Abstract
This article describes a newly recognized highly malignant neoplastic entity in young bearded dragons (Pogona vitticeps), gastric neuroendocrine carcinomas, which readily metastasize. Ten bearded dragons with histories of anorexia (8), vomiting (3), hyperglycemia (2), and anemia (3) were included in this study. All animals had neoplastic masses in their stomach, with metastasis to the liver. Microscopically, 6 of these neuroendocrine carcinomas were well-differentiated and 4 were poorly differentiated. For further characterization, immunohistochemistry for protein gene product 9.5, neuron-specific enolase, endorphin, chromogranins A and B, synaptophysin, somatostatin, insulin, glucagon, gastrin, pancreatic polypeptide, and vasoactive intestinal peptide was performed on 5 animals. Because only immunolabeling for somatostatin was consistently observed in all neoplasms, a diagnosis of somatostatinoma was made for these 5 bearded dragons. Some neoplasms also exhibited multihormonal expression. Electron microscopy performed on 1 tumor confirmed the presence of neuroendocrine granules within neoplastic cells. Gastric neuroendocrine carcinomas, and specifically somatostatinomas, have not been previously reported in bearded dragons, or other reptiles, and may be underdiagnosed due to inconsistent, ambiguous clinical signs. In humans, pancreatic somatostatinomas are associated with a syndrome of hypersomatostatinemia, which includes hyperglycemia, weight loss, and anemia, as observed in some of these bearded dragons. Somatostatinomas in humans are commonly associated with neurofibromatosis type 1 (Von Recklinghausen's disease), caused by a mutation in the tumor suppressor gene NF1, which results in decreased expression of neurofibromin. In all 5 animals examined, neoplasms exhibited decreased neurofibromin expression compared with control tissues, suggesting that decreased functional neurofibromin may play a role in the pathogenesis of somatostatinomas in bearded dragons.
Keywords
Bearded dragons, notably the species Pogona vitticeps, are popular pets and exhibit animals. They constituted approximately 3% of all reptile biopsy and necropsy submissions to a private veterinary pathology laboratory, Northwest ZooPath (Monroe, WA), from 1994 to 2006.6, 9 Commonly observed diseases include metabolic bone disease due to nutritional imbalances or inadequate exposure to ultraviolet light, egg binding, bacterial and fungal dermatitides, and gastrointestinal disease of various causes (intestinal impaction, coccidiosis, adenoviral infection). Neoplastic diseases are relatively uncommon in reptiles compared with other animals and have infrequently been reported in bearded dragons.6, 9, 15 To our knowledge, only peripheral nerve sheath tumors and myelocytic leukemias have both been described to occur with some frequency in bearded dragons.6, 7, 9, 13, 24 Herein we describe an apparent emerging trend in the development of gastric neuroendocrine carcinomas in this species, with a relative prevalence of this tumor of 1.5% of submitted cases to Northwest ZooPath. Given the relatively common occurrence of gastrointestinal disease processes affecting bearded dragons and the frequently similar, ambiguous clinical signs common to many of these diseases, in addition to the relative infrequency of neoplasia in these animals, the diagnosis of gastric neoplasia may be missed by clinicians who lack awareness of this entity. The objective of the present study was to describe the clinical signs and gross, histologic, immunohistochemical, and ultrastructural characteristics of these newly recognized neoplasms, and to compare and contrast these features with those of gastrointestinal neuroendocrine carcinomas in humans.
Materials and Methods
Animals
A review of the records at Northwest ZooPath and the Veterinary Medical Teaching Hospital at the University of Wisconsin identified 10 bearded dragons with gastric neuroendocrine tumors. Eight animals (Nos. 1, 2, 3, 4, 5, 7, 8, and 9) were from Northwest ZooPath, 2 animals (Nos. 6 and 10) were from the University of Wisconsin.
Histopathology
Sections 3–5 μm in thickness were prepared from formalin-fixed, paraffin-embedded tissue samples from each bearded dragon. Sections from each tissue were routinely stained with HE.
Immunohistochemistry
An extended immunohistochemical (IHC) panel was performed on tissue from 5 bearded dragons (Nos. 1, 2, 4, 5, and 6). This panel included IHC to detect protein gene product 9.5 (PGP9.5), synaptophysin, gastrin, glucagon, chromogranins A and B, insulin, neuron-specific enolase (NSE), endorphin, somatostatin, vasoactive intestinal peptide (VIP), pancreatic polypeptide (PPP), and neurofibromin. Some of these IHC markers have previously been shown to exhibit cross-reactivity in other reptile species.2, 5, 14 Deparaffinization, antigen retrieval, IHC staining, and counterstaining was performed on the Bond maX Automated Staining System (Vision BioSystems, Leica, Bannockburn, IL) using the Bond Polymer Detection System (Vision BioSystems, Leica). Antigen retrieval for a few antibodies was achieved off-line (Table 1, steam retrieval) by incubation of slides in antigen retrieval solution (Dako, Carpinteria, CA) in a steamer (Black & Decker, Towson, MD) for 20 minutes. Primary antibodies and their dilutions, source, host, type of antigen retrieval, and positive control tissue are listed in Table 1. The immunoreaction was visualized with 3,3-diaminobenzidine substrate (Vision BioSystems, Leica) and sections were counterstained with hematoxylin. For negative controls, the primary antibodies were replaced with homologous nonimmune sera or buffer. Only cytoplasmic staining within neoplastic cells was evaluated as positive.
Primary antibodies used for immunohistochemistry.
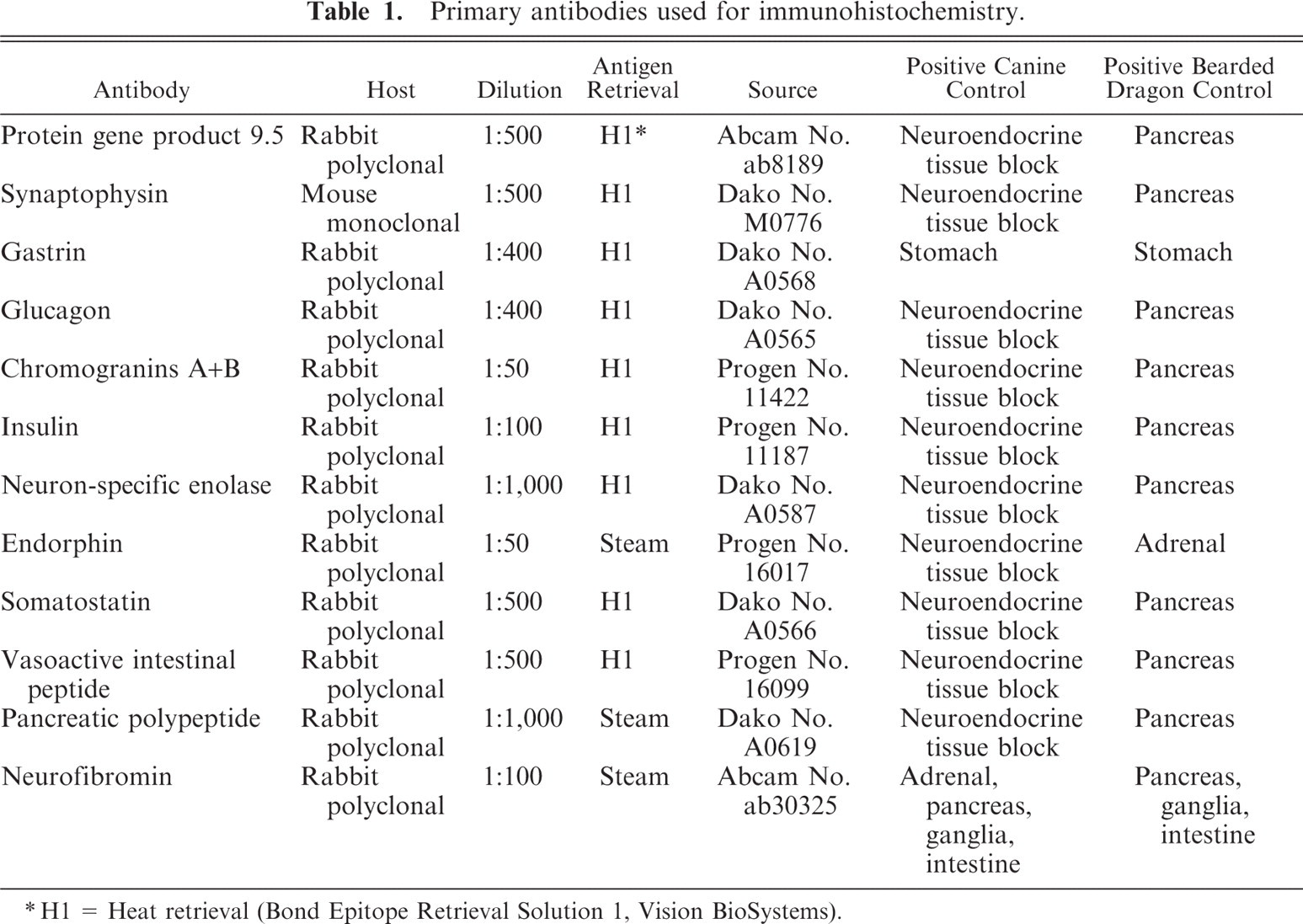
H1 = Heat retrieval (Bond Epitope Retrieval Solution 1, Vision BioSystems).
Electron microscopy
Electron microscopy was performed on tissue from bearded dragon No. 6. Briefly, formalin-fixed neoplastic tissue from the liver and stomach was subsequently retrimmed and submitted for transmission electron microscopy. After postfixation in 1% buffered osmium tetroxide, samples were washed in phosphate-buffered saline. Samples were then dehydrated in a graded ethanol series followed by 100% acetone, infiltrated in a graded ethanol/Spurr's resin (Spurr Embedding Kit, Ted Pella Inc., Redding, CA) series, and polymerized in 100% Spurrs at 60°C for 2 days. Thick sections were cut, mounted on glass slides, stained by toluidine blue O, and examined by light microscopy. Ultrathin sections (70 nm) were collected on 200-mesh copper grids and poststained with 2% uranyl acetate, followed by Reynolds lead citrate. Grids were examined on a Hitachi H-7000 transmission electron microscope (Hitachi High Technologies, Schaumburg, IL). Digital micrographs were acquired on an Olympus MegaView III (Olympus Soft Imaging Solutions Corp., Lakewood, CO).
Results
Clinical presentation
Table 2 summarizes signalment, clinicopathologic abnormalities when available, and gross and histologic findings for each case. Availability and completeness of clinical history varied among the 10 bearded dragons. Six animals were male, 4 were female. The age was known for 8 animals and ranged from 1.5 to 3 years. Clinical signs were provided for 8 animals. The most commonly reported sign was anorexia (8 animals, Nos. 1–7, 10). Others included weight loss (2 animals, Nos. 4, 7), weakness (2 animals, Nos. 4, 8), and vomiting (3 animals, Nos. 2, 9, 10). Clinicopathologic abnormalities were only reported for 4 animals and included anemia (3 animals) and severe hyperglycemia (2 animals).
Signalment, clinicopathologic abnormalities, and lesions associated with gastric neuroendocrine carcinomas in bearded dragons.
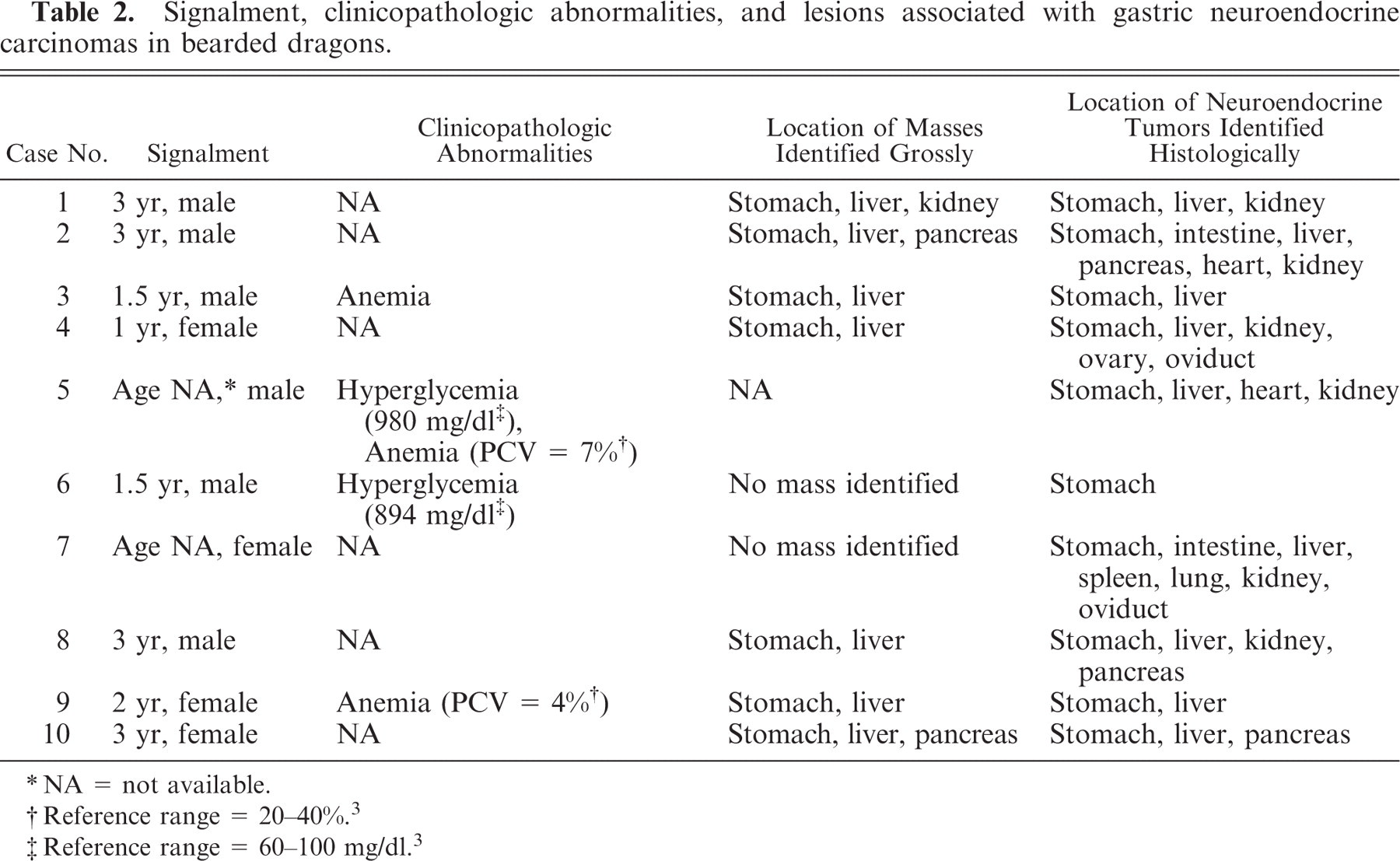
NA = not available.
Reference range = 20–40%.3
Reference range = 60–100 mg/dl.3
Gross findings
Gross necropsy findings were available for 9 of the 10 bearded dragons. Grossly, neoplastic masses were identified in 7 of the 9 animals, and all 7 had grossly visible masses in their stomach and liver. Stomach masses were variably sized, multinodular, but well-circumscribed, pale tan to white, and firm. They protruded into the gastric lumen, causing variable ulceration of the gastric mucosa (Fig. 1). Masses in the liver were smaller and more numerous and presented as discrete nodules that extended throughout the hepatic parenchyma (Fig. 2). Two of these animals also had grossly recognizable masses in their pancreas and in another animal, in its kidney. No masses were identified grossly in 2 animals.
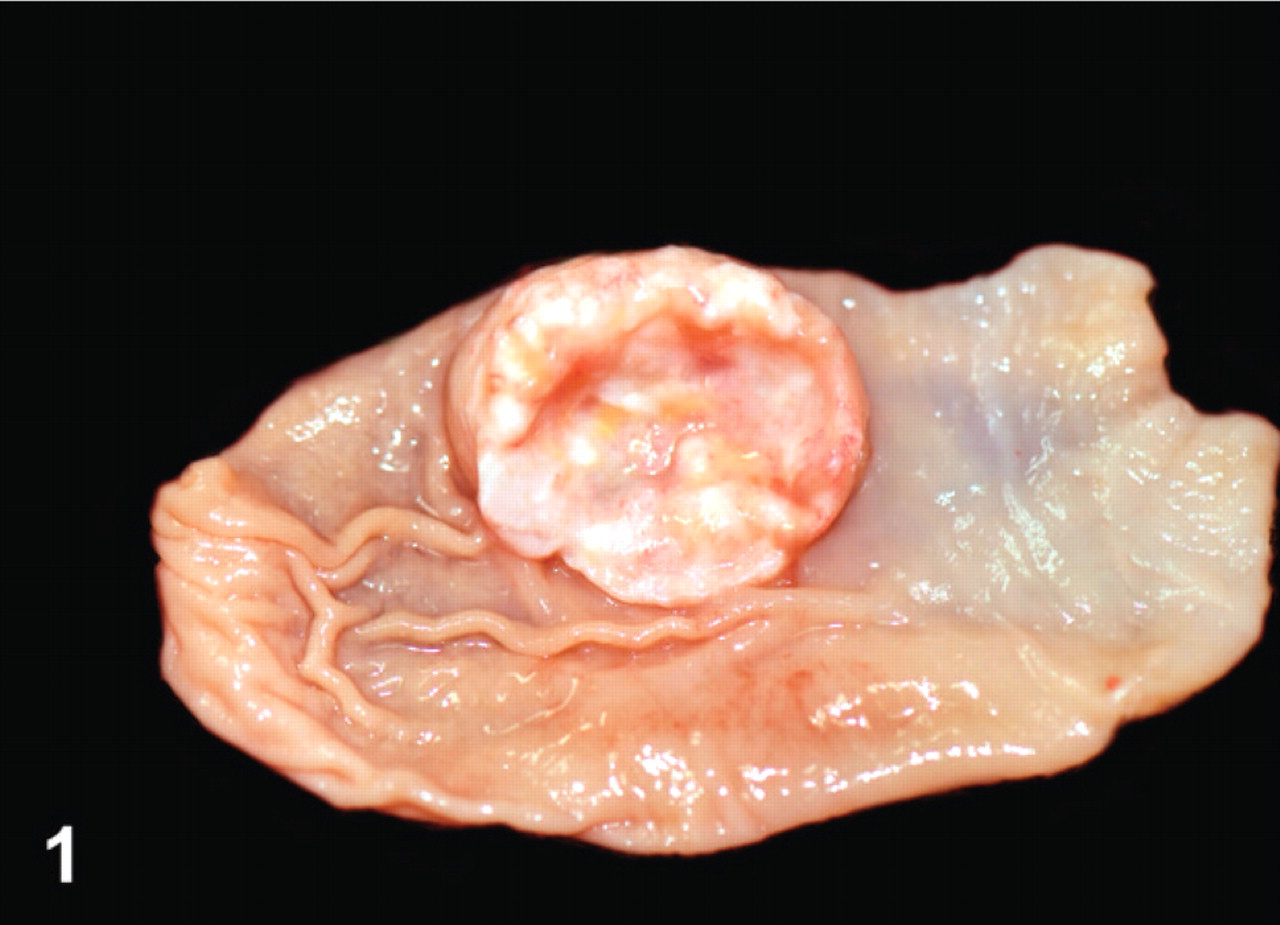
Stomach; bearded dragon No. 9. A single discrete neuroendocrine carcinoma protrudes from the luminal surface. Photograph courtesy of Dr. Steven Barten.
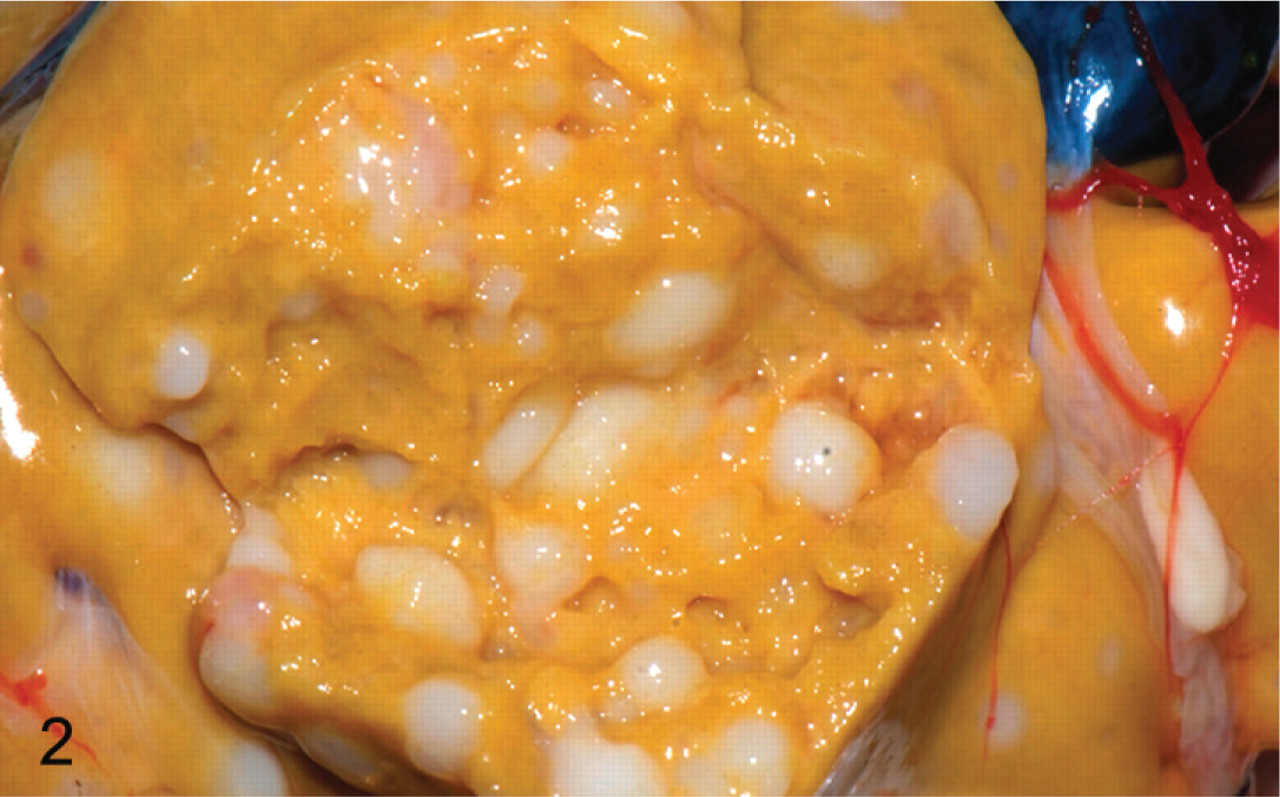
Liver; bearded dragon No. 9. Numerous discrete nodules representing hepatic metastases of neuroendocrine carcinoma. Photograph courtesy of Dr. Steven Barten.
Histopathology
Histologic examination revealed a neuroendocrine neoplasm within the gastric submucosa of all 10 animals. Metastases were identified in the liver of all 10 animals as well as in other organs in some animals: kidney (6), pancreas (3), intestine (2), heart (2), oviduct (2), and ovary (1). The neoplastic masses were located primarily within and expanded the gastric submucosa (Fig. 3). However, infiltration of neoplastic cells into the overlying mucosa (Fig. 3) or the muscle tunics of the gastric wall was commonly observed. Infiltration of submucosal veins and lymphatics was also present in all cases. Neoplastic cells were arranged in packets, acini, and trabecular cords, separated by a fine fibrovascular stroma (Fig. 4). They commonly formed rosettes and pseudorosettes. Cells were relatively monomorphic with round euchromatic nuclei and scant cytoplasm. In 4 bearded dragons (Nos. 4, 6, 7, and 8), neoplastic cells were severely anaplastic with marked nuclear pleomorphism and a high mitotic index (Fig. 5). The microscopic appearance of all neoplasms was consistent with a diagnosis of neuroendocrine carcinoma. We considered those tumors with relatively monomorphic cell populations to be well-differentiated carcinomas, and those with features of marked anaplasia to be poorly differentiated carcinomas.
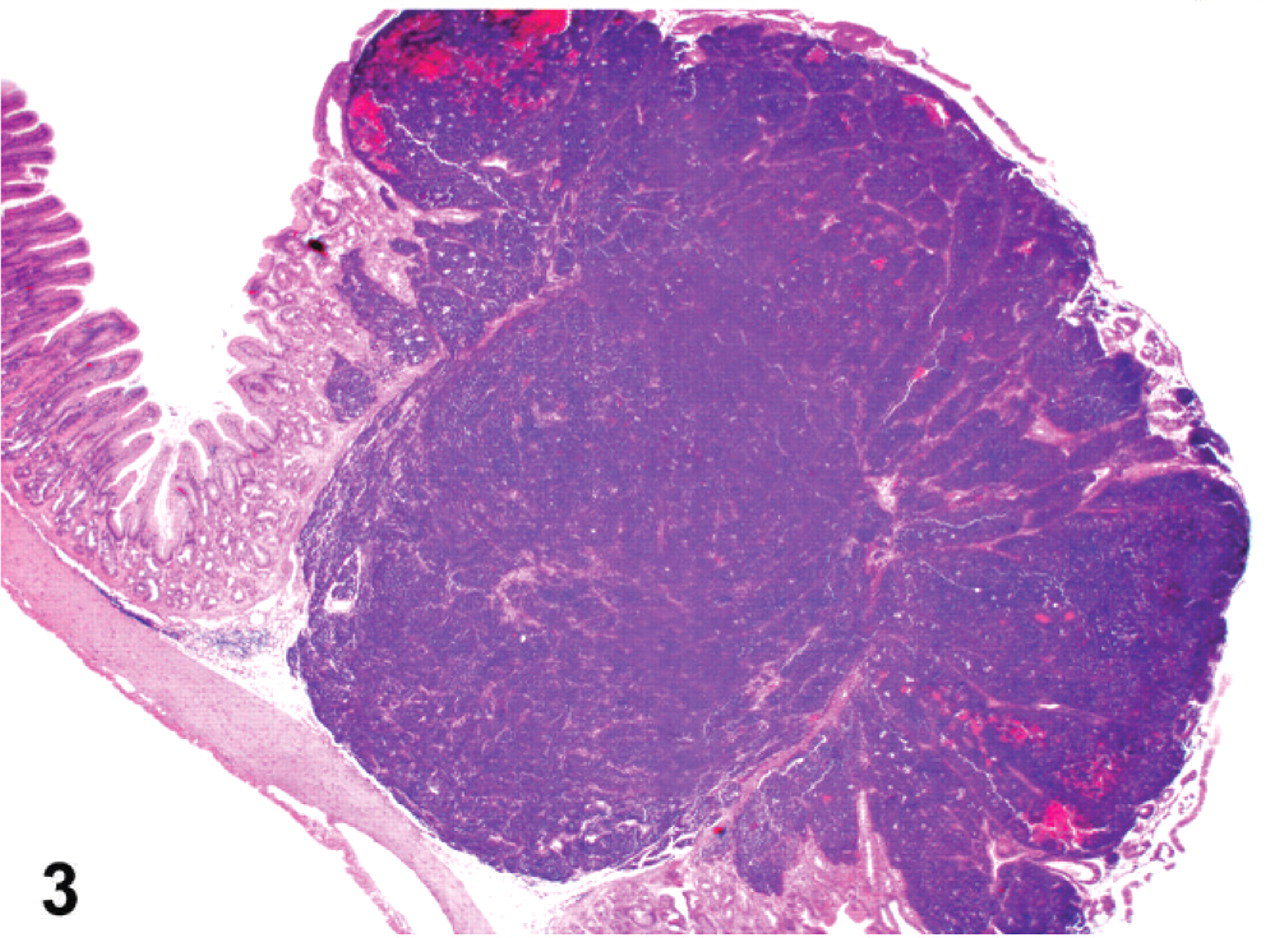
Stomach; bearded dragon No. 6. A neuroendocrine carcinoma expands the gastric submucosa and invades and destroys the mucosa. HE. Magnification = ×12.5.
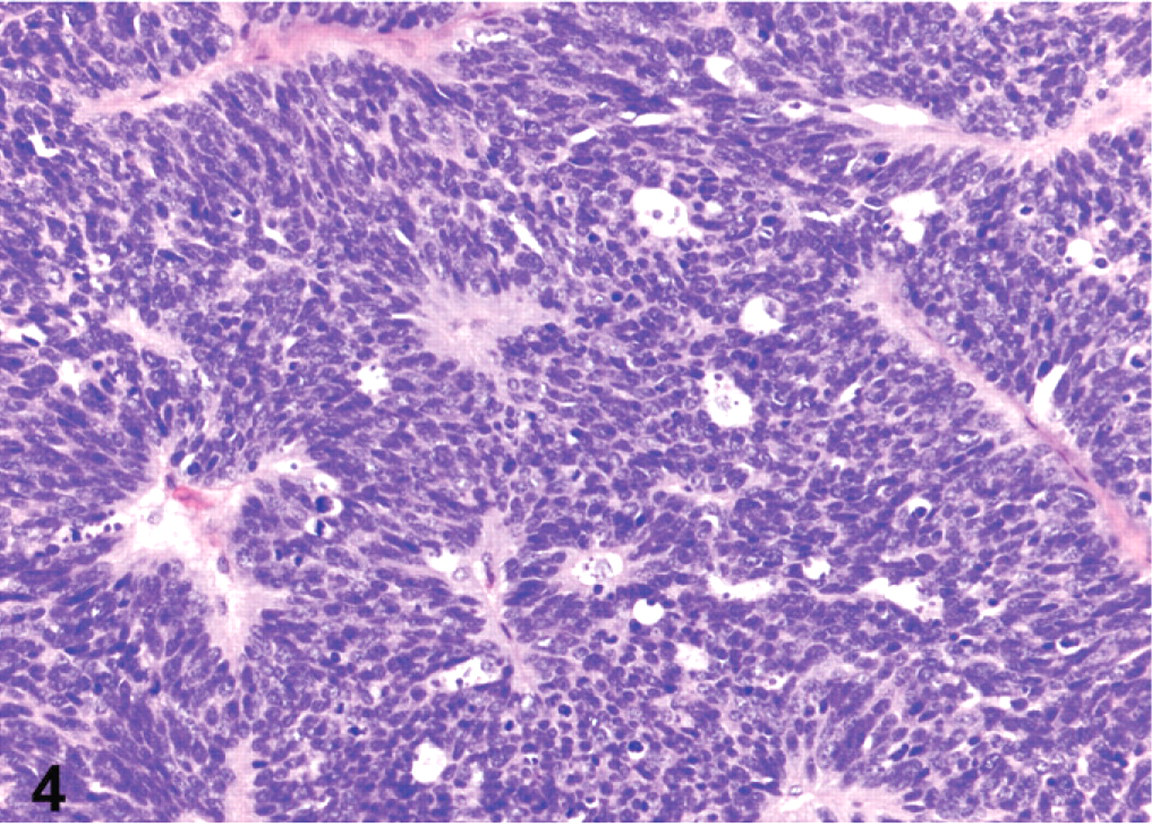
Stomach; bearded dragon No. 1. Well-differentiated gastric neuroendocrine carcinoma. HE. Magnification = ×200.
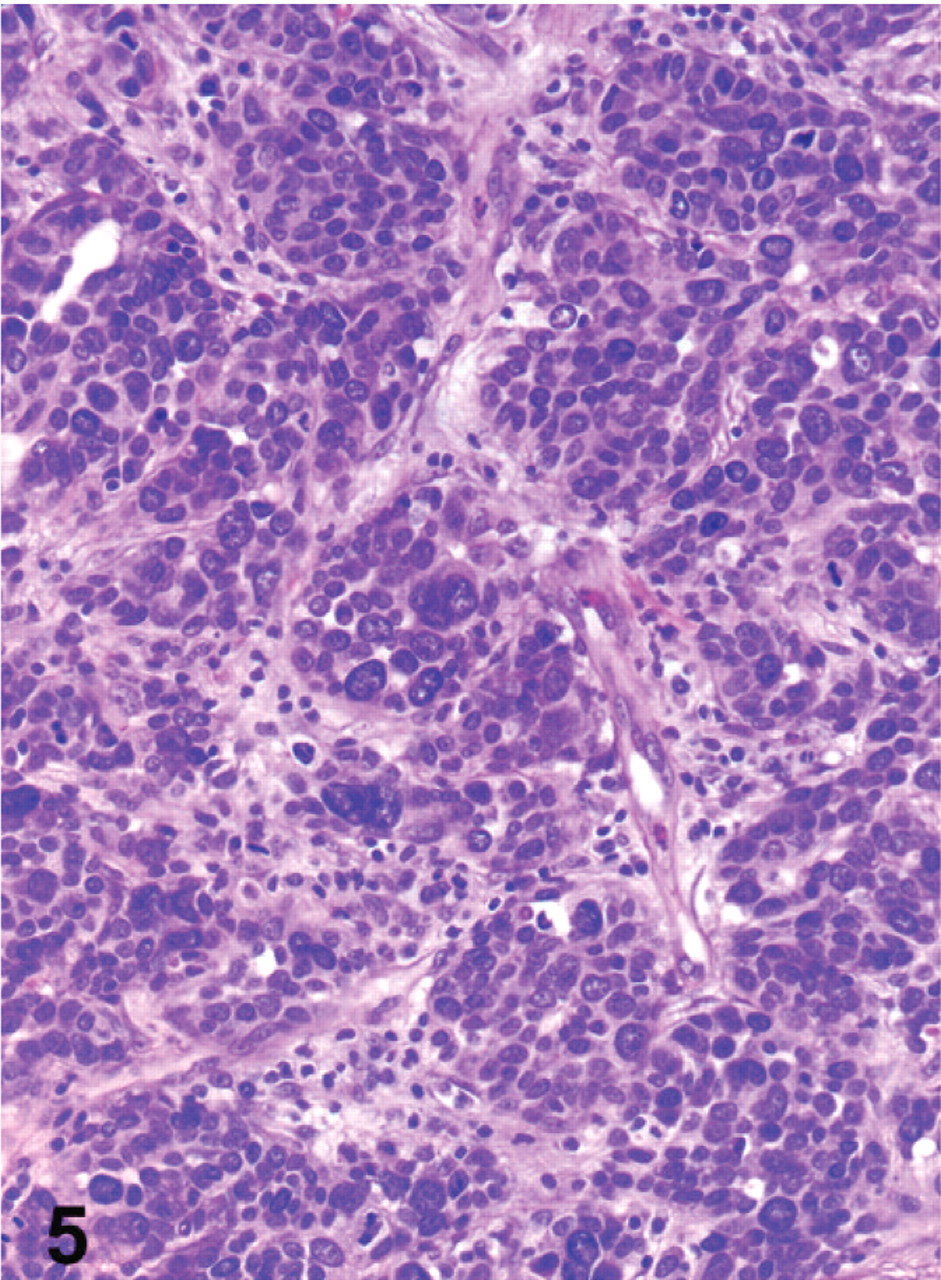
Stomach; bearded dragon No. 4. Poorly differentiated gastric neuroendocrine carcinoma in a bearded dragon. HE. Magnification = ×200.
Immunohistochemistry
In all 5 bearded dragons, neoplasms had variable antigen-specific labeling of neoplastic cells for somatostatin (Fig. 6). There was also rare, focal to multifocal labeling of 3 neoplasms for gastrin (Nos. 2, 4, and 5), of 2 neoplasms for synaptophysin (Nos. 5 and 6), and of 2 neoplasms for PPP (Nos. 4 and 5). Chromogranins A and B (case No. 4), and glucagon (case No. 5) were only detected in 1 of the 5 neoplasms. All neoplasms were negative for PGP9.5, NSE, insulin, endorphin, and VIP. Based on the consistent, variable expression of somatostatin observed in all 5 animals tested, a diagnosis of malignant somatostatinoma was made. However, multihormonal expression was common. IHC for neurofibromin was also performed for these 5 bearded dragons. In all animals, there was decreased immunoreactivity in the neoplastic cells compared with both canine and bearded dragon control tissues.
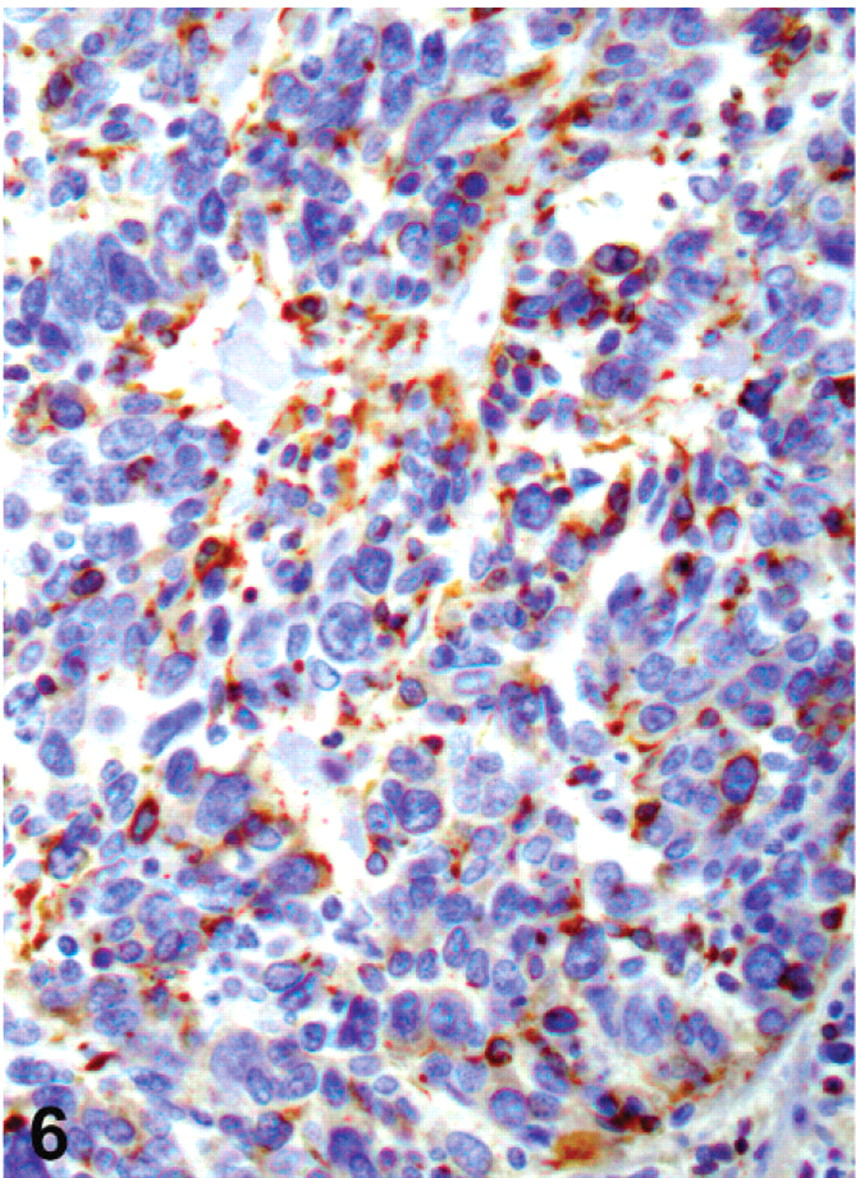
Stomach; bearded dragon No. 5. Gastric neuroendocrine carcinoma with immunohistochemical staining of neoplastic cells for somatostatin (dark brown). Hematoxylin counterstaining. Magnification = ×400.
Electron microscopy
Electron microscopic examination of a gastric and a liver mass revealed 130- to 250-nm diameter spherical, membrane-bound, dense-core granules typical of neuroendocrine granules in the cytoplasm of neoplastic cells (Fig. 7).
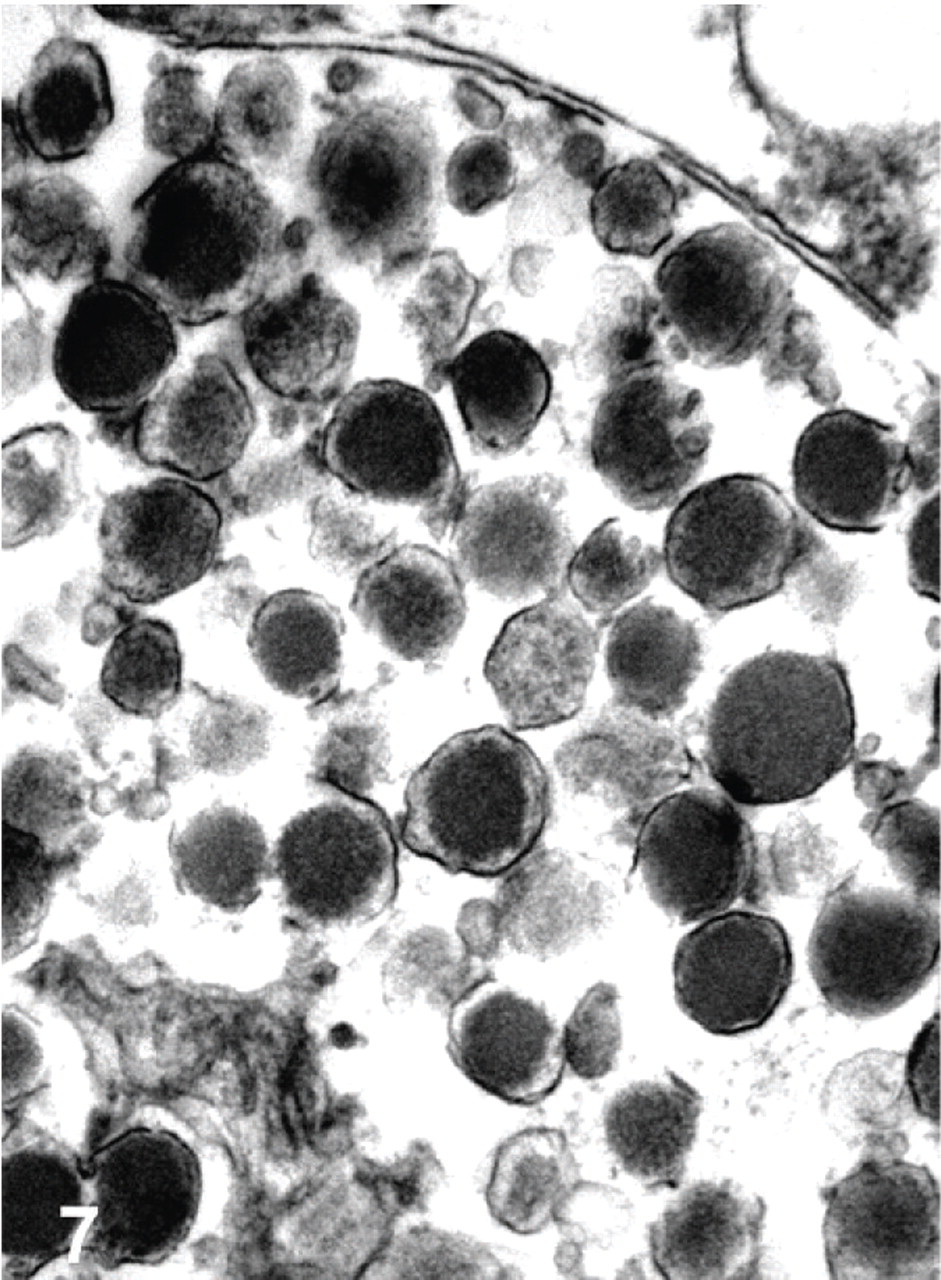
Transmission electron micrograph. Stomach; bearded dragon No. 10. Gastric neuroendocrine carcinoma with membrane-bound dense core granules within the cytoplasm of a neoplastic cell. Bar = 500 nm.
Discussion
Based on gross and microscopic findings, we conclude that the bearded dragons in this study had primary gastric neuroendocrine carcinomas with widespread metastasis, primarily to the liver. Results of IHC and electron microscopy support the diagnoses of neuroendocrine carcinomas, and IHC results further allow us to classify these tumors as somatostatinomas, some of which had multihormonal expression.
In reptiles, as in other animals, endocrine cells are either organized into discrete endocrine glands (pituitary, thyroid, parathyroid, adrenal, pancreatic islets) or distributed throughout many organ systems (e.g., lung, gastrointestinal tract, reproductive tract) in small clusters and individually as part of the dispersed neuroendocrine system.2, 5, 10, 12, 14 Neuroendocrine neoplasms of nonendocrine organs may arise from these dispersed endocrine cells. Such neoplasms occur throughout the gastrointestinal tract of humans and have been less frequently reported in other animals.12, 21 Classification of these neoplasms has proven difficult in both veterinary species and in humans, due to their morphologic and biologic heterogeneity.12, 17 Historically in humans, the term carcinoid was used, with subclassification based on embryologic origin of the tumor: foregut carcinoids (respiratory tract, stomach, duodenum, biliary system, and pancreas), midgut carcinoids (small intestine, appendix, cecum, and proximal colon), and hindgut carcinoids (distal colon and rectum).17, 26 Because these designations based solely on tumor location failed to convey the diverse morphologic and biologic spectrum of these neoplasms, the recent World Health Organization (WHO) classification scheme abandoned the term carcinoid for those that reflected various morphologic and biological characteristics of the neoplasms, resulting in the categories of well-differentiated neuroendocrine neoplasms, neuroendocrine carcinomas, poorly differentiated carcinomas, and mixed endocrine/exocrine carcinomas.12 Further subclassification can be based on hormone production by neoplastic cells, as can be demonstrated immunohistochemically, and the presence or absence of a clinical syndrome related to hormone secretion. Using this current WHO classification scheme, all of the neoplasms in these bearded dragons were classified as neuroendocrine carcinomas based on histomorphologic appearance and evidence of metastatic disease in all cases. Those that were composed of a relatively monomorphic cell population we termed neuroendocrine carcinomas; whereas those with features of marked anaplasia we termed poorly differentiated neuroendocrine carcinomas.
IHC provides the gold standard for confirmation of neuroendocrine origin and for functional subclassifications based on detectable secretory products. Neoplasms may be nonfunctional, secrete a single product, or exhibit multihormonal expression. In cases of multihormonal expression, one hormone typically predominates, with others present in minor, varying amounts. In such cases, the neoplasm is subclassified according to the hormone that predominates.12, 17, 21, 22 Generic markers to determine neuroendocrine differentiation include antibodies against secretory granule proteins, synaptic vesicle proteins, and enzymes specific to neuroendocrine cells. In this study, we performed IHC for chromogranins A and B (secretory vesicle proteins), synaptophysin (synaptic vesicle protein), and PGP9.5 and NSE (enzymes).23, 25 Staining for these generic neuroendocrine markers was rarely detected in the bearded dragon tumors, and we only demonstrated in 3 of the tested neoplasms synaptic or secretory vesicle proteins. The loss of immunoreactivity for these proteins in the other 2 neoplasms as well as for enzymes commonly found in neuroendocrine neoplasms in all 5 bearded dragons may indicate a poorly differentiated phenotype. This is supported by the high rate of metastasis and the marked anaplasia observed in numerous neoplasms. Additional IHC was performed for insulin, glucagon, gastrin, somatostatin, PPP, endorphin, and VIP in an effort to further subclassify these neoplasms based on hormone secretion. These choices reflect those hormones frequently secreted by neuroendocrine neoplasms of the gastroenteropancreatic tract in other species, all of which have been optimized in our laboratory for use in various species.21, 22 Consistent, variable expression of somatostatin within all 5 bearded dragons examined justifies a diagnosis of somatostatinoma. In addition, several neoplasms exhibited multihormonal expression, a feature not uncommon for human somatostatinomas or other neuroendocrine tumors.12, 21, 22 Primary pancreatic somatostatinoma with gastric and other metastasis was considered. However, neoplastic cells were identified in the pancreas in only 3 of the 10 bearded dragons, and in these animals neoplastic cells in the pancreas were multifocally distributed, more consistent with metastatic spread, whereas gastric neoplasms were present in all cases. The submucosal gastric neoplasms were solitary and presented as the largest mass in each animal, supporting the diagnosis of primary malignant gastric somatostatinomas.
In humans, functional subclassifications also relate neuroendocrine neoplasms to the presence or absence of clinical symptoms or syndromes resulting from hormone secretion by neoplastic cells. In humans, somatostatinomas most commonly occur in the pancreas and clinically manifest in up to 65% of human cases as a “somatostatin syndrome.”16 This syndrome is characterized by diarrhea, weight loss, cholelithiasis, diabetes mellitus, and anemia.12, 16 Interestingly, hyperglycemia and anemia were reported in this study in 2 and 3 bearded dragons, respectively, raising the possibility of a similar somatostatin syndrome in this species. Because the clinical history and clinicopathologic data were incomplete for many of the affected bearded dragons, the prevalence of these signs may actually have been higher than reported. If a somatostatin syndrome occurs in bearded dragons with gastric somatostatinomas, recognition of associated clinicopathologic abnormalities and related clinical signs may allow for earlier detection, and potentially treatment, of these neoplasms. Additional studies are needed to further investigate the possibility of such a syndrome in bearded dragons with somatostatinomas.
Somatostatinomas occurring in the duodenum are far less commonly reported in humans and are often associated with neurofibromatosis type 1 (NF-1, also called Von Recklinghausen's disease).1, 16 This autosomal dominantly inherited disease results from a mutation of the NF1 gene, whose protein product is neurofibromin. Neurofibromin acts a tumor suppressor gene, through negative regulation of the Ras-related G-proteins by increasing Ras GTPase activity.18, 19, 27 The resulting disease is characterized by formation of multiple peripheral neurofibromas, skin discoloration (café-au-lait spots) due to abnormal melanocyte distribution, iridal hamartomas (Lisch nodules), and an increased risk for the development of many different neoplasms, including somatostatinomas of duodenal origin.4, 8, 16, 20, 27 In one study, 26/55 (47%) of duodenal somatostatinomas were associated with NF-1.16 Unlike pancreatic somatostatinomas, duodenal somatostatinomas, whether associated with NF-1 or not, are rarely associated with a recognizable clinical syndrome, and are less frequently associated with metastases at the time of diagnosis.16
The primary gastrointestinal, rather than pancreatic, location of the somatostatinomas in these bearded dragons is reminiscent of those associated with NF-1 in humans; however, there was no evidence of the other clinical signs and lesions of Von Recklinghausen's disease in these bearded dragons. In humans, neoplasms, including neuroendocrine carcinomas, associated with Von Recklinghausen's disease have decreased expression of neurofibromin by IHC.11, 18 Interestingly, in all 5 bearded dragon neoplasms examined by IHC for neurofibromin expression, there was decreased immunoreactivity among neoplastic cells compared with both interspecies and intraspecies control tissues. The presence of immunostaining for neurofibromin in bearded dragon control tissues, along with the decrease or complete loss of expression in neoplastic cells, suggests that a decrease in functional neurofibromin protein may also be involved in the pathogenesis of these tumors in bearded dragons. Insufficient genetic data did not allow for an analysis of a potential mutation of the NF1 gene in these bearded dragons.
In summary, young bearded dragons appear predisposed to the development of gastric neuroendocrine carcinomas, specifically somatostatinomas, which are high grade malignancies that readily metastasize. In contrast to humans, somatostatinomas in bearded dragons appear to be primarily of gastric origin, but similar to their human pancreatic counterpart, exhibit widespread metastases and may be associated with a clinical syndrome of hypersomatostatinemia. The cause for the bearded dragon's apparent predisposition to this otherwise unusual neoplasm is unknown. Decreased functional neurofibromin protein may play a role in the pathogenesis of these tumors, and further genetic studies are necessary to investigate this pathway. Somatostatinomas may be underdiagnosed in bearded dragons due to inconsistent, ambiguous clinical signs and the lack of IHC evaluation of routine biopsy or necropsy material. Somatostatinoma should be considered in the differential for animals presenting with anorexia, weight loss, and anemia, particularly if there is concurrent, otherwise unexplained, hyperglycemia.
Footnotes
Acknowledgements
We thank Roy Brown of Histologic Consulting Service for slide preparation, and Jamie Kinion and Christie Buie of Northwest ZooPath for data retrieval and photo editing, respectively. We also thank the following for case submissions: Community Pet Hospital, Lakawana, New York; Pet Hospital of Penasquitos, San Diego, California; Exotic and Companion Animal Veterinary Services, Mooresville, Indiana; Department of Animal Health, Woodland Park Zoo, Seattle, Washington; Whispering Pines Veterinary Clinic, Magalia, California; Animal Health Department, Oakland Zoo, Oakland, California; Vernon Hills Animal Hospital, Vernon Hills, Illinois. We also thank Tom Wood of the Immunohistochemistry Laboratory at the DCPAH, Michigan State University for technical assistance. We also thank the Electron Microscopy Core Laboratory, Interdisciplinary Center for Biotechnology Research, University of Florida, Gainesville, Florida.
