Abstract
Equine viral arteritis (EVA) can cause prominent economic losses for the equine industry. The purpose of this review is to provide the pathologist some familiarity with the clinical history, lesions, pathogenesis, and diagnosis of EVA. EVA is caused by an arterivirus (equine arteritis virus, EAV), and the vascular system is the principal but not unique viral target. EVA has variable presentations, including interstitial pneumonia, panvasculitis with edema, thrombosis and hemorrhage, lymphoid necrosis, renal tubular necrosis, abortion, and inflammation of male accessory genital glands. EAV antigen (EAVAg) can be demonstrated within the cytoplasm of epithelial cells such as alveolar pneumocytes, enterocytes, adrenal cortical cells, trophoblasts, thymus stroma, renal tubular cells, and male accessory genital gland cells. It can be also demonstrated within endothelia, in vascular, myometrial, and cardiac myocytes, macrophages, dendritelike cells of lymphoid organs, and chorionic mesenchymal stromal cells. In young and adult horses, following colonization of macrophages, the virus spreads systemically using circulating monocytes and enters the endothelium and tunica media of blood vessels, histiocytes, and dendritelike cells. Eventually, the virus multiplies within renal tubular cells. Lesions are uncommon in the aborted fetus; if present, they are mild, and EAVAg is frequently not detectable within fetal tissues and placenta. The clinical presentation and lesions of EVA may resemble those of other diseases. Complete pathologic examination associated with immunohistochemistry, virus isolation, and, especially in cases of abortion, serology will guarantee a directed and accurate diagnosis.
Equine viral arteritis (EVA) is a global infectious disease of horses and is characterized by panvasculitis inducing edema, hemorrhage, and abortion in pregnant mares. 15 , 16 , 27 EVA can cause severe economic losses for the equine industry. The purpose of this review is to provide the pathologist some familiarity with the clinical history, lesions, pathogenesis, and diagnosis of EVA. EVA can be confused with other equine diseases and must be considered in sporadic and epizootic respiratory syndromes, foal death associated with respiratory and/or enteric signs, or sudden death in foals. Although not frequent, velogenic isolates of equine arteritis virus (EAV) can induce severe and fatal disease in adults. Natural outbreaks of EVA characterized by transient clinical signs and abortion in pregnant mares contrast with experimental disease, which features high mortality and prominent systemic vascular necrosis. Blood vessel cells are the major, but not exclusive, target of EAV. Lung, intestine, kidney, the reproductive tract, and occasionally the placenta are important viral replication sites, which favor the spread of the virus. Complete pathologic examination associated with the available ancillary procedures will help guarantee a directed and accurate diagnosis.
The Virus
EAV is an enveloped, spherical, positive-stranded RNA virus with a diameter of 50–70 nm. The virion is comprised of an isometric core surrounded by a lipid-containing envelope from which delicate spikes protrude. The viral genomic RNA, which is encapsulated by a single nucleocapsid protein, is contained within the core particle. Within the viral envelope at least three integral membrane proteins are incorporated. EAV is a non-arthropod-borne virus classified as a member of the new order Nidovirales, including also the bigeneric family Coronaviridae, within the family Arteriviridae with porcine respiratory and reproductive syndrome virus, simian hemorrhagic fever virus, and lactate dehydrogenase elevating virus. 5 , 14 Genetically, EAV is similar to coronaviruses but has a dissimilar viral structure and a complement-fixing antigen but no hemagglutinins. 6 Genetic diversity is recognized among field isolates. 1 EAV was first isolated from fetal lung collected during an epizootic of abortion in Bucyrus, Ohio (USA). 4 , 16 In Bucyrus, horses of both sexes and various age groups experienced an acute and febrile infectious disease characterized by mucopurulent rhinitis, stiffness, depression, and edema of conjunctiva and limbs. 9 , 15 The virus was isolated on primary kidney cells. 15 Only one strain of EAV is recognized, the Bucyrus strain; however, various isolates have different degrees of virulence. 39
Clinical Features and Carrier State
Clinical signs may be absent or may include pyrexia, depression, anorexia, leukopenia, limb edema, stiffness of gait, rhynorrhea and epiphora, conjunctivitis, and rhinitis. Edema of the periorbital and supraorbital areas, midventral regions, scrotum, prepuce, and mammary gland, urticarial rash, and abortion also occur. Less frequently, severe respiratory distress, ataxia, mucosal papular eruptions, submaxillary lymphadenopathy, and intermandibular and shoulder edema may be observed. 52 , 55 EAV can be associated with epidemic abortion, 8 , 51 , 52 is occasionally fatal in adults, 35 , 47 and more frequently can be fatal for foals. 13 , 18 , 21 , 54 , 56 When neonates are not protected by passive maternal immunity, they may present with sudden death or severe respiratory distress followed by death. 55 Affected intact males may become long-term carriers and may shed EAV in the semen. 53 Stallions shedding EAV in their semen serve as a reservoir for the virus within the equine population. Additionally, infected stallions and semen have resulted in restrictions for international movement of horses and semen.
Clinical Pathology
Clinical laboratory results and serology
Clinical pathology findings reported in affected foals include hypoxia, hypercapnia, respiratory acidosis sometimes complicated by metabolic acidosis, neutropenia/neutrophilia, lymphopenia/lymphocytosis, thrombocytopenia, and hyperfibrinogenemia. 13 , 18 , 54 , 56 However, abnormal values may be inconsistent, are highly variable, and are not diagnostic for EVA. Arterial blood gas values in affected foals are diagnostic of severe disease of the respiratory system, but these abnormalities may be seen in a variety of neonatal diseases of horses, including bacterial sepsis and other viral diseases such as equine herpesvirus 1 (EHV-1) infection.
In one report, 4 experimentally infected mature horses were consistently leukopenic. The horses in this study presented with a biphasic leukopenia and neutropenia. Lymphopenia was consistently present postinfection (PI) in this group of experimentally infected horses.
Cytology
Cytology of pulmonary washings has been described in one foal with severe pulmonary disease characterized by dyspnea, tachypnea, hypoxemia, and hypercapnia. The condition in this foal was initially reported in the literature as a case of bronchopulmonary dysplasia. 18 Subsequent reanalysis of the case revealed that the foal was infected with EAV, and a correction was published. 13 , 56 Cytologic abnormalities in bronchoalveolar lavage and tracheal aspirate reported for this foal included formed fragments of granular material, mucus, fibrin, a few small macrophages, and epithelial cells demonstrating cilia loss with retention of a prominent terminal bar and squamous metaplasia. In addition, there were papillary clusters of squamoid or cuboidal cells with focal loss of cilia. These cytologic findings corresponded to proliferative bronchointerstitial pneumonia with intraalveolar macrophages and a few neutrophils, fibrin and hyaline membrane formation, type 2 pneumocytic hypertrophy, and hyperplasia. Most foals with EVA die without cytologic evaluation of tracheal and bronchial aspirates. This case is the only one with such an evaluation and likely represents findings in severe cases.
Because the EAV pulmonary antigen is not as abundant as the antigen in EHV-1 infection, 10 , 11 , 41 its identification within the cytologic specimens may be difficult. However, renal tubular necrosis occurring during the late phase of the EAV infection is associated with florid intraepithelial intracytoplasmic viral growth, 13 , 15 making the urine sample a potential candidate for in vivo immunocytochemical testing. In addition to buffy coat and rhinopharyngeal swab, part of the fluid obtained with the tracheal lavage and urine should be submitted for ancillary virologic testing.
Anatomic Pathology
EAV-associated gross and histologic lesions have been described in naturally infected 13 , 18 , 21 , 27 , 35 , 45 , 47 , 54 , 56 and experimentally infected 7 , 15 , 17 , 22 , 36 , 39 , 45 equids. The EAV isolates differ in virulence and consequently induce lesions that differ in severity. 39 EAV antigen (EAVAg) can be identified in various tissues and organs either associated with lesions or occasionally in the absence of them. 9 , 13 , 35 , 36 , 56 EAVAg can be identified within the cytoplasm of the infected cell using equine polyclonal 9 and murine monoclonal 35 antibodies in fresh and fixed tissues.
Gross lesions
Gross lesions are the expression of the vascular pathologic changes. Edema, congestion, and hemorrhage of the subcutaneous tissues, lymph nodes, and viscera are the most frequent gross lesions in horses that die after natural or experimental EAV infection (Fig. 1). The body cavities may contain moderate to abundant amounts of yellowish clear exudate. Congestion and lymphadenomegaly, edema, and hemorrhages can be observed along the course of the colonic and cecal vessels but also are evident systemically. On the cut surface of lymph nodes, there may be a prominent subcapsular sinus and dilated medullary sinuses. Lungs, especially those of infected neonates (Fig. 1), are wet and increased in weight, with a prominent lobular pattern. The trachea may contain froth. On occasion, lungs can be multifocally or diffusely reddish because of congestion and hemorrhage. The uterine endometrial surface of aborting mares may be swollen and diffusely congested, sometimes with hemorrhages.
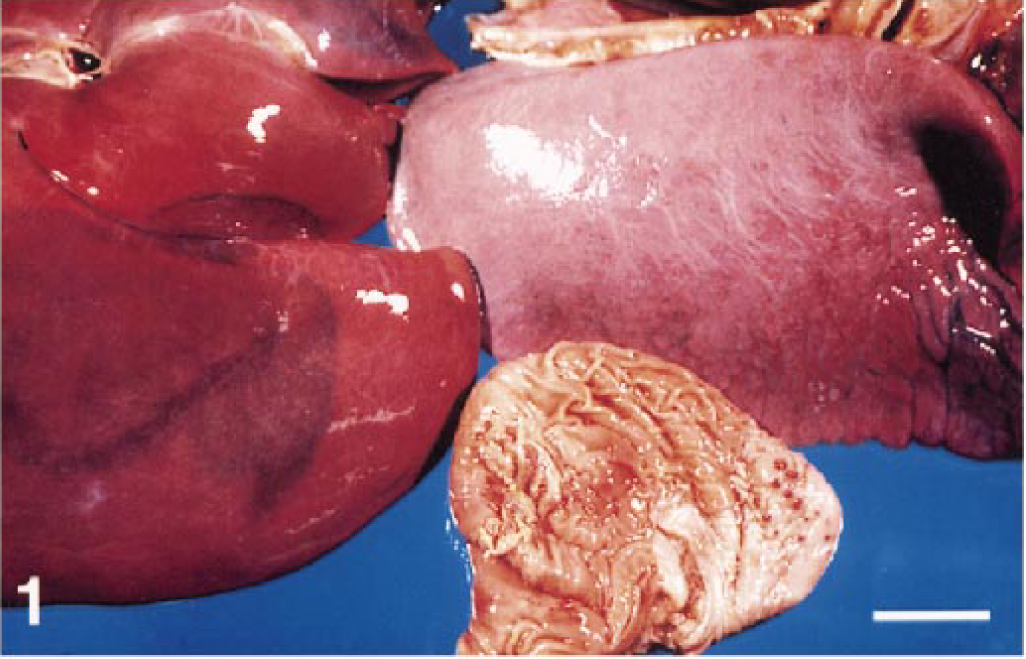
Lung, liver, and stomach; foal with EVA. Severe and diffuse edema of lung, Glisson hepatic capsule, and gastric mucosa, with petechiae. Bar = 8 cm. (Courtesy of J. M. King, Department of Pathology, College of Veterinary Medicine, Cornell University, Ithaca, NY.)
Microscopic lesions
The histologic lesions are observed in various systems of horses with EAV infection. The blood vessels are the principal target.
Blood vessels
All organs, including the skin, may contain vascular changes. Mild lesions include vascular and perivascular edema with occasional lymphocytic infiltrate and endothelial cell hypertrophy. Severe changes include vasculitis with fibrinoid necrosis of the tunica media, abundant vascular and perivascular lymphocytic and lesser granulocytic infiltration (Fig. 2) with karyorrhexis, frequent loss of endothelium, and formation of large fibrinocellular stratified thrombi. Occasionally, vascular portal vasculitis consists of severe inflammatory cell infiltrate that erodes the hepatocellular periportal limiting plate. Within vessels, EAVAg localizes in endothelium, medial myocytes (Fig. 4), and pericytes. Marginating macrophages containing intracytoplasmic EAVAg may be seen, occasionally associated with infected endothelial cells. Rarely, cardiac vasculitis with myocyte necrosis and associated EAVAg can be observed. Ultrastructurally, 17 infected endothelial cells appear hypertrophied, with expansion of cytoplasmic matrix in the absence of increased cytoplasmic organelles. Endoplasmic cisternae are distended, and mitochondria are increased in volume and their cristae are affected by degenerative changes. Specific cytoplasmic alterations associated with viral colonization of endothelial cells include dense crystalloid viral particles contained within endoplasmic cysternae and membranes of endoplasmic reticulum and vacuoles containing virions. These viral particles average 58 nm in diameter and are characterized by a 25-nm core and a less dense external membrane. Capillary lumina are often obliterated by swollen endothelial cells, platelet thrombi, or neutrophils. The nervous system is generally not affected, although cerebral vascular necrosis has been reported in fetuses.
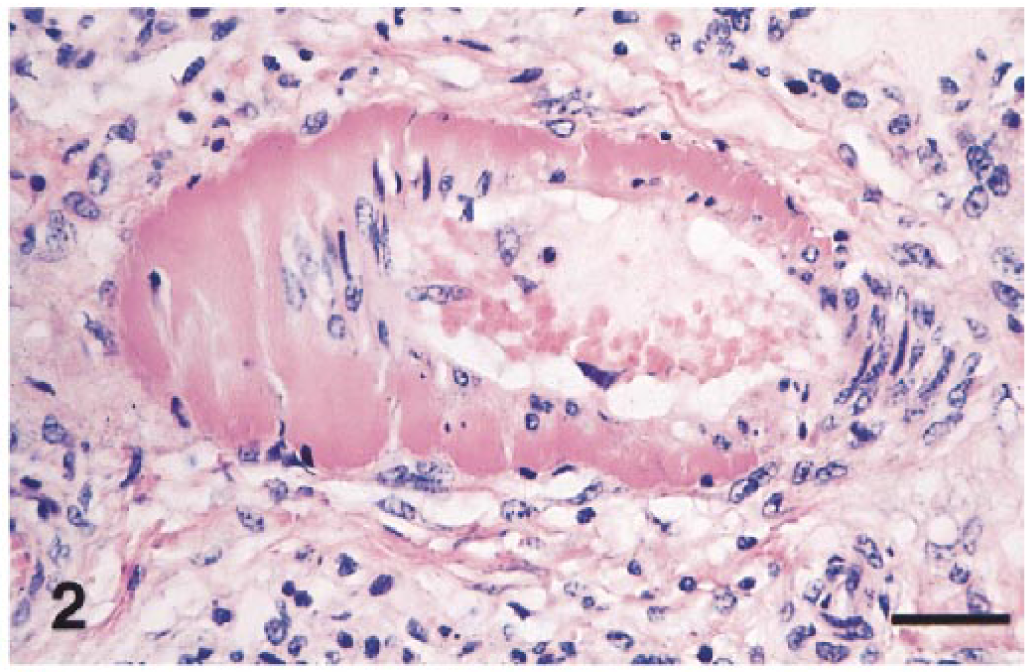
Small muscular artery; foal with EVA. Fibrinoid necrosis of the tunica media and perivascular edema with lymphocytic infiltrate. HE. Bar = 80 µm.
Lungs
Lungs may be affected by mild to severe interstitial pneumonia (Figs. 1, 3) characterized by alveolar infiltration with macrophages and lesser numbers of neutrophils, hyaline membrane formation, and fluid-filled alveoli. There is hypertrophy and hyperplasia of alveolar pneumocytes, which become rounded with a vesicular nucleus. In addition, there is pulmonary arteritis and phlebitis. Usually EAVAg localizes within the cytoplasm of pneumocytes (Fig. 3) and within alveolar macrophages. Later in the infection, EAVAg becomes identifiable within vascular endothelium and then extends to the cytoplasm of vascular myocytes.
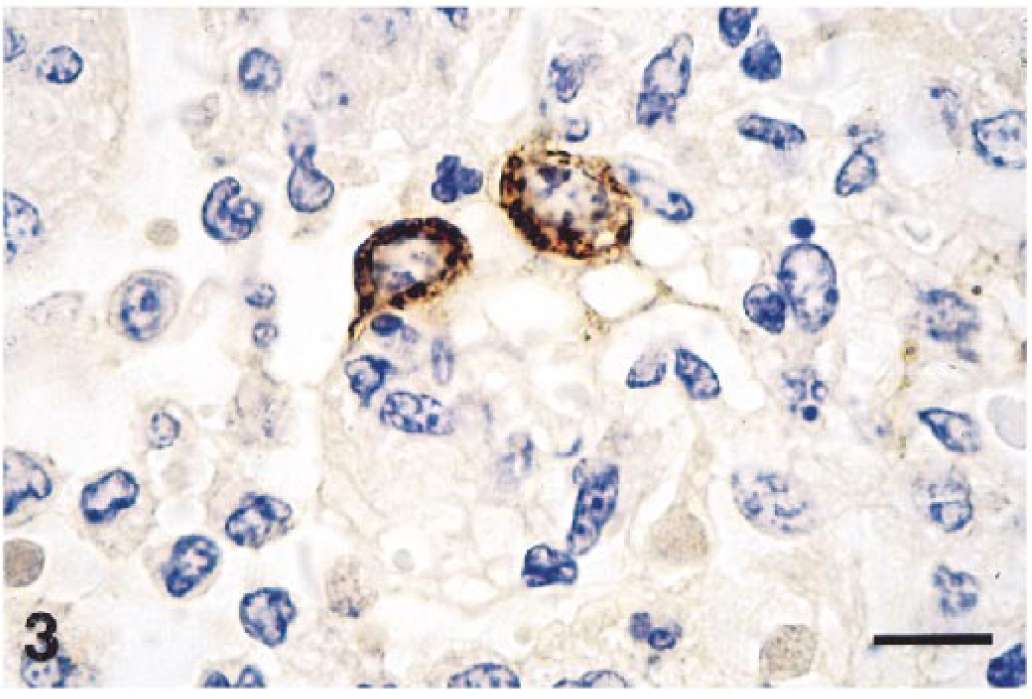
Lung; foal with EVA. Interstitial pneumonia with hypertrophied type 2 pneumocytes containing intracytoplasmic EAV antigen. Hematoxylin and avidin–biotin immunoperoxidase. Bar = 20 µm.
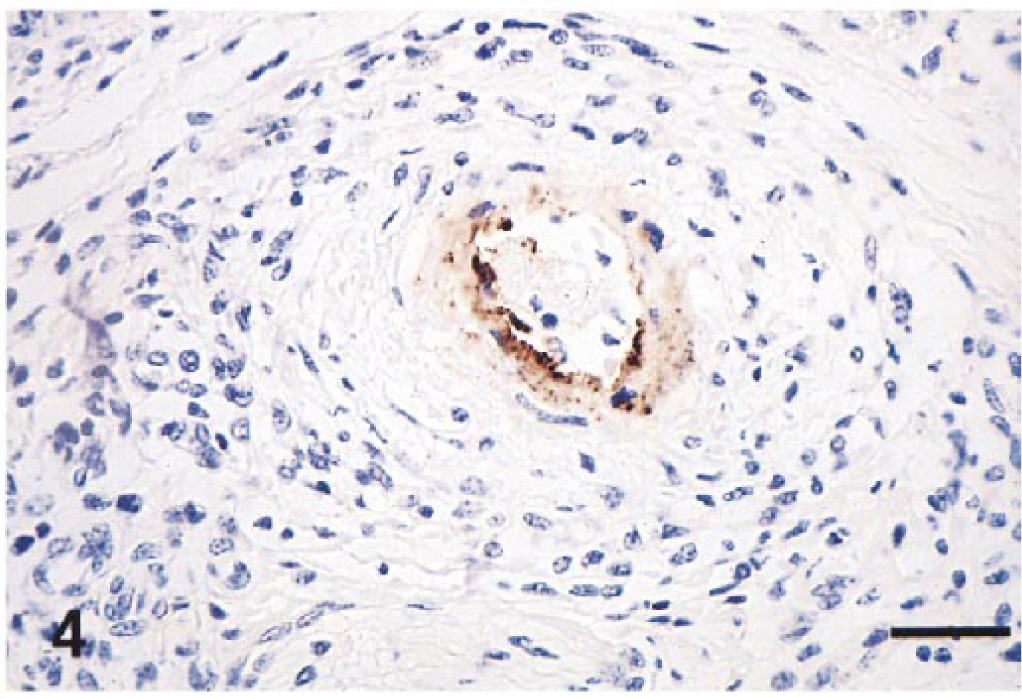
Small muscular artery; foal with EVA. Vascular intimal and medial necrosis with perivascular lymphocytic infiltrate and edema. EAV antigen is diffusing from the endothelium to the tunica media. Hematoxylin and avidin–biotin immunoperoxidase. Bar = 120 µm.
Lymphoid tissue
Within lymphoid organs, it is possible to observe lymphoid follicle necrosis, edema, and slight hemorrhage with histiocytic erythrophagocytosis. Lymph node sinuses may contain prominent and sometimes highly pleomorphic histiocytic cells and lymphocytes. EAVAg is contained within stromal dendritelike cells and within the macrophages of the lymph node sinuses and spleen.
Intestine
In adults, distention of the submucosal lymphatics of the large intestine, with mild crypt and lamina propria necrosis, may be observed. In foals, where the pulmonary lesions generally prevail, it is also possible to observe a pneumoenteric syndrome with pathologic changes involving crypts, intestinal mucosal blood vessels, and gastrointestinal tract–associated lymphoid tissue. Infarcts may be present in cecum and colon. In these cases, EAVAg may localize within enterocytes and vascular endothelial cells.
Adrenal glands
The adrenal gland may present with multifocal vasculitis, hemorrhages, and infarcts. The EAVAg can localize within cortical epithelial cells and capillary endothelium of cortex and medulla.
Kidneys
Renal lesions, which can be severe, occur when infection is at an advanced stage and consist of tubular necrosis, lymphocytic interstitial nephritis (Fig. 5), glomerular tuft disorganization, and hypercellularity. The viral antigen can localize within morphologically intact and necrotic tubular epithelial cells, intratubular cellular hyaline casts (Fig. 5), and glomerular endothelial and perhaps mesangial cells. Sometimes, in absence of morphologic changes and before the occurrence of the tubular lesions, it is possible to observe abundant intracytoplasmic viral antigen within stellate and fusiform cells located within the renal interstitium.
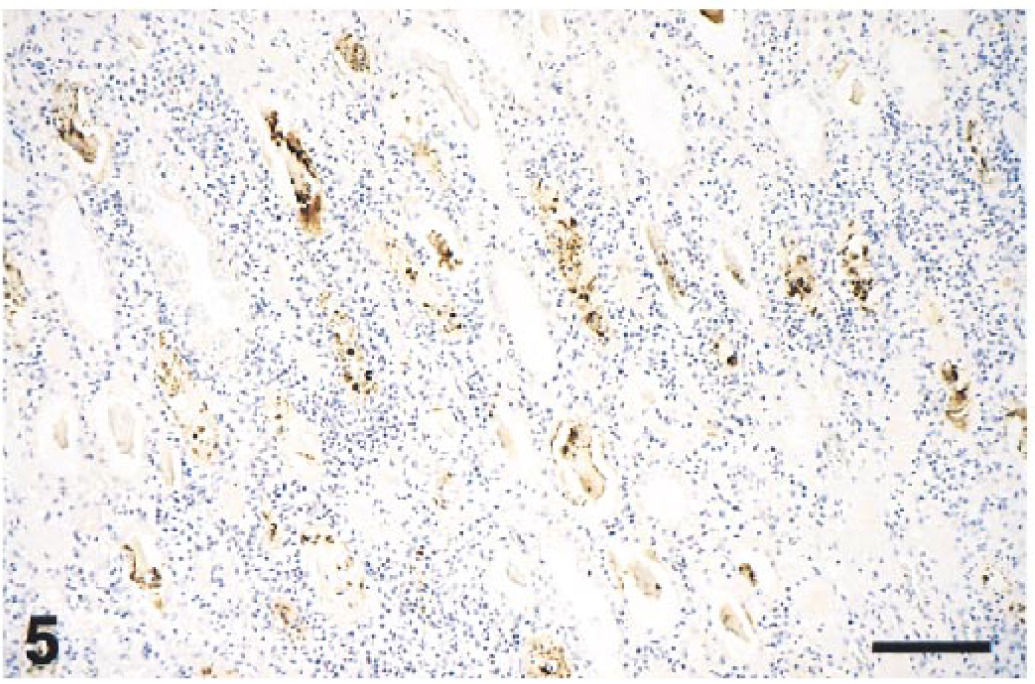
Kidney; mare with EVA. Severe diffuse interstitial lymphocytic nephritis with tubular necrosis and abundant EAV antigen contained in renal tubules and intraluminal casts. Hematoxylin and avidin–biotin immunoperoxidase. Bar = 300 µm.
Skin
The dermis may be involved with vasculitis, making skin biopsy suitable for diagnostic histopathology and immunohistochemistry. The vasculitis may be associated with thrombosis and ulcerative dermatitis.
Female reproductive tract and fetus
Fetuses and fetal membranes are often expelled without premonitory signs of abortion and can be autolyzed or well preserved. 10 , 16 , 35 Lesions in the fetus are only occasionally detected 7 , 10 , 29 and when present are represented by mild perivascular lymphocytic infiltrate and mild interstitial pneumonia. Rarely, they can be severe and consist of vasculitis involving the allantochorion, brain, liver, spleen, and lung. 29 EAVAg is inconsistently detectable within tissues of aborted fetuses and when present is localized within the cytoplasm of the areolar trophoblast (Fig. 6), allantochorionic mesenchyma, thymus epithelium, splenic reticular cells, endothelium of visceral blood vessels, and enterocytes. 10 , 13 , 36
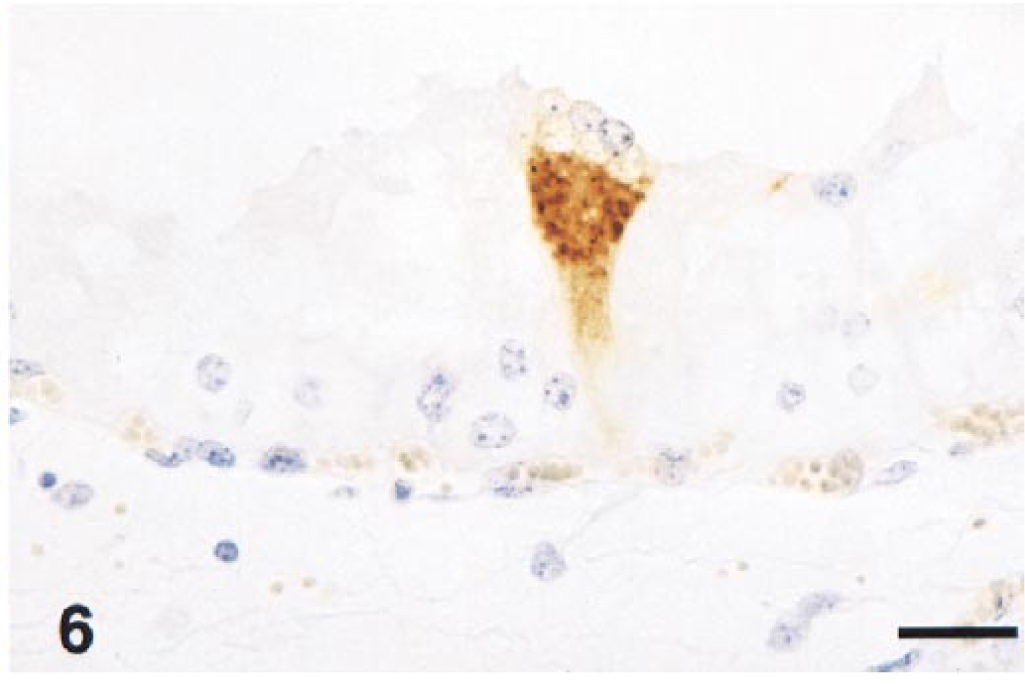
Chorioallantois; equine fetus with EVA. A syncytial trophoblast cell contains abundant intracytoplasmic EAV antigen. Hematoxylin and avidin–biotin immunoperoxidase. Bar = 25 µm.
In mares experiencing abortion following experimental infection, 7 the uterine epithelial cells are swollen, with fragmentation of cytosolic network, swollen granular mitochondria, residual bodies, and large phagolysosomes containing dense and finely granular material, membranes, and vescicles. The uterine propria–submucosa may be edematous with infiltration of neutrophils and macrophages and endothelial cell swelling. The myometrium may contain necrotic myocytes with ribosome clusters, macrophages, and swollen endothelial cells.
Male reproductive tract
EVA lesions associated with the male reproductive tract were studied in experimentally infected prepubertal and peripubertal colts. 22 Colts euthanized between the 7th and the 14th day following viral inoculation had thoracic and abdominal effusions, lymphadenopathy, and diffuse edema of the genital organs. Histologically, necrotizing vasculitis involving testes, epididymides, vasa deferentia, ampullae, prostate glands, and vesicular and bulbourethral glands were observed. This vasculitis was characterized by severe fibrinoid necrosis of small muscular arteries with edema and hemorrhage. Colts examined between the 28th and 180th day PI had lymphocytic and plasmacytic infiltrate within the lamina propria and tunica muscularis of the epididymides and accessory genital glands. One of the prepubertal foals, persistently infected for 15 months following viral inoculation, had marked lymphoplasmacytic infiltration of ampullae.
Experimental infection
Lesions observed following experimental infection have been arbitrarily divided into three groups: developmental, terminal, and chronic but active. 45 Lesions were subdivided on the basis of the progression of the macroscopic and histologic changes.
The developmental lesions, observed from the 4th through the 6th day PI, consist of excessive pleural and peritoneal fluid, congestion and enlargement of the lymph nodes, and edema along the course of the colonic and cecal vessels. Histologically, distention of the submucosal lymphatics of the large intestine, with mild necrosis of the mucosal epithelium, is evident. In addition, subcapsular and interstitial edema and congestion of lymph nodes, adrenal cortices, and kidneys have been reported.
The terminal lesions are considered extensions of the developmental lesions with subcutaneous edema and abundant quantities of fluid in the peritoneal and pleural cavities. All the lymph nodes of the body are swollen and hemorrhagic to some degree. Infarcts have been described in the cecum and colon. Severe submucosal edema and hemorrhage are frequent findings, and hemorrhage of the adrenal cortex has been observed. Microscopically, there is severe necrosis of the germinal centers of lymph nodes, with associated hemorrhage. Severe necrosis of the mucosa, submucosal edema, and necrotizing panvasculitis with neutrophils and lymphocytes mainly involving the small muscular arteries also has been reported. Multifocal thrombosis and infarction involving cecum and colon and hemorrhage of the renal medulla are seen. In the renal cortex, there is mild glomerulitis with swelling and disorganization of the glomerular tufts.
Chronic lesions are seen in horses from day 12 following virus inoculation. Lesions consist of mild swollen lymph nodes and excess of fluid within peritoneal and thoracic cavities. Microscopic lesions are dramatic and consist of generalized arteritis and severe glomerulonephritis with tubular necrosis and hyaline casts.
Lesions of natural EVA are rarely as severe as those observed following experimental infection.
Pathogenesis
It is possible to determine the pathogenesis of EAV infection in horses by following the distribution of viral antigen and lesions in experimental 2 , 9 , 36 , 40 and natural 10 , 13 , 35 , 56 infections (Fig. 7). Twenty-four hours PI or later, the virus invades the respiratory epithelium (Fig. 3) and alveolar macrophages. By 48 hours PI, the virus can be found in the satellite lymph nodes, especially bronchial lymph nodes. At 3 days PI, the virus replicates in bronchopulmonary lymph nodes, endothelium, and circulating monocytes. Systemic distribution of the virus follows, with localization within macrophages and dendritelike cells of lymphoid tissue. Approximately 6–8 days PI, the virus localizes within endothelium and medial myocytes of blood vessels and mesothelium. At day 10 PI, the most severe damage occurs to blood vessels. After 10 days PI, there is decrease of EAVAg in all the locations except the tunica media of small muscular arteries (Fig. 3). The last site to be invaded apparently is the renal tubular epithelium (Fig. 5), where the virus may persist for an additional 2 weeks. Infective EAV is no longer detectable in most tissues 28 days after experimental infection, except in the reproductive tracts of some colts and stallions. 19 , 43
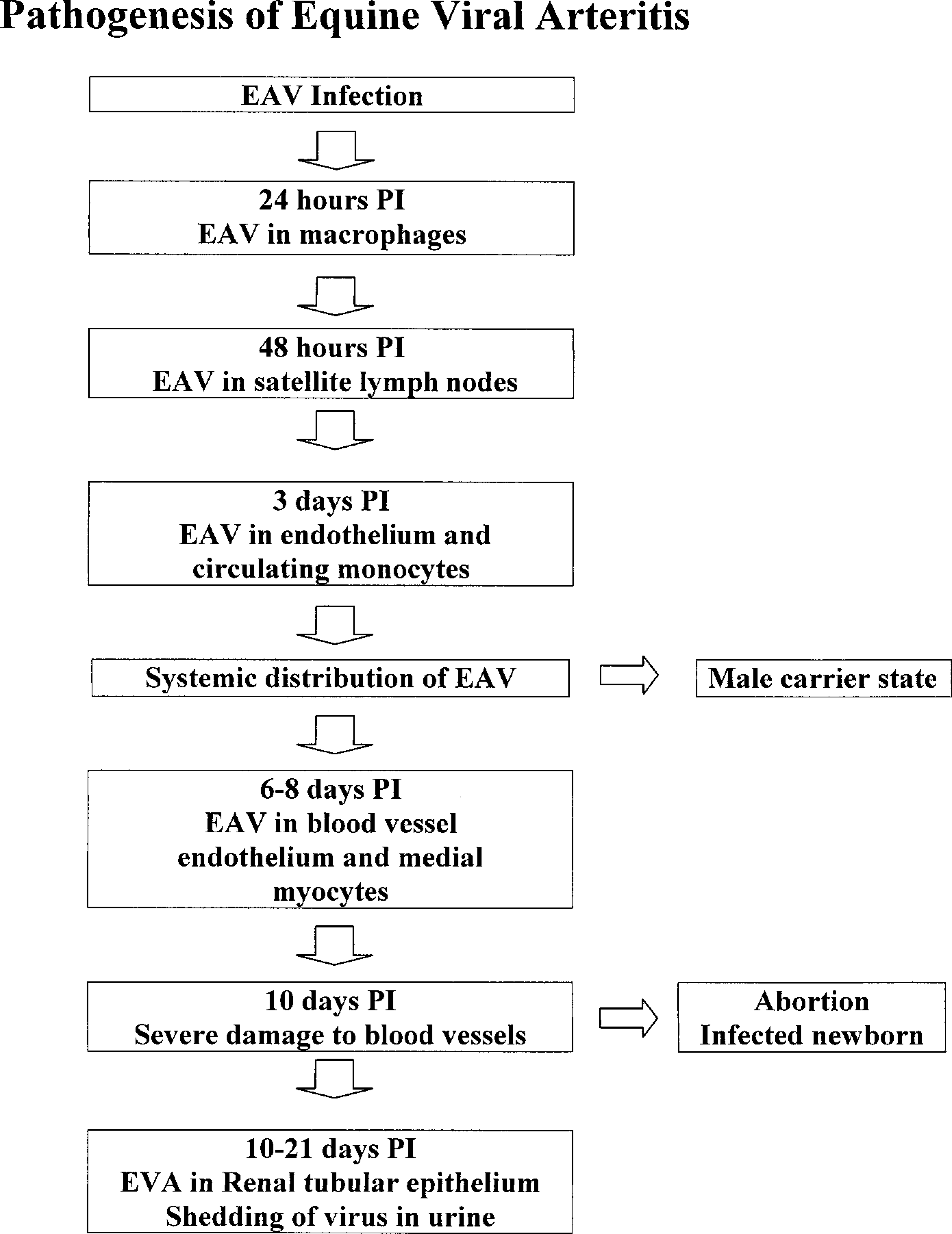
Pathogenesis of EVA.
The vascular lesions observed in EAV infection, particularly medial necrosis, may be due to a combination of factors. Virus localization may have a direct cytopathic effect on endothelium and medial myocytes. 13 The endothelial cell damage could induce anoxia 17 or thrombosis.
As previously speculated, 7 abortion may occur by myometritis. Reduction of blood flow to the fetus may occur due to blood vessel compression by endometrial edema or alteration of vascular tone by various inflammatory mediators. Serum progesterone levels constantly diminish from 6 to 48 hours before abortion and are not detectable at necropsy. This decreased progesterone production from a hypoxic placenta, combined with a local release of prostaglandins, may trigger chorionic detachment. 7 Vasculitis with associated thrombosis may also play a role inducing ischemia. Chorionic detachment and expulsion of an infected or uninfected fetus may follow (Fig. 8). In the infected fetus, EAVAg can be identified within the trophoblastic epithelium and mesenchyma and shortly after within pneumocytes, alveolar macrophages, thymic epithelium, and enterocytes.
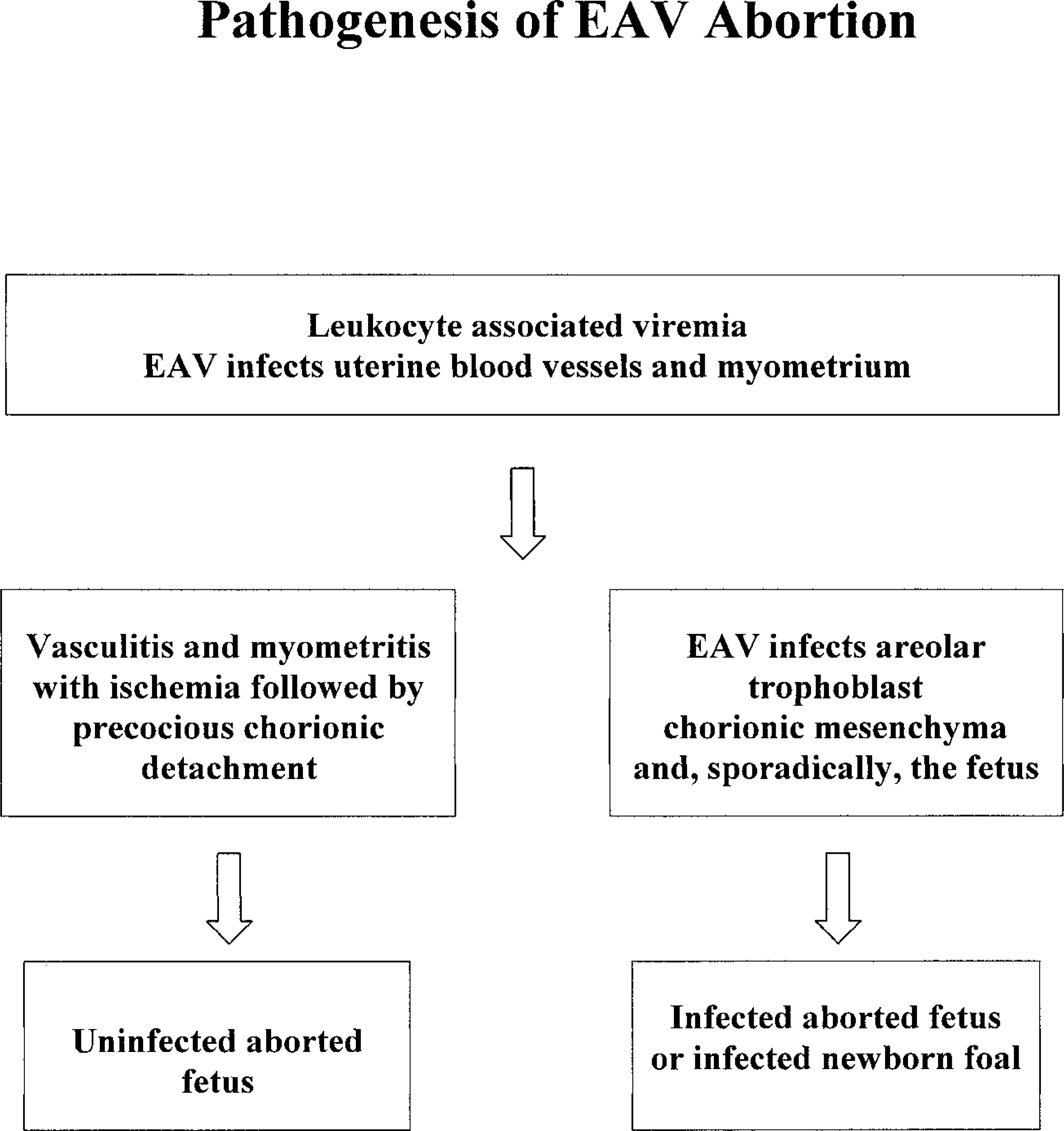
Pathogenesis of EAV abortion.
Diagnosis
The diagnosis of EVA is based on demonstration of lesions and the etiologic agent and/or seroconversion.
Serology and virology
The detection of seroconversion with complement-dependent virus neutralization performed using the Bucyrus strain in EAV-infected animals is a reliable method for identifing EAV infection in horses. Some foals 13 , 54 have presuckle positive serology tests for EAV, suggestive of in utero infection. Postsuckle testing would be invalid because of passive transfer of maternal immunity in seroconverted mares. 13
Tissue culture cell lines generally used to isolate EAV are RK-13 cells, Vero cells, and equine lung cells.
In addition to immunohistochemistry, other molecular techniques, such as reverse transcription polymerase chain reaction, have been used to identify the presence of EAV, especially in regard to genital transmission of the virus. 46 , 49 The application of these techniques to identify viral RNA in tissues may be very useful for diagnostic purposes and further studies, although sensitivity and specificity for routine diagnostic use still need to be proven.
Differential diagnoses
There are several infectious and a few noninfectious diseases that should be considered as differential diagnoses for EVA.
EHV-1 and, sporadically, equine herpesvirus 4 induce late abortion and stillbirth in horses. 57 , 60 In neonates it is difficult to distinguish between EHV and EAV pneumonia macroscopically, whereas at the microscopic level the difference is rather clear. EHV-1 induces necrotizing bronchiolitis and interstitial pneumonia with intranuclear viral inclusions, chromatin margination, and fragmentation. Large quantities of intranuclear and intracytoplasmic EHV antigen can be immunocytochemically observed within epithelial cells, macrophages, and pulmonary endothelial cells of fetuses. 11 , 42 In addition, EHV-1–infected fetuses and neonatal foals may present with multifocal coagulative necrosis of various organs, in particular liver, intestine, and lymphoid organs, which are also the ideal specimens for histologic and immunocytochemical diagnosis. 58 EHV-1 induces encephalomyelopathy secondary to vasculitis, 26 with virus antigen localizing within endothelial cells, myocytes, and pericytes. 12 , 59 Encephalomyelitis has not been associated with EAV infection. EHV-1 also may rarely induce nonneurologic fatal disease in young adult horses with severe vasculitis that is difficult to distinguish histologically from EVA infection (F. Del Piero, unpublished).
Equine adenovirus 37 is a rare cause of death in horses and almost exclusively involves Arabian and Thoroughbred foals affected by combined immunodeficiency characterized by severe atrophy of the lymphoid tissues. Foals die because of a severe necrotizing bronchopneumonia. Large intranuclear basophilic Cowdry type B viral inclusions are seen within the bronchial epithelium and are also occasionally located within the exocrine pancreas. The identifiable viral antigen is present in smaller quantities than in EHV-1 and EAV infection and is almost exclusively intranuclear.
Influenza orthomyxovirus infections accompanied by bacterial infections cause interstitial and bronchopneumonia. 28 Vasculitis is not a characteristic feature. The orthomyxovirus antigen is abundant within the cytoplasm and nucleus and can be detected in pulmonary specimens. 50
The acute and subacute forms of lentivirus-induced equine infectious anemia present with gross and histologic lesions 33 that, with the exception of bone marrow hyperplasia and dyserythropoiesis, the absence of vasculitis, and the possible occurrence of granulomatous meningoencephalomyelitis, 38 could be mistaken for EVA. Viral RNA can be identified systemically in cells of macrophage lineage. 44
The orbivirus of African horse sickness causes acute pulmonary, retroorbital, and muscle edema with ascites, hydrothorax, and hydropericardium. Histologically, the edema may be associated with mild perivascular mononuclear infiltrate, which may resemble an arterivirus lesion. The virus is localized within the cytoplasm of endothelium and macrophages. 3 , 34 , 61
The morbillivirus of Hendra disease 41 may cause a fatal infection and induces hemorrhagic pneumonia with typical endothelial cell syncytia. Vascular lesions resembling EVA can be systemically distributed, particularly in the kidney. The viral antigen is localized within endothelial cells, and filamentous paramyxovirus structures may be identified ultrastructurally. 24
Getah virus, of the alphavirus subgroup of the Togaviridae, can cause fever, rhinorrhea, and occasional exanthema, limb edema, lymphopenia or monocytosis, and abortion and must be included among the differential diagnoses for EVA. 30
Purpura hemorrhagica 31 and septic shock may induce systemic hemorrhages and occasionally vascular changes 48 resembling the severe lesions of the rare fatal EVA cases.
The toxic plant hoary alyssum (Berteroa incana) induces fever, hemolysis, limb edema, laminitis, gastroenteritis, and abortion and can mimic EVA clinical presentation and gross lesions. 20 , 25
Differential diagnoses should also include infectious and noninfectious abortion, and each of these forms will present more or less characteristic lesions. 23 , 32 , 57
Conclusions
Although not frequently diagnosed as a cause of abortion, neonatal mortality, and disease in adults, EVA is an important and perhaps emerging disease that can result in severe economic losses for the equine industry. Complete pathologic examination associated with the available ancillary procedures will guarantee a directed and accurate diagnosis.
Footnotes
Acknowledgements
I am very grateful to Mrs. A. Wensky-Roberts of the Image Lab for the photography.
