Abstract
Eighty-three canine cutaneous mast cell tumors were graded histologically and evaluated immunohistochemically for p53 tumor-suppressor protein expression. An avidin–biotin immunohistochemical protocol incorporated a rabbit polyclonal antibody (CM-1) directed against normal and mutant p53 protein. Positive staining was observed in 44.6% (37/83) of tumors and included 50% (12/24) of grade I (well differentiated) tumors, 46.9% (23/49) of grade II (intermediate differentiation) tumors, and 20% (2/10) of grade III (poorly differentiated) tumors. A statistically significantly higher proportion (P < 0.019) of tumors from the head and neck (83.3%, 10/12), stained positive for p53 than tumors from the thorax, back, abdomen, and axilla (39.4%, 13/33), legs (35.7%, 10/28), or prepuce, scrotal, or inguinal areas (44.4%, 4/9). No statistically significant difference between p53 labeling and histologic grade, breed, or tumor size was present. Survival data were available for 53/83 (63.9%) of dogs. Positive reactivity for p53 was observed in 47% (25/53) of tumors within this group, with 57.9% (11/19) of grade I, 43.3% (13/30) of grade II, and 25% (1/4) of grade III tumors labeled. Mean survival time for the 53 dogs was 12.1 months. The median survival time for dogs with grade III tumors or tumors >5 cm was statistically significantly shorter (P < 0.0001) than for dogs with grades I and II or smaller tumors. Although p53 protein abnormalities may play a role in tumor development or behavior in some canine cutaneous mast cell tumors, immunoreactivity was not associated with lack of tumor differentiation, tumor locations previously shown to demonstrate aggressive biological behavior, breed predisposition, or survival times.
Mast cells are of bone marrow origin and migrate via the blood to the skin, upper and lower respiratory tract, and gastrointestinal system.8,11,28 Neoplastic proliferations of mast cells represent 16–21% of all canine cutaneous tumors28 and 11–27% of all malignant canine cutaneous neoplasms.18 Although mast cell tumors have been reported in dogs as young as 3 weeks,11 they are most commonly reported in older dogs with a mean age of approximately 9 years.11,28 Breed predispositions and site predilections are well documented.11,28 The clinical presentation of canine cutaneous mast cell tumors is highly variable. However, arriving at a definitive diagnosis for the majority of mast cell tumors is easily accomplished with the use of cytologic preparations and routine histologic assessment utilizing histochemical stains.11,25 Establishing an accurate prognosis for canine cutaneous mast cell tumors is more challenging and has been based on a number of factors including histologic grade, clinical stage, tumor location, argyrophilic nucleolar organizer region counts, growth rate, breed, DNA ploidy, recurrence, and the presence of tumor-related systemic signs.28 Determining the fraction of cells that stain positive for anti-proliferating cell nuclear antigen has also been shown to be useful in predicting recurrence, probability of metastasis, and survival times.26 Histologic grade has been the most applicable and reliable factor in predicting clinical outcome.20,28 Establishing the histologic grade is a subjective process and is less useful for predicting clinical outcome in tumors of intermediate differentiation and when other clinical information, such as breed, site, and clinical stage of disease, is not known.1,17,18,26 Despite many efforts to define criteria of malignancy in canine cutaneous mast cell tumors, this group of tumors retains a degree of unpredictability that leads most diagnosticians to regard all canine cutaneous mast cell tumors as potentially malignant.1,11
The lesions, natural history, and clinical signs of mast cell tumors in the dog are well documented and excellent reviews are available.17,18,28 The etiology of canine mast cell tumors or molecular and genetic events contributing to tumor development are largely unknown. Mast cell tumor development at sites of chronic inflammation or irritation have been reported.28 Attempts to document a viral cause are limited and not convincing.3,28 Chromosomal fragile site expression as a possible genetic predisposition to development of cancer has been investigated in Boxers, a breed highly predisposed for mast cell tumor development.27 In that study, cells from Boxers with mast cell tumors did have a greater frequency of fragile site expression than those from controls; however, the greater frequency was interpreted to be an age-related change rather than an indication of a familial or breed-specific chromosomal fragility syndrome.27
A basic understanding of cellular events leading to tumorigenesis is the cornerstone of the modern approach to cancer therapy.4,29 Alterations in the functions and activities of cellular proto-oncogenes and tumor suppressor genes are pivotal events in the loss of normal homeostatic control of cellular proliferation and in the neoplastic transformation of cells. Future therapies are directed toward correcting or manipulating the functions or protein products of these types of genes.4,14,29 One of the most highly studied cancer-related genes is the p53 tumor-suppressor gene. The protein product of this gene is a nuclear transcription factor with functions that include cell cycle arrest in cells with damaged DNA, induction of transcription of DNA repair enzymes during this arrest, and the induction of apoptosis in cells that have sustained irreparable genetic damage, as well as the regulation of other transcription factors.5,6,21,24 These functions are essential for maintenance of the integrity of the genome and the prevention of malignant transformation. Mutation of the p53 gene is the most common genetic alteration known to occur in human cancers21 and has been associated with a shortened survival time or need for more aggressive therapy in some, but not all, types of human cancers studied.13,14 Single base pair substitutions leading to missense mutations are the most common type of mutation occurring in the human p53 gene.6 These mutations often lead to production of an altered p53 protein that binds to and inactivates the normal, wild-type p53 protein (WT p53), thereby promoting tumorigenesis. Mutant p53 protein is more stable and has an extended half-life in comparison with WT p53.14,21 These characteristics of the altered protein cause the protein to accumulate in the nucleus of affected cells and enable detection by immunohistochemistry.14,21 The very short half-life of WT p53 is thought to preclude detection via immunohistochemistry in normal cells. Immunohistochemical detection of p53 protein is equated to the detection of mutant p53 or otherwise stabilized abnormal p53 rather than due to overexpression of WT p53.14,23 Recently, alterations of the expression of the p53 protein have been the focus of a limited number of studies of naturally occurring tumors in the dog.9,10,22,23,30 Overexpression of p53 in osteosarcomas of the appendicular skeleton and in poorly differentiated and metastatic thyroid carcinomas has been interpreted as a possible indication of more aggressive tumor behavior.22,23 A retrospective study of p53 expression of colorectal tumors in the dog, however, was not useful prognostically and did not correlate with a diagnosis of malignancy.30 Still, the diverse role of this highly conserved tumor-suppressor protein in tumorigenesis and its potential usefulness as a prognostic indicator or as a target for future molecular biological approaches to therapy make further investigations of spontaneously occurring tumors in veterinary patients worthwhile.9
The objectives of this study were to investigate relationships between p53 tumor-suppressor protein immunoreactivity and the histologic grade, tumor location, tumor size, breed, and survival time in dogs with cutaneous mast cell tumors. The usefulness of p53 protein accumulation as a prognostic indicator for canine cutaneous mast cell tumors was evaluated.
Materials and Methods
Tumors and histologic grading
Excisional biopsy specimens of 83 canine cutaneous mast cell tumors from 83 dogs from the period of 1993–1994 were obtained from the University of Florida Veterinary Medical Teaching Hospital (VMTH) surgical pathology archives. No attempt was made to select cases for criteria such as sex, age, breed, or cutaneous location of the tumor. The average age at the time of diagnosis was 7.5 years (range 0.6–13 years). Genders were intact males (26/83, 31%) and females (15/83, 18%) and neutered males (15/83, 18%) and females (27/83, 33%). Purebred dogs were overrepresented (62/83, 75%). Breeds were mixed (21/83, 25%), Boxer (11/83, 13%), Labrador Retriever (9/83, 11%), Boston Terrier (6/83, 7.2%), Golden Retriever (5/83, 6%), Doberman Pinscher (5/83, 6%), Chinese Shar Pei (5/83, 6%), German Shepherd Dog (3/83, 3.3%), Pug (2/83, 2.4%), Miniature Schnauzer (2/83, 2.4%), Cocker Spaniel (2/83, 2.4%), and one each (1/83, 1.2%) of Bullmastiff, Bull Terrier, Yorkshire Terrier, Fila Brasil, Weimaraner, German Shorthaired Pointer, Beagle, Maltese, Scottish Terrier, Chihuahua, Rhodesian Ridgeback, and Lhasa Apso.
Formalin-fixed, paraffin-embedded sections of each of the above neoplasms were stained with hematoxylin and eosin and another with Giemsa. Each tumor was classified independently by two investigators (Ginn, Gaskin) as grade I (well differentiated), grade II (intermediate differentiation), or grade III (poorly differentiated to anaplastic) by criteria previously established.20,28 Grading criteria included degree of cellularity; discreteness of cellular borders; number, size, and staining characteristics of cytoplasmic granules; frequency of mitotic figures, and consistency of size and shape of tumor cell nuclei. Tumors were histologically graded as grade I (24/83, 29%), grade II (49/83, 59%), or grade III (10/83, 12%) tumors (Table 1).
Tumor, grade, size and p53 staining results of 83 dogs with mast cell tumors.
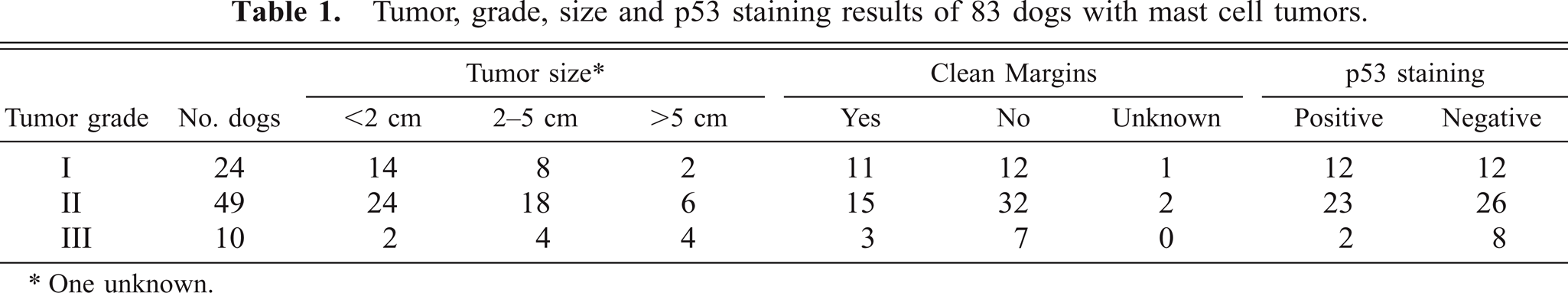
∗ One unknown.
Immunohistochemistry
A polyclonal antibody of rabbit origin (polyclonal antibody CM-1, Signet Laboratories, Dedham, MA) directed against wild and mutant forms of the p53 protein served as the primary antibody. An antigen retrieval technique designed for formalin-fixed, paraffin-embedded tissues was utilized according to the procedure outlined below (Citra Solution, Biogenex, San Ramon, CA).
Five-micrometer serial sections from paraffin blocks containing the above described neoplasms were mounted on Silane (Sigma Chemical Co., St. Louis, MO)-treated slides prior to immunostaining. Sections were immunostained by the standard avidin–biotin–peroxidase complex procedure.12 Avidin and biotinylated peroxidase (Vectastain Elite Kit PK 6102, Vector Laboratories, Inc., Burlingame, CA) and biotinylated goat anti-rabbit antibody (Vector Laboratories) served as the detection system. The chromagen was 3,3′ diaminobenzidine tetrahyrochloride (Sigma Chemical Co.).
Formalin-fixed tissues were deparaffinized and dehydrated by sequential immersion in three changes of xylene followed by four changes of graded ethanol concentrations. Sections were then rinsed in distilled water. Endogenous peroxidase activity was quenched by immersion in 0.3% hydrogen peroxide in phosphate-buffered saline (PBS) for 30 minutes at room temperature and subsequently washed three times in PBS. Tissue sections were then incubated in preheated (90 C) Citra solution diluted in PBS 1:10 for 10 minutes to facilitate unmasking of antigens. After 30 minutes of cooling, the slides were rinsed three times in PBS for 10 minutes per rinse. Nonimmune binding of antiserum was blocked by incubating sections for 30 minutes at room temperature in PBS containing 4% (1:10 dilution) normal goat serum. After washing in PBS, sections were incubated overnight at room temperature with primary antiserum at 1:75 dilution. After rinsing in PBS, the slides were incubated for 45 minutes with biotinylated goat anti-rabbit antibody prepared according to the Vectastain Elite ABC kit directions. PBS-washed sections were then incubated for 30 minutes in an avidin–biotin complex peroxidase solution made according to kit directions and again washed in PBS. Slides were developed for 5 minutes in a solution of 3,3′-diaminobenzidine (50 mg/250 ml) in PBS supplemented with hydrogen peroxide (three drops of 3%). Final steps consisted of additional rinsing in PBS, counterstaining with hematoxylin, clearing, and mounting. All incubations were carried out at room temperature unless otherwise indicated.
Positive controls consisted of formalin-fixed, paraffin-embedded sections of a canine colon carcinoma and a canine osteogenic sarcoma shown previously to label positive for p53 tumor-suppressor protein (Fig. 1).30 Negative controls for each case were performed in parallel by substituting normal rabbit serum for the primary antibody. Additionally, regions of normal skin with resident mast cells within many of the case sections provided additional internal negative controls.
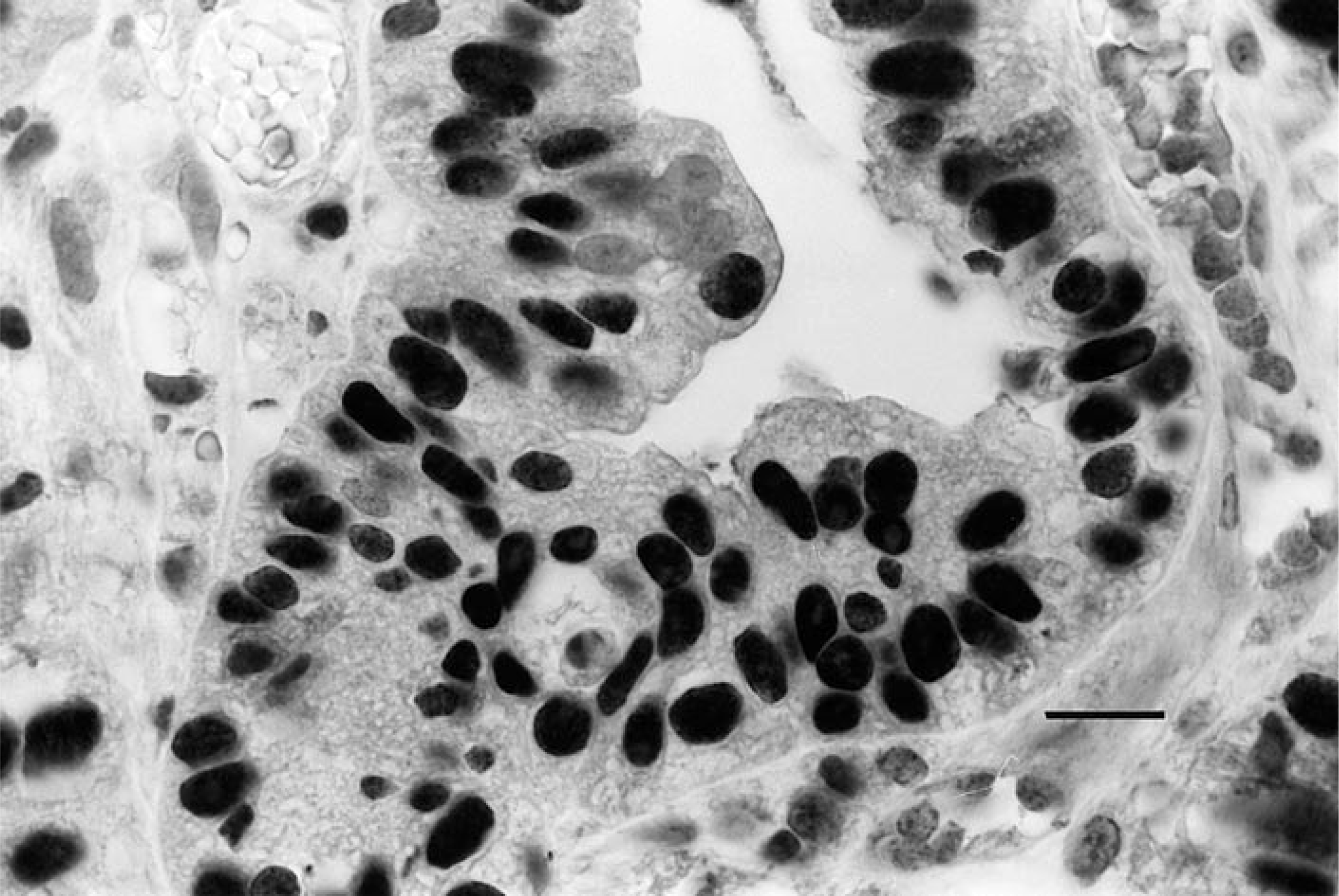
Colonic adenocarcinoma; dog. Tumor cell nuclei are labeled for p53. This tissue served as a positive control. Avidin–biotin–peroxidase complex method, CM-1 antibody, Mayer's hematoxylin counterstain. Bar = 12 μm.
Microscopic evaluation
Light microscopic evaluation of immunohistochemically stained sections was performed by two investigators (Ginn, Gaskin) independently. Any discrepancies were resolved by review of the case in question. The quality of each immunohistochemically stained batch was assessed by comparing the sections with an accompanying positive control slide. Estimates of the percentage of tumor cells with positively labeled nuclei were made, and a score of 0, 1, 2, or 3 was assigned according to the following system: 0 = no reactivity; 1 = <10%, 2 = 10–50%, 3 = >50% of tumor cells staining positively (Fig. 2).9,10,23 On the basis of previous reports linking p53 protein accumulation with mutation of the p53 gene, tumors scoring 2 or 3 were considered p53 positive.9,10,23
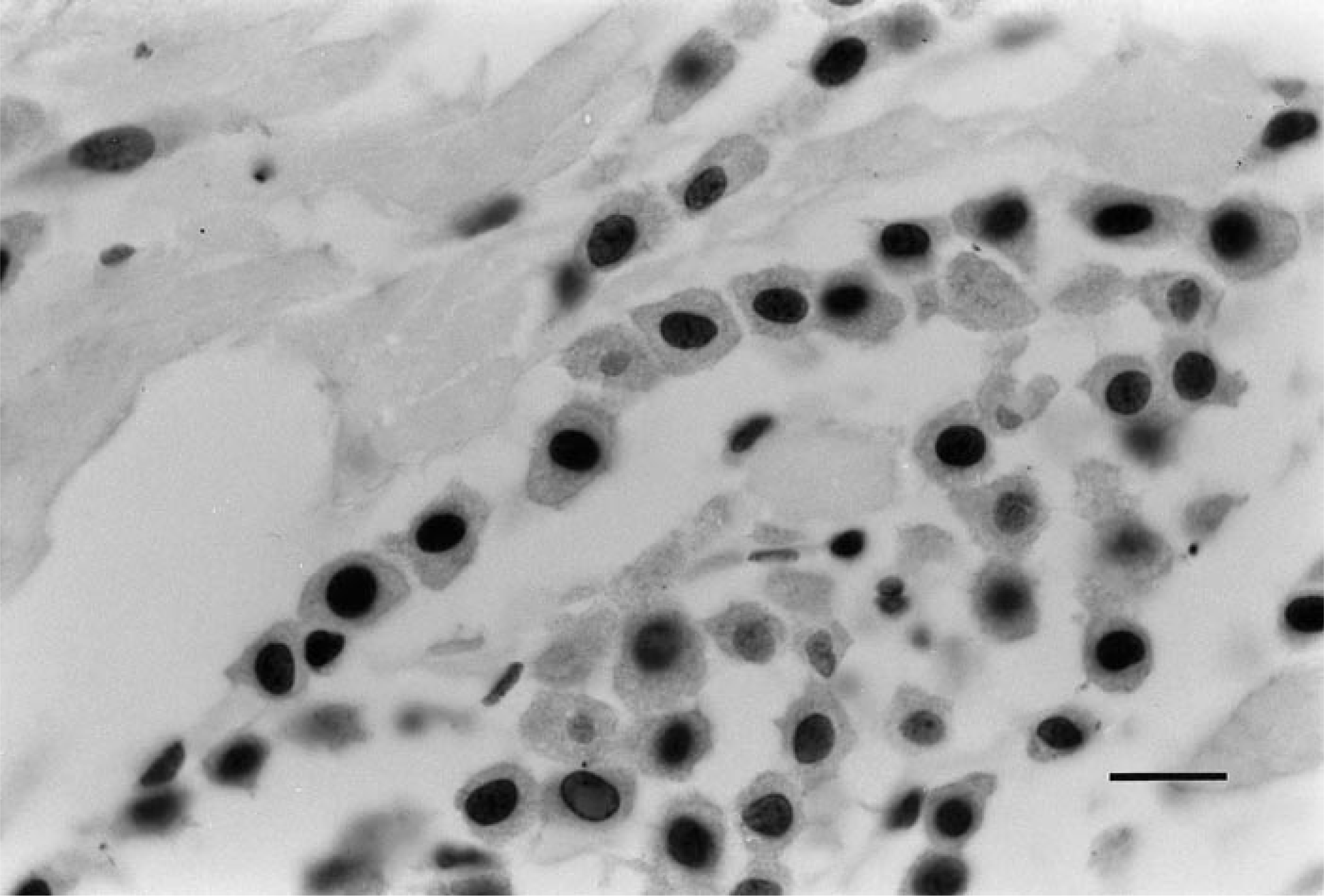
Well-differentiated (grade I) cutaneous mast cell tumor; dog. Note the nuclear staining of the majority of tumor cells within the field. No staining of the mast cell cytoplasm is present. Avidin–biotin–peroxidase complex method, CM-1 antibody, Mayer's hematoxylin counterstain. Bar = 12 μm.
Clinical evaluation
Retrospective data were obtained from the VMTH medical records and a telephone survey of cases mailed in to the VMTH surgical biopsy service. Signalment, tumor size and location, surgical margin status, and date and cause of death were abstracted. All attempted treatments involved surgical excision and margins were assessed as clean (no evidence of tumor cells [29/83, 34.9%]), dirty (tumor cells extend to the histologic margins of the biopsy sample [51/83, 61.4%]), or not determined (3/83; 3.6%). Tumors were grouped according to largest tumor diameter where a = <2 cm (40/83, 48.2%), b = 2–5 cm (30/83, 36.1%), and c = >5 cm (12/83, 14.5%). Tumor size was not available in one case (1/83, 1.2%). Tumor locations were grouped into four categories where 1 = head or neck (12/83, 14.5%), 2 = legs (28/83, 33.7%), 3 = thorax, back, abdomen, or axilla (33/83, 39.8%), and 4 = prepuce, scrotum, or inguinal area (9/83, 10.8%). Tumor location could not be determined in one (1.2%) of the cases. Presence of metastasis at the time of diagnosis and time to recurrence were not available. The p53 staining results for the four overrepresented breeds in the study (Boxers, Boston Terriers, Labrador Retrievers, and Golden Retrievers) were compared with the population as a whole. Survival time was defined as the time from surgical excision to death resulting from progression of disease. Median survival time was defined as the time at which 50% of the dogs had died or were euthanatized because of progression of disease. For statistical evaluation, survival times were grouped according to breed, histologic grade, tumor size and location, surgical margin status, and p53 staining.
Statistical analysis
The Kaplan–Meier product limit method was used to generate survival curves.16 The differences between groups were compared by the Breslow and Mantel–Cox tests of significance between survival curves.7 Tumors staining for p53 were compared according to breed, histologic grade, size, and location by Fisher's exact test. Linear trends in ordered proportions were detected by an Exact test.19 Data were analyzed with the Statxact program.19 Differences between groups were considered statistically significant at the 5% level (P < 0.05).
Results
Immunohistochemical evaluation of 83 dogs
No statistically significant difference with respect to p53 labeling was present between histologic grades, breeds, or tumor sizes. The relative frequencies of mast cell tumors graded as well differentiated, intermediate differentiation, or poorly differentiated in the group of 83 dogs were 29, 59, and 12%, respectively. Surgical margins were free of tumor cells in 29 of 80 (36%) tumors with specified margins.
Immunohistochemical staining for p53 was positive in 37 of 83 (44.6%) tumors. Positivity for p53 was demonstrated in 12 of 24 (50%) grade I (well differentiated) tumors, 23 of 49 (46.9%) grade II (intermediate differentiation) tumors, and 2 of 10 (20%) grade III (poorly differentiated) tumors (Table 1). A statistically significant (P < 0.019) higher proportion of tumors from category 1 (head and neck [10/12, 83.3%]) stained positive for p53 than tumors from category 2 (legs [10/28, 35.7%]), category 3 (thorax, back, abdomen, and axilla [13/33, 39.4%]), or category 4 (prepuce, scrotal, or inguinal areas [4/9, 44.4%]).
Of the nine tumors in our study from category 4, three (33.3%) were well differentiated, four (44.4%) were of intermediate differentiation, and two (22.2%) were poorly differentiated. Four of the nine (44.4%) category 4 tumors were p53 positive. Three of these four (75.0%) were of the well-differentiated type and one (25.0%) was of intermediate differentiation.
Eleven out of 83 (13%) of our cases were Boxers, and seven (63.6%) of these tumors were positive for p53. Three of the seven (42.9%) positive tumors were of the well-differentiated type. Eight of the 11 (72.7%) tumors from Boxers were of intermediate differentiation and four (50%) were p53 positive. No Boxer in our study had poorly differentiated tumors. Tumors were located on the head or neck (2/11, 18.2%), trunk (7/11, 63.6%), legs (1/11, 9.1%), or inguinal area (1/11, 9.1%). The majority (7/11, 63.6%) were less than 2 cm in largest diameter. The p53 reactivity of tumors in Boxers was not significantly different when compared with other breeds.
Clinical follow-up for 53 dogs
Survival data were available for 53 of 83 (63.9%) dogs. The average age at the time of diagnosis for these 53 dogs was 7.3 years (range = 2–12 years). The 53 cases included 19 (35.8%) grade I, 30 (56.6%) grade II, and 4 (7.5%) grade III tumors. Tumor cells labeled for p53 in 25 of 53 (47%) cases for which clinical follow-up was available and were observed in 11 of 19 (57.9%) grade I, 13 of 30 (43.3%) grade II, and 1 of 4 (25%) grade III tumors. When tumors were grouped according to size, 28 of 53 (52.8%) were <2 cm (group a), 17 of 53 (32.1%) were 2–5 cm (group b), and 8 of 53 (15.1%) were >5 cm (group c) in diameter. When grouped according to location, 6 of 53 (11.3%) were from the head or neck, 19 of 53 (35.8%) from the legs, 20 of 53 (37.7%) from the thorax, back, abdomen, or axilla, and 8 of 53 (15.1%) from the prepuce, scrotum, or inguinal area. Mean survival time for the group of 53 dogs was 12.1 months (range = 10 days–48 months). Survival data were available for 8 of 11 Boxers. Only one Boxer died as a result of progressive disease at the end of the study. No statistical differences in survival times were found between dogs that were positive or negative for p53 or that had clean or dirty surgical margins or that were in different tumor location subgroups (category 1, 2, 3, or 4). Mast cell tumors of the inguinal, prepucial, scrotal, and perineal locations (category 4) have been associated with more aggressive biological behavior and lesser differentiation than tumors in other locations.28 The only dogs with category 4 tumors with short survival times were the those with the poorly differentiated tumors. Reactivity for p53 did not correlate with a poor prognosis for dogs with tumors in category 4 locations. However, a trend toward a significant difference in survival was found between those dogs with dirty surgical margins and those without histologic evidence of tumor in the surgical margins (P < 0.0585). Median survival times could not be determined for grades I and II; however, median survival time for grade III was 1 month. Survival was statistically significantly shorter for dogs with grade III tumors than for dogs with tumors from grades I or II (P < 0.0001). Median survival time could not be determined for dogs grouped according to sizes a or b; however, median survival time for size c was 5 months. The survival time of dogs in group c was statistically significantly shorter than in groups a and b (P < 0.0001).
Discussion
The results of this study suggest that p53 immunoreactivity is not useful as a prognostic indicator for canine cutaneous mast cell tumors. Immunoreactivity for p53 has been associated with a poor prognosis in a number of tumors in humans. No comparative data for mast cell tumors in humans are available because mast cell tumors are rare in humans.28 A recent study reported that overexpression of p53 was common in a diverse group of spontaneously occurring tumors in dogs.9 Sixteen mast cell tumors were analyzed and 4 of 14 (25%) were positive for p53. The body site, breed, histologic grade, tumor size, and survival time were not indicated. The authors of the study concluded that positive immunoreactivity did not necessarily correlate with the expected clinical outcome because some tumors with known aggressive biological behavior did not label whereas some relatively innocuous tumors did label.
The lack of correlation of labeling with suggested biological behavior in the current study may reflect a lack of specificity of immunohistochemical detection of p53 for the tumor-suppressor gene mutation. Although immunohistochemical detection of p53 protein is accepted as indication of gene mutation, the link between immunohistochemistry and molecular genetic alteration is not conclusively established. The majority of studies have been conducted with human neoplasms. The relationship of immunolabeling and gene mutation in the dog may not exactly parallel that in man. That certain cellular and viral proteins may also bind and stabilize WTP53 and that, under certain conditions, stressed cells with intact p53 genes may overexpress p53 protein are well established. In addition, what degree of overexpression of mutant p53 is clinically significant has not been established. A tumor with <50% of cells labeled may not be clinically more aggressive, or the significance of percentage of cells labeled might vary by tumor type. Malignant transformation of cells almost invariably requires loss of normal function of a combination of tumor-suppressor genes and proto-oncogenes. Analyzing for one molecular change may not be sufficient to predict biological behavior of some tumors. False-negative results may also have occurred because of the negative effects of prolonged formalin fixation, nonprotein-producing p53 mutations, or lack of antibody recognition of epitopes of canine p53 proteins in some of the cases. The use of a panel of different antibodies to the p53 protein may have yielded additional positive cases.
The trend toward statistically significant differences in survival for the dogs in this study with dirty versus clean margins is consistent with previous reports and would likely be statistically significant if a larger number of dogs were evaluated. These findings support the widely held clinical impression that 3.0-cm margins are desirable for complete surgical excision and that complete excision results in improved survival times.2,18,20,28
Histologic grade has been the most consistent prognostic factor available for dogs with cutaneous mast cell tumors, but its reliability is not without controversy.11,28 Immunoreactivity for p53 was demonstrated in mast cell tumors of all histologic grades in this study. No statistically significant difference was found between tumor histologic grade and p53 immunoreactivity. Tumors of intermediate grade were most common, and poorly differentiated mast cell tumors were the least common in our study. This finding is similar to that reported by Patnaik et al.20 but in contrast to that reported by Bostock,2 where poorly differentiated tumors were most common.28 A study by Hottendorf and Neilson15 found well differentiated tumors to be most common. These differences most likely reflect the inherent subjectivity of the criteria used by various investigators for grading mast cell tumors.2,11,15,20 The subjectivity of the grading system may also have influenced the final outcome of this study because some tumors might have been placed in a different grade by another investigator.
The short median survival time of 1 month observed in our dogs with grade III tumors when compared with dogs with grades I and II tumors is consistent with the decreasing trend in longevity with increasing histologic grade. However, the median survival time for dogs with grade III tumors was shorter than the median survival times and survival rates reported by others.2,20 This difference may be because of the inability to determine the clinical stage of disease because of unavailable information or variable time between detection of tumor and excision.
Tumor diameter of greater than 5 cm was associated with a poor long-term median survival time of 5 months in this study. The association of large tumor diameter and poor prognosis suggests that size may be a useful determinant in clinical staging of the extent of disease. Clinical staging for mast cell tumors according to the currently used system from the World Health Organization for Tumors in Domestic Animals does not use size in the classification system.28
In summary, this study attempted to identify relationships between p53 protein immunoreactivity and the histologic grade, tumor location, tumor size, breed, and survival time in dogs with cutaneous mast cell tumors. Immunoreactivity for p53 was found in tumors of all histologic grades. The only significant difference between subgroups was that tumors located on the head and neck were positive for p53 more often than tumors from other locations. Differences in positive reactivity were not statistically significant within or between any other subgroups investigated. Although p53 tumor-suppressor protein abnormalities may play a role in tumor development or behavior in some canine cutaneous mast cell tumors, immunoreactivity was not associated with lack of tumor differentiation, tumor locations previously shown to demonstrate aggressive biological behavior, breed predisposition, or survival times. Immunoreactivity for p53 may not be a major determinant of biological behavior of canine cutaneous mast cell tumors.
Footnotes
Acknowledgements
We thank biological scientist Betty J. Hall for her expertise and assistance in immunohistochemistry.
