Abstract
Mortality of 20% of a flock of 1000 chukar partridge chicks occurred over a 6-week period in Northern California from August to September 2012. Affected birds were 2 to 42 days old and died without premonitory clinical signs or after showing ruffled feathers and anorexia for 24 to 72 hours. Three carcasses were submitted for necropsy, 2 birds had hemorrhagic tracheitis grossly, and all 3 had lymphoplasmacytic and histiocytic myocarditis with myocardial necrosis microscopically. The differential diagnoses and the diagnostic workup to achieve a final diagnosis are discussed. The detection of 2 zoonotic agents in these birds makes this an interesting case from a public health perspective.
History
The death of 200 of 1000 (20%) chukar partridge chicks (Alectoris chukar) occurred over a 6-week period in Glenn County, California, from August to September 2012. The chicks were 2 to 42 days old and housed in 4 hutches. Two days after arrival to the facility, 57 birds were found dead, and mortality progressed with 2 to 12 deaths daily. Affected birds died without premonitory clinical signs or after showing ruffled feathers and anorexia for 24 to 72 hours. Two female and 1 male frozen carcasses (bird Nos. 1–3) were submitted to the California Animal Health and Food Safety (CAHFS) Laboratory for necropsy and diagnostic workup.
Necropsy Findings
Tissues were in a moderate state of postmortem decomposition upon thawing. Necropsy revealed segmental red discoloration of the tracheal mucosa with intraluminal red-tinged mucus in bird Nos. 2 and 3, as well as multiple small intraluminal tracheal and oral blood clots in bird No. 2. The lungs of all 3 birds were irregularly reddened and had an increased consistency. All other organ systems were unremarkable.
Differential Diagnoses
Differential diagnoses for hemorrhagic tracheitis in birds include viral etiologies such as highly pathogenic avian influenza virus (HPAI, influenzavirus A), avian paramyxovirus 1 (APMV-1, Avulavirus [Newcastle disease]), infectious bronchitis virus (IBV, Gammacoronavirus), infectious laryngotracheitis virus (ILT, Gallid herpesvirus 1), bacterial etiologies (Pasteurella multocida, Ornithobacterium rhinotracheale, and Bordetella avium), and toxic aerosols (eg, ammonia gas).
Histopathology
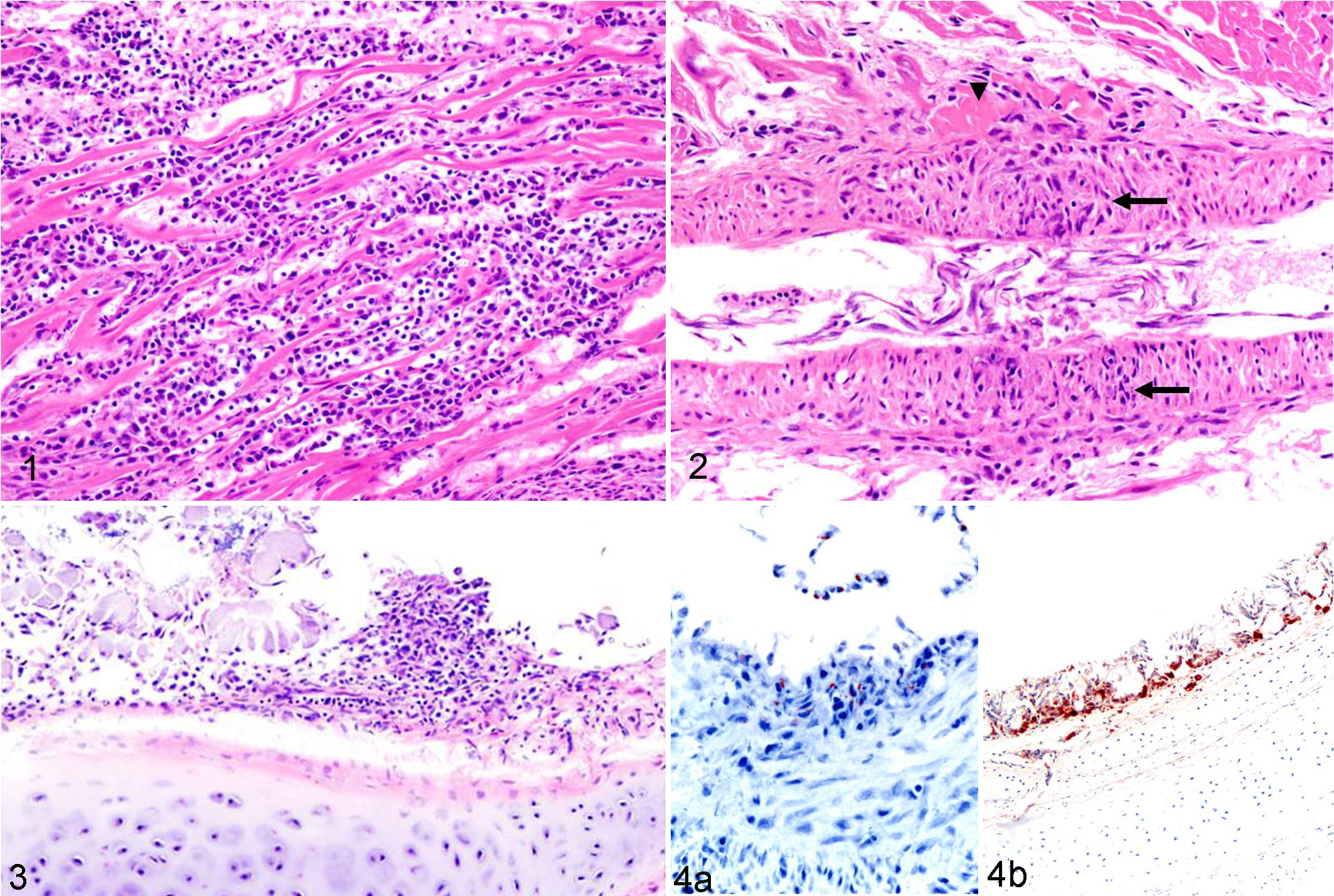
Histological examination revealed lymphoplasmacytic and histiocytic myocarditis with concurrent cardiomyocyte necrosis in all 3 birds (Fig. 1), coronary arteritis in bird No. 2 (Fig. 2), and multifocal mild lymphohistiocytic laryngotracheitis and conjunctivitis in bird No. 2 (Fig. 3). Bird No. 1 had caseogranulomatous laryngitis and moderate multifocal interstitial pneumonia with Gram-negative bacilli and moderate focal granulomatous pneumonia with unidentified intralesional fungal hyphae. Bird No. 3 had segmental marked submucosal tracheal edema and multifocal random acute necrotizing hepatitis with sinusoidal microthrombosis and intralesional Gram-negative bacilli.
Ancillary Test Results
Ancillary diagnostic test results are summarized in Table 1. Of note, West Nile virus (WNV, Flavivirus) was detected by real-time polymerase chain reaction (PCR) in kidney and immunohistochemistry (IHC) following procedures previously described. 6 WNV antigen was detected by IHC in the heart in all birds (Fig. 4a), skeletal muscle in bird Nos. 1 and 3, trachea and conjunctiva in bird No. 2, and trachea and larynx in bird No. 3 (Fig. 4b). In addition, Salmonella spp was detected by PCR and isolated by Salmonella enrichment culture from intestinal contents of all 3 birds. Salmonella was also isolated on blood agar by regular bacterial aerobic cultures from lung in bird No. 1 and liver in bird No. 3; both birds had either pulmonary or hepatic lesions consistent with septicemia. The Salmonella strain from bird No. 1 was serotyped as Salmonella Typhimurium.
Diagnostic Tests and Results.

AIV, avian influenza virus; APMV-1, avian paramyxovirus 1; DEM, direct electron microscopy; FAT, fluorescent antibody test; IBV, infectious bronchitis virus; IHC, immunohistochemistry; ILTV, infectious laryngotracheitis virus; NA, not applicable; OPS, oropharyngeal swab; RT-PCR, real-time polymerase chain reaction; vvIBDV, very virulent infectious bursal disease virus; WNV, West Nile virus; —, .
Diagnosis
The diagnosis was multifocal lymphoplasmacytic and histiocytic myocarditis and myocardial necrosis with intralesional WNV antigen in all 3 birds, hemorrhagic tracheitis with intralesional WNV antigen in bird Nos. 2 and 3, and concomitant Salmonella spp septicemia in bird Nos. 1 and 3.
Discussion
WNV is an avian Flavivirus transmitted by mosquitoes that can cause fatal disease in mammals (including humans), reptiles, and birds. Birds are the natural reservoir host in the life cycle of WNV, but among avian species, there is a broad range of pathogen-host responses. Many known avian species are highly susceptible to developing severe or lethal disease, including corvids, 4 raptors, 7 psittacines, 6 and waterfowl; 3 however this is very rare in birds of the order Galliformes. 1 To our knowledge, there is only 1 other published description of WNV infection in chukar partridges. 8 These authors described two 25% mortality events involving hundreds of captive juvenile chukar partridges in the fall of 2002 and 2003. In these cases, myocardial necrosis and myocarditis were the predominant microscopic lesions. Epidemiologic, clinical, and cardiac pathological findings were similar to those described in this report. In contrast to our cases, hemorrhagic tracheitis and coronary arteritis with intralesional detection of WNV antigen were not reported previously. Therefore, we propose that WNV should be included in the list of differential diagnoses for hemorrhagic tracheitis/tracheal bleeding in chukar partridges.
WNV is endemic in California, with transmission occurring primarily in summer months, 2 and our case fits this temporal pattern. According to the California Department of Public Health, the number of symptomatic human cases reported in 2012 (479) was the highest since 2005 and third highest since the disease was first detected in the state in 2003 (http://www.cdph.ca.gov/programs/vbds/Documents/VBDSAnnualReport12.pdf). Interestingly, in 2012, the incidence was highest in Glenn County, the same county in which these birds were raised.
In addition to WNV infection, bird Nos. 1 and 3 had evidence of systemic salmonellosis, but bird No. 2 was considered only a carrier, since no compatible systemic lesions accompanied the intestinal Salmonella. Bird No. 1 had concurrent respiratory infections (bacterial granulomatous laryngitis and focal fungal pneumonia). Immunosuppression and stress were possibly critical contributors to disease progression and mortality in these birds, since stress increases mortality in individuals exposed to WNV. 5 Age might have contributed in these cases, since the mortality affected juvenile birds less than 6 weeks old. This matches the previously published cases of WNV in chukars. 8
HPAI and Newcastle disease are highly contagious reportable viral diseases that affect many avian species and can result in rapid high mortality attributable to respiratory, alimentary, and/or systemic disease; thus, both diseases were high on the differential diagnoses list in this outbreak. Infectious bronchitis can manifest as acute respiratory signs in growing chickens, and the severity is enhanced by concurrent pathogens. Although IBV is a disease that primarily affects chickens (and rarely pheasants), coronaviruses have been detected in partridges and thus was investigated. ILT is characterized by lesions in the upper respiratory tract, affects chickens and pheasants, but has not been reported in chukars. Thus, even though the disease was unlikely, it was ruled out. Infectious bursal disease is a highly contagious disease of young chickens. Recently, very virulent strains causing severe mortality events have emerged. The disease is characterized by immunosuppression and necrohemorrhagic lesions, particularly in the bursa of Fabricius. Although the condition has not been reported in chukars and the lesions in these birds were not consistent with this disease, the fact that it can result in high mortality events in young galliforms prompted investigation. All 4 aforementioned viral diseases were ruled out by specific diagnostic testing.
In conclusion, this report further confirms chukar partridges are highly susceptible to WNV and can result in high mortality, particularly in young birds. The detection of WNV and Salmonella Typhimurium makes this an interesting case from a public health perspective. Finally, this report highlights the importance of vigilant care of young animals, reducing stress and other causes of immunosuppression of intensely raised chukar partridges, and supports the possible need for insect-vector control, vaccination, and surveillance in this species.
Footnotes
Acknowledgement
We thank all pathology, histology, bacteriology, and biotechnology technicians from the CAHFS laboratory.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
