Abstract
Salivary gland diseases are well characterized in human medicine but are uncommonly reported in dogs. Herein we describe the clinical and pathologic features of 179 canine salivary gland biopsy submissions to the Athens Veterinary Diagnostic Laboratory from 2010 to 2018. The mean age of affected dogs was 8.5 y; no sex or breed predisposition was evident. The main reason for sample submission was regional swelling (107 cases; 59.7%). Extraoral (major) salivary glands were affected in 125 cases (69.8%), and oral (minor) salivary glands were affected in 43 cases (24% of cases). The location of the lesion (extraoral or oral) was not specified in 11 cases (6.1%). The diagnoses included nonspecific sialoadenitis (89 cases; 49.7%), normal salivary gland (42 cases; 23.4%), neoplasia (36 cases; 20.1%), salivary gland lipomatosis (7 cases; 3.9%), necrotizing sialometaplasia (4 cases; 2.2%), and traumatic hemorrhage (1 case; 0.5%). Most cases of sialoadenitis (63 cases), neoplasia (23 cases), and lipomatosis (5 cases), as well as all cases of necrotizing sialometaplasia and the only case of traumatic hemorrhage, affected extraoral glands. Most neoplasms (32 cases, 88.8% of the tumors) were epithelial and malignant, followed by round cell tumors (2 cases; 5.5%), a carcinosarcoma (1 case; 2.7%), and a tumor of undetermined histogenesis (1 case; 2.7%).
In human medicine, salivary gland disorders are associated with systemic disease (diabetes, stroke, AIDS, Alzheimer’s disease, Parkinson’s disease, among others), certain medications (such as chemotherapy), head and neck radiotherapy, local infections, sialolithiasis, sialocele and mucocele, and neoplasia. 23 Depending on the severity and clinical course, salivary gland dysfunction can cause xerostomia and consequent oral or systemic problems, including altered taste, dysphagia, halitosis, predisposition to aspiration pneumonia, and nutritional imbalances. 23 Human salivary gland neoplasms are classified into > 40 distinct subtypes based on morphology, which typically correlates well with clinical behavior, prognosis, and specific therapeutic protocols. 27
In veterinary medicine, there is a scarcity of studies characterizing the clinical and pathologic changes in salivary glands.16,26 Salivary gland diseases are described uncommonly and mainly in dogs and cats, with an overall prevalence of 0.3% of diagnostic case submissions.12,25,26 In dogs, the main salivary gland disorders include primary or secondary neoplasia,3,5 –7,9,11,13,16,24,26 followed by nonspecific sialoadenitis, sialocele, and necrotizing sialometaplasia.4,10,12,14,15,18,21,26 However, most of these descriptions are single case reports, and there have been only a few population studies assessing the prevalence of different salivary gland diseases in dogs.12,13,21,25,26 Herein we describe the clinical and pathologic features of 179 salivary gland tissue samples from dogs submitted to the biopsy service at the Athens Veterinary Diagnostic Laboratory (AVDL; Athens, GA) over a period of 8 y.
Our laboratory web-based archive system was searched for salivary gland biopsy cases submitted between 2010 and 2018 using the keyword “salivary gland”. Submission forms and biopsy reports from selected cases were assessed for patient signalment, clinical signs, and anatomic location of the sample (major extraoral salivary gland [i.e., mandibular, sublingual, parotid, and zygomatic] or minor oral salivary gland), as well as pathology diagnosis. Archived hematoxylin and eosin–stained slides were reviewed concomitantly by both authors and tissue changes were categorized as 1) non-pathologic, 2) inflammatory, 3) infectious, 4) neoplastic, or 5) other. Neoplasms were reviewed and diagnosed after a consensus was reached between the authors. All neoplasms were classified according to the World Health Organization Histological Classification of Tumors of the Alimentary System of Domestic Animals and the fifth edition of Tumors in Domestic Animals.11,19
We selected and included 179 cases in our study (Suppl. Table 1). Ninety-three samples (51.9%) were from male dogs (69 castrated, 24 intact) and 86 (48%) were from female dogs (73 spayed, 13 intact). The mean age of affected dogs was 8.5 y, with a range of 6 mo to 20 y. The most common affected breeds were mixed breed (17), Dachshund (16), Labrador Retriever (12), Shih Tzu (10), German Shepherd (8), Golden Retriever (8), and Chihuahua (7). The most common reason for sample submission was regional swelling (107 cases), which often raised suspicion of sialocele or neoplasia. Other less frequently reported clinical signs included halitosis (2 cases), gagging, and fight wound (1 case each). The clinical signs were not specified in the other cases. Extraoral (major) salivary glands were examined in 125 cases (69.8%); the most commonly affected glands were mandibular (52 cases), sublingual (14), parotid (11), and zygomatic salivary gland (1); the affected extraoral site was not specified in 47 cases. Oral (minor) salivary glands were evaluated in 43 occasions (24%); the main sites included the gum (13 cases), oropharynx (9), tongue (8), palate (7), cheek (1), and lip (1); the affected oral site was not specified in 4 cases. The location of the lesion (extraoral or oral) was not available in 11 cases (6.1%).
The most common diagnosis in the examined data set was nonspecific sialoadenitis (89 cases; 49.7%), which consisted of lymphoplasmacytic or neutrophilic inflammatory infiltrates that expanded the periglandular, interlobar, and interlobular spaces (Table 1). Inflammatory cells occasionally effaced the salivary gland acini and were admixed with necrotic cell debris or fibrosis. Cavitated areas with accumulation of mucoid secretory material were detected in 24 (26.9%) of these cases, which was consistent with a diagnosis of sialocele; no cause for inflammation was evident. No pathologic changes were detected in 42 (23.4%) of the cases.
Diagnoses and anatomic location of salivary gland conditions in 179 dogs between 2010 and 2018.
NS = specific gland not specified.
Neoplasia was diagnosed in 36 cases (20.1%); 32 of 36 (88.8%) of the neoplasms were of epithelial origin, consisting of 14 adenocarcinomas, 10 mucoepidermoid carcinomas, 7 acinic cell carcinomas, and 1 squamous cell carcinoma. These malignant epithelial neoplasms consisted of solid lobules of neoplastic cells that partially or completely effaced the salivary gland architecture.
Adenocarcinomas were poorly demarcated and confined to one or more salivary gland lobules. Tumors were composed of multiple groups of neoplastic cells forming solid lobules or small acini and ducts supported by a fine fibrovascular stroma. Neoplastic acini often contained faintly basophilic or eosinophilic secretory material (Fig. 1). Neoplastic cells had low-to-moderate pleomorphism with a moderate amount of polygonal-to-columnar, eosinophilic or less often vacuolated, mucin-rich cytoplasm (Figs. 2, 3). Nuclei were round with finely stippled chromatin and 1 or 2 nucleoli. There was low-to-moderate anisocytosis and anisokaryosis.
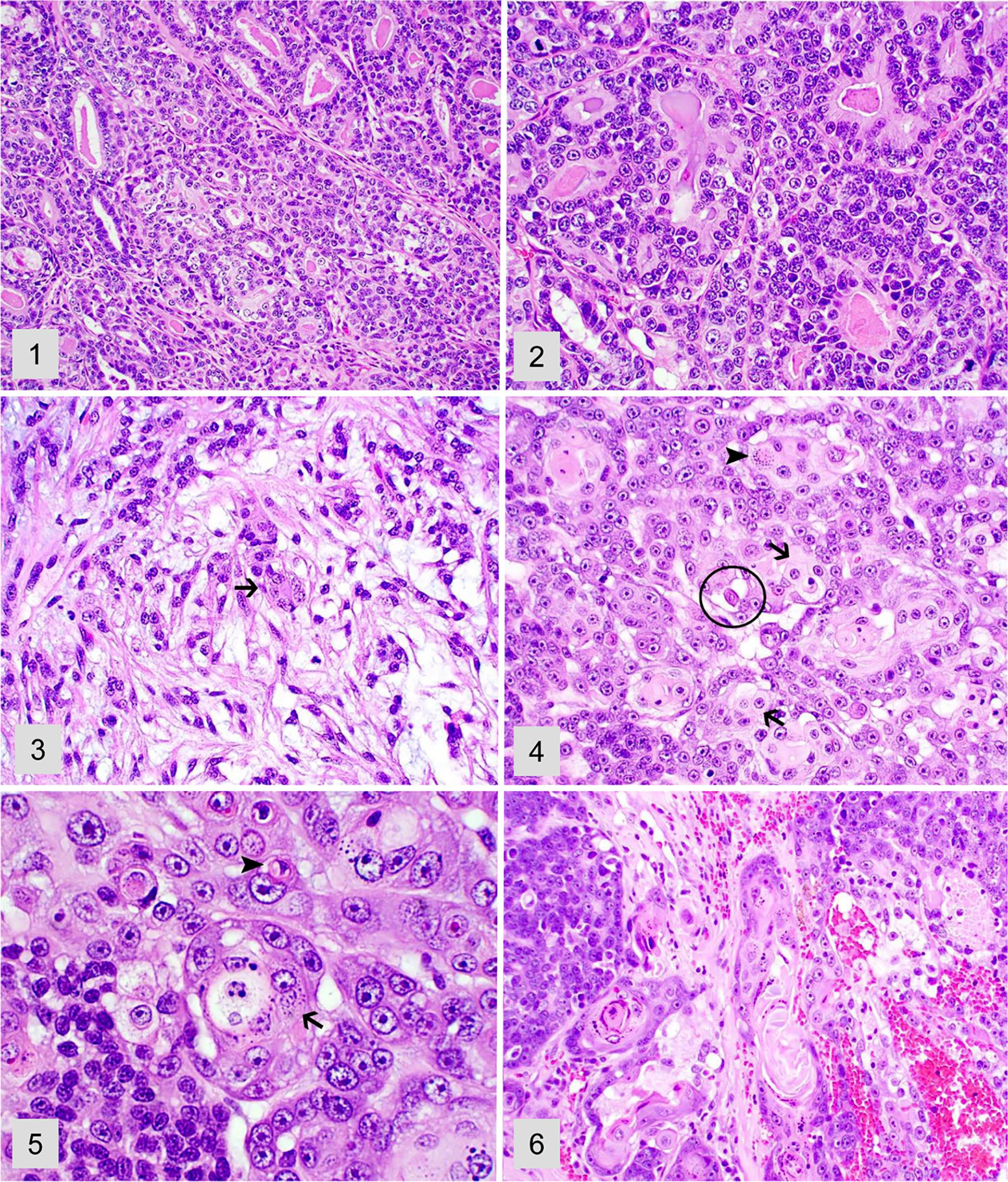
Various salivary gland neoplasms in dogs. H&E.
Mucoepidermoid carcinomas were poorly demarcated tumors and consisted of solid lobules of neoplastic cells supported by a moderate amount of fibrovascular and collagen stroma. Neoplastic cells had moderate pleomorphism and abundant, round-to-polygonal, finely vacuolated, mucin-rich cytoplasm with indistinct cell borders (Fig. 4). Scattered throughout were neoplastic cells with epidermoid morphology (present of cytoplasmic keratohyalin granules and occasional dyskeratosis) arranged in clusters of small nests (Fig. 5). Overt keratinization was rare but also occurred within these tumors (Fig. 6). Nuclei were round and had finely stippled chromatin with 1 or 2 nucleoli.
Acinic cell carcinomas were poorly demarcated and were invasive tumors that consisted of multiple distinct solid lobules of neoplastic cells supported by a moderate amount of fibrovascular and collagen stroma (Fig. 7). Neoplastic cells had moderate pleomorphism and abundant, round-to-polygonal, finely vacuolated cytoplasm with distinct cell borders (Fig. 8). Nuclei were round with finely stippled to coarse chromatin and one nucleolus. There was low anisocytosis and anisokaryosis.
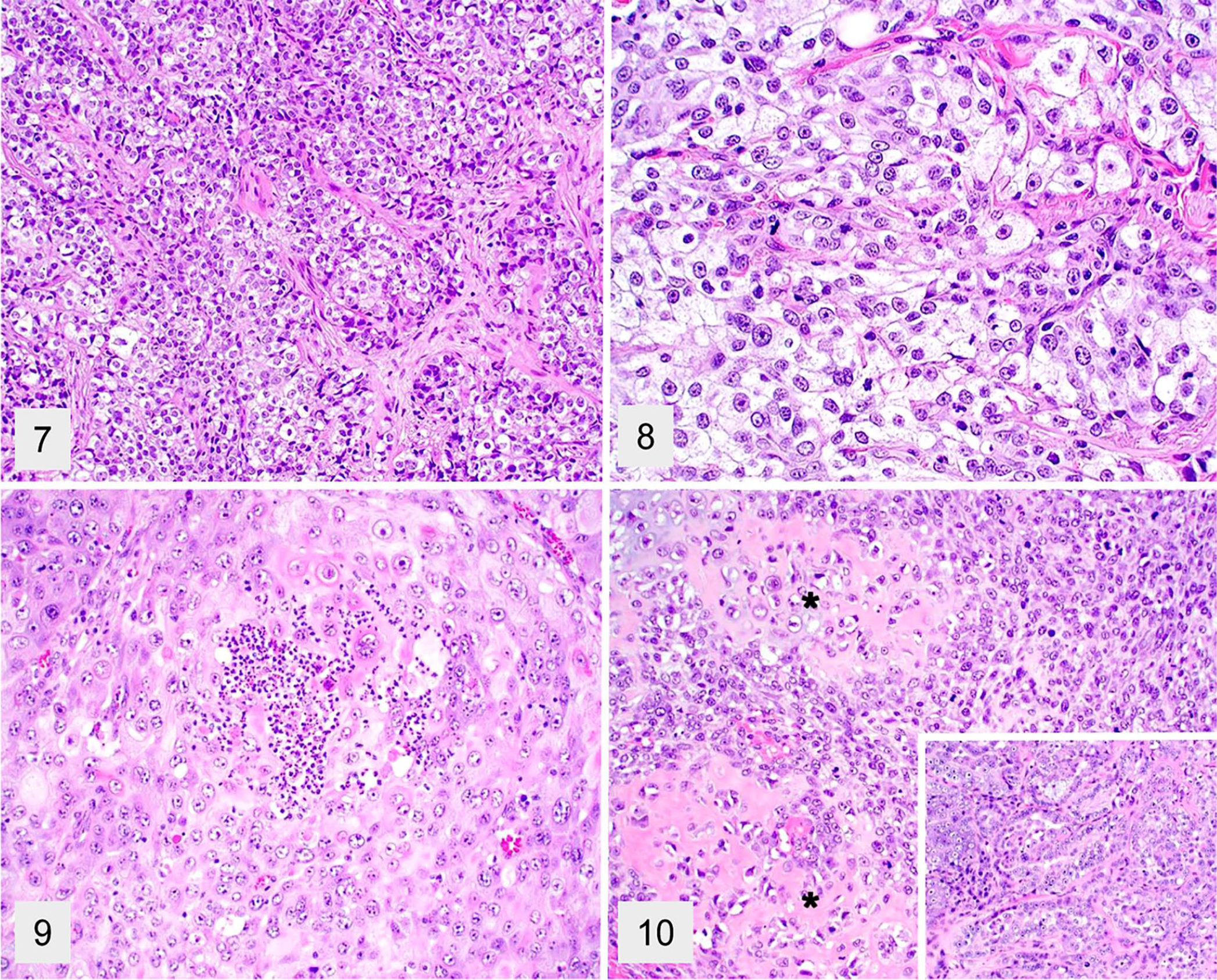
Various salivary gland neoplasms in dogs. H&E.
The squamous cell carcinoma was poorly demarcated and effaced most of the affected salivary gland (Fig. 9). Mitotic activity was common in all epithelial tumors, with an average of 1–12 per high power (FN22/40×) field. Areas of necrosis, hemorrhage, and fibrosis were also common findings.
One tumor (2.7% of the neoplasms) was a carcinosarcoma that effaced the salivary gland tissue and consisted of clusters of chondroid and osteoid matrix surrounded by spindle-shaped cells admixed with solid areas of epithelial cell proliferation with scattered acini (Fig. 10). The mitotic count was 59 per 2.4 mm2 (equivalent to 10 FN22/40× fields). One tumor (2.7% of the neoplasms) had undetermined histogenesis, and 2 (5.5% of the neoplasms) had round cell origin (a salivary lymphoma and a salivary plasma cell tumor). The final consensus diagnosis for neoplasia after review differed from the original diagnosis in 20 cases (Suppl. Table 1).
Other changes consisted of salivary gland lipomatosis (7 cases; 3.9%), necrotizing sialometaplasia (4 cases; 2.2%), and disseminated hemorrhage (1 case; 0.5%). Lipomatosis (Fig. 11) was characterized by widespread expansion of the salivary gland by sheets of adipocytes and subsequent lobular atrophy. Necrotizing sialometaplasia was characterized by extensive, well-demarcated areas of coagulative necrosis (Fig. 12) bordered by mixed inflammation and fibrosis (Fig. 13), vascular thrombosis, and duct hyperplasia with squamous metaplasia (Fig. 14). Salivary gland hemorrhage was characterized by abundant extravasated erythrocytes within the salivary gland interstitium and glandular architecture; the hemorrhage was attributed to physical trauma to the site. No infectious agents were identified during routine histology evaluation during the study period. However, additional ancillary testing (e.g., special stains and bacterial or fungal culture) was not attempted in any case.
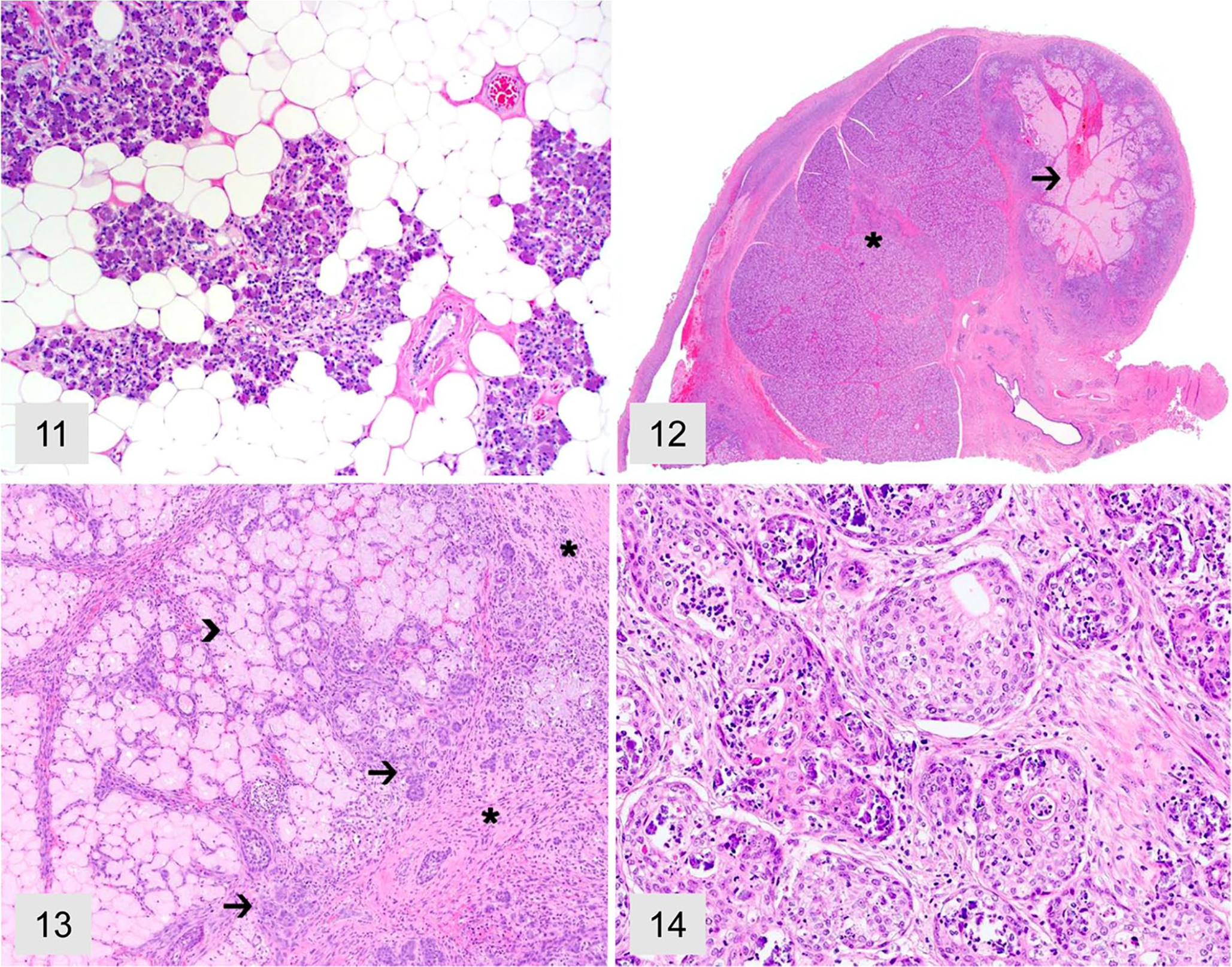
Salivary lipomatosis and necrotizing sialometaplasia in dogs. H&E.
The 179 salivary gland submissions in our study comprised 0.3% of 59 canine biopsy submissions to our laboratory within the studied period, confirming the overall low frequency of these routine submissions.2,10 –12,26 Affected dogs were mainly adults, and no sex or breed predisposition was evident.
Almost 70% of the changes in our case series affected a major extraoral salivary gland, most commonly the mandibular gland. When in the oral cavity, minor salivary glands associated with the gum tissue were most frequently involved. However, it is not possible to determine whether the inflammatory changes affecting minor salivary glands were primarily affecting these glands or reflected an extension of inflammatory processes occurring within the adjacent oral tissues. There have been studies describing sialocele (also referred to as mucocele) affecting mainly the cervical and sublingual location,1,2,20 with rare cases occurring in the minor salivary gland tissues present in the pharyngeal mucosa.10,28 However, other population studies have not assessed the anatomic distribution of non-neoplastic salivary gland changes in dogs, and it remains undetermined whether a predisposition may exist for specific disorders to affect major versus minor salivary glands. 26
Nonspecific sialoadenitis constituted the most frequent pathologic change in our study and was characterized by nonspecific inflammatory infiltrates with no discernible cause. Inflammation in most cases was attributed to a possible underlying sialocele, perhaps secondary to duct obstruction by sialoliths, which was confirmed by the presence of necrosis, hemorrhage, and fibrosis associated with cavitated areas filled with mucoid secretory material in 24 cases.2,10,28 Extraoral salivary glands were affected in 63 of our cases, confirming a predisposition for inflammatory changes, often associated with sialocele, to affect the extraoral salivary glands.1,2,20
Most cases in our study had been submitted for histologic evaluation because of local swelling near the salivary glands. However, a surprising number of tissue samples had no pathologic changes and consisted of normal salivary gland. The reason for this discrepancy is elusive, but it has been attributed to the compact nature of the cervical area, which hosts multiple and closely associated structures, prompting salivary gland samples to be incidentally submitted in cases in which other tissues (such as lymph node) were affected. 26 This hypothesis was confirmed in 10 of our cases, which were submitted by the referring veterinarian as lymph node but contained only salivary gland tissue. Another possibility for such cases includes sialoadenosis, a rare and poorly understood salivary gland disorder of laboratory animals, dogs, and humans. 25 Although sialoadenosis is clinically characterized by an enlarged salivary gland, no morphologic, histochemical, or immunohistochemical changes are evident histologically, and the condition is exceedingly difficult if not impossible to diagnose based on histologic examination. 25
A total of 36 neoplasms (20.1% of cases) were diagnosed in our case series. A substantial number of salivary gland neoplasms are associated with extraoral salivary glands, particularly the parotid and mandibular gland.9,11,16 In our data set, extraoral salivary glands were affected in 23 cases in which neoplasia was diagnosed, with 20 tumors occurring in the mandibular or parotid gland. Approximately 88% of the neoplasms in our study were malignant and of epithelial origin, similar to findings reported previously.9,11,13,16,26 Dogs with salivary gland neoplasia were an average of 11 y old, and no breed or sex predisposition was apparent. The neoplasms diagnosed most commonly were adenocarcinomas (14), mucoepidermoid carcinomas (10), and acinic cell carcinomas (7). These findings are similar to previous reports.8,9,11,16,26,29 One of the limitations of our study is the lack of clinical follow-up after the diagnosis, which prevented us from assessing the clinical behavior of these tumors. An investigation of 24 dogs with malignant epithelial tumors of the salivary gland indicated a median survival time of 550 d after a diagnosis, with improved survival associated with lower clinical stage. 9 Further, histologic grading of malignant salivary gland neoplasms in humans has been found to correlate well with tumor prognosis, 22 but the same has not been found in canine cases based on limited data. 9 Because postdiagnosis clinical information was not available and, more importantly, given that no standard grading system has been proposed in veterinary medicine, we did not assess the tumor grade in the current data set. It is also unknown whether affected dogs in our study underwent any type of treatment, but surgical excision alone or in combination with radiation therapy has been shown to offer a longer survival time in dogs with salivary adenocarcinomas. 9
Salivary lipomatosis or lipomatous infiltration are rare lesions that need to be differentiated from infiltrative lipomas. 17 Although lipomatosis results in widespread infiltration and swelling of the salivary gland without a discrete tumor mass, infiltrative lipoma tends to form a tumor mass and infiltrate adjacent tissues. 17 However, lesions referred to as salivary lipomatosis forming a tumor mass have been described in dogs. 3 These inconsistencies raise the suspicion that the morphologic features of salivary lipoma and lipomatosis may overlap and lead to confusion in terms of diagnosis. 17 In our case series, lipomatosis was originally diagnosed in 5 cases, with 2 other similar cases originally diagnosed as salivary lipoma. After our histologic reexamination, we reassigned the 2 lipomas to the lipomatosis group based on the widespread salivary infiltration by adipose tissue and absence of a tumor mass.
Necrotizing sialometaplasia was diagnosed in nearly 2% of our cases. As seen in 2 of these cases, these lesions have been shown to affect predominantly, but not exclusively, the mandibular salivary gland.4,14,15,26 Numerous comorbidities have been described in dogs suffering from necrotizing sialometaplasia, but no causal link has been established. 21 Although an underlying vascular injury leading to ischemia and subsequent infarct has been proposed as the main mechanism for the development of the characteristic lesions in such cases, the cause of necrotizing sialoadenitis remains unknown.4,15,18 As seen in our cases, the distinct areas of duct hyperplasia with prominent squamous metaplasia closely resemble a salivary gland carcinoma or a squamous cell carcinoma, particularly if only small tissue sections are examined. Although we considered these possibilities, the extensive, well-demarcated infarcts associated with normal adjacent salivary gland tissue associated with the lack of solid acinar proliferation are key findings in the differentiation between necrotizing sialometaplasia and epithelial salivary gland neoplasia.4,15
Supplemental Material
Supplemental_material – Supplemental material for A retrospective study of salivary gland diseases in 179 dogs (2010–2018)
Supplemental material, Supplemental_material for A retrospective study of salivary gland diseases in 179 dogs (2010–2018) by Danielle E. Lieske and Daniel R. Rissi in Journal of Veterinary Diagnostic Investigation
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
