Abstract
Bilateral anophthalmia was discovered in a male rat (No. 1) and unilateral anophthalmia was found in the left eye of two female rats (Nos. 2 and 3) derived from a Fischer 344 inbred colony. One male rat (No. 4), a littermate of No. 3, had externally normal eyes, but his left eye had severe retinal atrophy, which was detected by ophthalmoscopy. The eyelids in both eyes of No. 1 and in the left eyes of Nos. 2 and 3 were present. Radiography of the skull revealed small optic foramina on both sides of No. 1 and on the affected side of Nos. 2, 3, and 4. Histologically, both globes and optic nerves (ONs) of No. 1 and the left globe and ONs of Nos. 2 and 3 were completely missing. Diffuse retinal degeneration in the left globe and axonal degeneration in the left ON and the right optic tract were observed in No. 4. Dysplasia of the sphenoid bone associated with stenosis of the optic foramen was detected on the affected side of all rats. Thus, ON aplasia in anophthalmic rats and atrophic ON in a rat with retinal degeneration seem to be closely related to stenosis of the optic foramen.
Anophthalmia and microphthalmia are rarely occurring, severe congenital ocular malformations in humans and animals. 13 , 16 The distinction of anophthalmia from microphthalmia can be made only by histological examination, because the former is characterized by no trace of any ocular structures, whereas the latter retains some remnants of ocular tissues. 16 In rats with similar ocular anomalies, it has been described that anophthalmia may be preceded by microphthalmia 10 or that anophthalmia represents the most severe manifestation of a spectrum of microphthalmia. 9 Thus, both conditions do not necessarily relate to entities of different cause or pathogenesis. 10 Spontaneous anophthalmia and microphthalmia have been reported in almost every species of laboratory and farm animal. 16 Similar conditions have been experimentally induced in rats with many physical and chemical agents, as reviewed by Szabo. 16 On the other hand, retinal degeneration in rats has been detected in association with inherited diseases, phototoxicity, chemical administrations, infectious diseases, and aging. 4 , 9 Stenosis of the optic foramen has been described in anophthalmia or optic nerve (ON) hypoplasia in humans 12 , 15 and ON hypoplasia in a pup. 5 Furthermore, the relationship between the changes occurring in the central nervous system, particularly the ON and its associated bony covering, has been well documented in vitamin A–deficient mammals 1 , 6 and birds. 7 In rats, however, none of previous workers focused their attention on the relationship between morphological changes in the optic foramen and anophthalmia or ON defects. In the present paper, we describe morphological changes in the oculovisual system and the optic foramina in Fischer 344 rats with anophthalmia and retinal degeneration.
The rats were originally derived from the F344/DuCrj strain of Charles River Japan, Inc. (Kanagawa, Japan). They were housed in polycarbonate cages in a barrier-sustained room controlled at 23 ± 2 C, 50 ± 20% relative humidity, and a 12-hour light–dark cycle. The animals had free access to a standard laboratory diet for rats (CRF-1, Charles River Japan, Inc.) and tap water. A male rat (No. 1) with bilateral anophthalmia was found in a colony. To evaluate possible inheritance of the lesions, the rat was mated with a female littermate and then mated with five female rats from the resultant offspring. In litters from the parent–offspring mating, a female rat (No. 3) with unilateral anophthalmia was discovered in one litter (seven males and four females). A male rat (No. 4) with unilateral retinal degeneration was a littermate of No. 3. No ocular abnormalities were found in the other offspring or in litters obtained by four consecutive parent–offspring matings. A female rat (No. 2) with unilateral anophthalmia was from another litter.
Clinically, none of the rats in the colony showed abnormal signs except for ocular lesions. The eyelids in both eyes of No. 1 and the eyelids of the left eyes of Nos. 2 and 3 were present but closed and stained reddish-brown with ocular discharge. The face on the affected side of these rats appeared somewhat flattened. All the offspring were examined ophthalmoscopically at 10 weeks of age using an ophthalmoscope (AU-12205, Welch Allyn, NY) and a fundus camera (RC-2 model-621, Kowa Co., Ltd., Tokyo, Japan). No. 4 had diffuse retinal atrophy in his left eye, showing narrowed retinal vessels, enhanced fundal reflection, and edematous optic disc (Fig. 1). Radiographs of the skulls of the four affected rats and two normal littermates of Nos. 2 and 3 were taken by placing a film underneath the head of each rat anesthetized with thiopental sodium, 2 ml/kg, i.p. Both optic foramina of No. 1 were indistinct due to increased X ray absorption, suggesting altered growth of the sphenoid bone. The left optic foramina of rat Nos. 2, 3, and 4 were smaller in diameter than the right ones (Fig. 2).
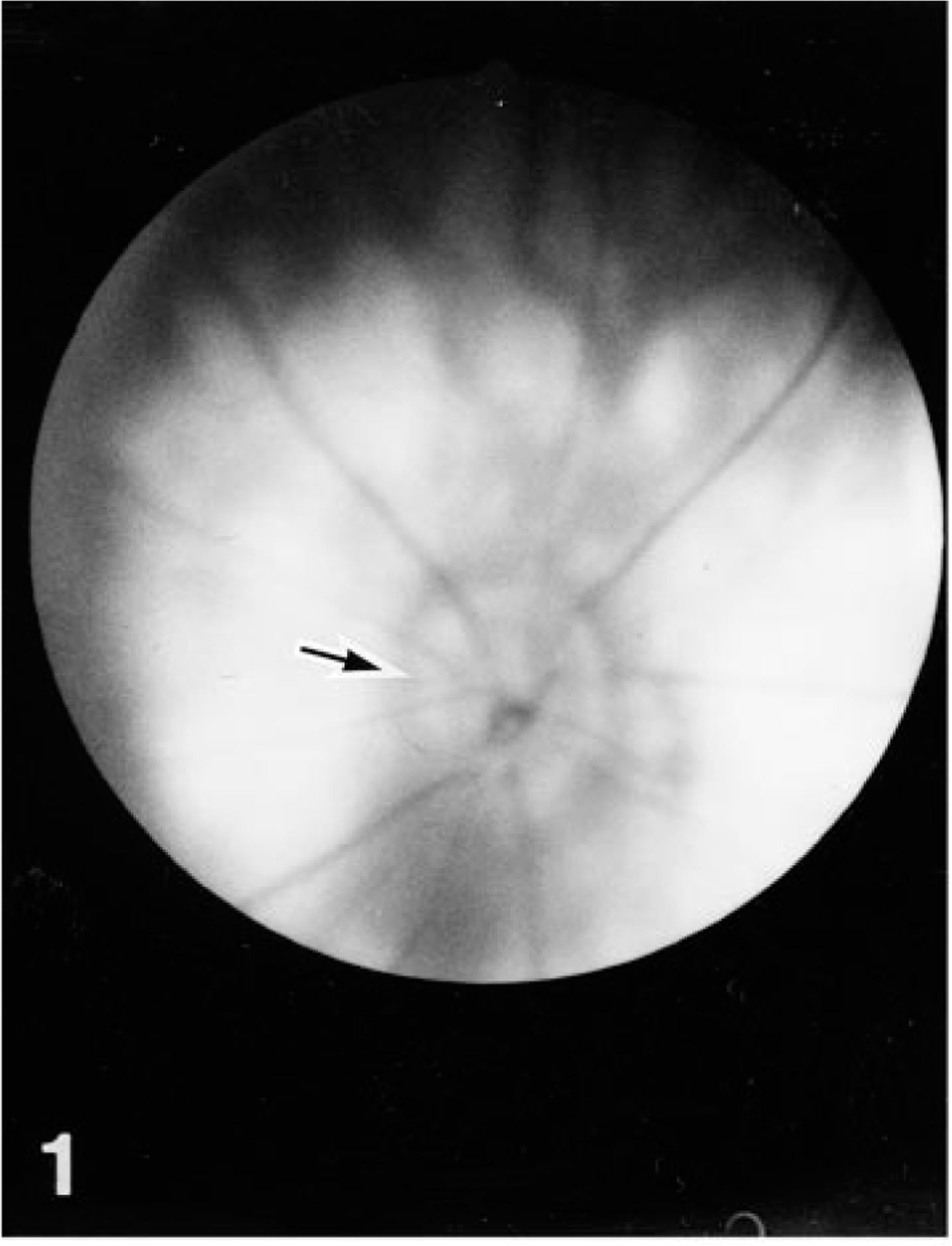
Fundus; rat No. 4. The left fundus reveals diffuse retinal atrophy with high retinal reflectance, slender retinal vasculatures, and slightly swollen optic disc (arrow).
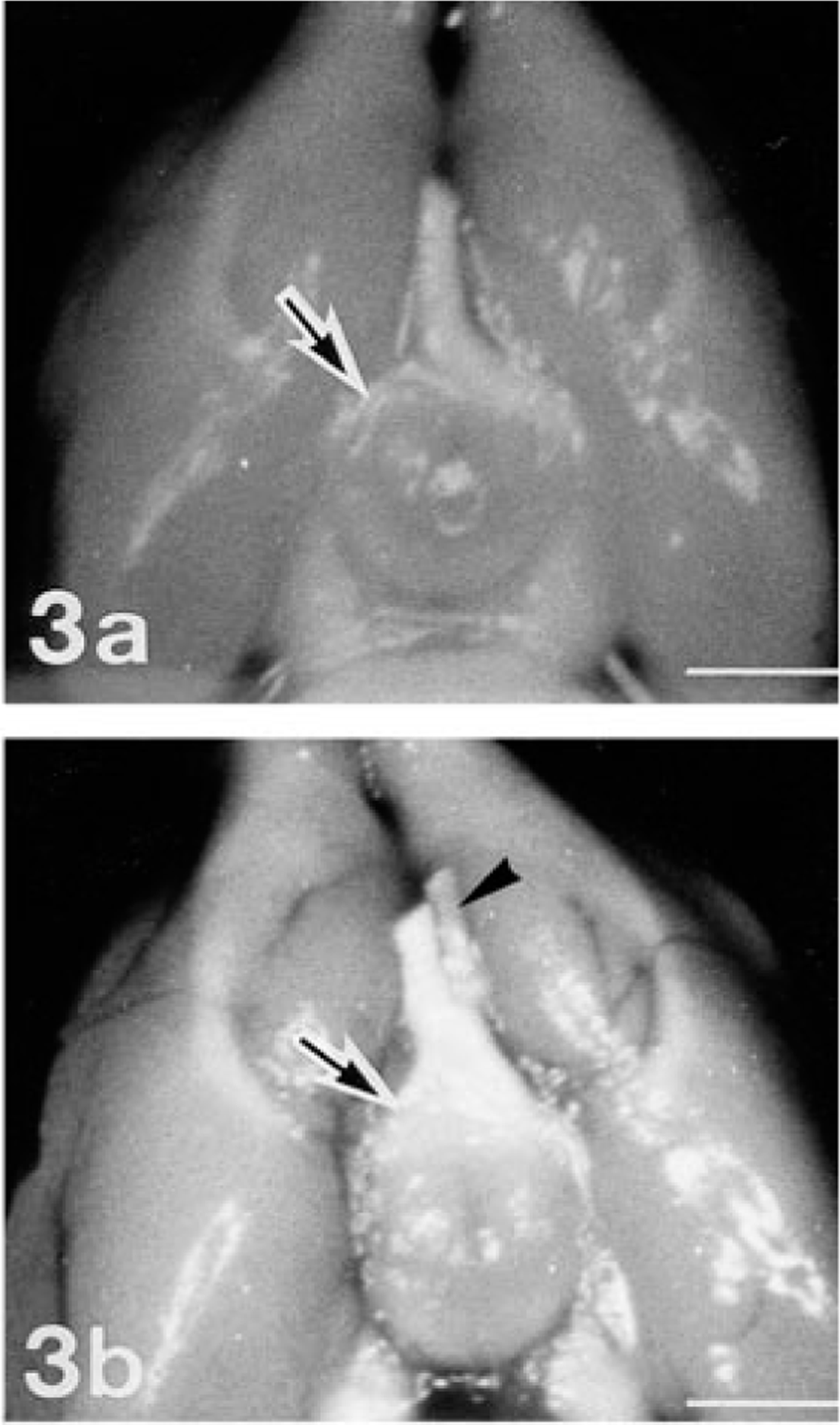
These rats and the other offspring were euthanatized by ether inhalation and subjected to a complete necropsy. Grossly, both ONs of No. 1 and the left ON of Nos. 2 and 3 were completely missing in the intracranial cavity (Fig. 3a). The optic chiasm and optic tract on both sides in No. 1 and those on the right side in Nos. 2 and 3 were rudimentary. In No. 4, the left ON and the right half of the optic chiasm and optic tract were reduced in volume (Fig. 3b). The brain with the intracranial portion of ONs and the skull with the globes and extracranial portions of the ONs were fixed in 10% neutral-buffered formalin. Three tissue blocks containing the orbit, globe, optic foramen, and ON were trimmed after the skull had been decalcificated with 5% trichloroacetic acid. Three tissue blocks comprising the proper anatomic position of the optic chiasm, optic tract, lateral geniculate body, and superior colliculus were trimmed from the brain. The specimens were embedded in paraffin, sectioned serially, and stained with hematoxylin and eosin (HE). Selected sections were stained by the Azan, Bodian, and Klüver-Barrera (KB) methods. 3 In addition, an avidin–biotin–peroxidase complex technique 8 with antibodies to glial fibrillary acidic protein (GFAP), myelin basic protein (MBP), and ubiquitin (DAKO Corp., Carpinteria, CA) was applied.
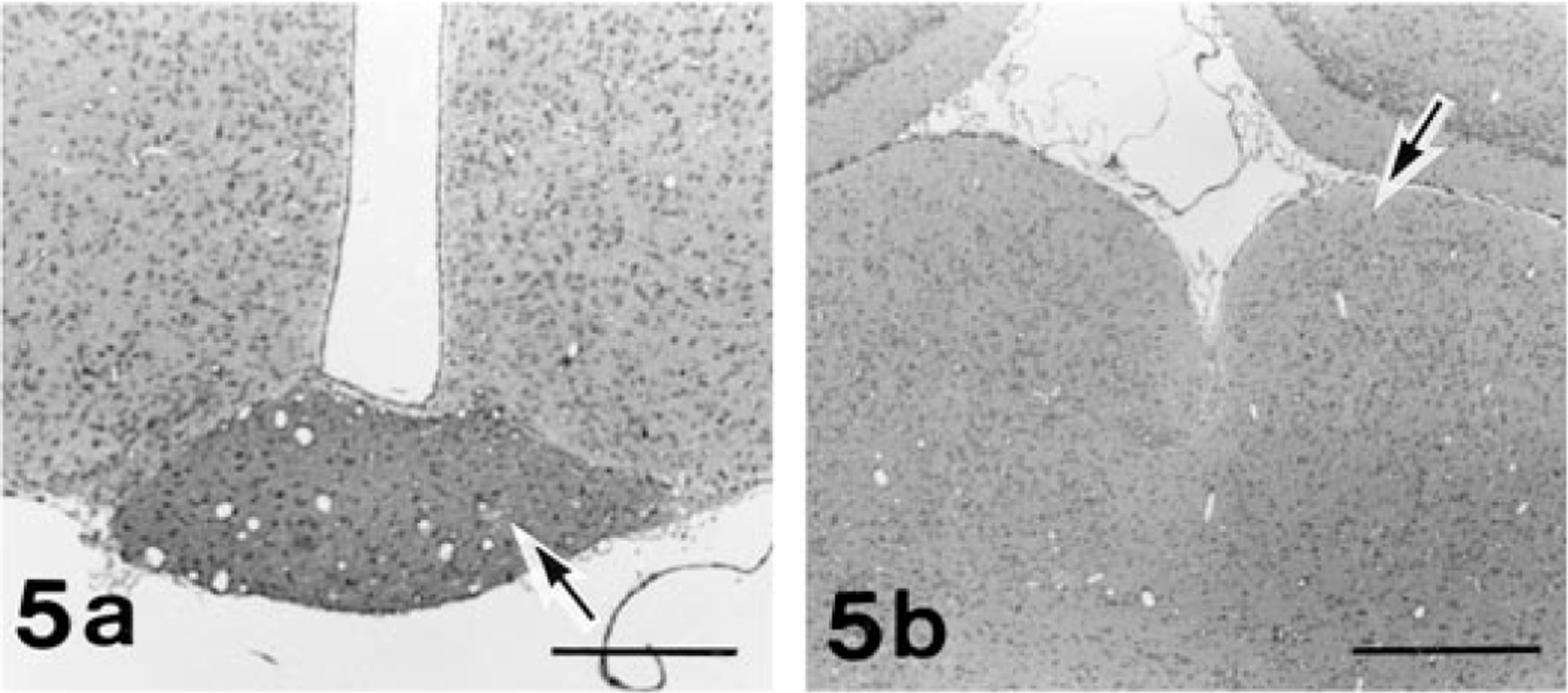
Histologically, no traces of any ocular structures were detected despite an extensive search with multiple histological sections of both orbits of No. 1 and of the left orbit of Nos. 2 and 3. Within the orbits on both sides of all the cases examined, normal-appearing Harderian glands and lacrimal glands were observed. In the left orbit of No. 1, there were irregular ductlike structures surrounded by connective tissue that appeared to be vestigial mesodermal elements such as the nerve sheath. The left retina of No. 4 was characterized by severe atrophy, fragmentation, and loss of all retinal layers in the whole retina. There were no noticeable changes in any structures of the contralateral globes of Nos. 2, 3, and 4. In the portion of the optic foramen, the sphenoid bone on the affected side of Nos. 1, 2, 3, and 4 was irregularly shaped, and dimensions of the optic canal were reduced (Fig. 4). These observations coincided with the radiographic findings. The dysplastic bone consisted of normal mature osseous tissue. Both ONs of No. 1 and the left ON of Nos. 2 and 3 were completely absent in the optic foramen. The optic chiasm and optic tract were rudimentary, and the lateral geniculate body and superior colliculus were hypoplastic on both sides of No. 1 and on contralateral side to the affected globe of Nos. 2 and 3 (Fig. 5). Neither degenerative nor reactive changes were detected in these areas. In contrast, examination of rat No. 4 revealed atrophy of the left ON and the right optic tract, lateral geniculate body, and superior colliculus (Fig. 6). In this ON pathway, the nerve fibers decreased in number, and demyelination of the ON fibers was demonstrated by the KB method and MBP immunostaining. Degenerated axons were fragmented or swollen and were accompanied by vacuolation in the affected area. Marked astrogliosis and deposits of ubiquitin-positive granules were observed in the degenerative lesions (Fig. 6).
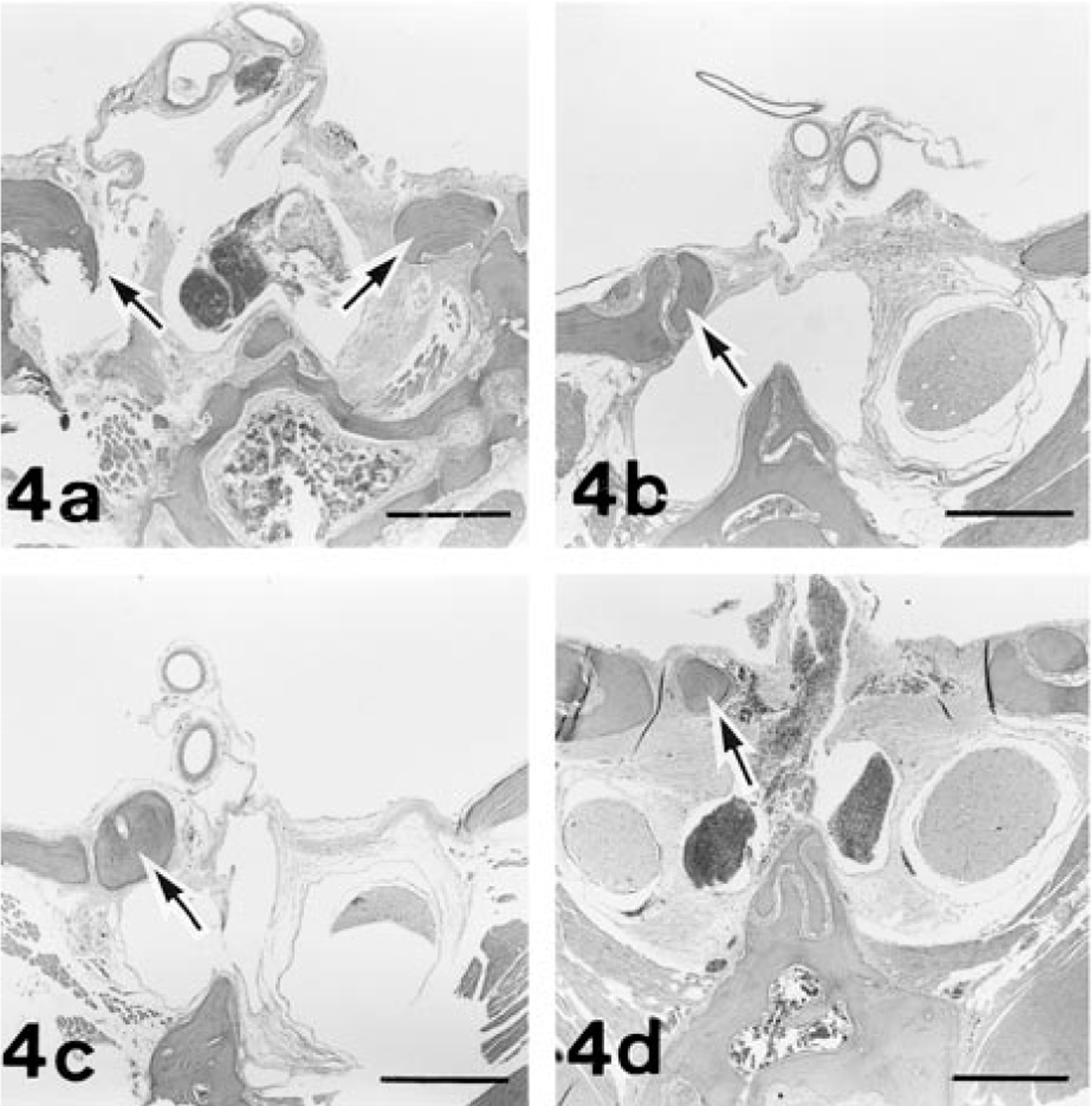
Optic canals; frontal section. Fig. 4a. Rat No. 1. Irregularly shaped sphenoid bone on both sides (arrows) is causing stenosis of the canals. No optic nerves on both sides are seen. Fig. 4b represents rat No. 2; Fig. 4c represents rat No. 3. The left canal of each rat is stenotic due to irregularly shaped sphenoid bone (arrow). No left optic nerve is seen. Fig. 4d. Rat No. 4. The dysplastic sphenoid bone (arrow) is present on the left side, and the atrophic optic nerve is in the stenotic left canal. HE. Bar = 500 µm.
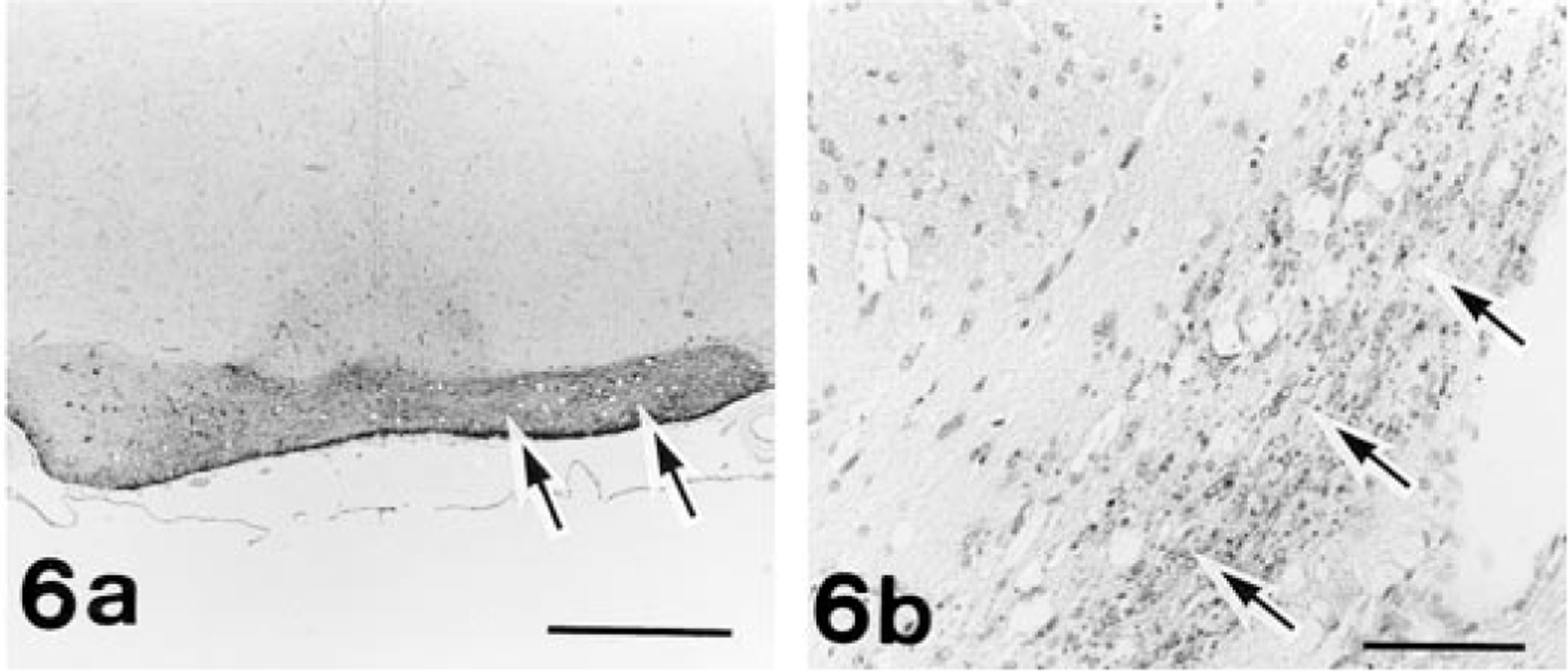
Brain, frontal section; rat No. 4. Fig. 6a. Optic chiasm. An area of the reduced right half shows a marked increase in immunoreaction for GFAP (arrows). Bar = 500 µm. Fig. 6b. Optic tract. An area of the degenerative right tract shows granular immunoreaction for ubiquitin (arrows). Bar = 300 µm. Avidin–biotin complex method with hematoxylin counterstain.
Ocular anomalies in rat Nos. 1, 2, and 3 were clinically and morphologically diagnosed as anophthalmia because no remnants of ocular tissues were detected in the affected orbits. Neither severely deformed anomalies nor degenerative lesions were detected in the brains of these cases, suggesting that the present anophthalmic cases may have begun during early forebrain differentiation as described in the primary type of anophthalmia. 13 On the other hand, retinal degeneration was observed in the left globe of No. 4 at 10 weeks of age. Retinal degeneration in rats is caused by phototoxicity, aging, chemical administration, or inherited diseases, and these conditions have been described as affecting both eyes. 4 , 9 Externally normal appearances of the globe and degenerative changes of the affected ON pathway in No. 4 suggest that the globe had formed normally and then underwent degeneration of the retina and ON pathway. Hence, it appears that unilateral retinal degeneration in No. 4 may also be associated with the foraminal dysplasia.
All four rats had stenosis of the optic foramen on the affected side, which was presumed to have resulted from dysplasia of the sphenoid bone. No inflammatory reaction was observed in the sphenoid bone and its surrounding tissues, supporting the idea that these changes may represent a developmental deformity. Optic nerve lesions and bone changes similar to those described herein have been induced in calves 6 and chickens 7 by raising them on a vitamin A–deficient diet. The present cases could not be attributed to vitamin A deficiency because the defect occurred sporadically in the colony, although all of the rats were fed with the same formulated diet and no abnormal components were detected in the diet. Moreover, no metaplastic change to stratified squamous epithelium typical of vitamin A deficiency was seen in any part of the body. It has been reported that the human sphenoid bone consists of at least 13 ossification centers at birth and that most of them are assimilated into the sphenoidal body during the first 2 years. 11 Analysis of the results obtained by the techniques of ultrasonography and axial tomography suggested that the optic canal dimensions corresponded closely to the size of the optic nerve in the human hypoplastic eyes. 2 The present cases of ON defect in the anophthalmic rats and ON atrophy associated with retinal degeneration appeared to have developed during late fetal development or early postnatal life. Hence, it seems that defects in both ON and sphenoid bone may be closely related to each other in their development. However, the etiology and pathogenetic mechanisms of the present cases should be clarified by further studies.
A genetic involvement in the occurrence of anophthalmia and microphthalmia has been suggested in many species, but the evidence for a genetic basis is not conclusive. Breeding studies on microphthalmic and anophthalmic rats indicated that the conditions were inherited as an autosomal recessive trait. 10 , 14 Our test mating, conducted using limited numbers of rats and generations, suggested that the present ocular anomalies were not hereditary.
Footnotes
Acknowledgements
The authors are grateful to Y. Ogata, M. Kaneko, H. Suzaki, and S. Ishikawa for their technical assistance.
