Abstract
The aim of the current study was to evaluate the expression of vascular endothelial growth factor (VEGF) and the microvascular density in canine soft-tissue sarcomas. Immunohistochemistry for VEGF expression was performed on 20 canine neoplasms by the streptavidin–biotin–peroxidase method using an anti-VEGF mouse monoclonal antibody (ab-119). The volume fraction of microvessels in the sarcomas was quantified in hematoxylin and eosin–stained tissue sections. At least 10 fields of view (40x magnification) per neoplasm were analyzed by positioning a grid with 100 points and counting the microvessels that fell into the intersection points. This percentage was considered the volume fraction of these microvessels in the tumor section. VEGF expression was detected in 65% of the neoplasms. In 92.3% of the neoplasms, the expression occurred in the peritumor region; in 46.15%, in the intratumor region; and in 38.46%, the expression was present in both regions. The cells responsible for VEGF expression were fibroblasts and macrophages in the peritumor region or in the pseudocapsule and neoplastic cells in the intratumor region. Greater intratumoral VEGF was expressed in hemangiopericytomas (
Sustained angiogenesis is considered one of the hallmarks of cancer 6 and plays an important role in tumor progression 7 and development of metastases. 11 Vascular endothelial growth factor (VEGF) has been implicated as the major factor in tumor angiogenesis 5,19 and has been proposed as a prognostic indicator in several types of cancer. 12,18 Human sarcomas are highly vascularized, 3 and angiogenesis can be involved in neoplastic progression because sarcoma cells produce large amounts of VEGF in vitro. 20 Comparatively, human and canine soft-tissue sarcomas have a similar histologic appearance, clinical presentation, and behavior. 4 Therefore, it can be inferred that canine sarcomas have a similar degree of vascularization as human sarcomas. Thus, the objective of the present study was to investigate VEGF expression and microvessel density in soft-tissue sarcomas of dogs.
Tissue samples were taken from 20 canine soft-tissue sarcomas and fixed in 4% buffered formalin, processed routinely, and embedded in paraffin. Four-micrometer histologic sections were prepared, mounted on poly-L-lysine–coated slides, and stained with hematoxylin and eosin (HE) for histopathologic analysis. Immunohisto-chemistry was performed on replicate tissue sections to detect the presence of VEGF expression. Tissue sections were dewaxed in xylene, dehydrated in graded alcohols, and washed in 0.01 M phosphate buffered saline (PBS; pH 7.2–7.4). Endogenous peroxidase activity was blocked with 0.3% hydrogen peroxide in absolute methanol for 30 min. Immunohistochemistry was performed using the streptavidin–biotin–peroxidase method, a and a monoclonal, mouse, anti-VEGF primary antibody. b Before incubation with anti-VEGF, sections were heated in a microwave oven for 3 cycles of 5 min at 37°C. A mixture of biotinylated anti-mouse, anti-rabbit, and anti-goat immunoglobulins, a diluted in PBS, was used as a secondary antibody. This mixture was applied, and the tissue sections were incubated for 30 min. After washing in PBS, the tissue sections were incubated for 30 min in streptavidin-conju-gated horseradish peroxidase in Tris–HCl buffer containing 0.015% sodium azide. a To identify sites of primary antibody binding, 3,3′-diaminobenzidine tetrahydrochloride was used as a chromogen, and hematoxylin dye was used as the counterstain. Expression of VEGF was assessed by determining the percentage of immunoreactive cells from a total of 1,000 cells, as previously described. 17 Immunoreactivity was graded as follows: positive staining was defined as >10% immunoreactive cells, whereas negative staining was defined as <10% immunoreactive cells or an absence of detectable staining. 17
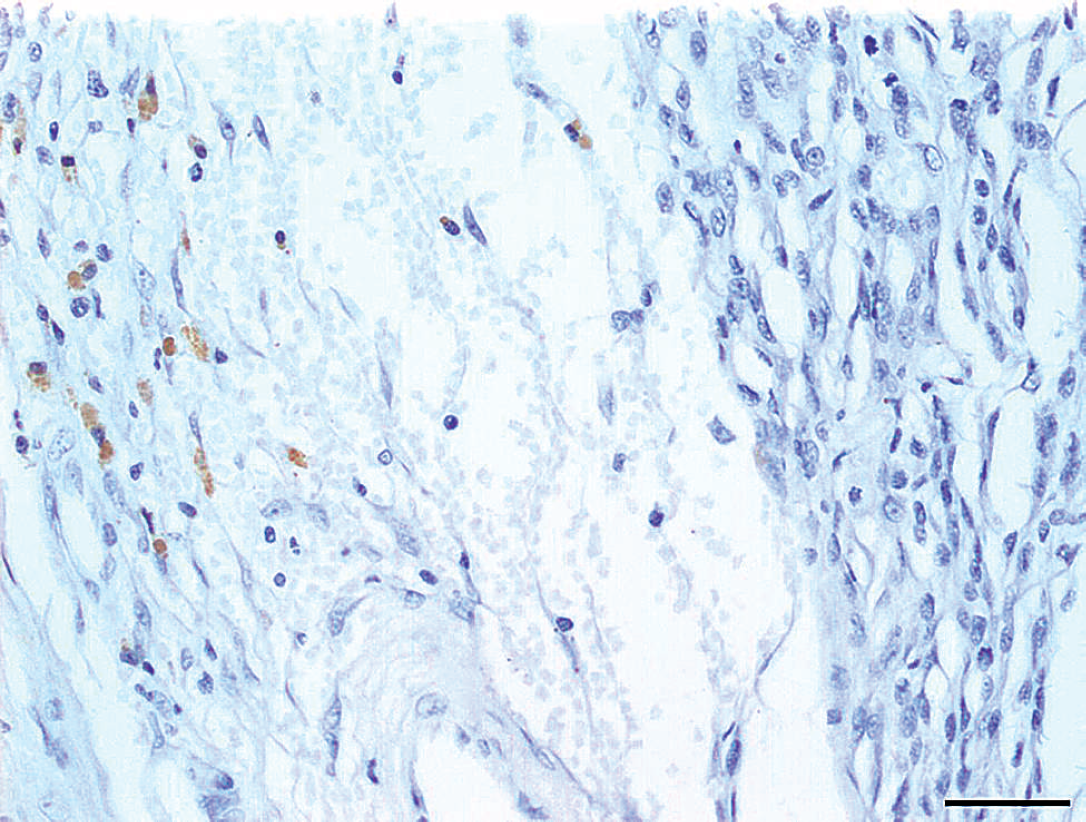
Section of a malignant peripheral nerve sheath tumor demonstrating peritumoral expression of vascular endothelial growth factor. Bar = 50 mm.
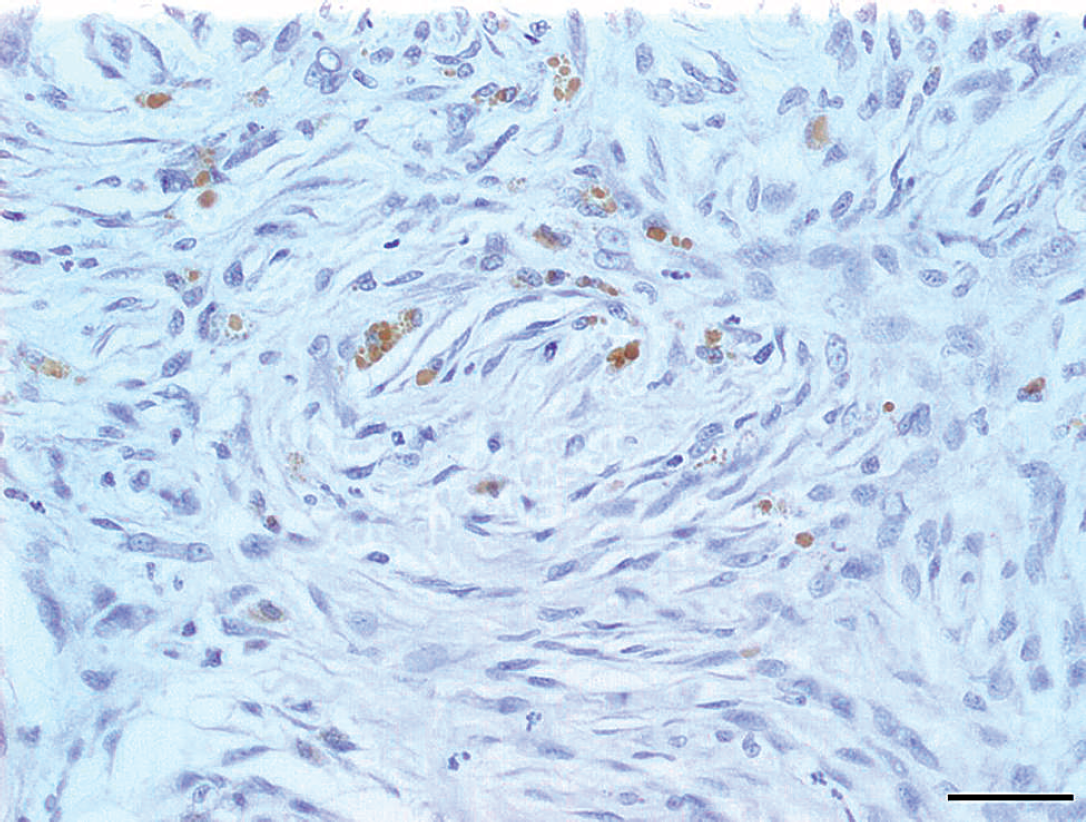
Section of a myxosarcoma showing intratumor expression of vascular endothelial growth factor by neoplastic cells. Bar = 50 μm.
The volume fraction of tumoral microvessels was quantified in HE-stained slides of the neoplasms, as previously described. 1 At least 10 fields of view (403 magnification) were analyzed per neoplasm by positioning a grid with 100 points, and counting the microvessels that fell onto the intersection points. This percentage was considered the volume fraction of microvessels in the tumor section.
The Mann–Whitney
The VEGF expression and microvessel density were investigated in 20 sarcomas, as presented in Table 1. VEGF expression was present in the intratumor or peritumor regions in 13 out of 20 neoplasms (65%). Peritumoral VEGF expression was observed in 92.3% of the neoplasms (Fig. 1); intratumor VEGF expression was observed in 46.15% of the neoplasms (Fig. 2); and combined intratu-mor and peritumor VEGF expression was observed in 38.46% of the neoplasms. Hemangiopericytomas had higher intratumoral VEGF expression than malignant peripheral nerve sheath tumors (
The quantity of microvessels was 134.85 ± 61.12 (mean ± standard deviation). No difference in microvascular density was present between VEGF-positive and VEGF-negative neoplasms (
VEGF expression in human neoplasms seems to be governed by factors such as hypoxia, insulin-like growth factor 1, inflammatory mediators, cell differentiation, and genetic changes. Studies have shown that hypoxia is a potent inducer of VEGF expression in vitro and in vivo. 8,13 In the current study, peritumoral cells that stained positively for VEGF could be resident, local cells, or they could have migrated in response to a tumor hypoxia to promote tumor angiogenesis. VEGF is expressed in many tumor types, correlates with human soft tissue sarcomagrading, 2,15 and has been proposed as a prognostic indicator correlated with decreased survival in several types of cancer. 19 In the present study, VEGF expression in hemangiopericytoma, hemangiosarcoma, malignant peripheral nerve sheath tumor, and myxosarcoma may provide important information to predict the biologic behavior of soft-tissue sarcomas in dogs. However, further studies comparing histologic grade with VEGF expression will be necessary to provide information about the behavior of soft-tissue sarcomas in dogs.
. Histologic type, vascular endothelial growth factor (VEGF) expression, and microvessel density of selected soft tissue neoplasms of dogs. *
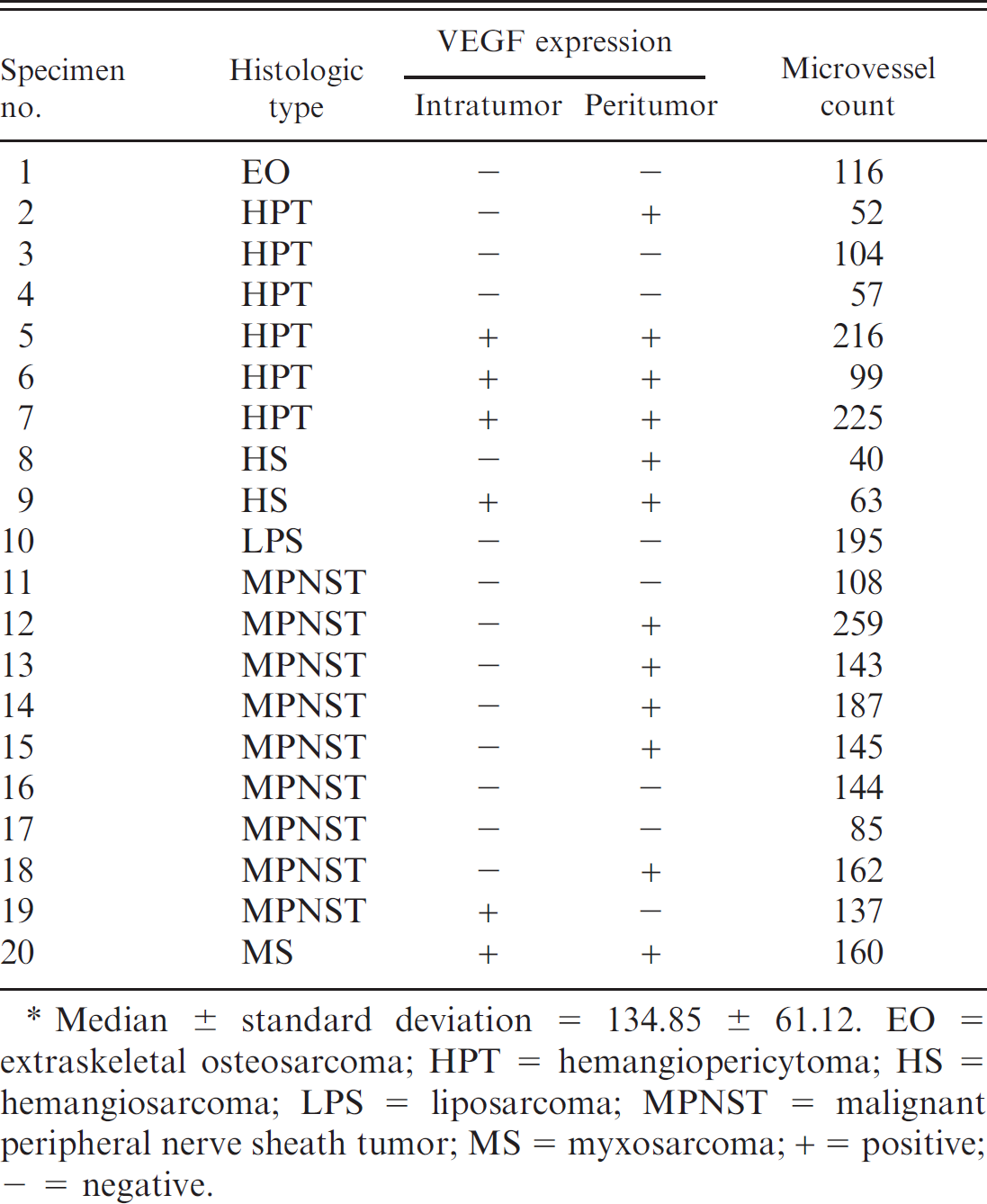
Median ± standard deviation = 134.85 ± 61.12. EO = extraskeletal osteosarcoma; HPT = hemangiopericytoma; HS = hemangiosarcoma; LPS = liposarcoma; MPNST = malignant peripheral nerve sheath tumor; MS = myxosarcoma; + = positive; + = negative.
Microvessel density has been investigated in solid tumors of humans to establish criteria for evaluating malignancy and growth potential. In a study of 119 patients with high-grade soft-tissue sarcomas of the extremities, the microves-sel density did not correlate with risk of distant metastasis or overall survival. 16 Similar findings have been reported in patients with malignant peripheral nerve sheath tumors and synovial sarcomas. 9,14 Results of the present study did not reveal any difference regarding microvessel counts between VEGF-positive and VEGF-negative neoplasms or between VEGF expression and tumor type. These observations suggested that the number of neoplasms evaluated was too small to provide meaningful information about VEGF expression and microvessel count or tumor type. In addition, the possibility exists that VEGF expression may affect prognosis by a mechanism other than the driving of tumor vessel growth. For example, VEGF may increase vascular permeability and promote the spread neoplastic cells in the circulation. Increased vascular permeability might also stimulate tumor growth by increasing nutrient availability. A larger number of neoplasms should be evaluated to determine whether VEGF expression increases vascular permeability and microvessel counts in soft-tissue sarcoma of dogs.
Footnotes
a.
LSABTM, Dako North America Inc., Carpinteria, CA.
b.
Clone VG76e, Abcam Inc., Cambridge, MA.
c.
Microsoft Corp., Redmond, WA.
d.
Minitab Inc., State College, PA.
