Abstract
A 12-year-old male Boxer dog presented with a 5 X 5 X 7-cm partially encapsulated mass in the right mandibular salivary gland. Histologically, the mass was composed of neoplastic epithelial and mesenchymal cells. The mesenchymal component consisted of two cell populations arranged in different patterns: coalescing nodules of neoplastic mononuclear cells with rare osteoid and numerous osteoclastlike giant cells; and sheets of neoplastic spindle cells intermingled with neoplastic epithelial cells and containing osteoid and well-formed bone trabeculae lined by osteoblasts and few osteoclastlike giant cells. On the basis of these histological features, two malignant salivary tumors were diagnosed: a malignant fibrous histiocytoma (giant cell type) and a malignant mixed tumor. Immunohistochemical studies demonstrated keratin 5 and 8 expression by the neoplastic epithelial cells, indicating a probable salivary ductal origin, and vimentin expression by all mesenchymal elements, suggesting a fibroblastic line of differentiation.
Keywords
Primary neoplasms of the major salivary glands are not common in domestic animals 4 , 6 and are considered rare in dogs. 5 The majority of salivary gland tumors are carcinomas, 12 and mixed tumors are rare in domestic animals. 6 , 12 However, tumors with both adenocarcinomatous and sarcomatous components have been described in dogs, 6 cats, 1 horses, 6 and cows. 4 In contrast, mixed tumors are the most common salivary gland neoplasms in humans. 9 Likewise, mesenchymal tumors, which comprise 50% of salivary gland tumors in children, 4 are rare in dogs. 12 Neoplasms histologically similar to malignant fibrous histiocytoma (MFH; giant cell type) have been reported in human salivary glands either in a pure form 7 or admixed with an epithelial malignancy, 13 but to the authors' knowledge, this type of salivary gland tumor has never been reported in domestic animals.
A 12-year-old male Boxer dog was presented to a private veterinary clinic from León (Spain) with an enlarging subcutaneous mass in the right submandibular region that had been noticed 4 weeks before. Radiographic examination of the head and neck showed no abnormalities of the mandibles and revealed multiple bone densities in the mass. The clinician decided to perform a surgical excision because of the mass's rapid growth. At surgery, a 5 × 5 × 7–cm mass was located within the mandibular salivary gland, showing adherences to the surrounding tissues without involving the mandibular bone. The biopsy was submitted to the anatomic pathology department of the Veterinary School of León for pathologic evaluation. On cut section, the tumor was circumscribed with an incomplete capsule and was surrounded by remmants of normal gland; it exhibited a lobulated appearance with multiple cysts. Dense portions were firm and gritty in some areas. The major lymph nodes of the head and neck appeared unaffected.
The mass was fixed in 10% neutral-buffered formalin, and some samples were decalcified in formic acid solution. Paraffin-embedded tissues were sectioned at 3 µm and stained with hematoxylin and eosin (HE) and with the Alcian blue pH 2.6 periodic acid–Schiff method (Alcian blue–PAS). The standard avidin–biotin complex (ABC) immunoperoxidase method (Vector Laboratories, Burlingame, CA) was applied to additional sections of the tumor. The primary antibodies used were monoclonal mouse anti-vimentin (Sigma Chemical Co., St. Louis, MO), monoclonal mouse anti-keratin wide-spectrum screening (Zymed Laboratories, Inc., San Francisco, CA), rabbit polyclonal anti-keratin 5 (BAbCO, Richmond, CA), monoclonal mouse anti-keratin 8 (Becton Dickinson, Erembodegem, Belgium), rabbit polyclonal anti-lysozyme (Concepta Biosystems SA, Barcelona, Spain), rabbit polyclonal anti–factor VIII–related antigen (Sigma Chemical Co.), and rabbit polyclonal anti-actin (Sigma Chemical Co.). The specificity of the immunoreactions was verified by staining negative and positive control tissue sections.
Histologically, the mass involved most of the mandibular salivary gland and was composed of neoplastic epithelial and mesenchymal cells. Alcian blue–PAS stained normal mucous gland but did not stain the tumor cells. The predominant cell component throughout the tumor was mesenchymal and consisted of two cell populations arranged in different patterns. The first was characterized by neoplastic mononuclear cells arranged in coalescing nodules with rare osteoid (Fig. 1) and numerous osteoclastlike giant cells unassociated with the osteoid (Fig. 2). These neoplastic cells were oval and showed features of malignancy, such as pleomorphic nuclei and frequent mitotic figures, some of them atypical (Fig. 2). The histologic features of this mesenchymal component were consistent with a diagnosis of a MFH (giant cell type). The second mesenchymal component consisted of sheets of spindle cells admixed with frequent osteoid and well-formed bone trabeculae lined by osteoid-producing osteoblasts and few osteoclasts. In some areas, the spindle cells were intermingled with neoplastic epithelial cells (Fig. 3), which formed ductlike structures with focal squamous metaplasia (adenocarcinomatous growth pattern) or were solidly packed in sheets and nests without a structural arrangement (solid growth pattern). The presence of bone trabeculae among and near neoplastic epithelial cells justified a diagnosis of malignant mixed tumor of salivary gland.
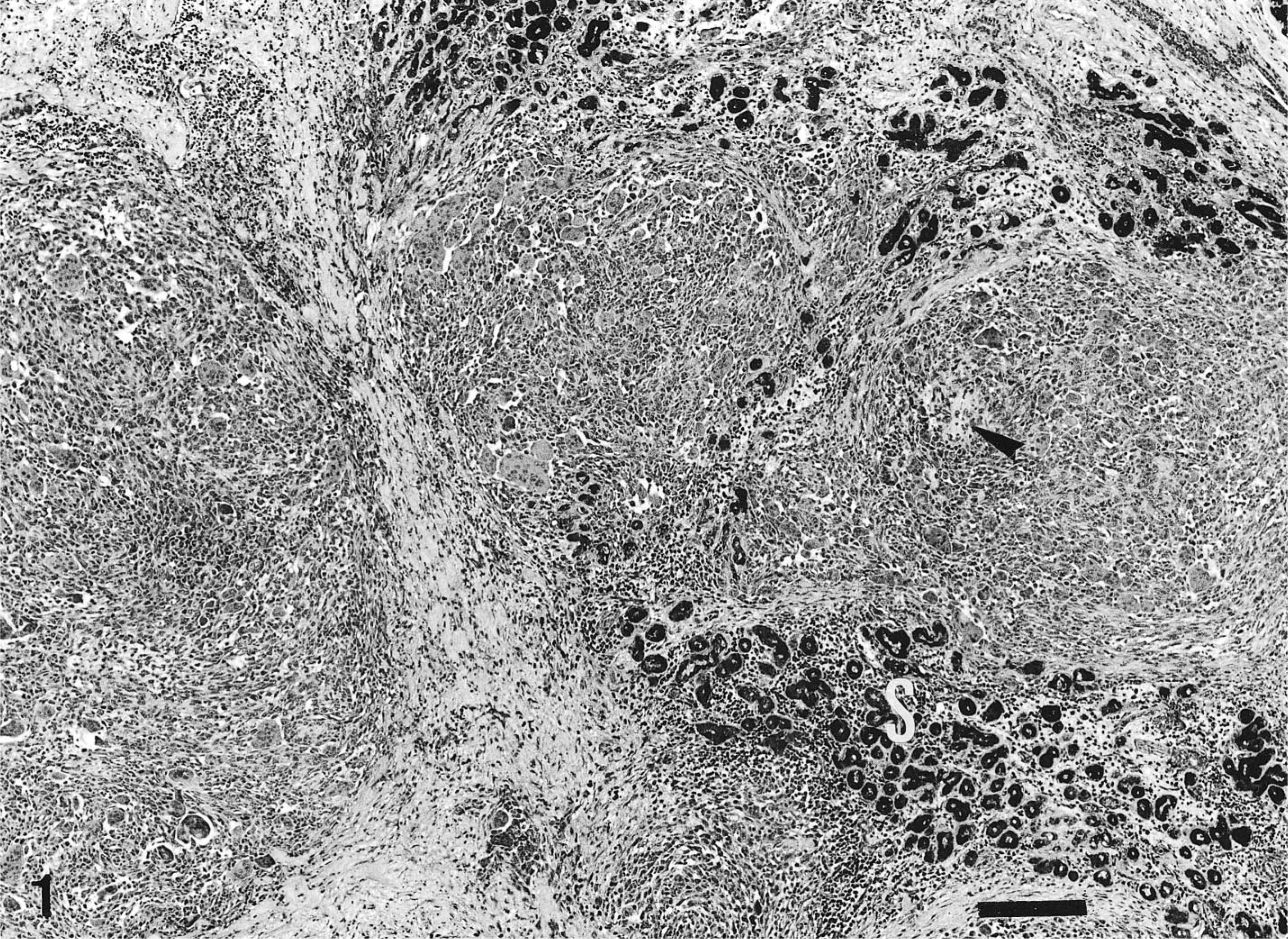
Mandibular salivary gland; dog. MFH (giant cell type) involving the salivary gland (S) and showing a multinodular growth pattern, with paucity of osteoid (arrowhead). Blue Alcian–PAS. Bar = 220 µm.
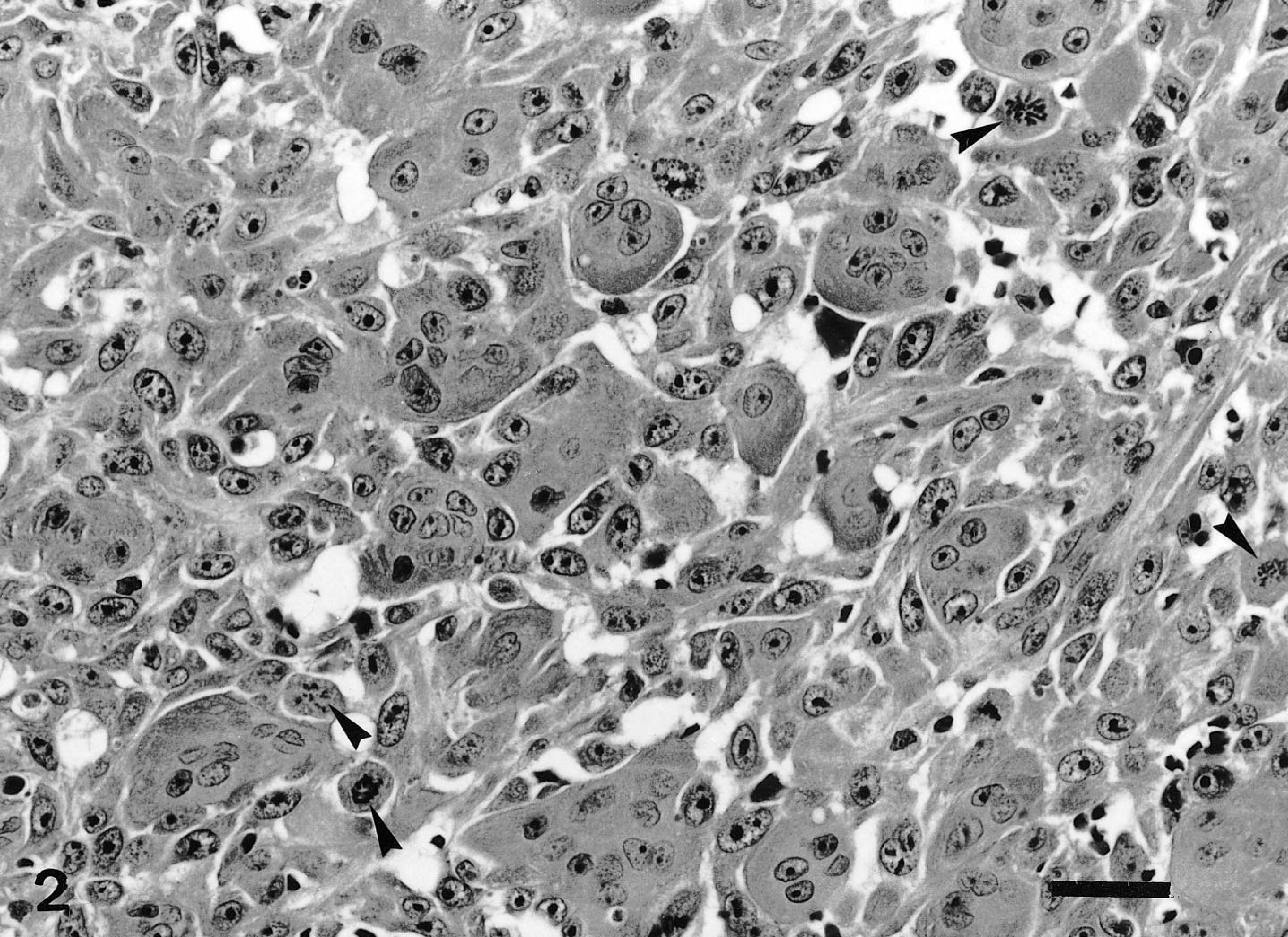
Mandibular salivary gland; dog. MFH (giant cell type) showing numerous osteoclastlike multinucleated giant cells among pleomorphic mononuclear cells. Note the frequent atypical mitoses (arrowheads). HE. Bar = 30 µm.
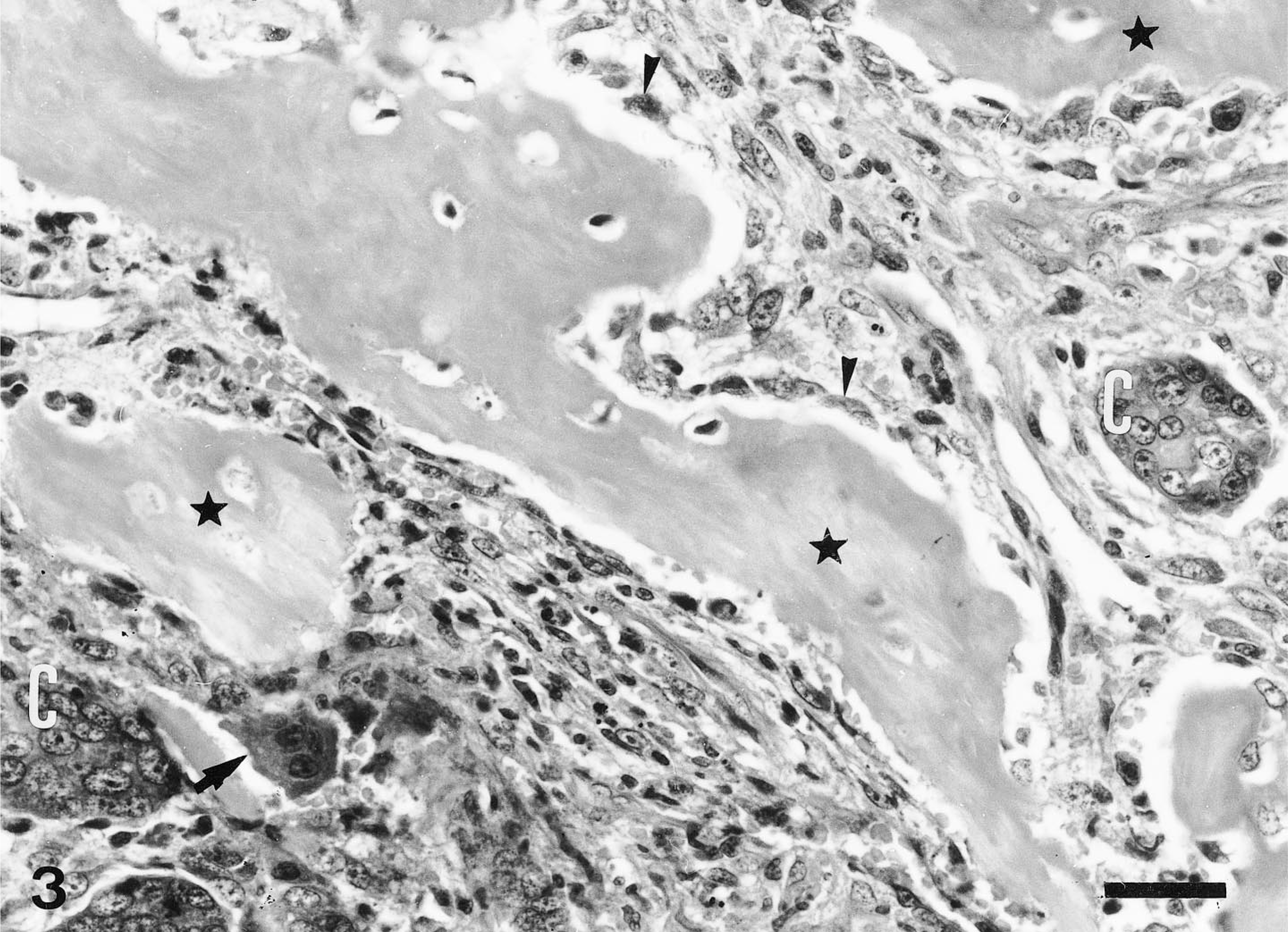
Mandibular salivary gland; dog. Islands of neoplastic epithelial cells (C) intermingled with eroded bone trabeculae (∗) lined by osteoblasts (arrowheads) and osteoclasts (arrow). HE. Bar = 30 µm.
Both neoplastic mononuclear and osteoclastlike giant cells of the MFH (giant cell type), as well as the mesenchymal portion of the malignant mixed tumor, were only immunoreactive for vimentin. Carcinoma cells were positive for keratins (K), showing K5 or K8 immunolabeling, but gave no reaction with the other antibodies used. Lysozyme expression was restricted to scattered neutrophils and macrophages, factor VIII–related antigen expression to endothelial cells of the well-formed vessels, and actin expression to the smooth muscle of blood vessels. The residual nonneoplastic salivary gland tissue showed K5-positive basal cells and K8-positive luminal cells in normal ducts and positivity for actin and K5 in myoepithelial cells.
On the basis of the most prominent histological features, two different malignant tumors were diagnosed in this salivary gland: MFH (giant cell type) and malignant mixed tumor. Because of the lack of primary tumor of bone in this case, the differential diagnosis of MFH (giant cell type) included mainly the extraskeletal osteosarcoma, 8 which has been described in the human 7 and canine 12 salivary gland. The main histological features that led us to the diagnosis of MFH (giant cell type) in this case (versus extraskeletal osteosarcoma) were the multinodular growth pattern and the paucity of osteoid formation. 3 Likewise, the presence of numerous osteoclastlike giant cells without relation to the scanty osteoid was a useful diagnostic feature in the diagnosis. Another important difference was the positive actin reactivity described in canine spleen 10 and salivary gland 12 extraskeletal osteosarcoma, in contrast to the negative actin reactivity in our case. The diagnosis of malignant mixed tumor was based on the presence of neoplastic epithelial cells intimately admixed with a mesenchymal component in some areas, as has been described in dogs and cats. 6
The origin of the neoplastic cells of MFH (giant cell type) in the salivary gland is unknown. In human pancreas, this tumor is positive for vimentin, supporting a fibroblastic line of differentiation. 11 Likewise, positive vimentin reactivity has been demonstrated in the MFH (giant cell type) component of human salivary gland carcinosarcomas. 13 In the mass we studied, the immunolabeling of neoplastic cells with vimentin, but not with the other antibodies used, supports this fibroblastic origin. Immunohistochemical results also suggested a mesenchymal differentiation for the giant cells. With regard to the origin of the mesenchymal component from mixed salivary gland tumor, two cell types have been cited: myoepithelial cells 2 or undifferentiated mesenchymal cells. 13 In the mass we studied, the positive vimentin reactivity, as well as the lack of the expression of K5 and actin (two well-known myoepithelial markers), supported an undifferentiated mesenchymal origin. The conservation of salivary duct–keratin immunolabeling in the carcinoma cells suggested a ductal origin, as previously proposed by other authors, 13 in contrast to a myoepithelial origin. 2
Footnotes
Acknowledgements
We thank Dr. J. F. García-Marín, Dr. A. Escudero-Diez, and Dr. J. Espinosa-Alvarez for invaluable help and scientific advice.
