Abstract
A 12-year-old female polar bear (Ursus maritimus) developed a sudden onset of muscle tremors, erratic circling, increased blinking, head shaking, and ptyalism, which progressed to partial and generalized seizures. Ancillary diagnostic tests were inconclusive, and the only significant laboratory finding was nonsuppurative pleocytosis of cerebrospinal fluid. Euthanasia was elected. Microscopic evaluation demonstrated multifocal, random nonsuppurative meningoencephalitis involving most prominently the rostral cerebral cortex, as well as the thalamus, midbrain, and rostral medulla. Lesions consisted of inflammation, neuronal necrosis, gliosis, and both neuronal and glial basophilic intranuclear inclusion bodies. Immunohistochemistry with a polyclonal antibody reactive to several equine herpesviruses was positive within affected areas of the brain, and polymerase chain reaction conclusively demonstrated the presence of only equine herpesvirus 9. The clinical and morphologic features of this case resemble other fatal herpesvirus encephalitides derived from interspecies transmission and underscore the need for extreme caution when managing wild or captive equids.
Introduction
Equine herpesvirus 9 (EHV-9) is a newly recognized herpesvirus previously called gazelle herpesvirus 1 (GHV-1). GHV-1 was first described in an epizootic of encephalitis in Thomson's gazelles (Gazella thomsoni) in Japan and caused nonsuppurative meningoencephalitis, neuronal necrosis, intranuclear inclusion bodies, and gliosis, most commonly in the rostral regions of the cerebrum.8, 35 The virus was isolated from the brain of gazelles and was found to serologically cross-react with EHV-1 and EHV-4.8, 30 Analysis of nucleotide sequences of the glycoprotein G gene and a conserved region of the glycoprotein B (gB) gene of this virus revealed that it had the highest genetic similarity to EHV-1, but restriction enzyme cleavage profiles were significantly different. The virus was recognized as a new equine herpesvirus, designated EHV-9.3, 8, 16, 30, 32, 35 Thomson's gazelles involved in the outbreak shared a field and water source with zebras, which were considered to be a possible source of the virus.35 Serologic studies indicated that wild Burchell's zebras developed neutralizing antibodies to EHV-9, and a recent molecular epidemiologic survey showed that Grevy's zebras and polar bears are susceptible to infection and resultant disease from EHV-9.26 The natural host of EHV-9 and the complete host range are still unknown.3, 8, 20, 26, 30, 31
Experimental infection with EHV-9 has shown tropism for neural and respiratory tissues.16 Intranasal inoculation of EHV-9 has been documented to cause clinical neurologic disease and nonsuppurative encephalitis with neuronal necrosis, intranuclear inclusion bodies, and gliosis in a wide variety of animals, including dogs, cats, goats, hamsters, BALB/c mice, and marmosets.7, 8, 16, 31, 34, 36 Intranasal inoculation was a common method of virus administration in the experimental studies, some of which revealed a strong affinity of this virus for the olfactory system as a site of viral replication.36 The molecular aspects of EHV-9 latency are unknown.
This report describes the clinical, morphologic, and immunohistochemical features of meningoencephalitis in a polar bear naturally infected with EHV-9. Nearby zebras were considered as possible reservoirs for EHV-9.26
Materials and Methods
Case history
A 12-year-old female polar bear was housed with 2 other polar bears in an indoor/outdoor enclosure containing a pool at the Zoological Society of San Diego in California. The polar bear had a sudden onset of altered mentation, tremors, increased blinking, head shaking, drooling, abnormal chewing, and tachypnea. Early in the illness, the polar bear tended to rest the top of her head on the cage bars between events. Clinical signs increased in frequency and severity, including circling and partial seizures, consisting of uncontrolled asymmetric muscle fasciculations and opisthotonus. Later in the clinical course, the polar bear also had flexor rigidity and was less responsive to stimuli. The bear was anesthetized on 3 separate occasions for numerous diagnostic tests. Excepting cerebrospinal fluid aspirates, no significant findings were revealed by complete blood count; chemistry panel; blood gases; fungal titers (Cryptococcus neoformans, Blastomyces dermatitidis, Histoplasma capsulatum, Coccidioides immitis, Aspergillus spp.); serology for Toxoplasma gondii, canine distemper virus, West Nile virus, and St. Louis encephalitis virus; vitamin and mineral panels; radiographs; ultrasound; and magnetic resonance imaging. Cytologic evaluation of cerebrospinal fluid on 2 occasions revealed nonsuppurative pleocytosis. The T. gondii titer (1:2048) had doubled in 4 months but was interpreted to more likely reflect exposure, rather than active infection. The polar bear was hospitalized for 48 hours under sedation and later a drug-induced coma using continuous-rate infusions of initially diazepam and subsequently pentobarbitol. Additional supportive care was given, including anti-inflammatory and antimicrobial therapy, but the polar bear developed hyperthermia, tachypnea, pulmonary edema requiring tracheal suctioning, and episodes of twitching (at times of the whole body but more often limited to the head and face, interpreted as generalized seizures) that could not be controlled with medications. Due to this severe decline in condition and poor prognosis, the polar bear was euthanatized, 6 days after the initial onset of clinical symptoms.
A complete necropsy was performed, and samples from the brain; cervical, thoracic, and lumbar spinal cord; peripheral nerves; eyes; skin; and all visceral organs were immersion fixed in 10% neutral buffered formalin and routinely processed for histology. The brain was routinely processed for electron microscopy as previously described.12, 25, 24 Fresh sections of brain and liver were also frozen at −20°C or −70°C. Genomic DNA was obtained from frozen tissues and from paraffin-embedded fixed brain using standard extraction methods as previously described15 and analyzed with conventional polymerase chain reaction (PCR) assays for chlamydia,5 adenovirus,14 rabies,21 paramyxovirus,23 and West Nile Virus.2 PCR for the herpesvirus polymerase gene segment32 and equine herpesvirus6, 8 was performed as previously described, and PCR for the EHV-9 gB gene segment was performed as previously described.4 PCR-generated products were cloned, sequenced, analyzed, and assembled using MacVector v. 7.0 and AssemblyLIGN v. 1.0.9 software packages (Accelrys, San Diego, CA) and compared with products from the GenBank database using the basic local alignment search tool.1 DNA samples were additionally tested with a Taqman (Applied Biosystems, Foster City, CA) real-time PCR specific for EHV-1.6 Immunohistochemistry was performed on brain tissues using a rabbit polyclonal antibody cross-reactive with EHV-1 and EHV-4 as previously described.9, 28, 33 Positive control tissues were from a confirmed case of EHV-1–induced abortion in a domestic horse.
Results
On gross examination, the polar bear was in good body condition and the meninges of the brain and spinal cord were diffusely congested. The caudodorsal lung lobes were edematous. Microscopic examination revealed severe nonsuppurative meningoencephalitis, predominantly in the grey matter of the cerebrum. The rostral telencephalon, particularly the rhinencephalon, was the most significantly affected region of the brain. Although no specific anatomical structure of the brain was targeted, scattered regions of inflammation involved the olfactory bulbs, cerebrum (including the cingulate gyrus, basal nuclei, and pyriform lobes), thalamus (including the hypothalamus, thalamic nuclei, cingulate gyrus, amygdala, fornix, hippocampus, and geniculate nuclei), midbrain (including the rostral colliculi, periaqueductal grey matter, occulomotor nuclei, reticular formation, and caudal colliculi), and rostral medulla. Inflammatory cells consisting of lymphocytes, plasma cells, macrophages, and fewer eosinophils formed perivascular cuffs within the meninges overlying the brain and Virchow-Robin spaces within the parenchyma, as well as more poorly delineated inflammatory cell infiltrates within the subjacent meninges, neuropil, and neuroparenchyma (Figs. 1, 2). The neuropil was edematous, with increased vascular branching, endothelial hypertrophy and perivascular clear space. Neuronal degeneration was characterized by peripheral localization or complete loss of Nissl substance, and neuronal necrosis consisted of cytoplasmic hyperosinophilia, loss of cytoplasmic detail, nuclear pyknosis, karyolysis, and karyorrhexis. Glial proliferation accompanied the inflammation, consisting of microgliosis and astrogliosis.
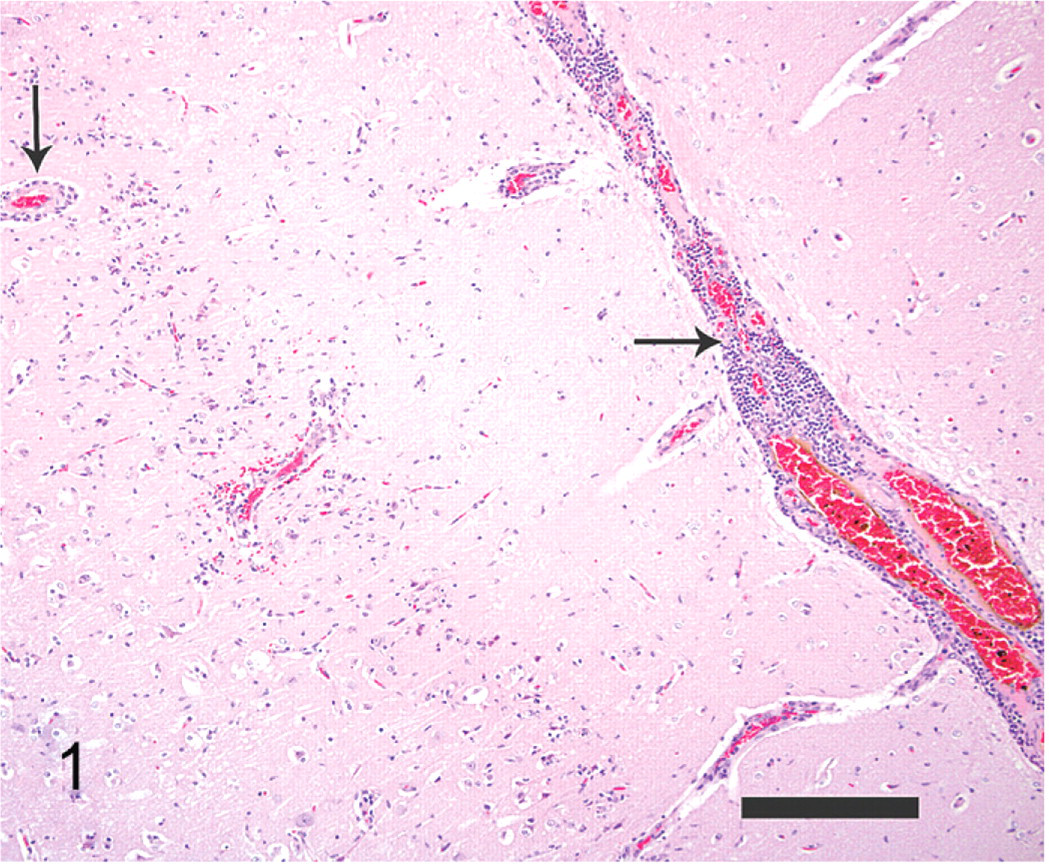
Brain; polar bear. Large numbers of inflammatory cells expand the leptomeninges and Virchow-Robbin spaces (black arrows). HE. Bar = 250 μm.
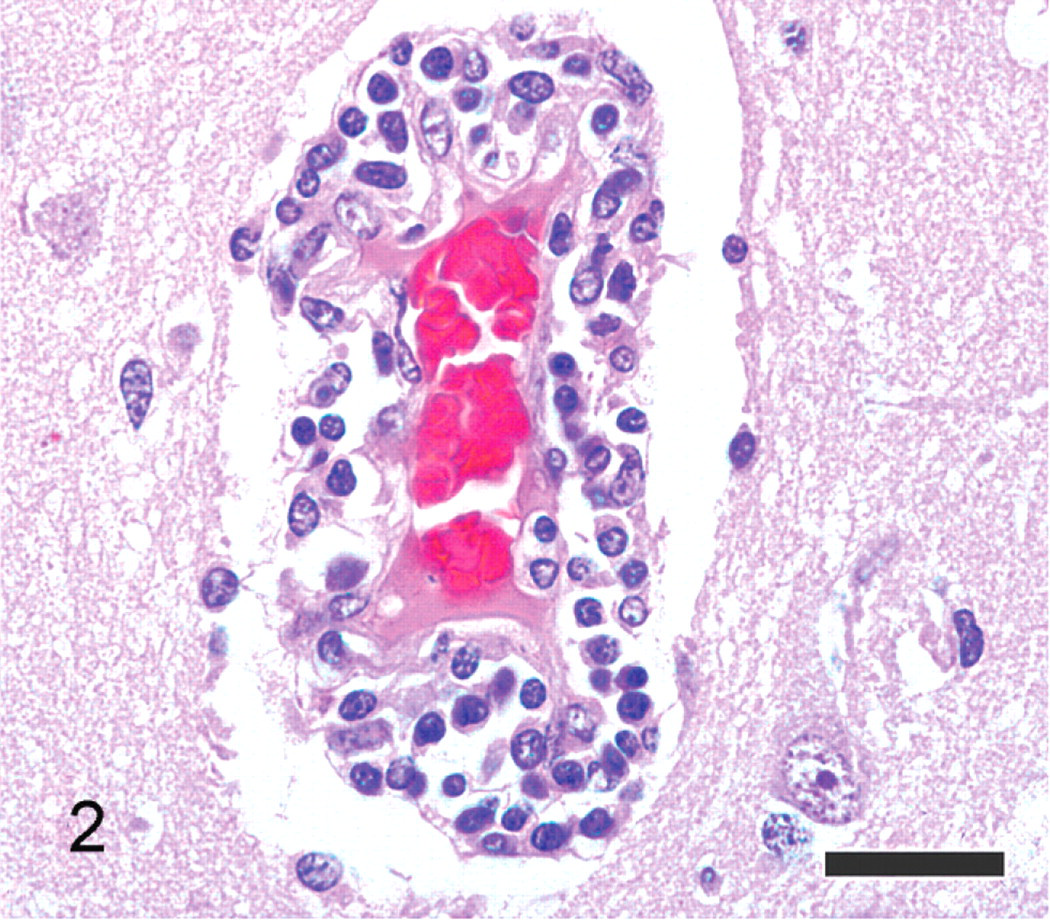
Brain; polar bear. Higher magnification view of inflammatory cells within the Virchow-Robbin space, occasionally infiltrating the neuropil. Inflammatory cells are predominantly composed of lymphocytes and macrophages. HE. Bar = 25 μm.
Microglial cells exhibited nuclear rod-shaped elongation (reactive microglia), satellitosis, and neuronophagia (Fig. 3). Within the nuclei of neurons and astrocytes, there were smudgy to distinct basophilic to amphophilic intranuclear inclusion bodies with chromatin margination and occasional clear space around the inclusion bodies (diffuse type and Cowdry type A inclusions; Fig. 4). White matter tracts had random areas of vacuolation, axonal degeneration characterized by formation of axonal spheroids, demyelination, edema, and microglial and astrocytic gliosis. Additional histologic findings included mild, multifocal neutrophilic bronchiolitis, mild neutrophilic rhinitis, moderate pulmonary edema, mild lymphocytic myocarditis, and mild membranoproliferative glomerulonephritis.
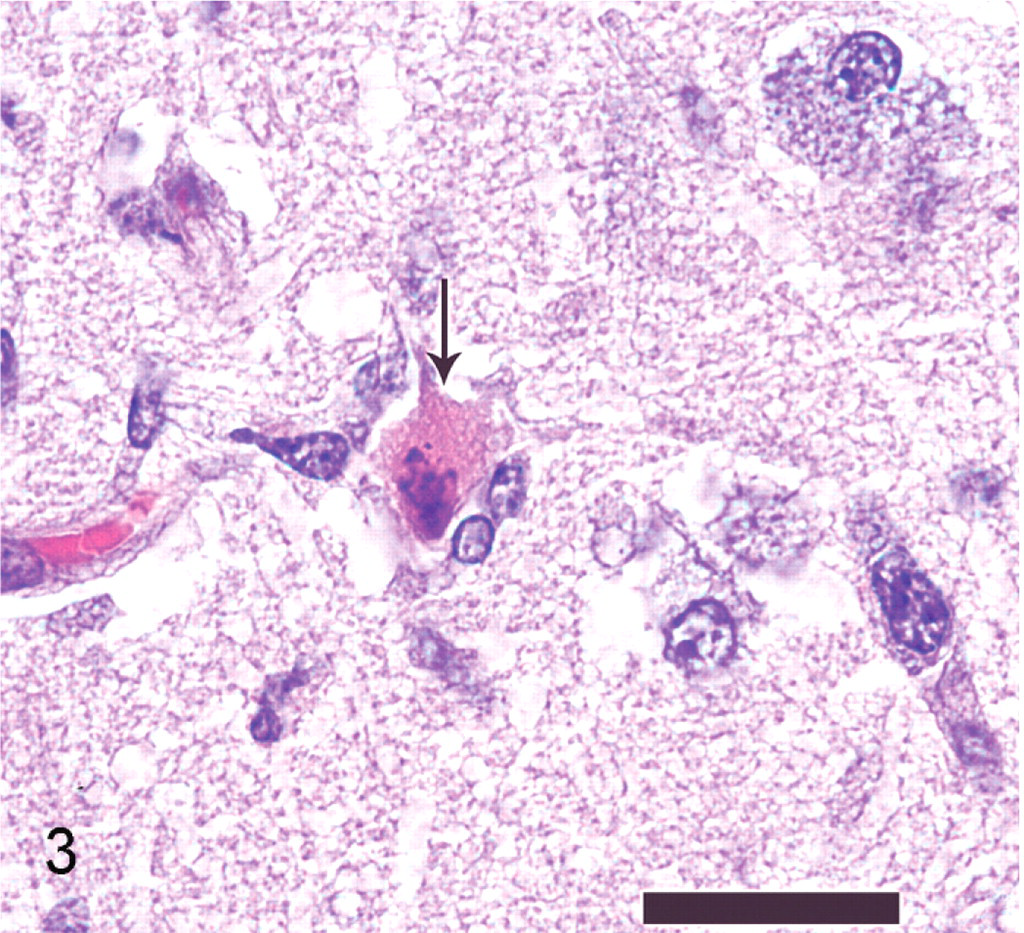
Brain; polar bear. Necrotic neuron within the grey matter of the cerebral cortex (black arrow). Note cytoplasmic hypereosinophilia, loss of cytoplasmic detail, the pyknotic neuronal nucleus undergoing karyorrhexis, and adjacent glial cells, which surround (satellitosis) and phagocytose the neuron (neuronophagia). HE. Bar = 25 μm.
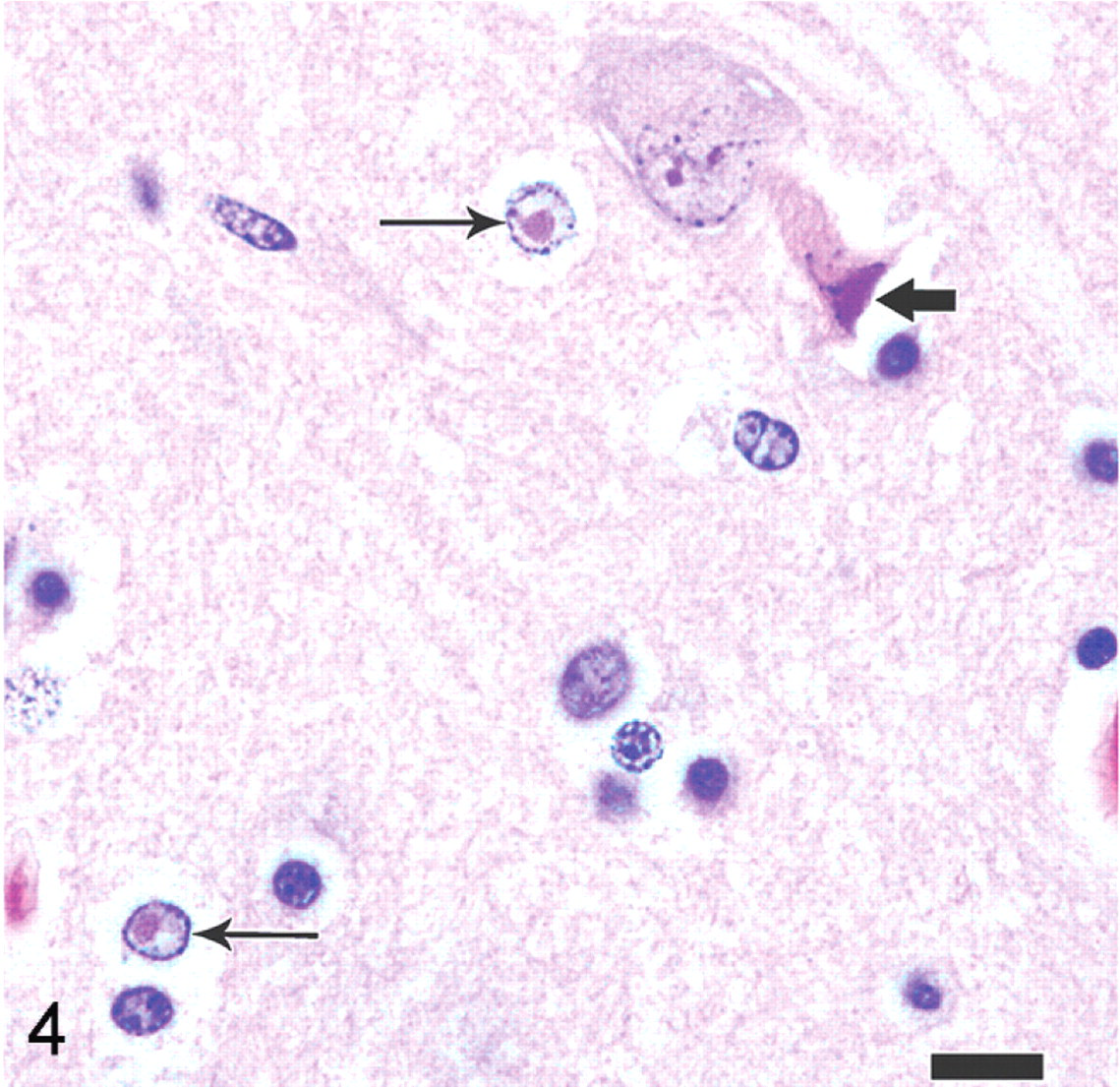
Brain; polar bear. Inclusion bodies within neurons and astrocytes. Note both the indistinct, smudgy inclusion that fills the nucleus (diffuse type inclusion body, thick black arrow) and the distinct inclusions surrounded by clear space (Cowdry type A inclusion bodies, thin black arrows). HE. Bar = 12.5 μm.
PCR for herpesvirus was positive, and basic local alignment search tool analysis of the amplified gene segment identified the virus as EHV-9. The sample was also found to be positive by real-time PCR designed to detect EHV-1, demonstrating the extensive homology between EHV-1 and EHV-9. Two subsequent PCR assays targeting an additional region of the DNA polymerase gene confirmed the presence of EHV-9 only.8 PCR tests for chlamydia, adenovirus, rabies, paramyxovirus, and West Nile virus were negative.
Immunohistochemistry revealed detectable EHV-1 antigen within neurons and glial cells (Fig. 5). Immunohistochemistry for pseudorabies, rabies virus, canine distemper virus, West Nile virus, adenovirus, Toxoplasma, and Sarcocystis was negative. Transmission electron microscopy of the brain demonstrated numerous 86–93-nm-diameter unenveloped virions with electron lucent cores within neuronal nuclei (Fig. 6). Virions were consistent with herpesvirus.
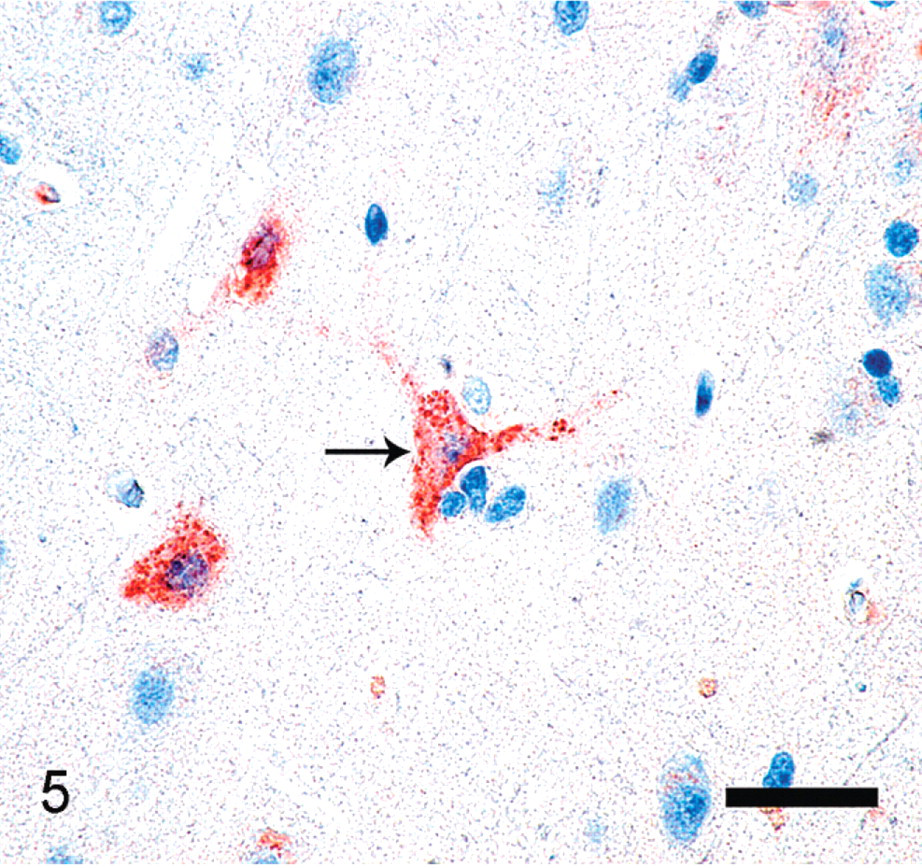
Brain; polar bear. Immunostaining is present within the cytoplasm of neurons and astrocytes using rabbit polyclonal anti-equine herpesvirus 1 antibody that cross-reacts with equine herpesvirus 4 (black arrow). Diaminobenzadine, hematoxylin counterstain. Bar = 50 μm.
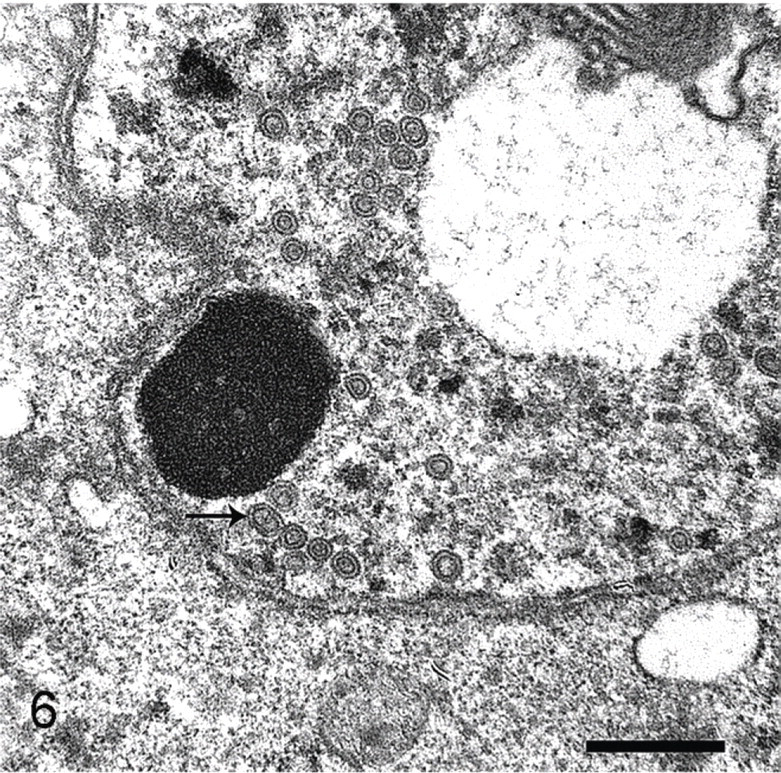
Brain; polar bear. Transmission electron microscopy reveals numerous 86–93-nm-diameter unenveloped virions with electron lucent cores within neuronal nuclei, consistent with herpesvirus virions (black arrow). Bar = 500 nm.
Discussion
The histopathologic alterations in the polar bear brain were consistent with herpesviral-induced meningoencephalitis, and molecular data confirmed EHV-9 as the cause. The lesions were analogous to those described in Thomson's gazelles naturally infected with EHV-9 and numerous animals experimentally infected with EHV-9. Initial immunohistochemical and molecular analysis revealed that the polar bear virus was closely related to EHV-1. Therefore, the possibility that meningoencephalitis and neuronal necrosis were a manifestation of EHV-1 in an aberrant host was considered. However, the histopathologic lesions were not consistent with those typical of EHV-1 myeloencephalopathy in equids (nonsuppurative necrotizing vasculitis and thrombosis with secondary injury to the neuroparenchyma).10, 18, 27, 29 Instead, morphologic changes closely resembled herpesvirus-induced encephalitis in an aberrant host, which has been documented to cause acute and lethal disease. Well-known examples related to the nervous system include Cercopithecine herpesvirus-1 (herpes B virus), which can result in fatal encephalomyelitis in humans,17 and Herpesvirus suis (Aujeszky's disease), which is typically asymptomatic in adult pigs but can cause peracute fatal neurotropic encephalitis in dogs, cats, cattle, sheep, and goats.29 Other investigative studies showed that nearby Grevy's zebras were the likely source of EHV-9 in this polar bear.26
Zebras have been implicated as a possible source of EHV-9 in several reports.3, 8, 10, 16 In one study of a population of Burchell's zebras in the Serengeti National Park in Tanzania, because of a high prevalence of neutralizing antibodies to EHV-9, it was hypothesized that Burchell's zebras in East Africa are a natural host for EHV-9.3 In contrast to previous reports of cross-neutralization of EHV-9 with EHV-1 and EHV-4, Burchell's zebras had low levels of EHV-1– and EHV-4–neutralizing antibodies.3
EHV-1 and EHV-1–like viruses, such as EHV-9, have rarely been described to cause natural disease in nonequid species. Reported cases include neurologic disease in alpacas and llamas.8, 10, 11, 13, Llamas naturally and experimentally infected with a virus serologically indistinguishable from EHV-1 had nonsuppurative encephalitis with neuronal degeneration, satellitosis, gliosis, vasculitis, cerebral edema, intranuclear inclusion bodies in neurons and endothelial cells, choroiditis, optic neuritis, and retinitis.11, 22 The experimentally administered virus was derived from the brain of a naturally infected alpaca, which had been housed with zebras (species unspecified). These histologic findings are interesting, because they include aspects of both EHV-9 and EHV-1 encephalitis. Molecular phylogeny was not reported for the llama cases.11, 22 Therefore, the true molecular nature of this herpesvirus remains unknown.
The olfactory bulbs of the polar bear were the most severely affected portion of the brain, and it is plausible to consider that EHV-9 may have entered the central nervous system from the nasal mucosa. It has been previously hypothesized that a possible route of EHV-9 infection is from the nasal mucosa along the olfactory pathway, vomeronasal organ, and/or trigeminal nerve31 and then transsynaptically via its connections to the hippocampus, amygdala, and cerebral cortex.7 The presence of EHV-9 antigens in axons and dendrites suggests possible transsynaptic spread, which has been shown in pseudorabies and EHV-1 infections.19, 31, 35 Intranasal inoculation of EHV-9 in pigs suggested viral replication in nasal epithelial cells, with entry to the brain via the olfactory pathway.19 One study compared various techniques of experimental EHV-9 inoculation in Syrian hamsters, including intranasal, intravenous, intraperitoneal, intramuscular, intraocular, and subcutaneous routes, but only intranasal inoculation induced disease.7 EHV-9 was recovered from nasal secretions of intranasally inoculated pigs, implying that once EHV-9 has infected the nasal mucosa, infective virus particles may be shed from the nasal cavity.16, 20
Numerous EHV-9 experimentally infected animals, typically inoculated intranasally, commonly developed interstitial pneumonia and rhinitis.7, 8, 16, 31, 34, 36 Mild neutrophilic rhinitis and pulmonary edema were noted in the polar bear. However, no interstitial pneumonia was found. The mild neutrophilic bronchiolitis was interpreted to be caused by aspiration during sedation. A definitive cause of the mild myocarditis and glomerulonephritis was not identified, and the relationship between these lesions and the virus is unclear.
This case report illustrates the devastating effects of cross-species viral transmission. In a zoologic institution, animals from significantly dissimilar regions of the world may be housed short distances from one another, as was the case here. Cross-species disease transmission may be facilitated under these circumstances. Because the range of species susceptible to EHV-9 may be extremely wide, caution should be taken in a zoologic institution when considering movement of all animals, particularly exotic ruminants, camelids, and carnivores, into or adjacent to enclosures containing zebras. Information from this case report may assist other institutions in preventing potentially detrimental animal interactions and cross-species viral transmission, particularly those involving zebra.
Footnotes
Acknowledgements
This work was supported by the Zoological Society of San Diego in California. Outstanding technical assistance was provided by April Gorow, Julie Albright, Marc Hammond, and Yvonne Cates, and valuable information was graciously provided by Gaylene Thomas. We thank Dr. Ilse Stalis for consultation on this paper, the Beckman Coulter Corporation for donation of the automated capillary DNA sequencer and high-speed centrifuge used in these studies, and Charles and Shirley Sykes of San Diego for financially supporting our laboratory.
