are known to be of nonsynovial origin.6,12,14
In summary, we have identified that both subcutaneous (perivascular wall type) and articular sarcomas can express cytokeratin. Cytokeratin expression does not indicate synovial origin and was not correlated to overall survival in articular or subcutaneous perivascular wall sarcomas in this small study. The term “synovial sarcoma” should not be used in veterinary medicine, because it indicates a nonsynovial sarcoma in humans that has not been identified in animals.
Footnotes
Acknowledgements
We thank the participating referral clinics for providing follow-up information, as well as the UTCVM Histology Department for slide preparations.
Author Contributions
A.C.W. recorded case history, collected follow-up information, and prepared the manuscript. L.E.C. designed the study, selected the cases, reviewed the histopathology on each case, and revised the manuscript. C.S. and R.B. performed the statistical analysis of data.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the University of Tennessee College of Veterinary Medicine Faculty Educational Advancement and Research Fund.
