Abstract
Malignant mixed Müllerian tumor (MMMT) is a rare neoplasm of the female genital tract. We report a case of bilateral ovarian MMMT in a 10-year-old female dog. Ovaries were only moderately enlarged with a papillary surface and firm nodules. Multiple metastases were observed in the abdominal cavity and pulmonary parenchyma. Histologically, both ovaries had intermingled carcinomatous and sarcomatous components with cartilage and bone. Metastatic lesions were not mixed. The peritoneal metastases were carcinomatous; pulmonary metastases were sarcomatous. Carcinomatous elements of the MMMT were immunohistochemically positive for cytokeratin (CK) AE1/AE3, CK7, and vimentin and estrogen receptors. Conversely, the sarcomatous cells were positive for vimentin, but negative for CKs. Chondrocytes also expressed S-100 protein. On the basis of similarities to human ovarian MMMT, the diagnosis was heterologous malignant mixed Müllerian tumor of the ovary.
Primary ovarian neoplasms are classified as epithelial, sex cord–stromal, germ cell, and mesenchymal tumors. 9 Ovarian adenocarcinomas are commonly encountered in aged bitches and frequently involve both ovaries. 15 In humans, ovarian adenocarcinoma is relatively common, but mixed tumors, such as carcinosarcomas (also termed sarcomatoid carcinomas), malignant mixed mesodermal tumors, or malignant mixed Müllerian tumors (MMMTs), are rarely reported, representing less than 1–4% of all malignant ovarian tumors. 3 These neoplasms are generally highly aggressive and develop principally in postmenopausal women.
Histologically, MMMTs are composed of variably admixed malignant epithelial and nonepithelial elements and have been classified according to the nature of the mesenchymal tissue. Homologous MMMT contains malignant stromal elements native to the ovary, such as spindle cells; heterologous MMMT contains sarcomatous tissue not found in the normal ovary, such as bone or cartilage. 11 The epithelial component is usually serous, endometrioid, or undifferentiated carcinoma. 2 Few MMMTs have been described in the veterinary literature and mainly in the uterus of cats, rodents, and a rabbit. 5, 6, 8, 13, 14
We describe light microscopic and immunohistochemical findings in a canine heterologous ovarian MMMT. A 10-year-old mixed-breed bitch with several days of dyspnea, night cough, and hemoptysis was submitted to electrocardiographic and echocardiographic examination. Arrhythmia was attributed to the presence of a 10-cm-long polypoid mass in the left atrium, which prevented closure of the left atrioventricular valve. The dog died from left-sided cardiac failure, and a complete postmortem examination was performed.
At postmortem examination, the right ovary was 3.8 cm in diameter and the left, 1.8 cm. Both ovaries were mottled red to pale tan, nodular, and partially contained within the bursa. The ovarian surfaces were irregularly roughened with a papillary appearance (Fig. 1). The uterus and regional lymph nodes were uninvolved, but numerous, variably sized, firm nodular masses were scattered throughout the peritoneum and were attached to the omentum and to serosal surfaces of the intestine and urinary bladder. Nodules were also found in pulmonary parenchyma; a polypoid mass protruded into the lumen of a pulmonary vein. Samples from ovaries and other organs were fixed in 10% buffered formalin and embedded in paraffin. Sections were examined by light microscopy after HE staining. Immunohistochemistry was performed with antibodies raised against cytokeratin (CK; clone AE1/AE3; Dako, Glostrup Denmark), CK7 (clone OVTL 12/30; Dako), vimentin (clone V9; Cell Marque, Rocklin, CA), S-100 protein (Dako), α-smooth muscle actin (α-SMA clone 1A4; Dako), human desmin (clone D33; Dako), calponin (clone Calp1; Dako), Ki-67 (clone MIB-1; Dako), E-cadherin (clone NCH-38; Dako), and estrogen α and β receptor (rabbit polyclonal antibody; Zymed, San Francisco, CA).
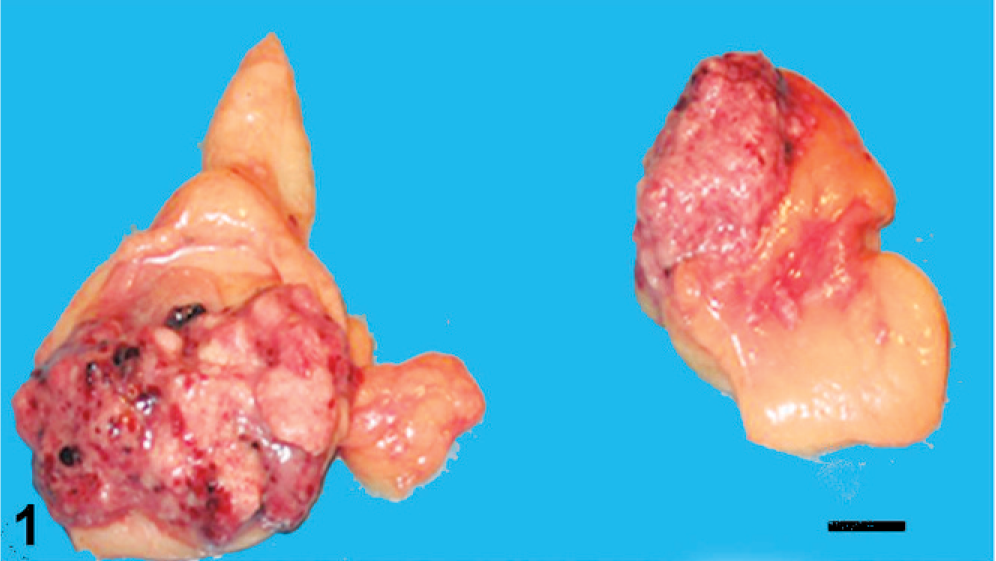
Ovaries; dog. Enlarged ovaries with roughened surface, multiple nodules and hemorrhagic areas. Bar = 1 cm.
Histologically, both ovaries were infiltrated by a neoplasm with both malignant epithelial and stromal components, mostly separated, but focally admixed (Fig. 2). Epithelial elements were arranged mainly in tubular and papillary structures supported by a fibrovascular stroma (Fig. 3). Papillae were lined by cuboidal to columnar cells with abundant pale eosinophilic cytoplasm, distinct cell margins, ovoid and vesicular nuclei, and prominent nucleoli. Mild to moderate nuclear and cellular pleomorphism and 5 mitotic figures per high-power field were detected. Psammoma bodies were scattered among the epithelial component. Stromal proliferation consisted of closely packed malignant polygonal cells with distinct cell margins and prominent nucleoli, embedded in a chondroid and osseous matrix (Fig. 4). Nuclear and cellular pleomorphism was moderate to severe with 4 mitotic figures per high-power field. The neoplasm was not clearly demarcated from adjacent nonneoplastic ovarian tissue.
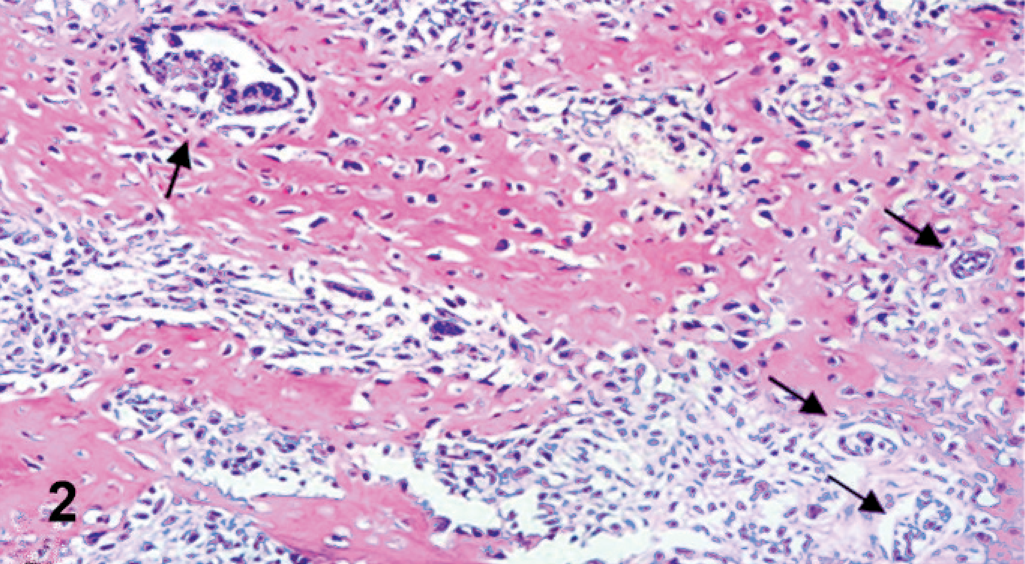
Ovary; dog. Sarcomatous components and carcinomatous structures (arrows) intimately admixed. HE.
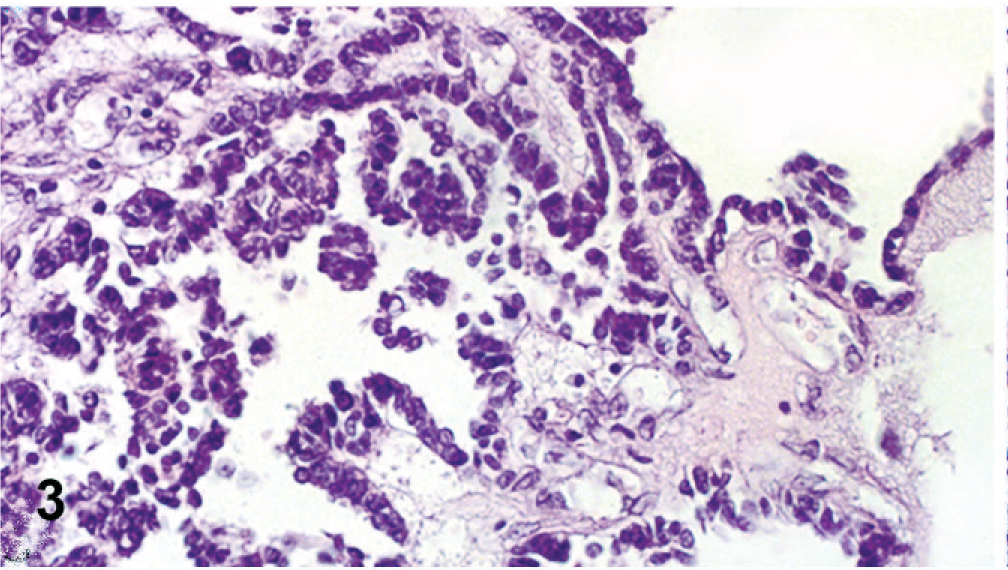
Ovary; dog. Detail of carcinomatous component with papillary structures supported by a fibrovascular stroma. HE.
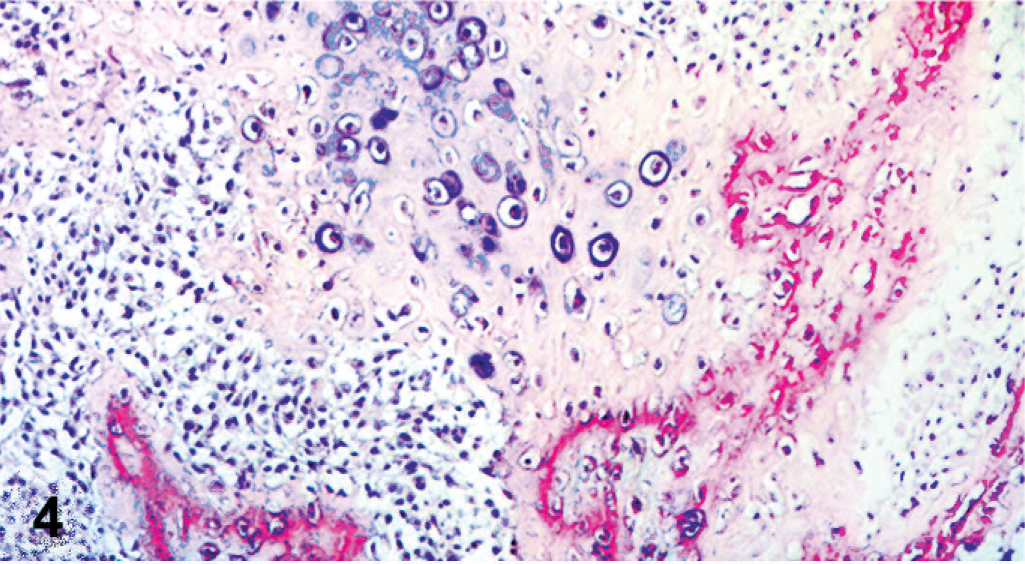
Ovary; dog. Detail of sarcomatous component composed of polygonal cells embedded in cartilage and bone lacunae. HE.
Peritoneal, serosal, and omental metastatic lesions were composed of the same malignant epithelial proliferation found in the ovaries. Conversely, pulmonary metastases and the mass in the pulmonary vein had only sarcomatous elements in a predominantly cartilaginous matrix. Immunohistochemically, both nonneoplastic and neoplastic ovarian surface epithelia had strong and diffuse reactivity for cytokeratin AE1/AE3 (>50% of cells) and strong and focal positive immunostaining for CK7 (<20% of cell positive) (Fig. 5). These cells were also diffusely and faintly positive for vimentin and estrogen receptors, but negative for α-SMA, calponin, and desmin. Malignant mesenchymal cells expressed vimentin strongly and diffusely, whereas cartilaginous elements also had strong immunoreactivity for S-100 protein. No cytokeratins (Fig. 6), α-SMA, calponin, or desmin was detected. Ki-67 reactivity was higher in carcinomatous than in the sarcomatous component. Additionally, E-cadherin membranous immunoreactivity was recognized in carcinomatous elements and, focally, in nonneoplastic ovarian surface epithelium, whereas the sarcomatous proliferation was negative. Peritoneal metastases were positive for cytokeratins, whereas pulmonary metastases and the mass in the pulmonary vein expressed vimentin and S-100.
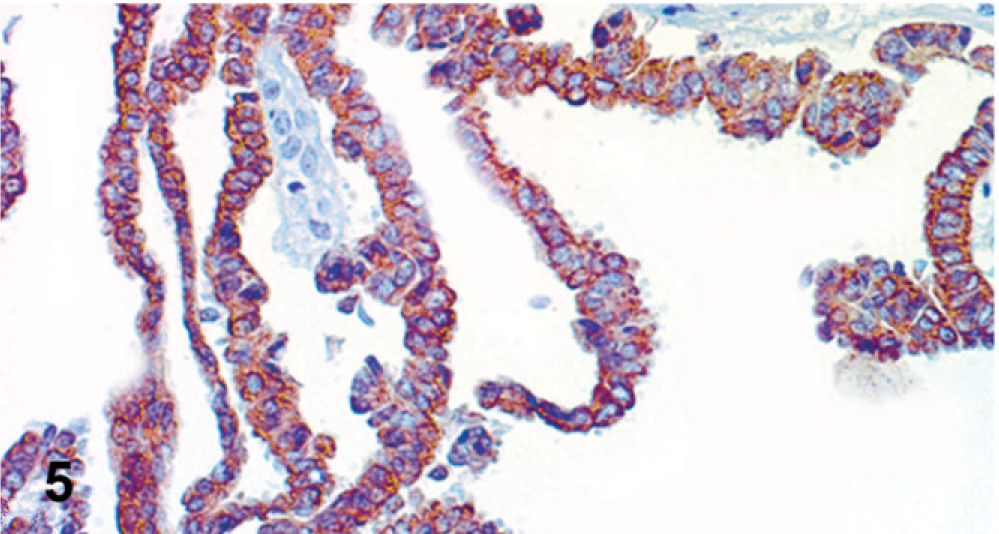
Ovary; dog. Antibody to cytokeratin 7 is strongly expressed in cytoplasm of carcinomatous cells. Streptavidin-biotin-peroxidase method, Harris' hematoxylin counterstain.
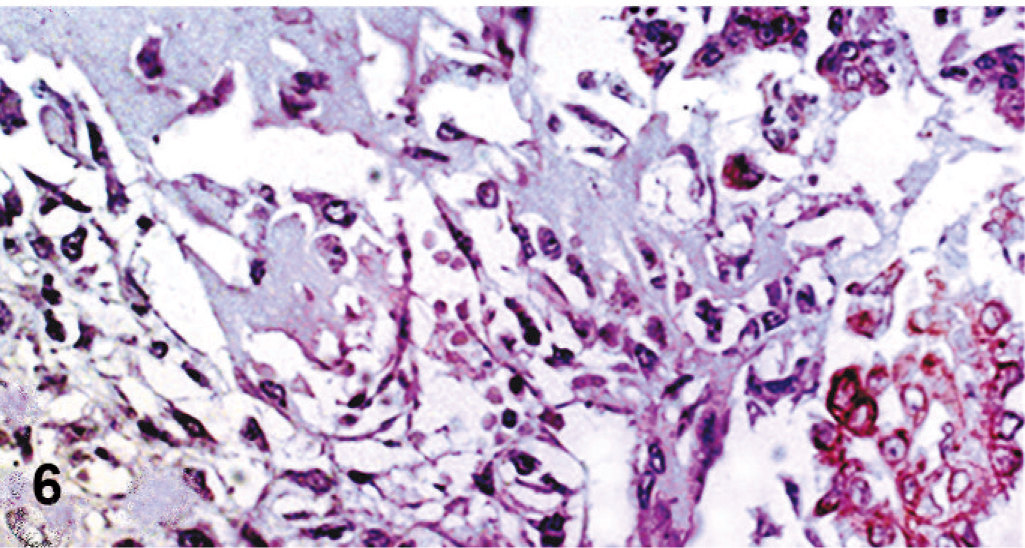
Ovary; dog. Cytokeratin (AE1/AE3) immunoreactivity is evident in malignant epithelial cells, but not in the sarcomatous component. Streptavidin-biotin-peroxidase method, Harris' hematoxylin counterstain.
In the veterinary literature, MMMTs are rarely described and only in the uterus. 5, 6, 8, 13, 14 Heterologous components, such as rhabdomyosarcomatous and cartilaginous differentiation, have been reported exclusively in rat uterine MMMTs. 8 In our case, the heterologous component included malignant osseous and cartilaginous matrices. Muscular differentiation was not detected, as evidenced by the absence of immunoreactivity for calponin, desmin or α-SMA. Uterine MMMT malignant epithelial components were described as poorly differentiated endometrioid adenocarcinoma in a cat 5 and a well-differentiated adenocarcinoma in a rabbit. 6
We report a bilateral solid primary ovarian neoplasm, classified histologically as a moderately differentiated serous papillary adenocarcinoma with heterologous differentiation as commonly seen in human ovarian MMMTs. 11 The differential diagnosis included malignant immature ovarian teratoma, another tumor with both epithelial and mesenchymal components. However, an immature teratoma should have a more embryonal appearance than MMMT, and in bitches, the ectodermal component of ovarian teratomas usually appears as hair-filled follicular cysts. Furthermore, the expression of estrogen receptors by the carcinomatous component of the tumor in this case is consistent with Müllerian origin and tends to rule out teratoma with malignant transformation.
Immunohistochemically, coexpression of cytokeratins and vimentin in neoplastic and nonneoplastic epithelia was consistent with the coelomic origin of this tumor. Peculiarly, in our case, ovarian surface epithelium expressed little or no E-cadherin. Detection of this marker in carcinomatous elements of human epithelial ovarian carcinoma suggested a functional role in the earliest stages of tumor development. 1 In addition, estrogen receptor expression in neoplastic ovarian epithelium might indicate involvement of steroid hormones in the pathogenesis of canine ovarian MMMT. Nevertheless, the tumor of this communication had highly aggressive behavior, with both carcinomatous peritoneal and sarcomatous pulmonary metastases, and a poor clinical outcome as in human ovarian MMMT. 2
The histogenesis of MMMTs is still controversial, and different theories have been proposed to explain their biphasic appearance. Recent evidence indicates that most MMMTs are monoclonal neoplasms and are in reality metaplastic carcinomas; however, a few carcinosarcomas are considered to be true collision tumors. 7, 10, 12 A true collision tumor cannot be ruled out in our case, with its mixture of carcinomatous and sarcomatous components in the ovaries but purely carcinomatous or sarcomatous nature in the metastases. 16 Although one third of human ovarian MMMTs is bilateral, 4 to the best of our knowledge, no studies have associated the biclonality of MMMT with its distribution. Further studies with genetic-molecular techniques are needed to elucidate canine ovarian carcinosarcoma histogenesis.
Footnotes
Acknowledgements
We are deeply grateful to Dr. R. Monaco at the Division of Anatomic Pathology—A.O.R.N. “A. Cardarelli”—Naples for his useful and critical suggestions.
