Abstract
Brucella abortus infection has not been documented in llamas. This report describes the abortion of the only pregnant animal in a group of 12. The llama was infected by inoculating 1 × 108 viable B. abortus organisms into the conjunctival sac. Forty-three days postinfection, the llama aborted a fetus of approximately 8 months gestational age. Brucella organisms were isolated from the placenta and all fetal specimens examined. These organisms were also isolated from the dam's mammary gland and numerous lymph nodes when the llama was necropsied 42 days later. Microscopically, there was a moderate, multifocal, lymphocytic and histiocytic, subacute placentitis with marked loss of trophoblastic epithelial cells. The superficial chorioallantoic stroma contained abundant necrotic and mineralized debris as well as numerous swollen capillaries protruding multifocally from the denuded surface. Immunohistochemistry revealed that these capillaries, as well as sloughed and intact trophoblasts, were expanded by large numbers of Brucella organisms. Brucellar antigen was also detected in occasional macrophages in the fetal kidney and lung. Ultrastructurally, bacteria labeled by an antibody-based colloidal gold procedure were located within degenerate capillaries, within necrotic leukocytes, and extracellularly in the placental stroma.
Naturally occurring brucellosis caused by Brucella abortus exists in dromedary and bactrian camels in Asia and North Africa.4 Abortion with excretion of the organism has been reported.4 Camels are routinely tested for brucellosis with bovine serum agglutination tests, and their serologic response to infection is considered to be similar to that of cattle.4 Brucella melitensis infection was diagnosed in three llamas in the London Zoo and in a Peruvian alpaca herd.6,7 In the alpaca herd, 21% of the animals were serologically positive (standard plate agglutination titers greater than 1:25) and more than 25% of the caretakers had serologic evidence of infection.
Brucella abortus infection in llamas has not been reported, and its infectivity in new world camelids has not been determined.6 Approximately 65,000 llamas are in the United States. The significance of these animals in the epidemiology of bovine brucellosis is unknown. The purpose of this report is to describe abortion in a llama after experimental infection with B. abortus biovar 1.
Twelve llamas were part of an experimental study to determine pathologic, serologic, and bacteriologic effects of B. abortus strain 2308. Rectal palpation at the National Veterinary Services Laboratories revealed an unanticipated 6-month pregnancy in one of the llamas; the remaining llamas were nongravid. Hematologic examination, serum biochemical evaluation of hepatic and renal function, and fecal parasite evaluation demonstrated no abnormalities. The animal was pastured with its cohorts with access to shelter for approximately 1 month prior to the beginning of the study. Antibodies were not revealed by any of the conventional Brucella serologic tests (standard tube, standard plate, card, rivanol, complement fixation, and the buffered acidified plate) conducted with serum collected 30 days and again 7 days prior to the initiation of the study.
One hundred microliters of phosphate-buffered saline (0.2 M, pH 6.4) containing 1 × 108 viable B. abortus strain 2308 organisms was divided and half was instilled into the conjunctival sac of each eye. Housing was provided in an animal biosafety level 3 facility. Ten milliliters of heparinized blood was collected daily for the first 5 days postinfection (PI), weekly for 7 weeks PI, and on weeks 10 and 12 PI. Heparinized blood samples were cultured within 48 hours or stored at −70 C until cultured.
On day 43 PI, the llama aborted a fetus of approximately 8 months gestational age (normal gestational period is 345 days) with a crown rump measurement of 35 cm. The placental membranes were diffusely and moderately opaque and contained numerous small white flecks 0.25 mm in diameter. Specimens of placenta and fetal brain, small and large intestine, spleen, kidney, liver, stomach fluid, heart blood, and lung were collected. One half of each specimen was cultured for Brucella spp. and one half was fixed in 10% neutral buffered formalin for histopathologic and immunohistochemical evaluation.
On day 85 PI, the dam was euthanatized (ketamine and xylazine anesthesia followed by exsanguination) and necropsied. Tissue specimens included mammary gland; palatine tonsil; palpebral conjunctiva and third eyelids; eyes; lung; trachea; heart; kidney; adrenal gland; liver; spleen; reproductive tract; brain; cervical, thoracic, and lumbar spinal cord; and representative sections from the gastrointestinal tract. Specimens also included the following lymph nodes: dorsal and ventral superficial cervical, popliteal, supramammary, mandibular, medial retropharangeal, parotid, mediastinal, tracheobronchial, mesenteric, ileoeecal, medial and lateral iliac, hepatic, and prefemoral. Lymph nodes, periorbital tissues, lung, kidneys, spleen, reproductive tract, liver, uterus, and brain were divided and submitted for microbiologic and histologic evaluation. The remainder of the tissues were evaluated only histologically. The formalin-fixed counterparts of Brucella culture-positive tissues were examined by immunohistologic techniques.
Tissues were processed for culture by the stomacher technique for solid tissues with multiple selective media, and isolates were identified by standard techniques.1,5 All tissues for histologic evaluation were stored in 10% neutral buffered formalin and processed by standard paraffin-embedding techniques. Sections were cut at 4 μm and stained with hematoxylin and eosin. Giemsa, Von Kossa, and a tissue Gram stain were used on selected sections. A labeled streptavidin–biotin system available in a commercial staining kit was used for immunohistochemical staining of brucellar antigen (DAKO Corp., Carpenteria, CA). The primary antibody was prepared in rabbits hyperimmunized with whole irradiated B. abortus strain 2308 and was used at a dilution of 1:10,000.11 Nonimmunized rabbit serum was used for the negative control. Infected BALB/C mouse liver and spleen were used as positive controls. Samples of the placenta for transmission electron microscopic (TEM) examination were originally fixed in 10% neutral buffered formalin and transferred to 2.5% glutaraldehyde in 0.1 M cacodylate buffer. Ultrathin sections were cut and stained with uranyl acetate and lead citrate. Antibrucella antibody combined with 20 nm colloidal gold was used to identify the bacteria in the TEM examination.2
Bacteremia in the dam was detected on days 16 and 21 PI. Brucella abortus strain 2308 was isolated from all fetal tissue specimens, placenta, and milk collected from the dam 1 day after the abortion. After necropsy of the dam, B. abortus strain 2308 was isolated from milk, mammary gland, and the following lymph nodes: dorsal and ventral superficial cervical, popliteal, supramammary, prefemoral, mandibular, medial retropharangeal, parotid, tracheobronchial, iliac, and hepatic.
Histologically, trophoblastic epithelial cells were missing from greater than 90% of the placental surface, leaving the superficial chorioallantoic stroma exposed (Fig. 1) Approximately 20% of this exposed stroma contained multifocal, variably sized areas of necrosis with occasional foci of mineralization. Adherent and sloughed trophoblasts contained fine, gram-negative coccobacilli. Multifocal infiltrates of low numbers of lymphocytes and macrophages were within the superficial chorioallantoic stroma, which often extended into the subjacent stroma (Fig. 2). In areas of denudation, the superficial chorioallantoic stroma contained numerous, variably sized (10–100 μm in diameter), round to oval capillaries containing gram-negative coccobacilli. These capillaries protruded multifocally from the denuded, superficial surface of the chorioallantoic stroma. In intact areas, numerous capillaries located immediately subjacent to the trophoblastic epithelium were distended with coccobacilli. Larger vessels located within the chorioallantoic stroma often contained similar bacteria. Edema of the placental stroma, especially superficially, caused marked, diffuse pallor and separation of collagen.
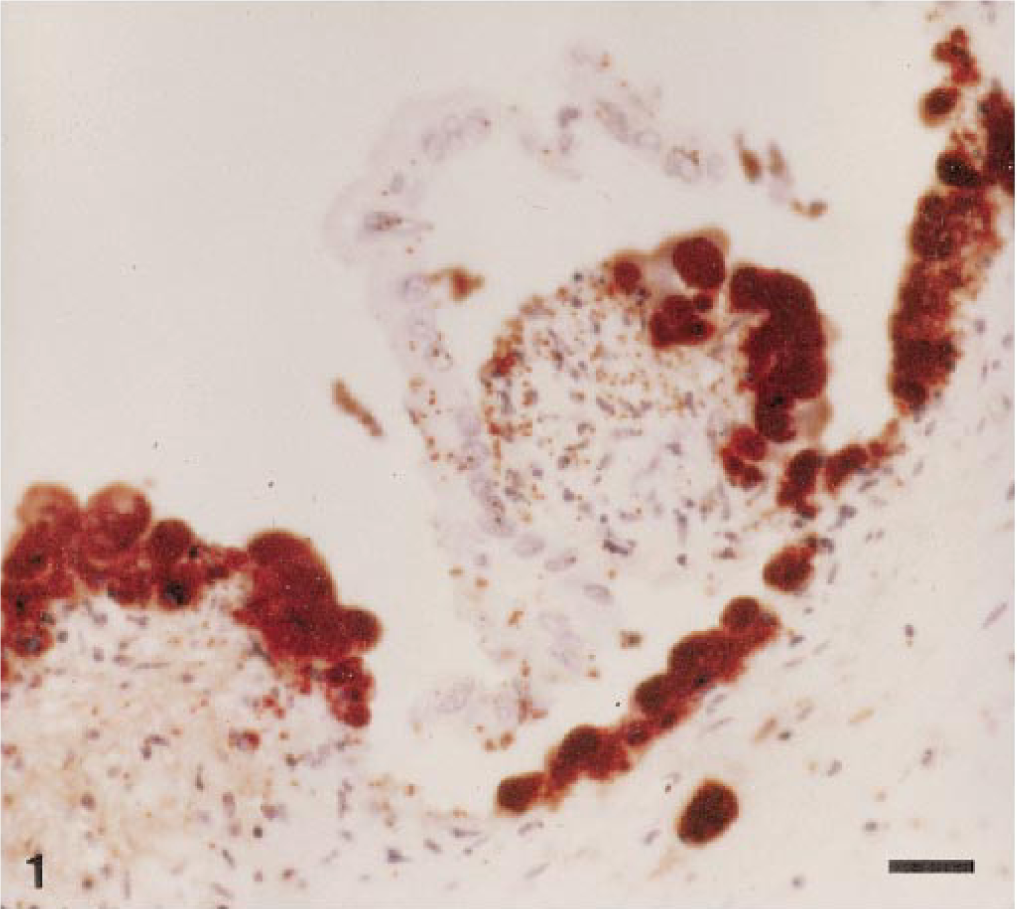
Placenta; llama abortion. Note loss of trophoblastic epithelial cells and superficial capillaries expanded with organisms immunoreactive for B. abortus. Peroxidase-labeled streptavidin–biotin method, Gill II hematoxylin counterstain. Bar = 20 μm.
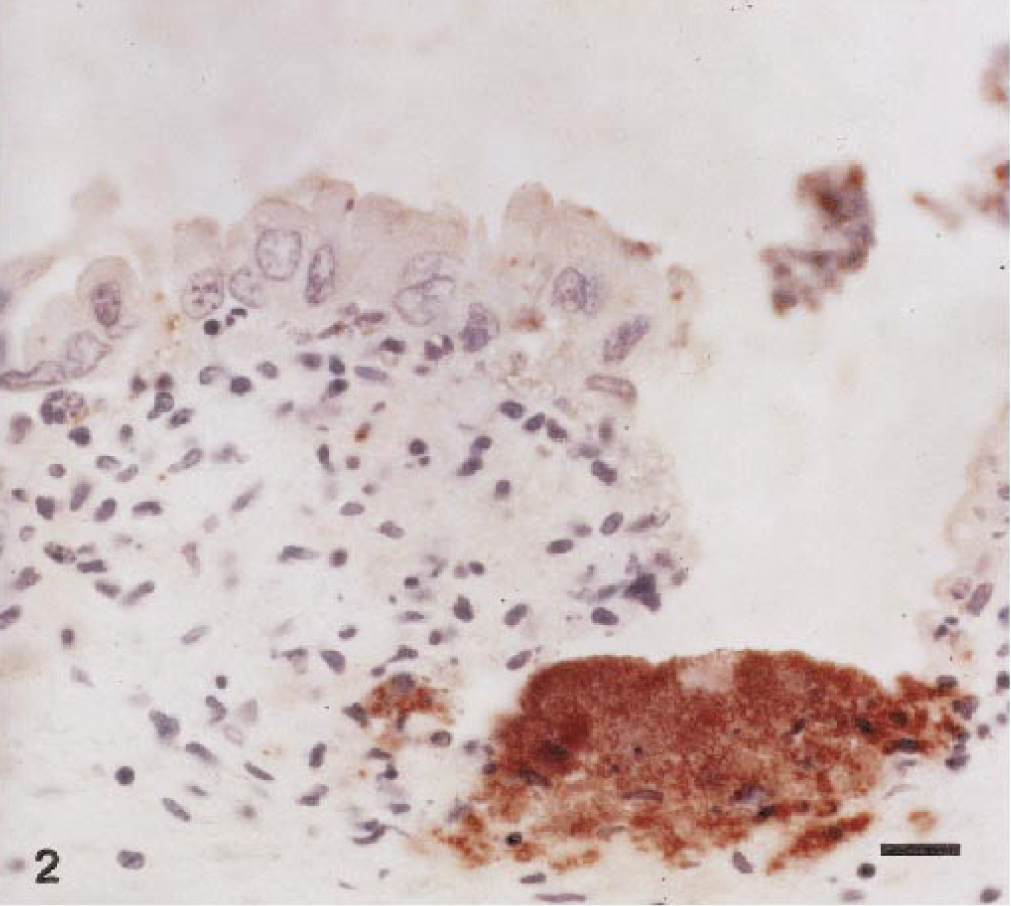
Placenta; llama abortion. Note adherent and sloughed trophoblastic epithelial cells containing low numbers of immunoreactive bacteria and the mixed cellular infiltrate in the superficial chorioallantoic stroma. Peroxidase-labeled streptavidin–biotin method, Gill II hematoxylin counterstain. Bar = 10 μm.
Lesions were not observed in the fetal tissues examined. The mammary gland contained moderate, diffuse, interstitial infiltrates of lymphocytes and plasma cells with numerous variably sized lymphoid follicles. Numerous alveoli and intralobular ducts contained intraepithelial lymphocytes, and alveolar and ductal lumina contained numerous swollen macrophages and fewer lymphocytes, plasma cells, and occasional neutrophils. There was moderate multifocal alveolar and ductal epithelial erosion and ulceration with occasional replacement of the normal epithelium with fewer flattened epithelial cells. Some lobules contained interstitial lymphocytes, whereas other lobules had large areas of acinar loss and replacement by areas of pallor and lymphoid follicles.
The endometrium contained numerous, diffuse, interstitial lymphocytes and plasma cells with occasional lymphoid follicles. Uterine glands were filled with degenerate neutrophils, eosinophilic amorphous material, and cellular debris. Variably sized aggregates of lymphocytes were found between occasional muscle bundles of the myometrium. The perimetrium was moderately and diffusely edematous and congested.
Brucellar antigen was detected immunohistochemically within superficial chorioallantoic capillaries, and lesser amounts of antigen were present within occasional trophoblasts (Figs. 1, 2). Macrophages and neutrophils contained low numbers of organisms, and multifocal, variably sized colonies of Brucella were free in the placental stroma. The fetal kidney and lung contained occasional large mononuclear cells with low numbers of organisms immunoreactive for B. abortus (Figs. 3, 4).
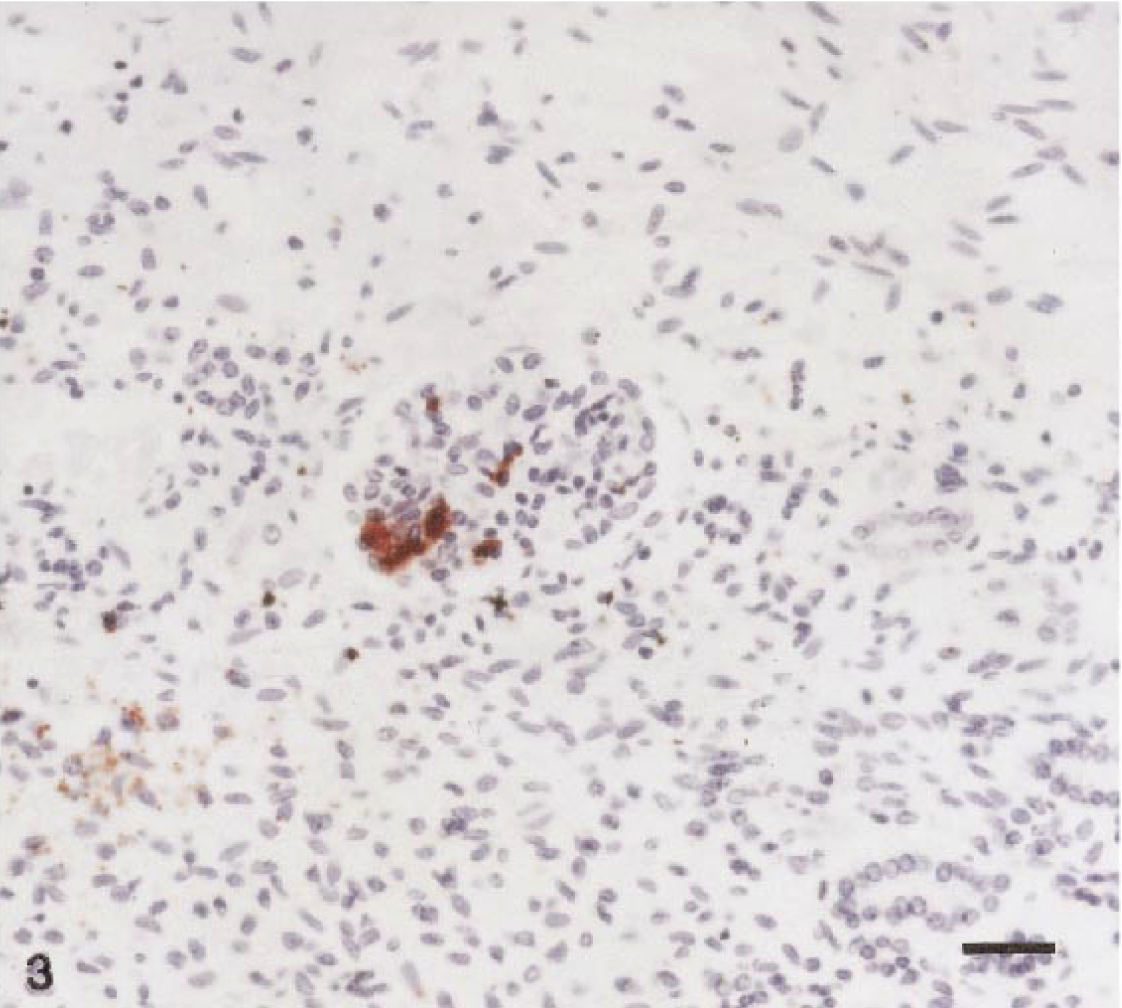
Kidney; llama fetus. Note expansion of glomerular capillary tufts by colonies of Brucella abortus. Scattered cells throughout the section contain occasional immunoreactive bacteria. Peroxidase-labeled streptavidin–biotin method, Gill II hematoxylin counterstain. Bar = 20 μm.
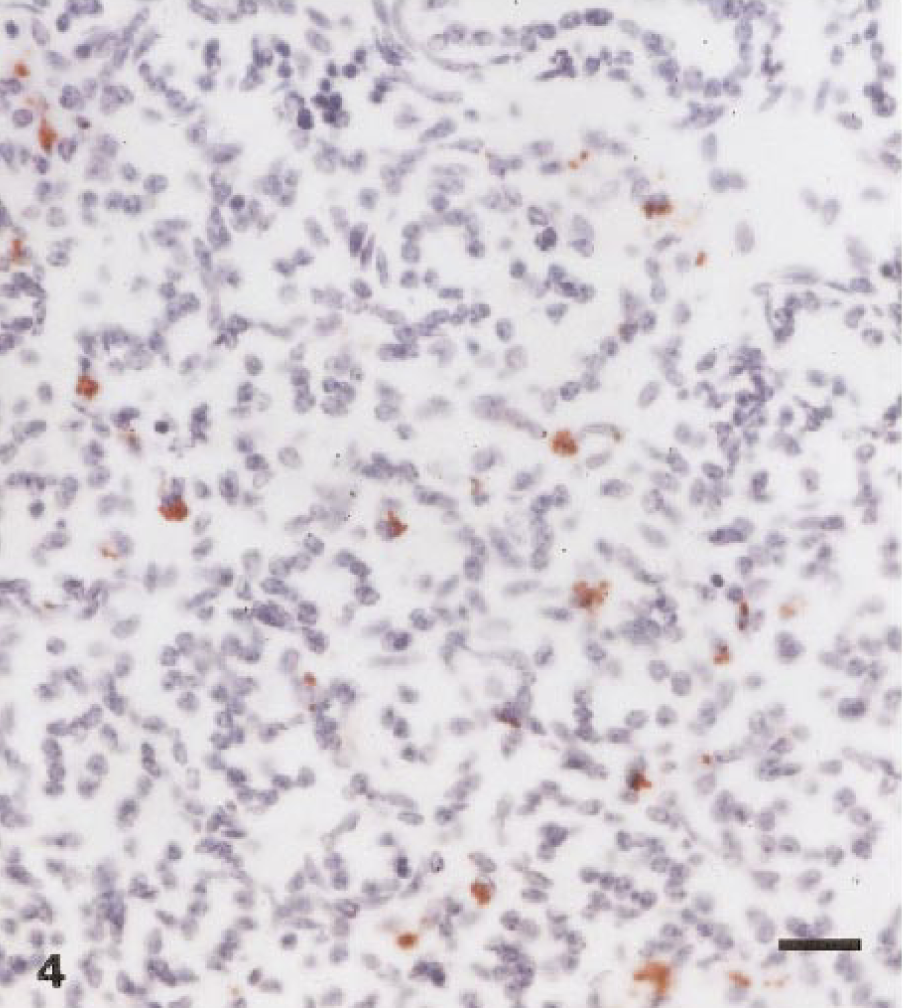
Lung; llama fetus. Numerous macrophages throughout the section contain small bacterial aggregates. peroxidase-labeled streptavidin–biotin method, Gill II hematoxylin counterstain. Bar = 10 μm.
Ultrastructural analysis revealed large aggregates of intact bacilli throughout the placental membranes (Fig. 5). Bacillary surfaces stained with the specific immunogold reagent with antibody against B. abortus (Fig. 6). In placental tissue, greater than 98% of the bacilli were structurally intact. Bacilli were present as free individual organisms in interstitial areas and as small groups embedded within electron-dense granular material. The latter were interpreted as necrotic leukocytes within distorted vascular lumina (Fig. 7). The largest aggregates of bacilli, which were present at intervals along the denuded surface of the placental membrane, were surrounded by a continuous basement membrane of variable width. The large aggregates appeared to be necrotic remnants of dilated placental capillaries. This interpretation was based on the irregularity, spherical shape, and continuity of the basement membrane, on the presence of necrotic erythrocytes and leukocytes among bacterial cells in the lumenal areas, and on the existence of several hundred bacteria in each group (Fig. 8).
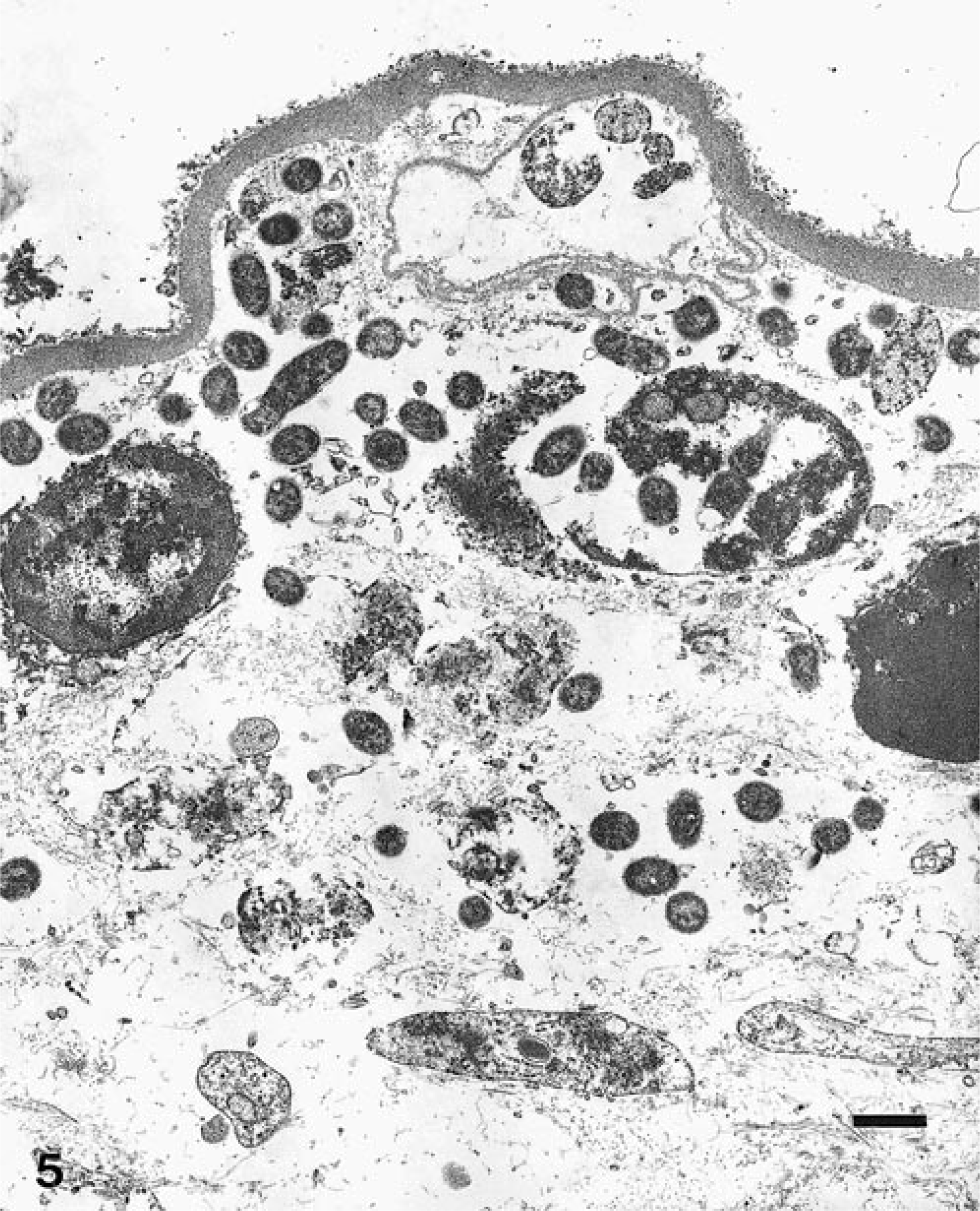
Electron micrograph. Placenta surface; llama abortion. Note aggregates of intact bacilli intra- and extravascularly. The basement membrane is completely devoid of trophoblastic epithelial cells. Bacilli are specifically labeled with colloidal gold. Bar = 1 μm.
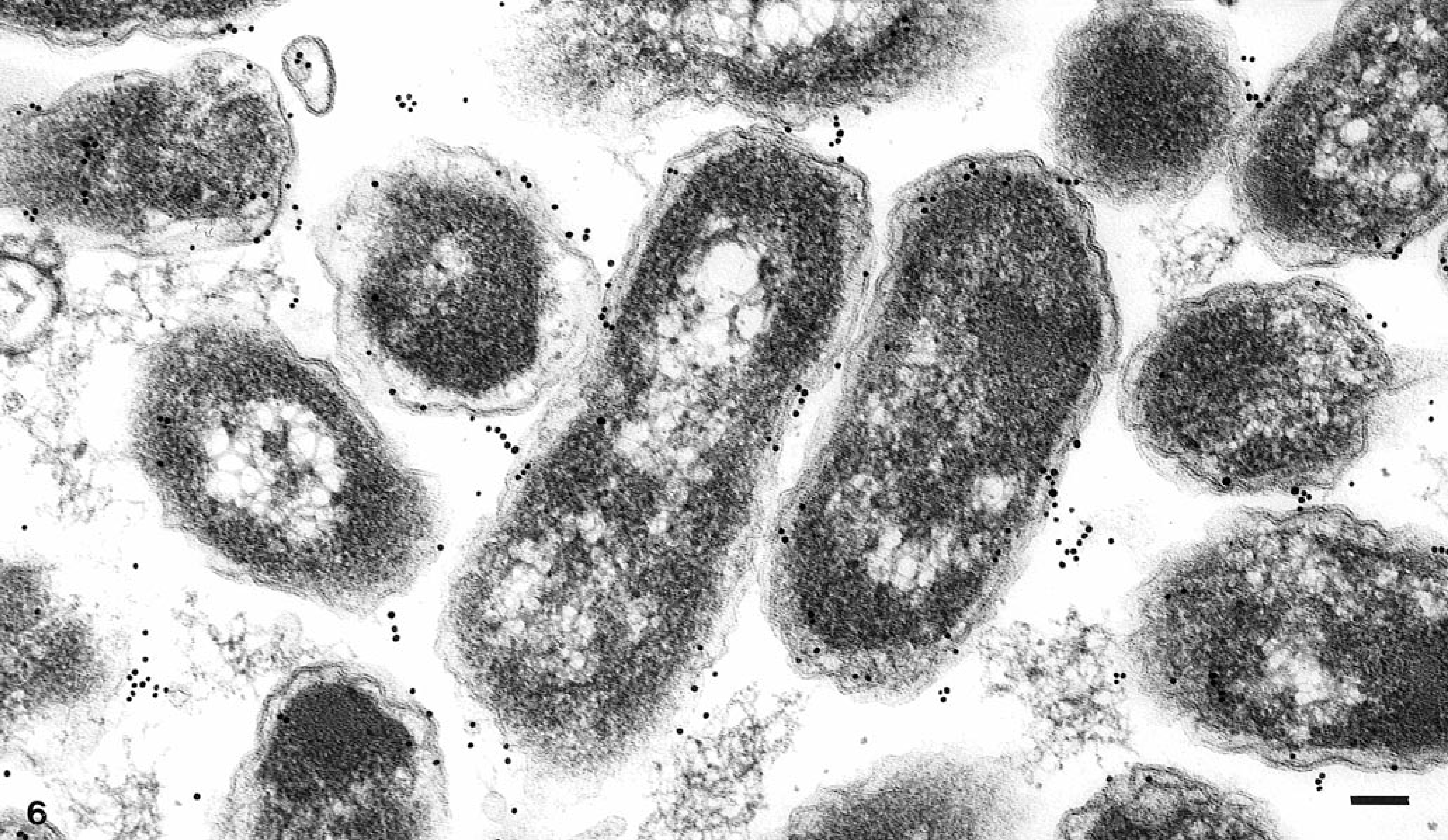
Electron micrograph. Superficial placental stroma; llama abortion. Note cross and longitudinal sections of bacilli stained with the specific immunogold reagent with antibody against B. abortus. Bar = 0.1 μm.
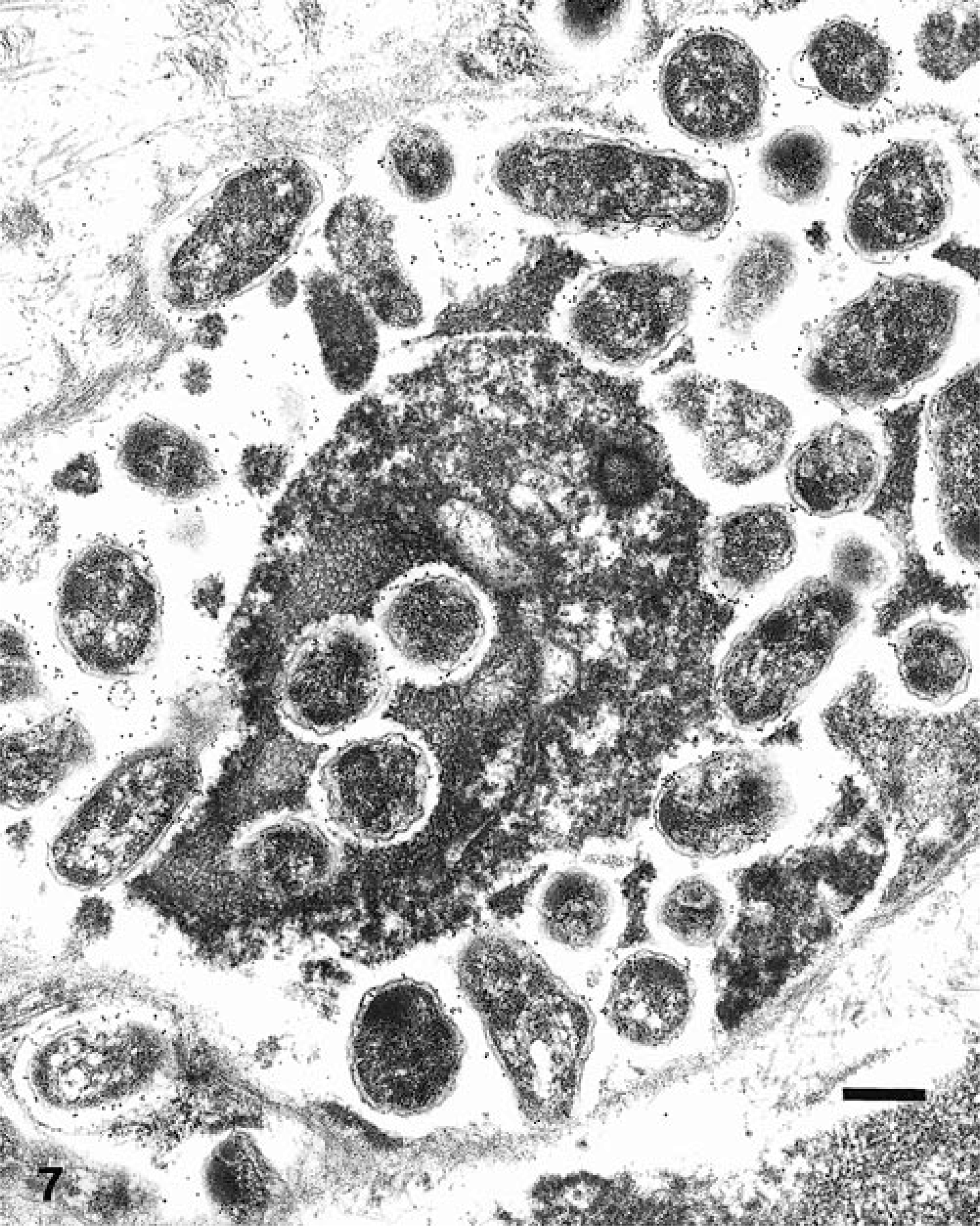
Electron micrograph. Superficial placental stroma; llama abortion. Note numerous colloidal gold-labeled organisms within a vascular lumen and four bacilli within a necrotic leukocyte. Bacilli are specifically labeled with colloidal gold. Bar = 0.5 μm.
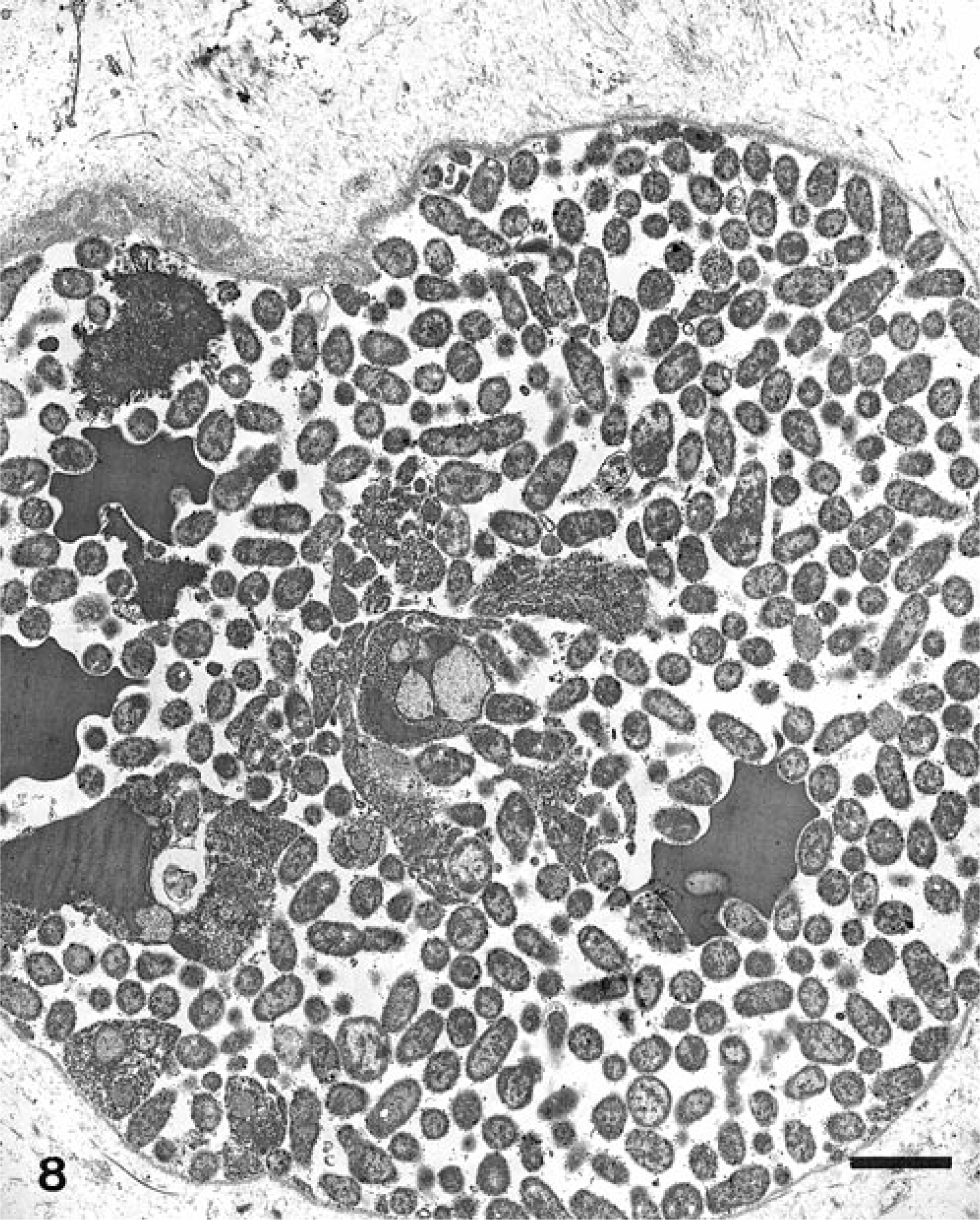
Electron micrograph. Superficial placental stroma; llama abortion. Note aggregate of numerous colloidal gold-labeled bacilli and occasional necrotic erythrocytes and leukocytes. This aggregate is surrounded by an irregular basement membrane (capillary lumen). Bar = 2 μm.
The placentation of the llama is diffuse epitheliochorial, similar to the placenta of the pig. The fetal chorioallantois is arranged in discontinuous or periodic unbranched folds or projections that coincide with uterine epithelial indentations. A feature in pregnant Camelidae is the presence of numerous placental and uterine intraepithelial capillaries. In late gestation, these capillaries are located so that fetal capillaries may be only 2 μm from their maternal counterparts.13 This appears to be the shortest intercapillary fetal–maternal distance of any ungulate with epitheliochorial placentation.13 This anatomical relationship may predispose the llama fetus to Brucella infection.
Anderson et al.2 suggested that the caprine erythrophagocytic trophoblastic epithelial cells internalize Brucella from the maternal circulation. Brucellae replicate within the rough endoplasmic reticulum, resulting in secondary infection of adjacent trophoblastic epithelial cells.2,8 After necrosis of infected trophoblasts, large numbers of brucellae are released. The proximity of the fetal capillaries in the ulcerated placenta to the lumenal bacteria is proposed as the source of the fetal bacteremia and further placental infection.2,3 Molello et al.10 proposed a similar pathogenesis in the ewe. In cattle, Payne12 suggested that the maternal bacteremia localizes the brucellae to the uterus and subsequently to the uterine glands and the lumen. The chorioallantoic trophoblastic epithelial cells then internalize the luminal brucellae.12
The loculi present on the denuded placental surface and in the superficial chorioallantoic stroma were remnants of capillaries and venules distended with bacteria. The unusual appearance was caused by the superficial location of the numerous intraepithelial capillaries in the llama placenta. Intravascular brucellae have been described in cattle and goats but not to the extent noted in the present study.9,12
The abortion and lesions produced by B. abortus strain 2308 in this llama were similar to those recognized in cattle, sheep, goats, and bison. The histologic, immunohistochemical, and ultrastructural results suggest that the pathogenesis of brucellosis in the llama is consistent with the course proposed by Anderson et al.2, Molello et al.10, and Payne12 in goats, sheep, and cattle, respectively. The differences observed in placental pathology between the other species and llamas are probably the result of different placentation. The lack of reports of the transmissibility of brucellosis between livestock and llamas may be because llamas and livestock do not usually share common range in South America. Llamas are traditionally raised in the higher elevations. With the heightened popularity of new world Camelidae, the possibility of interspecies communicability of brucellosis should be considered.
Footnotes
Acknowledgements
We gratefully acknowledge the assistance provided by the Pathobiology, Diagnostic Bacteriology, and Scientific Services Laboratories of the National Veterinary Services Laboratories, especially Darla Ewalt, Brad Pithan, and Kristin Wilson. The technical and professional assistance provided by these laboratories was crucial. The llamas used in this study were graciously donated by the International Llama Association.
