Abstract
Twelve cases of adult-onset blindness were identified in a flock of 130 polled Wiltshire sheep in New Zealand over a 3-year period. Affected sheep developed night blindness between 2 and 3 years of age, which progressed to complete blindness by 4 to 5 years of age. Fundic examination findings included progressive tapetal hyperreflectivity and attenuation of retinal blood vessels. Histologically, the retinas had a selective loss of rod photoreceptors with initial preservation of cone photoreceptors. Retinal degeneration was not accompanied by any other ocular or central nervous system abnormalities, and pedigree analysis suggested an inherited basis for the disease. Mating an affected Wiltshire ram to 2 affected Wiltshire ewes resulted in 6 progeny that all developed retinal degeneration by 2 years of age, while mating of the same affected ram to 6 unaffected ewes resulted in 8 unaffected progeny, consistent with autosomal recessive inheritance. Homozygosity mapping of 5 affected Wiltshire sheep and 1 unaffected Wiltshire sheep using an OvineSNP50 Genotyping BeadChip revealed an identical-by-descent region on chromosome 5, but none of the genes within this region were considered plausible candidate genes. Whole-genome sequencing of 2 affected sheep did not reveal any significant mutations in any of the genes associated with retinitis pigmentosa in humans or progressive retinal atrophy in dogs. Inherited progressive retinal degeneration affecting rod photoreceptors has not been previously reported in sheep, but this disease has several similarities to inherited retinal dystrophies in other species.
Keywords
Blindness in sheep is a rarely reported clinical entity, although visual impairment may be underrecognized as blindness does not appear to negatively impact the feed intake or productivity of sheep. 1 Reported causes of blindness in sheep include toxic causes such as chronic ingestion of bracken fern, 19 other plant toxicities 44 and overdosage of the anthelmintic closantel, 15,45 various infectious diseases, 7 nutritional deficiencies (particularly vitamin A deficiency), 14,17 and inherited disorders including achromatopsia (day blindness) in Awassi sheep, 37,41 microphthalmia in Texel sheep, 3,38 and cataracts in Romney sheep. 8 Blindness can also be observed as part of more generalized disorders, such as the neuronal ceroid lipofuscinoses in South Hampshire, 16,26 Borderdale, 21 and Merino 11 sheep. Apparent blindness can be the primary presenting sign of some noninherited diseases primarily affecting the central nervous system, such as polioencephalomalacia. 33
Sporadic cases of adult-onset blindness were first identified in a single breeding flock of Wiltshire sheep in New Zealand in 2008. Over time, the number of affected sheep in this flock gradually increased, with 7 out of 130 ewes identified as having visual impairment in 2011 on the basis of farmer observations and ocular examinations of sheep by a veterinarian. Affected sheep appeared to have normal vision until 2 to 3 years of age, when visual impairment became evident as an inability to negotiate gateways or recognize fences in low-light conditions. Vision loss progressed over time and affected sheep 5 years of age or older exhibited complete or near-complete blindness, and were difficult to muster in full daylight. Initial clinical examinations of affected sheep revealed that abnormalities were confined to the retina, with lesions suggestive of retinal degeneration or atrophy.
The aim of this study was to describe the clinical signs, and ophthalmoscopic and histopathological findings associated with adult-onset night blindness in Wiltshire sheep in this flock. In addition, the underlying genetic basis of this disease was investigated through a small breeding trial, homozygosity mapping using an OvineSNP50 Genotyping BeadChip in 5 affected and 1 clinically normal sheep, and whole-genome sequencing of 2 affected sheep and 1 clinically normal sheep.
Materials and Methods
Evaluation of Visual Impairment in Sheep
On the affected property, sheep were exclusively grazed outdoors on ryegrass/white clover pastures, with supplementary winter feed of grass silage that was made on farm. All paddocks were well-fenced and no known toxic plants were observed in or adjacent to any of the land grazed by the sheep. 20 To identify visually impaired sheep, all adult sheep on the affected property were run through an outdoor maze. Maze testing was performed at dusk in low ambient light conditions over several evenings, as prior observations suggested that scotopic (low light) vision was lost prior to photopic (bright light) vision in affected animals. Sheep performance in the maze was assessed by masked evaluators, and any sheep that showed slight hesitation or completed the maze more slowly than their cohorts were assessed individually including fundic examination. Ophthalmic examinations were conducted in a darkened indoor pen using a binocular indirect ophthalmoscope (Welch Allyn Inc) and a Pan Retinal 2.2 lens (Volk Optical Inc). A series of retinal images were obtained from each sheep using a handheld digital fundus camera (ClearView Optical Imaging System, Optibrand). Where necessary, a drop of tropicamide (Mydriacyl 1.0%, Alcon Laboratories) was applied to the corneal surface of each eye to facilitate pupillary dilation. A heparinized blood sample was collected via jugular venipuncture from each sheep that underwent ophthalmic examination. These animal manipulations were performed as part of the veterinary medical evaluation of the blindness on this farm and were not part of a research trial requiring ethical approval.
Ocular Histopathology
On the farm of origin, ocular tissue samples were collected postmortem from sheep slaughtered for human consumption. The eyes were enucleated shortly following death using a transconjunctival approach, and the posterior pole of each eye was separated from the anterior portion using a cross-sectional incision a few millimeters behind the limbus, posterior to the lens. The posterior part of each eye was immersed in Bouin’s solution for 36 to 48 hours before being washed with water, placed in 10% formalin, and subsequently dehydrated in graded alcohol and embedded whole in paraffin wax. The posterior portions of each eye were then trimmed to the size of a standard tissue cassette using a 6-inch mini hacksaw, and the smaller sections were each re-embedded in paraffin. From these blocks, 3-µm-thick sections were cut and stained with hematoxylin and eosin (HE). Brain samples were fixed in 10% buffered formalin prior to routine trimming and processing for histopathology.
Breeding Trial
A 6-year-old Wiltshire ram and two 4-year-old Wiltshire ewes, all with retinal degeneration on fundoscopic examination, were moved from the farm of origin in Canterbury, New Zealand, to a farm in Palmerston North, New Zealand, and were bred over 2 consecutive years. Both ewes were daughters of ewes that also had confirmed retinal degeneration. The same affected ram was also mated to 6 clinically normal, mixed-breed sheep on the Palmerston North farm. Progeny were closely observed during regular mustering for any signs of visual impairment, and fundoscopic examination and retinal photography as described previously was performed at approximately 1, 3, 6, 12, and 24 months of age. A heparinized blood sample was collected via jugular venipuncture from each sheep involved in the breeding trial. Any sheep in the breeding trial that showed severe visual impairment were euthanized by intravenous barbiturate overdose or captive bolt and exsanguination, with the eyes collected for histopathology as previously described. All animal manipulations (including euthanasia) on sheep that were part of the breeding trial were approved by the Massey University Animal Ethics Committee (approval numbers 14-24 and 17-17).
Molecular Genetics
DNA used for homozygosity mapping was extracted from heparinized blood of 6 Wiltshire sheep, including 5 sheep with confirmed retinal degeneration and 1 ophthalmoscopically normal sheep, using a Roche MagNA Pure automated analyzer. The Ovine SNP50 BeadChip (Illumina) was used with standard protocol at Iowa State University, Iowa, USA. SNPs were retained if they had a call rate of >80% and a GenTrain score of 0.25 or greater.
Homozygosity mapping was performed using an IBD program with awk, join, and sort UNIX commands to identify any strings of consensus homozygous markers with a length of more than 10 SNPs (single-nucleotide polymorphisms). All markers that were monomorphic in both the affected and normal sheep were excluded from the analysis, in order to define homozygous regions with consecutive SNP loci only existing in all the affected sheep. Genes in regions with at least 10 contiguous homozygous loci in affected sheep were identified and examined for potential involvement through comparison to the reference sheep genome (Oar v3.1).
DNA to be used for whole-genome sequencing was extracted from the stored heparinized blood of 2 affected Wiltshire sheep and 2 unrelated adult sheep with normal vision using the MagAttract HMW genomic DNA extraction kit (Qiagen GmbH) as per the manufacturer’s instructions. Sequencing was performed at Iowa State University DNA sequencing facility, where a fragment library was prepared using the Illumina TruSeq DNA PCR free library preparation kit (Illumina Inc) with a 550 bp insert size. Two lanes of paired-end reads (2 × 100 bp) were then obtained using an Illumina HiSeq 3000 machine (Illumina Inc). The raw reads were filtered and trimmed to remove adaptor sequences, and low-quality reads using FastQC (http://www.bioinformatics.bbsrc.ac.uk/projects/fastqc/) and TrimGalore! Cutadapt with paired-output and quality-base phred score 20. 25
The resulting fastq files, designated R1 and R2, were concatenated and then aligned to the reference sheep genome (Oar v3.1) using BWA-MEM 0.7.15 with –M (marks shorter split hits as secondary) and –R (read group ID attached to every read in output). The resulting SAM file was sorted, converted to BAM format, and duplicates marked in the BAM files using Picard tools 1.139 (https://broadinstitute.github.io/picard). Variants were called using GATK 3.4-46 HaplotypeCaller in DISCOVERY genotyping mode, using emitRefConfidence GVCF, variant_index_type LINEAR, and variant_index_parameter 12800. Otherwise default parameters were used. Individual g.vcf files for each animal were merged using GATK GenotypeGVCFs mode with the default parameters and a minimum confidence threshold for calling and emitting variants of Phred quality score of 30 (https://www.broadinstitute.org/gatk/guide/tooldocs). Percentage of reads mapped was determined using Samtools flagstat, and GATK DepthOfCoverage mode was used to determine read coverage.
Genetic variant annotation and effect was predicted using SnpEff 4.3 and reference genome Oar_v3.1.86. 10 The results were then filtered using SnpSift in order to list the variants that resulted in a stop gain, stop lost, frame shift, or splice site variant, considered to be high-impact variants, that were homozygous in the 2 blind sheep, but homozygous wild type in the 2 unrelated sheep. 40 Genetic variant annotation and effect was also predicted using Ensembl Variant Effect Predictor (VEP 90.3) and reference genome Oar v3.1 with sift prediction. 27 The results were then filtered using VEP 90.3 filter mode for all genes associated with lysosomal storage diseases and variants within these genes considered deleterious by SIFT. Each variant was searched for in the Ovis aries 3.1 genome at either the Ensembl database or the European Variant archive (www.ensembl.orgorhttp://www.ebi.ac.uk/eva/). In addition, the function of each gene, or its likely involvement in human disease, was determined by searching in Online Mendelian Inheritance of Man (http://www.omim.org/) and National Center for Biotechnology Information, US National Library of Medicine (https://www.ncbi.nlm.nih.gov/gene). All variants in known human retinopathy genes (from Online Mendelian Inheritance of Man) and canine retinopathy genes (from Online Mendelian Inheritance of Animals; https://omia.org/home/) were also examined for all levels of potential disease causing variants (104 genes in total).
Results
Clinical and Histological Features
Visual impairment was identified in 11 adult ewes and 1 adult ram on the farm of origin. Six of these sheep were 4 years of age or less at the time of examination and were able to navigate a simple maze in bright light conditions, but not in low ambient light conditions at dusk. The remaining 6 sheep were 5 years of age or older when examined, and were frequently observed to follow fence lines, misjudge gateways, and were unable to complete the maze in both low- and bright-light conditions. All affected sheep had normal mentation and ambulation and were in good body condition, with no other significant findings on physical examination.
On ophthalmic examination, all affected sheep had bilaterally dilated pupils with variably diminished pupillary light reflexes. No abnormalities were observed in the cornea, anterior chamber or lens of most sheep, but a 4-year-old ewe was noted to have a small subcapsular cataract in the right eye. Retinal changes were bilateral and increased in severity with advancing age. All affected sheep exhibited attenuation and narrowing of retinal blood vessels, most notably the dorsal cilioretinal arterioles and venules, along with their smaller tributaries (Figs. 1–6). Affected sheep also exhibited patchy tapetal hyperreflectivity, which radiated laterally, with large, multifocal areas of the tapetum appearing brighter than usual. Sheep that were 5 years of age or older had depigmentation of the non-tapetal fundus (data not shown), which was not observed in younger sheep.
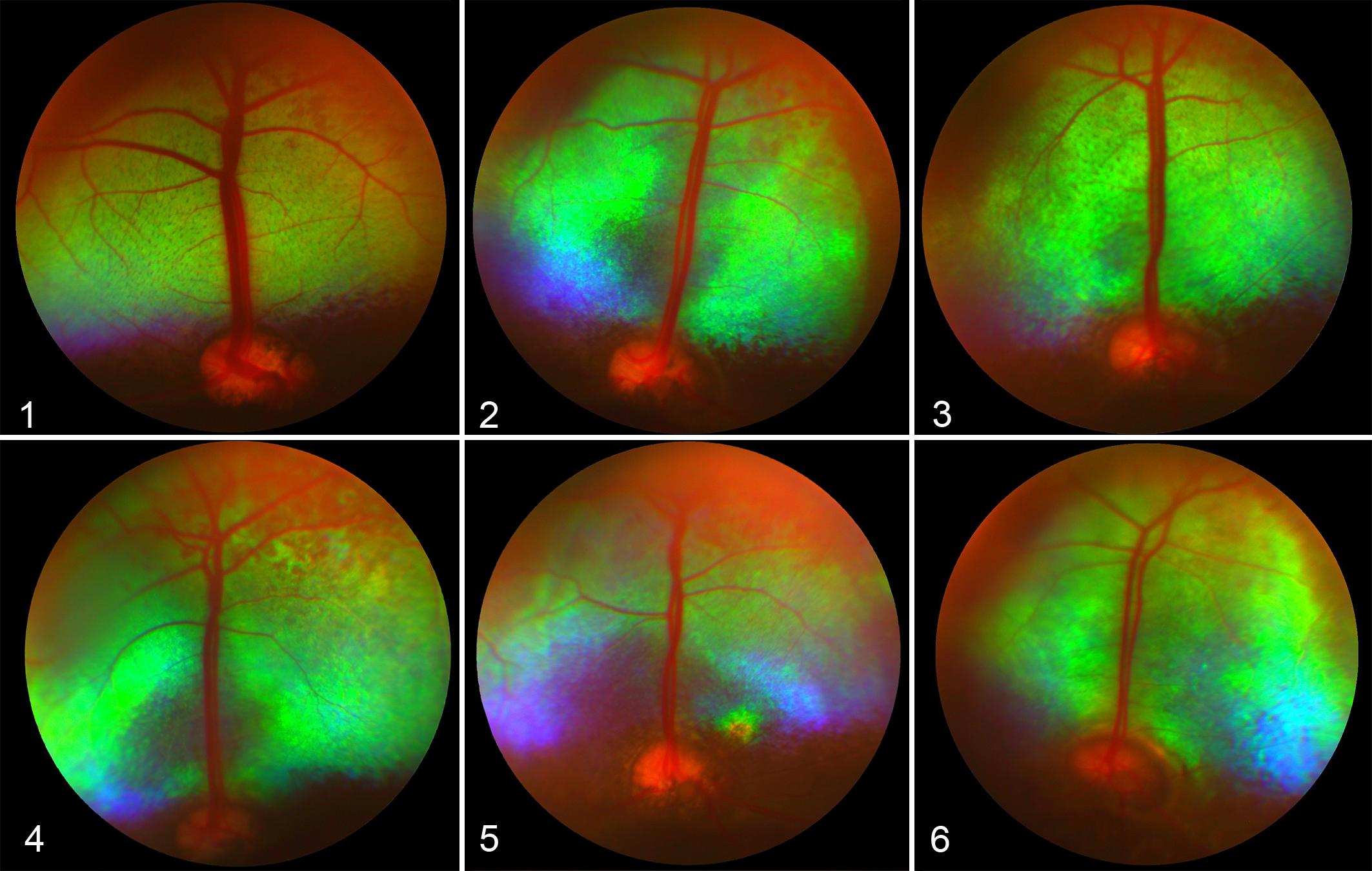
Fundus, 2-year-old Wiltshire sheep with normal vision (control).
The retina was examined histologically in 7 of the 12 initial cases. Compared with sheep with normal vision (Fig. 7), affected sheep exhibited loss of rod photoreceptors by 2 years of age, with no identifiable rod outer segments, inner segments or nuclei in both the tapetal and non-tapetal regions of the retina (Figs. 8, 9). Cone photoreceptors, characterized by broad eosinophilic inner segments, were present in these young sheep, but there was shortening of the outer segments of remaining cone photoreceptors. The outer nuclear layer was reduced to 1 to 3 layers of cone nuclei adjacent to the external limiting membrane, and there was mild attenuation of the outer plexiform layer. The inner nuclear, inner plexiform, ganglion cell, and nerve fiber layers appeared microscopically normal.
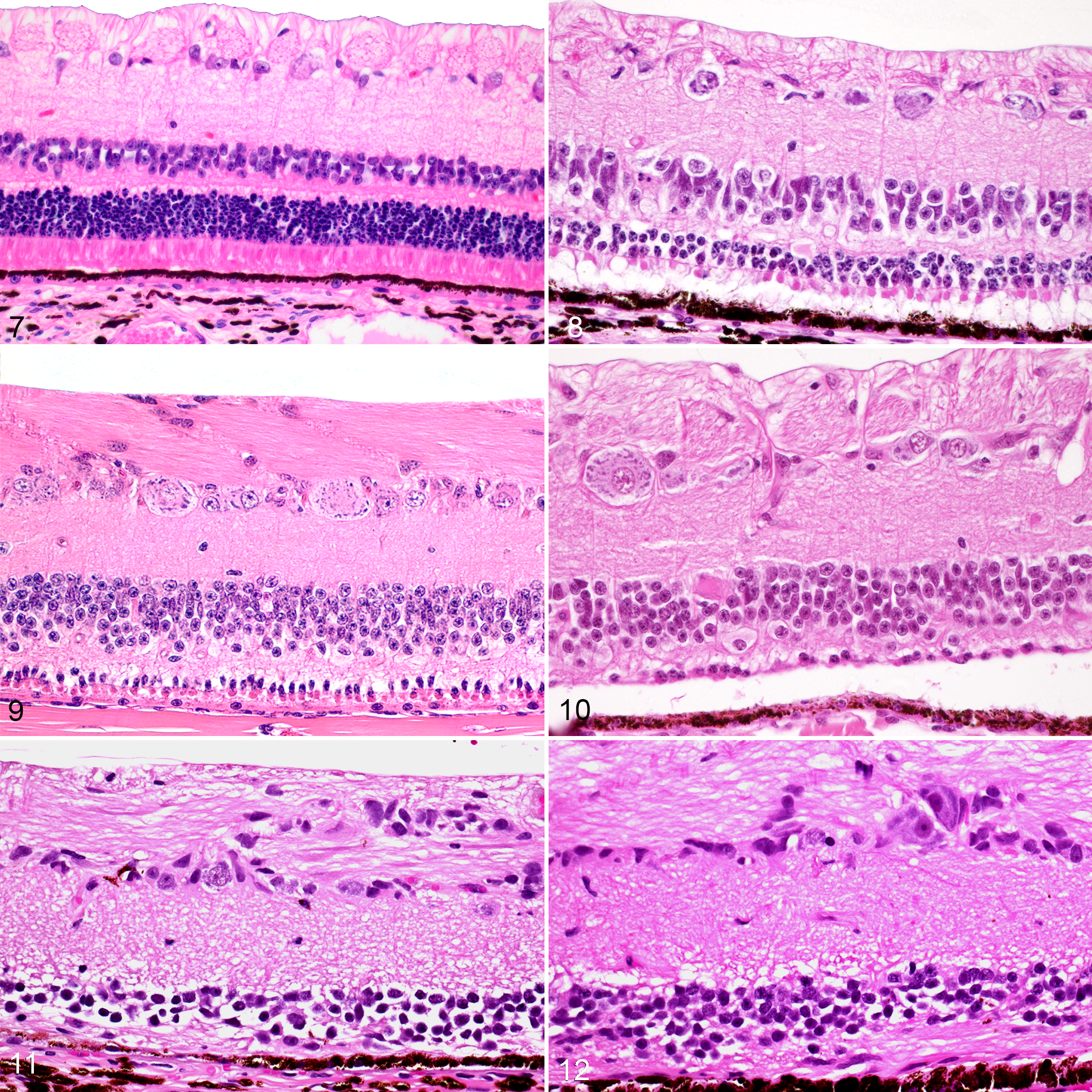
Retina, 4-year-old Wiltshire sheep with normal vision (control). Hematoxylin and eosin (HE).
With advancing age, retinal lesions became more severe and consisted of near-complete loss of all photoreceptors, accompanied by loss of the outer nuclear layer. Occasional small islands of shortened cone photoreceptors were present in the central aspect of the retina in sheep culled between 4 and 5 years of age (Fig. 10). Multifocally, the outer plexiform layer was thin or not appreciable in sheep 5 years of age or older, and in some sections the retinal pigment epithelium communicated directly with the inner nuclear layer, which was mildly to moderately attenuated (Figs. 11, 12). The inner plexiform, ganglion cell, and nerve fiber layers were histologically normal. Mild Wallerian degeneration was seen in the proximal aspect of the optic nerve in sheep aged 5 to 7 years. Sections of brain examined, including the visual cortex, did not contain significant microscopic lesions.
Breeding Trial
Matings between the affected ram and 2 of the affected ewes over 2 seasons produced 6 lambs that survived to weaning, as well as 5 lambs that were stillborn or died in the early neonatal period. Stillborn lambs had histologically normal retinas for their stage of development, but over the first 2 years of life all 6 surviving progeny had developed retinal lesions. The earliest change observed on fundoscopic examination was attenuation of retinal blood vessels, which was first detectable at 12 months of age on comparison of sequential retinal photographs. Small, focal areas of tapetal hyperreflectivity developed between 12 and 24 months of age. Histologically, the retina of 9- and 12-month-old lambs had mild attenuation of the outer nuclear layer and shortening of photoreceptor outer segments, but rod photoreceptors were still identifiable. One of the progeny was culled at 2 years of age, and in the retina of this sheep the photoreceptor outer segments were significantly shortened and the outer nuclear layer was reduced to 1 to 3 cells thick. The remaining 3 progeny were culled between 2.5 and 5 years of age and showed similar histologic retinal lesions to those described earlier in affected sheep of a comparable age on the original farm.
Mating the same affected ram to 6 clinically normal mixed-breed ewes resulted in 8 progeny that survived to weaning. One of the progeny was euthanized at 4 years of age and the retina was histologically normal. The remaining 7 progeny are currently 6 years old and all have normal retinal appearance on fundoscopic examination, with no clinical evidence of visual impairment. This supports a recessive mode of inheritance. If the disease was dominant and the sire was heterozygous, 4 of the progeny would be expected to be affected and 4 normal, in contrast to the observed 8 normal and zero affected sheep. A χ2 test for dominant inheritance gives χ2 = 8, df = 1, and P < .005, indicating that the hypothesis of dominant inheritance should be rejected.
SNP Analysis
All markers that were monomorphic in the affected and normal sheep were excluded from the analysis, leaving 1907 markers that were monomorphic in the 5 affected Wiltshire sheep. The largest homozygous segment consisted of 45 consecutive SNP loci, starting from SNP OAR5_96674157.1 and extending to SNP s59216, covering a region of 4.85 Mbp (OAR_ v3.1: 88 634 982 to 93 480 111) on the long arm of chromosome 5. There were 29 genes located in this region based on the ovine reference sequence within the NCBI Map Viewer (OAR_v3.1, annotation release 100). To include any genes located in the immediate vicinity of the homozygous SNP segment, the search area was extended from 88 000 000 to 94 000 000, and an additional 8 genes were identified (Supplemental Table S1). None of the genes within this 45-SNP region were considered plausible candidate genes based on their biological functions. However, a gene in the extended search area, G-protein coupled receptor 98 (GPR98), has been implicated in Usher syndrome type 2 in human patients, which is characterized by loss of rod photoreceptors (retinitis pigmentosa) and sensorineural hearing loss. All other homozygous segments encompassed 10 or fewer consecutive loci and were not further analyzed.
Whole-Genome Sequencing
Preliminary analysis of whole-genome sequencing produced a large number of variants between the affected sheep and the reference ovine genome. Filtering for high-impact recessive variants (including frame shift, stop gain/loss, and splice site mutations) produced 120 variants, all of which were either in genes not associated with the eye and retina or were known variants recorded in the European Variation Archive. In addition, the sequencing results were examined for any variants in 104 genes associated with retinal degeneration in human patients and dogs, including 77 genes known to be associated with retinitis pigmentosa in humans, an additional 24 genes associated with cone-rod dystrophy and a further 3 genes associated with progressive retinal atrophy in dogs but not retinitis pigmentosa in human patients. All variants detected in these genes were known normal variants that were included in the EVA database, and not known to cause disease. No disease-causing variants were detected in the GPR98 gene that was identified in the IBD region on homozygosity mapping.
Discussion
The ophthalmologic and histologic lesions in this flock of Wiltshire sheep are consistent with progressive retinal degeneration primarily affecting rod photoreceptors. Affected sheep had apparently normal vision at birth but developed detectable night blindness from 2 years of age onward. The severity of visual impairment, fundic examination findings, and retinal lesions worsened with advancing age, and the left and right eyes were equally affected in all sheep.
In normal sheep, rod photoreceptors predominate over cones, with 30 to 40 rods present for every cone. 7 In affected Wiltshire sheep, the number of rod photoreceptors progressively decreased over time, and in sheep with end-stage disease the cone photoreceptors were also lost. Rod photoreceptors have a very high sensitivity to light and are important in scotopic (low light) vision, 6 consistent with the affected sheep developing night blindness prior to day blindness. It is thought that the later changes observed in cone photoreceptors may be secondary to loss of trophic factors or lateral support normally provided by rod photoreceptors, 31 as similar findings have also been reported in primary rod dystrophies in human patients. 29
The initial presentation of retinal degeneration in these Wiltshire sheep had similarities to bright blindness due to chronic ingestion of bracken fern, 19 as sheep with bright blindness are typically 3 to 4 years old and have retinal atrophy with narrowing of retinal blood vessels and increased tapetal reflectivity. 47 However, the 2 diseases differ histologically, as in bright blindness there is swelling and fragmentation of both rod and cone photoreceptors, particularly cones, whereas in the disease in Wiltshire sheep there is significant loss of rod photoreceptors with relative sparing of cones. In addition, these affected Wiltshire sheep had never been exposed to bracken fern, and bright blindness has not been reported previously in New Zealand. 42 Other retinal diseases reported in sheep include closantel toxicity, 15 ocular toxoplasmosis, 35 vitamin A deficiency, 32 ceroid lipofuscinosis, 16,26 and other plant toxicities. 23,44 The features of retinal degeneration in these Wiltshire sheep were not compatible with any of these diseases, particularly as most also affect the central nervous system, whereas the brain was histologically normal in affected Wiltshire sheep.
Three of the 12 affected sheep in the original flock were daughters of other affected ewes, suggesting that the disease had an inherited basis. In the breeding trial, all progeny that developed retinal degeneration had 2 blind parents, while progeny resulting from matings between the blind ram and normal ewes all had normal fundoscopic appearance and normal vision at 6 years of age. This supports an autosomal recessive mode of inheritance. Inherited retinopathies are rare in sheep, 48 and to the authors’ knowledge, day blindness in Awassi sheep is the only inherited primary retinal disease reported in sheep to date. 41 In this disease, cone dysfunction is detectable in young lambs, but normal retinal histology is preserved, in contrast to the disease in Wiltshire sheep where severe loss of rod photoreceptors was observed histologically. Inherited retinopathies with selective loss of rod photoreceptors are well described in human patients, where they are referred to as retinitis pigmentosa. 46 The clinical course of retinal degeneration in the Wiltshire sheep closely parallels retinitis pigmentosa in humans, and both diseases have similar ophthalmic examination and histological findings. 18,29 Most commonly, human patients with retinitis pigmentosa experience the onset of night blindness during adolescence, with loss of the mid-peripheral visual field. Over time, far peripheral vision is lost leading to tunnel vision, and most patients are considered legally blind by 40 years of age, with complete blindness by 60 years. 18,29 Shortening of the outer segment of rod photoreceptors is the initial histological lesion observed in all forms of retinitis pigmentosa, and as rod photoreceptors degenerate and die there are reduced numbers of nuclei in the outer nuclear layer, as was observed in affected sheep. 29 Cone degeneration occurs following the loss of neighboring rods, 31 and loss of photoreceptors is accompanied by hypertrophy of Müller cells and migration of cells from the retinal pigment epithelium into the inner retina, reducing the area of the subretinal space. 29 Retinitis pigmentosa is a genetically heterogeneous group of diseases and to date over 70 genes have been implicated, 12 with genetic overlap between retinitis pigmentosa and other types of hereditary retinal dystrophies such as cone-rod dystrophies. Inherited retinal degeneration is also well described in more than 100 breeds of dog, 30 and is generally referred to as progressive retinal atrophy. As in human patients with retinitis pigmentosa, progressive retinal atrophy in dogs is phenotypically and genotypically heterogeneous, with wide variation in the age of onset and progression of disease. At least 18 mutations in 16 genes have been identified in cases of progressive retinal atrophy in dogs, and of these, 3 genes have not yet been implicated in retinal degeneration in humans. 9,13,30
As the retinal degeneration in the Wiltshire sheep was suspected to be a recessive trait, homozygosity mapping was chosen as the initial method to identify possible causative mutations. The 5 affected sheep used for SNP analysis shared a large identical-by-descent region on the long arm of chromosome 5, but none of the genes within this region are involved in the phototransduction cascade or reported to be specifically expressed in the retina. The GPR98 gene located adjacent to this region was considered as a potential candidate gene as it encodes G protein coupled receptor 98 (also known as very large G protein-coupled receptor 1), which is associated with the stereocilia of the inner ear and the ciliary calyx of photoreceptors of the eye. 28 Mutations in GPR98 in humans have been associated with Usher syndrome type 2C, which is inherited in an autosomal recessive manner and is characterized by congenital moderate hearing loss, normal vestibular function, and late-onset retinitis pigmentosa. 4,49 Hearing was not specifically assessed in the Wiltshire sheep, but the affected ewes used auditory cues (bleating) to help them locate their lambs following separation, which suggests they were able to hear other sheep vocalizing nearby. Further analysis of GPR98 sequences in the 2 affected sheep used for whole-genome sequencing failed to identify any disease-causing variants, making it unlikely that this gene plays a role in retinal degeneration in Wiltshire sheep.
Whole-genome sequencing of 2 affected Wiltshire sheep did not identify any significant mutations in the genes known to cause retinitis pigmentosa in human patients and progressive retinal atrophy in dogs. In addition, analysis to identify high-impact mutations throughout the genome did not reveal any significant variants in genes known to be associated with retinal function. As only 2 affected sheep were used for whole-genome sequencing, further analysis of genome data to identify low- to moderate-impact mutations (such as base substitutions and small insertions and deletions) is not practical due to the high degree of nucleotide variability between individual animals. Despite the wealth of data generated by whole-genome sequencing, it is important to note that the genetic basis of a significant proportion of retinitis pigmentosa cases in human patients is currently unknown, 5 and a recent study using whole-exome sequencing in 186 Korean patients with inherited retinal disease detected causative variants in only 51.2% of cases. 22 Similarly, an open access genetic testing program for inherited retinal diseases reported an overall diagnostic yield of 59.4% using a retinal dystrophy panel of 322 genes. 24
The main limitation of the current study is the small number of sheep in the original flock, which limited the number of animals available for use in the breeding trial and genetic testing. Investigations into the inheritance and molecular basis of retinal degeneration in these sheep was also difficult due to the relatively late age of onset of vision loss, which meant that many affected animals (particularly rams) had been sold or culled at a young age, prior to the possible development of any visual impairment. Blindness has not been reported in other Wiltshire flocks in New Zealand, but the incidence of the disease may be significantly underestimated as early night blindness may go unrecognized in large extensive farming operations. Whole-genome sequencing of additional affected sheep in future may aid in identification of a causative mutation, which would enable screening of young animals and sheep in other flocks for carrier and affected animals.
The current study relied on maze testing and fundoscopic examination to identify affected animals, with histopathology used to characterize the associated pathological changes and progression. While the findings were most consistent with a primary rod degeneration, this cannot be differentiated from a rod-cone dysplasia or degeneration on the basis of histology alone, and genes associated with rod-cone dysfunction were searched for variants as part of the whole-genome sequencing of affected sheep. Electroretinography (ERG) has been used in sheep to characterize retinal dysfunction in a range of diseases, including day blindness in Awassi sheep, 41 scrapie, 36 and ceroid lipofuscinosis, 16 and normal peak amplitudes and latencies have been reported in sheep. 43 ERG would have been useful to determine the relative contributions of rod and cone dysfunction to blindness in the current study, but on both the original farm and during the breeding trial, sheep were exclusively grazed outdoors and on-farm facilities were limited. Complete darkness and electrical isolation are required to conduct ERG studies and sheep must be anesthetized for the procedure, 34 which limits its usefulness in large animals outside of experimental studies. In future studies, another useful diagnostic tool could be evaluation of chromatic pupillary light reflexes using bright red, blue, and white light to assess rod and cone function independently. This technique is minimally invasive and was recently used to identify cone dysfunction in day-blind Awassi sheep, which showed a lack of response to bright red light. 39 In addition, immunohistochemistry of retinal sections could aid in confirming the primary involvement of rod photoreceptors 2 rather than relying on photoreceptor outer segment and nuclear morphology, and the use of electron microscopy could improve characterization of the early photoreceptor lesions.
In summary, this is the first report of a novel recessively inherited night blindness in Wiltshire sheep. Characteristic histologic changes in the retina included progressive thinning of the outer nuclear layer due to loss of rod photoreceptors, which has similarities to progressive retinal atrophy in dogs and retinitis pigmentosa in humans. Further research is planned to investigate the underlying molecular basis of this disease in Wiltshire sheep.
Supplemental Material
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211067461 - An inherited night blindness in Wiltshire sheep
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211067461 for An inherited night blindness in Wiltshire sheep by Hayley Hunt, Keren E. Dittmer, Dorian J. Garrick, Robert A. Fairley, Stephen J. Heap and Robert D. Jolly in Veterinary Pathology
Footnotes
Acknowledgements
The authors would like to thank Stephen and Lorraine Sheen for their significant contribution throughout all stages of the project, and Alex Ashworth and Tara Buxton for their assistance with sample collection.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This authors received funding from the Massey University School of Veterinary Science Postgraduate Research Fund toward this research.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
