Abstract
This study was designed to experimentally reproduce enterotoxemia by Clostridium perfringens type D in cattle and to characterize the clinicopathologic findings of this disease. Fourteen 9-month-old calves were inoculated intraduodenally according to the following schedule: group 1 (n = 4), C. perfringens type D whole culture; group 2 (n = 3), C. perfringens type D washed cells; group 3 (n = 5), C. perfringens type D filtered and concentrated supernatant; group 4 (n = 2), sterile, nontoxic culture medium. In addition, all animals received a 20% starch solution in the abomasum. Ten animals from groups 1 (4/4), 2 (3/3), and 3 (3/5) showed severe respiratory and neurologic signs. Gross findings were observed in these 10 animals and consisted of acute pulmonary edema, excessive protein-rich pericardial fluid, watery contents in the small intestine, and multifocal petechial hemorrhages on the jejunal mucosa. The brain of one animal of group 2 that survived for 8 days showed multifocal, bilateral, and symmetric encephalomalacia in the corpus striatum. The most striking histologic changes consisted of perivascular high protein edema in the brain, and alveolar and interstitial proteinaceous pulmonary edema. The animal that survived for 8 days and that had gross lesions in the corpus striatum showed histologically severe, focal necrosis of this area, cerebellar peduncles, and thalamus. Koch's postulates have been met and these results show that experimental enterotoxemia by C. perfringens type D in cattle has similar clinical and pathologic characteristics to the natural and experimental disease in sheep.
Keywords
Spontaneous and experimentally induced Clostridium perfringens type D enterotoxemia has been relatively well characterized in small ruminants.2, 4, 6, 9, 16, 26, 28 However, there are few reports about natural cases of enterotoxemia type D in cattle, and information about clinical and pathologic findings of the disease in this species is scant and frequently contradictory.14, 15, 17 Niilo et al.24 induced disease by intraduodenal inoculation of whole cultures of C. perfringens type D and dextrin in 8 calves. Two of the animals presented acute neurologic signs and severe pulmonary edema, hydrothorax, hydroperitoneum, and serosal hemorrhages, while the other 6 did not show clinical or pathologic changes. However, no histologic lesions were described in any of the animals.24 Uzal et al.29 reported acute loss of consciousness, hyperesthesia, intermittent tonic and clonic convulsions, recumbency, and dyspnea in calves inoculated intravenously with C. perfringens type D epsilon toxin (ETX). These calves showed severe acute pulmonary edema and varying degrees of perivascular proteinaceous edema in the internal capsule, thalamus, and cerebellum.29 While the clinical, gross, and histologic findings described in the calves inoculated with ETX29 were very similar to changes in acute enterotoxemia of sheep, the experimental procedures involved intravenous injection of ETX; therefore this cannot be considered a model of natural disease.
A condition called enterotoxemia of cattle, allegedly produced by C. perfringens type D, is described in textbooks,3, 4 but to the best of our knowledge, Koch's postulates have not been fulfilled and confirmation of the etiology of this condition remains to be established. The occurrence of focal symmetrical encephalomalacia (FSE) in cattle has been described by several authors,1, 7, 11, 19, 20 but to date no causal relationship between this lesion and C. perfringens type D has been established. The objectives of this study were to develop an infectious experimental model of enterotoxemia in cattle, resembling as closely as possible the natural disease, and to characterize the clinical and pathologic changes of the experimental disease in this species.
Materials and Methods
Animals
Fourteen crossbred, 9-month-old, unvaccinated calves, with average weight of 120 kg, born from cows that had never been vaccinated against enterotoxemia, were used. A serum sample was obtained from each animal before the experiments and tested for anti-ETX antibodies using a slight modification of an indirect enzyme-linked immunosorbent assay (ELISA) technique previously described.31 Briefly, plates were coated with purified ETX before the serum samples were added, followed by addition of horseradish peroxidase (HRP)-conjugated antibovine IgG antibodies and substrate-chromogen solution. Different dilutions of a hyperimmune bovine serum of known anti-ETX concentration were used as positive controls, while sera from 11 colostrum-deprived calves were used as negative controls. No anti-ETX antibodies were detected in the serum of any of the animals collected before inoculation.
Conventionally reared male Webster mice (20–25 g body weight) were used for the mouse tests. All experiments using animals conformed to an accepted code of practice for the care and use of animals for scientific purposes in the state of Minas Gerais, Brazil.
Inocula
Four different inocula were assayed: 1) C. perfringens type D whole cultures (WHOLE); 2) C. perfringens type D washed cells (WASH); 3) C. perfringens type D filtered and concentrated supernatant (SUPNT); and 4) sterile, nontoxic triptose-glucose-yeast (TGY) culture medium (Anaerobe Systems, Morgan Hill, CA).
A strain of C. perfringens type D originally isolated from an acute case of type D enterotoxemia in goats was used.25 The freeze-dried strain was reconstituted in 50 ml of fluid thyoglicollate broth (Barcelona, Dignolab, Spain) and incubated for 24 hours at 37°C under anaerobic conditions. A 10% inoculum of this first culture was seeded into bottles containing 500 ml of TGY and incubated anaerobically for 10 hours. Some of these cultures were used as WHOLE inoculum. Other cultures were centrifuged at 10,000 × g for 25 minutes at 4°C and filtered through a 0.22-μm filter; the filtered supernatant was concentrated 10 times utilizing an Amicon concentration device (Millipore, Billerica, MA) with retention membrane of >10 kD and stored at −80°C until used as SUPNT inoculum. Then, the sediment was resuspended in 100 ml of phosphate-buffered saline (PBS), pH 7.2, and centrifuged again as described above. The sediment was resuspended in 300 ml of PBS and used as WASH inoculum. Nontoxic, sterile TGY was used as inoculum in the control animals.
Before inoculation, samples of the inocula were obtained and processed as described below for the following determinations. Samples of all the inocula were inoculated on blood agar and incubated aerobically at 37°C for 72 hours to check for contamination. Samples of WHOLE and WASH were used to determine the purity of the culture and colony forming units (CFU)/ml in a standard spread technique on blood agar after overnight anaerobic culture at 37°C. Samples of SUPNT and of TGY were also inoculated into cooked meat medium (CMM; Anaerobe Systems, Morgan Hill, CA) and blood agar plates, and incubated anaerobically at 37°C for 72 hours to check for sterility. A sample of WHOLE was centrifuged at 10,000 × g for 25 minutes at 4°C and filtered through a 0.22-μm filter. This filtrate and an aliquot of CONC were activated with 0.1% trypsin (Trypsin 1:250, Difco, Detroit, MI) in sterile 1% peptone water, pH 7.2, at 37°C for 40 minutes. Minimal lethal dose (MLD) and the presence of ETX toxin was then determined for both aliquots using 2 mouse tests, as previously described.28
The amount of ETX and the CFU/ml present in the corresponding inocula are shown in Table 1. A pure culture of C. perfringens type D was obtained when WHOLE and WASH were cultured anaerobically on blood agar. No aerobic bacteria were found in any inocula, and no anaerobic bacteria grew from SUPNT or TGY.
Inocula and clinical features in cattle experimentally inoculated with Clostridium perfringens type D whole culture (WHOLE), washed cells (WASH), concentrated culture supernatant (SUPNT), or sterile, nontoxic culture medium (TGY). ∗
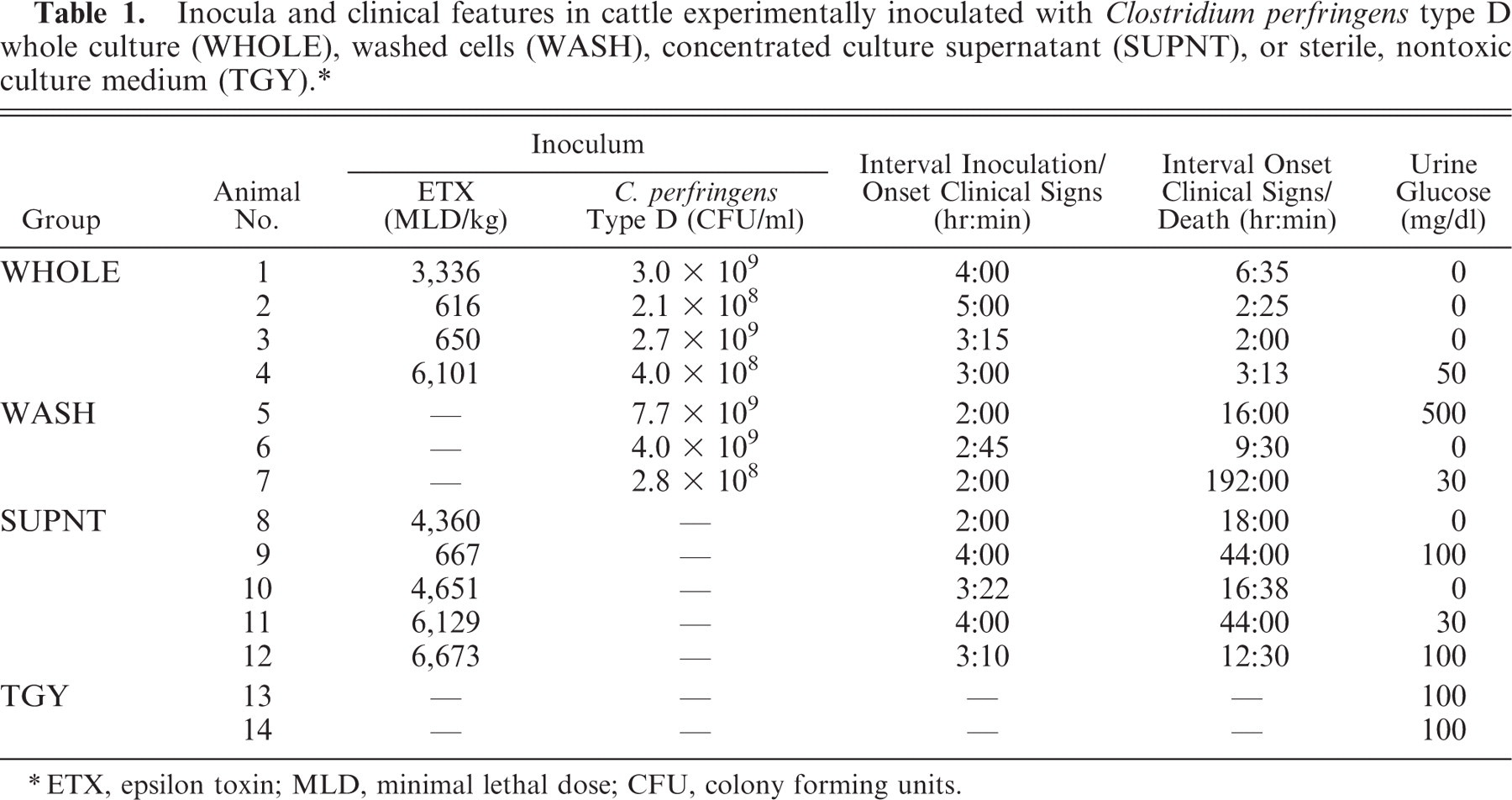
ETX, epsilon toxin; MLD, minimal lethal dose; CFU, colony forming units.
Experimental design
The calves were randomly distributed into 4 groups: WHOLE (n = 4), WASH (n = 3), SUPNT (n = 5), and TGY (n = 2), and inoculated intraduodenally as previously described.28 Briefly, the calves were fasted for 24 hours before surgery. They were tranquilized by an intravenous injection of xylazine (Bayer, São Paulo, Brazil). The animals were placed in a left lateral recumbency position and a blockage of the spinal nerves innervating the abdominal cavity was performed with 2% xylocaine (Betapharma, Shanghai, China). A laparotomy was performed via the right flank, and the pyloric area of the abomasum and the first portion of the duodenum were exposed. Two hundred milliliters of a 20% solution of cornstarch (Meta Therm Corp., New York, NY) in 0.85% saline was injected into the abomasum of all animals in each group to provide a substrate for these sacrarolytic bacteria to proliferate rapidly as in clinical cases with starch overload. Then, 300 ml of WHOLE, SUPNT, WASH, or TGY was inoculated into the duodenum through a drip, over a period of approximately 15 minutes. All animals were clinically examined before and periodically after inoculation.
All animals that developed severe respiratory or neurologic signs were euthanatized as soon as possible after the onset of these clinical signs. The animals that did not develop clinical alterations or those that developed only mild and transient clinical signs were euthanatized 48 hours after inoculation. One animal, which was inoculated with WASH and developed mild but progressive neurologic signs, was euthanatized 8 days after inoculation. Euthanasia was performed with an overdose of sodium pentobarbital (Abbott, Abbot Park, IL) in all cases. Necropsies were performed immediately after death. Samples of abomasum, duodenum, jejunum, ileum, cecum, colon, heart, lung, liver, spleen, kidney, and adrenal gland were collected and fixed in 10% buffered formalin for 48 to 72 hours. The whole brains were removed and fixed for 24–72 hours in the same fixative, cut into approximately 0.5-cm thick slices, and fixed in fresh formalin for an additional 7-day period before blocks from cerebral cortex, corpus striatum, thalamus, midbrain at the level of superior colliculi, pons, cerebellum, cerebellar peduncles, and medulla at the level of obex were prepared. All tissues were dehydrated through graded alcohols to xylene, embedded in paraffin wax, cut at 4 μm and stained with HE. Urine samples were collected from all animals during necropsy by bladder puncture and analyzed for density, pH, glucose, and protein, using urine sticks (Uridiag, Bayer Diagnostics, Bridgend, UK).
Statistical analyses were performed using Minitab 15 and Microsoft Excel. Pearson's and regression analysis were performed to determine if there was any significant correlation between ETX dose (MLD/kg) or bacterial numbers (CFU/ml), and the interval inoculation/onset of clinical signs or the interval onset of clinical signs/death.
Results
Clinical alterations
The time elapsed between inoculation and onset of clinical signs and between onset of clinical signs and death are shown in Table 1. All the animals inoculated with WHOLE and WASH and 3 of the animals inoculated with SUPNT (calves Nos. 8, 10, and 12) presented severe clinical alterations commencing between 2 and 5 hours after inoculation. Severe clinical signs necessitating euthanasia occurred in all but one of these animals between 2 and 18 hours after onset of clinical signs (Table 1). Initially, clinical signs in most animals consisted of dyspnea, lethargy, and decrease of ruminal motility, but progressively the animals developed ataxia with wide base stance followed by recumbency, paddling, opisthotonous, hyperesthesia, blindness with loss of pupillary reflex, strabismus, bruxism, decreased muscular tone, hypersalivation, and convulsions. One animal inoculated with WASH presented only mild neurologic alterations that worsened progressively over an 8-day period, after which it was also euthanatized (calf No. 7; Table 1). Two animals inoculated with SUPNT (calves Nos. 9 and 11; Table 1) developed mild ataxia starting 4 hours after inoculation, which resolved spontaneously within 2 hours of onset. These 2 animals were euthanatized 48 hours after inoculation.
There were no significant correlations between ETX dose or bacterial numbers in the inocula and the interval inoculation/onset of clinical signs or the interval onset of clinical signs/death, (Pearson correlation value [r ≤ 0.49, P > .05]).
Postmortem findings
Gross changes
All animals inoculated with WHOLE, WASH, or SUPNT that presented severe clinical alterations (calves Nos. 1–8, 10, and 12; Table 1) had similar gross lesions that differed only slightly in intensity. They consisted of pulmonary edema, characterized by distension of the interlobular septa by gelatinous fluid (Fig. 1), a large amount of stable froth in the trachea and lower airways, excessive pericardial fluid that usually contained strands of fibrin, watery contents in the small intestine and multifocal petechial hemorrhages on the jejunal mucosa. In addition, the mesenteric lymph nodes were moderately edematous and congested. No significant gross abnormalities were seen in the central nervous system, except for the brain of the animal that survived 8 days (calf No. 7; Table 1), which showed multifocal, bilateral and symmetric cavitation, and softening of the internal capsule within the corpus striatum (focal symmetric encephalomalacia [FSE]) (Fig. 2). No other gross abnormalities were observed in any other animal.
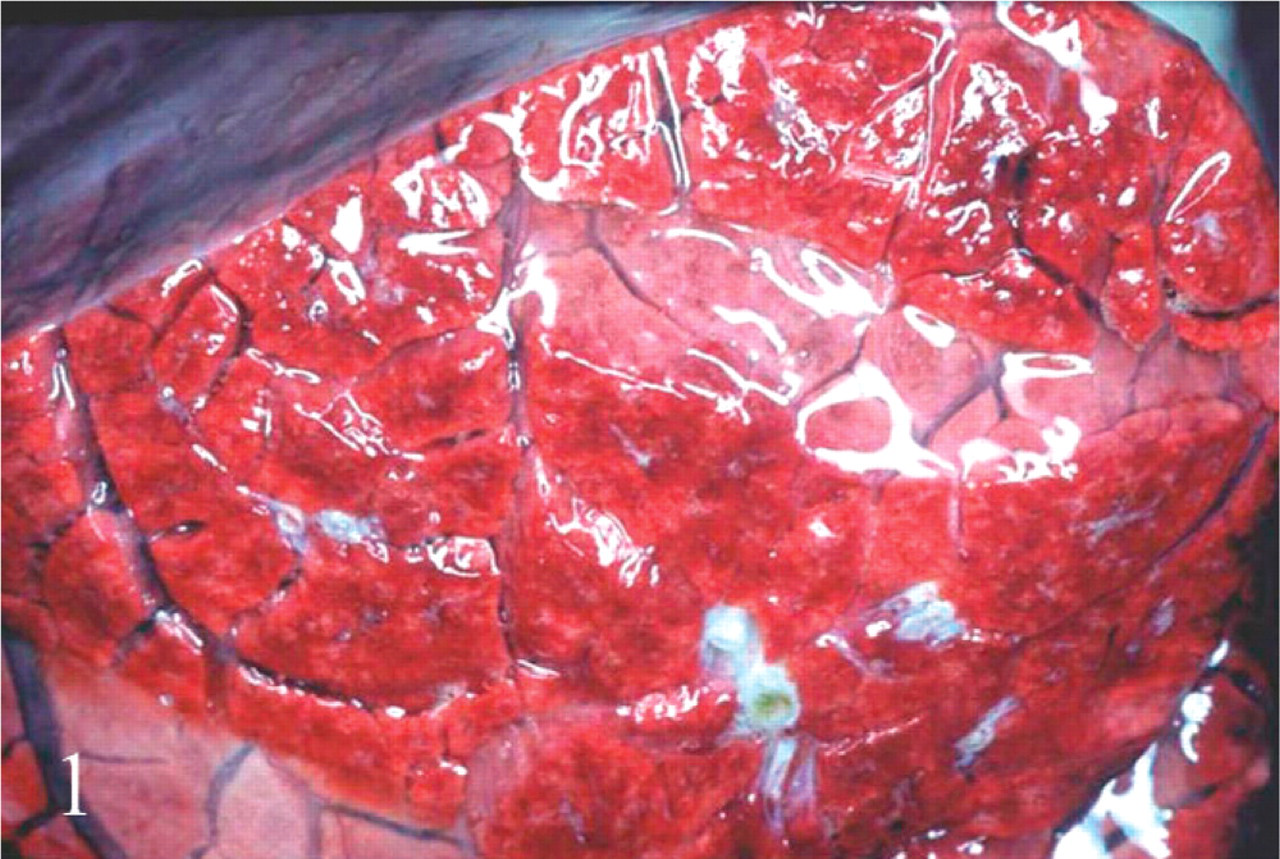
Lung; calf No. 5. Acute pulmonary edema. Note the interlobular septa severely distended with edema fluid.
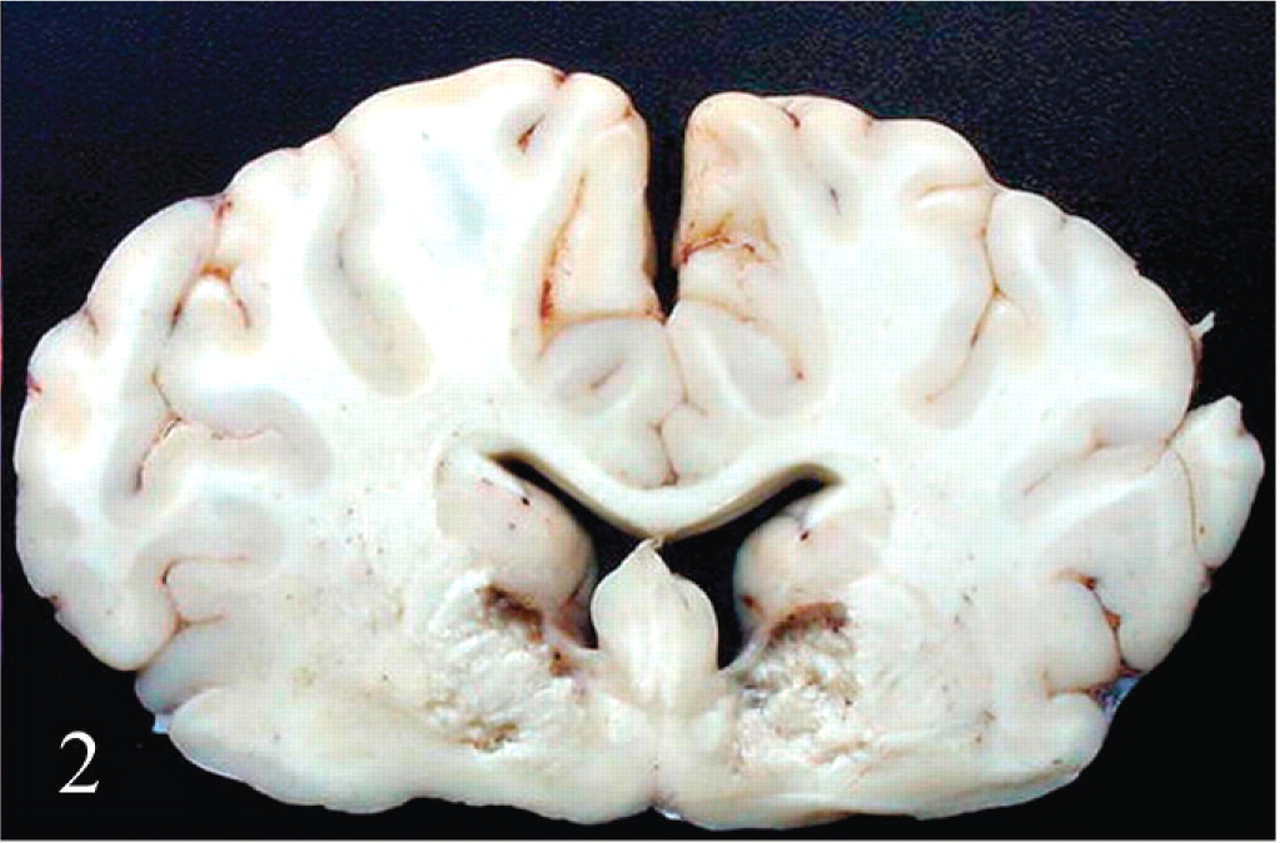
Corpus striatum (internal capsule); calf No. 7. Focal symmetrical encephalomalacia. This animal developed progressive neurologic signs that became very severe 8 days after inoculation, at which time the calf was euthanatized.
Histologic changes
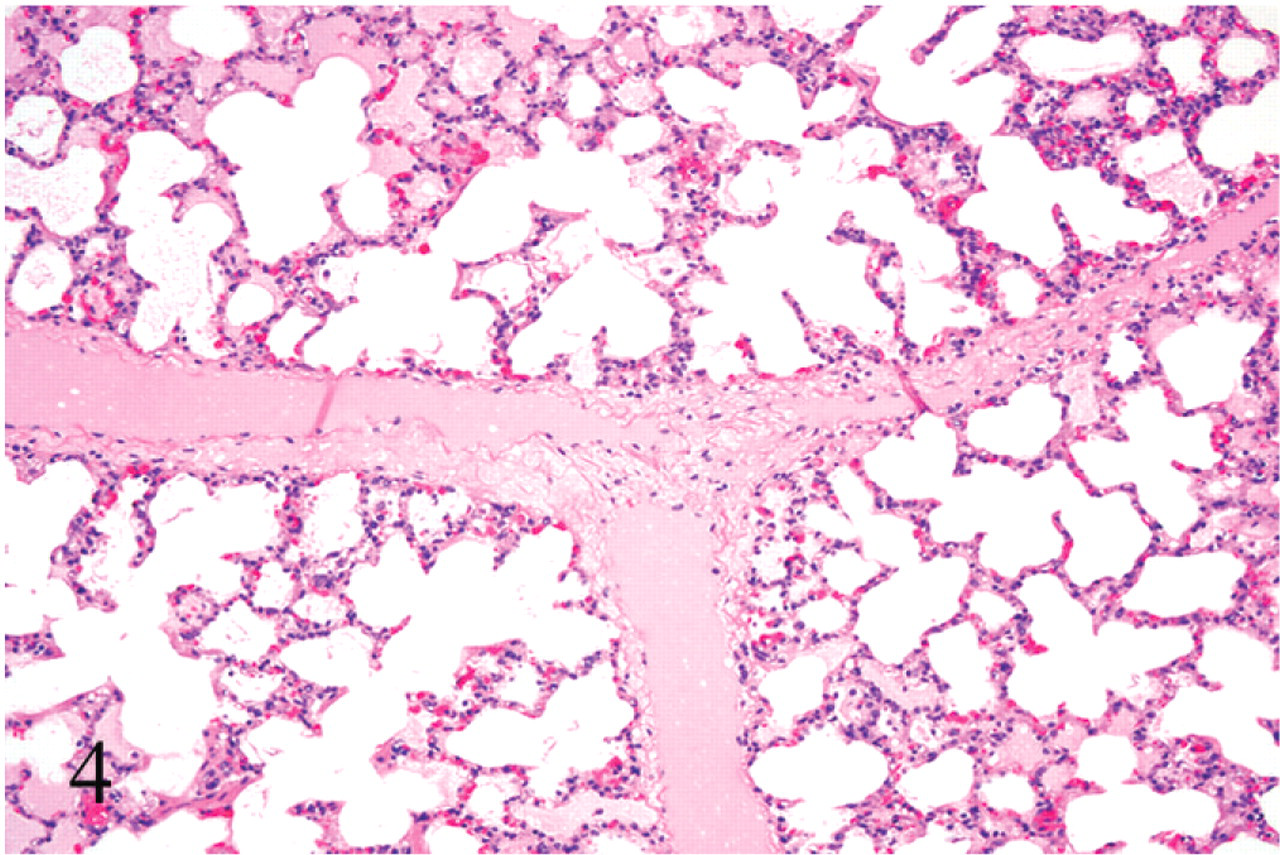
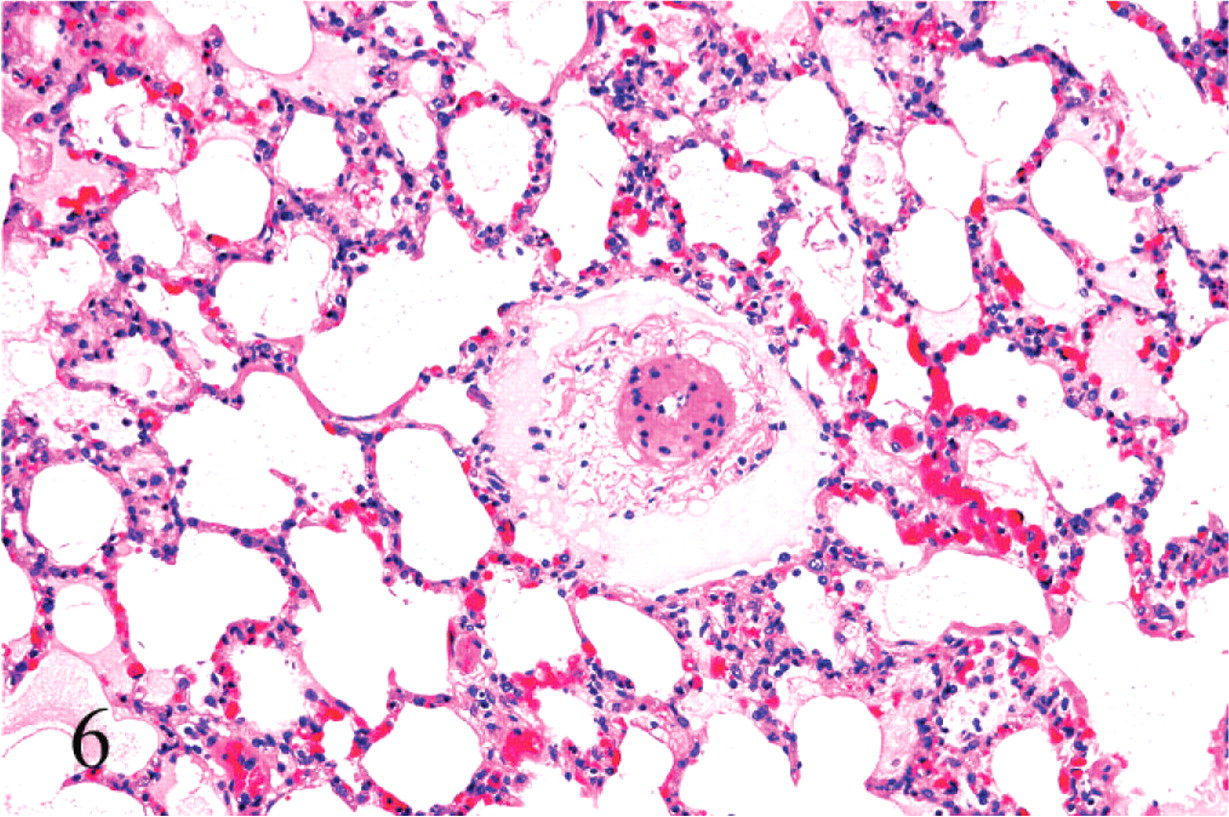
Light microscopic examination of the brain of the animals that died within 24 hours of inoculation revealed proteinaceous perivascular edema that affected both arteries and veins of small to medium size, with the perivascular spaces of several blood vessels being distended by eosinophilic, homogeneous fluid (Fig. 3). Both arteries and veins were affected in a similar proportion, and the maximum caliber of the vessels that exhibited this change was approximately 500 μm. No degeneration was evident in the wall of any of the vessels examined. These lesions were most commonly observed in the corpus striatum, mesencephalon, cerebellar white matter, and cerebellar peduncles (Table 2). Perivascular proteinaceous edema was mostly restricted to the white matter, and in the cerebellar white matter, these lesions were consistently located in the area immediately adjacent to the gray matter. Severe proteinaceous interstitial and intra-alveolar edema of the lung was observed in all these animals. The high protein pulmonary edema consisted of filling of alveoli and interlobular septa by dense, deeply acidophilic, and homogeneous fluid (Figs. 4, 5). Perivascular proteinaceous edema (microangiopathy) similar to that described in the brain was observed surrounding arteries and veins within the lungs (Fig. 6). Multifocal, acute, and mild hemorrhage of the lamina propria was observed in the jejunum of all calves of groups 1, 2, and 3, except for calves Nos. 7, 9, and 11. The animal that survived for 8 days and that had grossly FSE in the corpus striatum showed histologically severe, focal necrosis of the internal capsule within the corpus striatum, cerebellar peduncles (Fig. 7), and thalamus. Necrotic foci were characterized by severe vacuolation of the white matter, with dilated myelin sheaths, axonal swelling (spheroids), neuronal necrosis, perivascular and interstitial edema, diffuse microgliosis, and presence of large, lipid-laden macrophages (Gitter cells) (Fig. 8). Surrounding the area of degeneration and necrosis, the astrocytes were hypertrophic, with large, vesicular nuclei, and there was also hypertrophy of endothelial cells in both veins and arteries. No other significant histologic abnormalities were observed in any of the other organs examined of any of the test animals. No histologic abnormalities were observed in any of the calves that were clinically healthy 24 hours after inoculation, including those that developed only transient clinical alterations (calves Nos. 9 and 11) and the TGY-inoculated controls.
Histopathologic changes in brains of 14 calves experimentally inoculated with Clostridium perfringens type D whole culture (WHOLE), washed cells (WASH), concentrated culture supernatant (SUPNT), or sterile nontoxic culture medium (TGY). ∗
NL = no lesions; MAP = microangiopathy; FSE = focal symmetrical encephalomalacia.
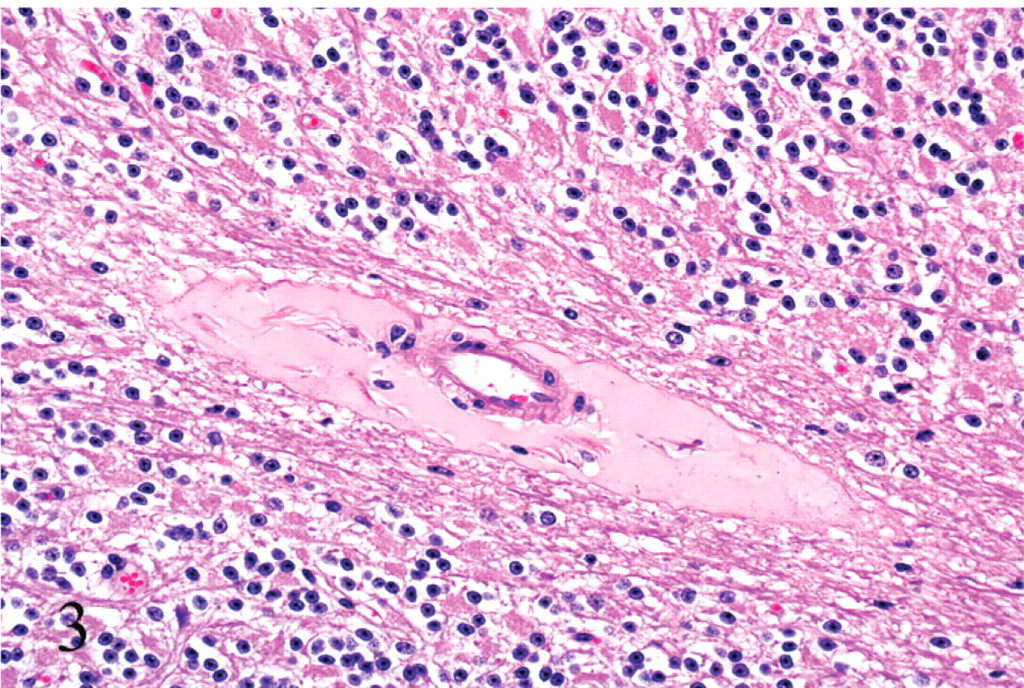
Cerebellum; calf No. 5. Perivascular proteinaceous edema in the white matter. HE; original magnification 250×.
Lung; calf No. 5. Acute edema. Note the interlobular septa severely distended with proteinaceous edema fluid. HE; original magnification 100×.
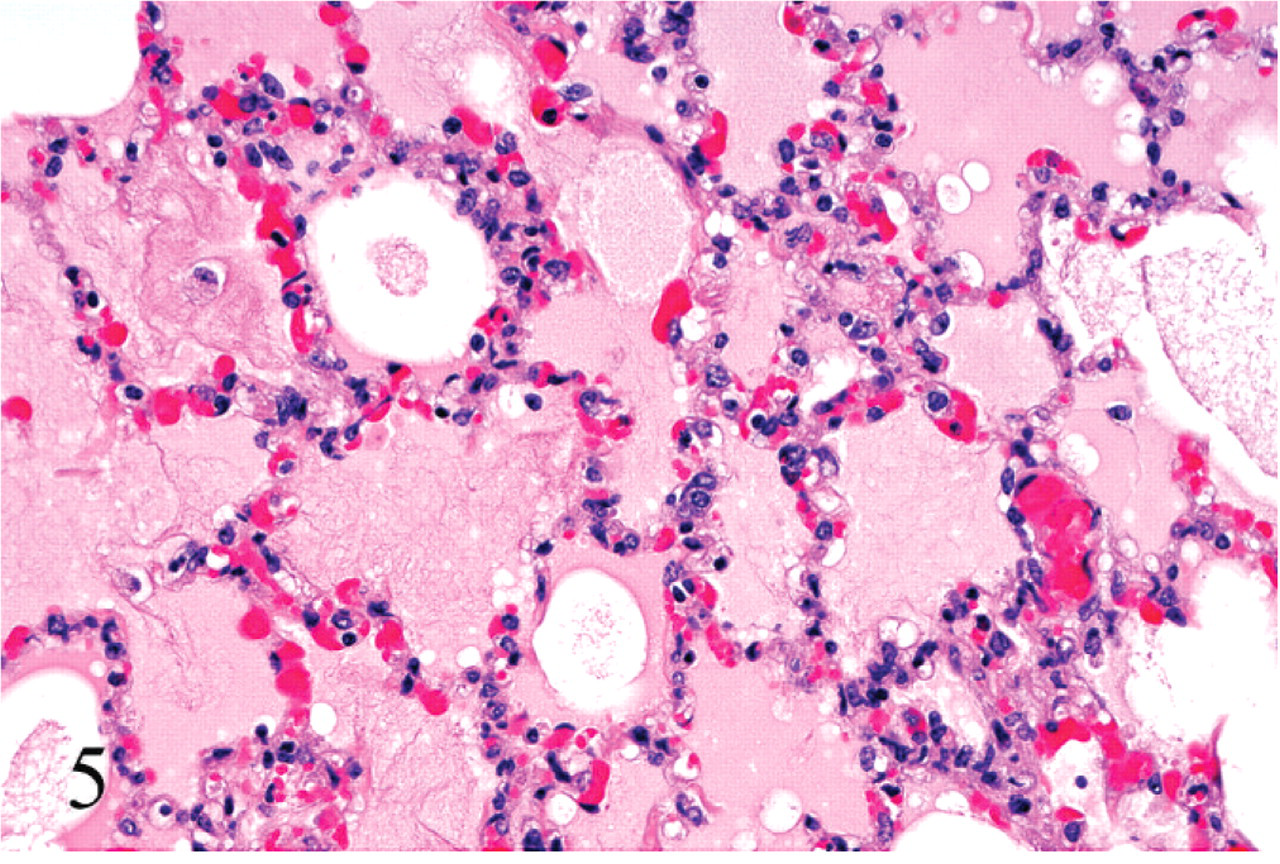
Lung; calf No. 5. Acute edema. Note the alveoli flooded with proteinaceous edema fluid. HE; original magnification 250×.
Lung; calf No. 5. Perivascular proteinaceous edema. HE; original magnification 100×.
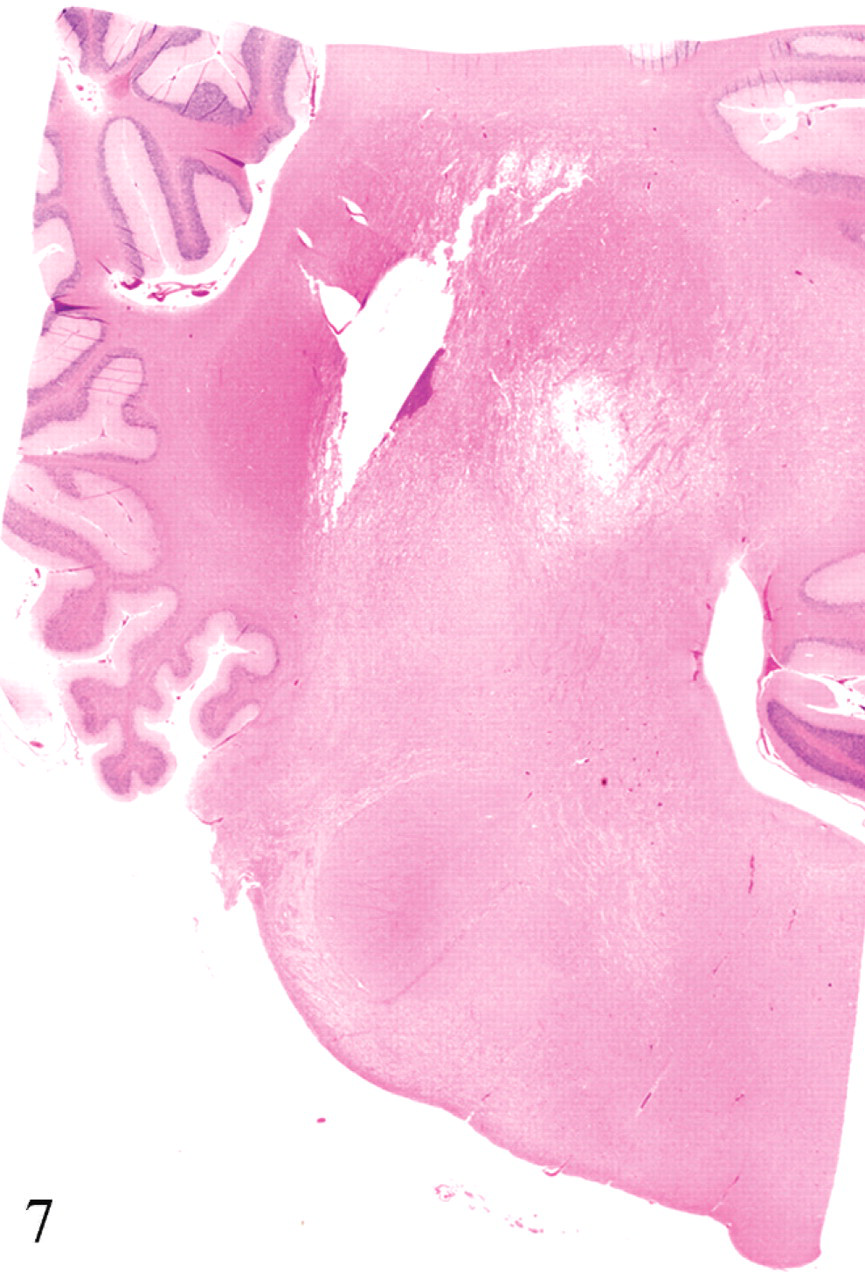
Cerebellar peduncles; calf No. 7. Focal symmetrical encephalomalacia. HE; original magnification 100×.
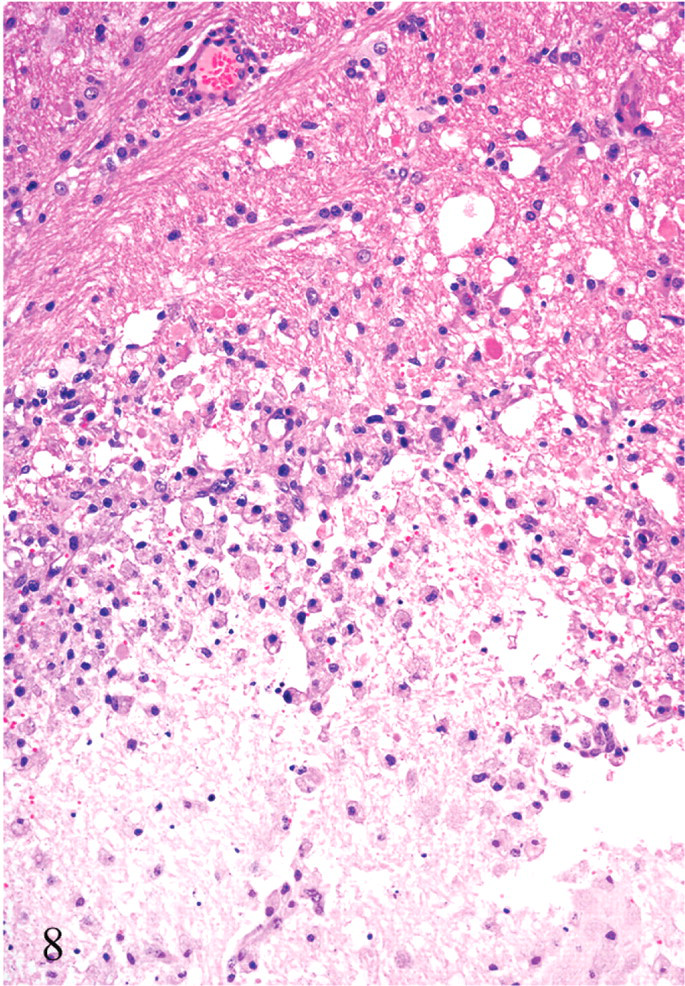
Corpus striatum (internal capsule); calf No. 7. Focal symmetrical encephalomalacia. Observe the accumulation of vacuolated macrophages (Gitter cells) in the periphery of the lesion. Vacuolation and cavitation of the white matter and diffuse gliosis are also seen. HE; original magnification 250×.
Urinalysis
The main (although not consistent) biochemical alteration in animals with clinical disease was glucosuria (Table 1). No significant alterations were observed in urine density, pH, or protein concentration of any of the animals.
Discussion
Information on enterotoxemia by C. perfringens type D in cattle is scant. In part, this paucity of information is owing to the lack of a model for the disease in this species. The method used here induced a disease with clinical signs and gross and histologic findings similar to those ascribed to natural type D disease in cattle.4, 14, 15, 17 The disease produced in calves in this study was also almost identical to type D enterotoxemia in sheep, a species in which this disease has been extensively studied and for which Koch's postulates have been fulfilled.15, 30
The inoculum containing bacteria plus supernatant (WHOLE) and the one with only C. perfringens type D cells (WASH) consistently induced disease in all animals inoculated (Table 1). However, the use of concentrated filtered supernatant without bacteria (SUPNT) produced severe disease in only 3 out of 5 animals inoculated, while the other 2 animals developed only mild and transient disease. These results suggest that the environment of the duodenum facilitated proliferation and toxin production by the bacteria present in WHOLE and WASH, which contributed to disease production. In this regard, it is well established that a starch overload in the small intestine, such as the one created in this model, provides a favorable substrate for rapid clostridial multiplication and abundant toxin production. Also, our results suggest that ETX can be absorbed from the intestine of cattle into the systemic circulation, since the histologic lesions observed in the brain of our calves were identical to those produced experimentally with ETX in sheep27 and cattle.29
Our clinical findings are similar to those described by other studies in cattle24 and lambs2, 5, 30 using intraduodenal inoculation of C. perfringens type D. They consisted mostly of neurologic and respiratory signs with only minor signs of gastrointestinal alterations. Although these signs are not specific of enterotoxemia, this disease should be included in the list of differential diagnosis for acute neurologic and respiratory diseases of cattle.
Perivascular proteinaceous edema (microangiopathy) is considered a characteristic change of C. perfringens type D enterotoxemia in sheep.7, 8, 30, 31, 33 This change was found in most sick animals in the present study, and, to the best of our knowledge, it is not a change observed in other neurologic diseases of cattle. This suggests that it might be useful for the diagnosis of enterotoxemia in this species. However, 2 of the animals that developed acute neurologic disease did not develop histologically visible microangiopathy. These animals were euthanatized for humane reasons, and it is possible that they would have developed microangiopathy had they lived longer.
The similarity in clinical and pathologic findings observed in the experimental disease induced by ETX in several species23, 27, 29 and the cases of experimental enterotoxemia in cattle presented here suggest that ETX was the principal factor in the pathogenesis of this disease in cattle in our study. However, C. perfringens type D produces, in addition to ETX, alpha toxin and at least 15 so-called minor toxins, thus a synergistic role of other C. perfringens toxins can not be ruled out.
Several authors have described the effects of ETX in the brain of mice,33 rats,12 and lambs.27 It has been suggested that this toxin binds to a specific receptor of the brain endothelial cells, causing increase of the vascular permeability and cerebral edema.8, 9, 18, 22 Gardner13 showed, by electron microscopy, progressive swelling of astrocytes with predominant alterations in the astrocyte foot processes around blood vessels and neurons. This pathogenesis is consistent with the main histologic alteration observed in our study, perivascular proteinaceous edema, which suggests damage to the vascular endothelium. FSE similar to what was observed in one of our experimental animals was previously described in cattle and speculatively attributed to ETX of C. perfringens type D.1, 9, 11, 19, 20 Our results lend support to that speculation. Two animals inoculated with SUPNT recovered after developing transient mild CNS clinical alterations, which is notable because no such spontaneous recovery has been reported in ruminants with type D enterotoxemia.
ETX also accumulates in the kidney of mice.10, 21 However, in ruminants, the production of renal lesions by ETX is controversial, and many authors agree that the so-called “pulpy kidney” is an autolytic change and that no morphologic antemortem changes occur in the kidney of ruminants with type D enterotoxemia.26, 31
Glycosuria was found in 4 out of 12 calves inoculated with C. perfringens type D or its products in this study. Glycosuria is supportive of a diagnosis of enterotoxemia in sheep.29, 30 These results confirm that glycosuria may also be associated with enterotoxemia in cattle, although this is not consistently observed.
In summary, Koch's postulates have been met, and the results of this study show that experimental enterotoxemia by C. perfringens type D in cattle has similar clinical and pathologic characteristics to the natural and experimental disease in sheep, including perivascular proteinaceous edema of the brain and FSE. As in sheep,30 lesions affected the internal capsule, thalamus, midbrain, cerebellum, and cerebellar peduncles.1
Footnotes
Acknowledgements
We thank Ms S. Fitisemanu for typing this manuscript and Ms S. J. Uzal for reviewing it.
