Abstract
Enterotoxemia caused by Clostridium perfringens type D usually affects sheep and goats ≥ 2-wk-old. The main clinical signs and lesions of the disease are produced by the epsilon toxin (ETX) elaborated by this microorganism. However, ETX is produced in the form of a mostly inactive prototoxin that requires protease cleavage for activation. It has traditionally been believed that younger animals are not affected by type D enterotoxemia given the low trypsin activity in the intestinal content associated with the trypsin-inhibitory action of colostrum. Two Nigerian dwarf goat kids, 2- and 3-d-old, with a history of acute diarrhea followed by death, were submitted for postmortem examination and diagnostic workup. Autopsy and histopathology revealed mesocolonic edema, necrosuppurative colitis, and protein-rich pulmonary edema. Alpha toxin and ETX were detected in intestinal content, and C. perfringens type D was isolated from the colon of both animals. The isolates encoded the gene for lambda toxin, a protease that has been shown previously to activate ETX in vitro. Type D enterotoxemia has not been reported previously in neonatal kids, to our knowledge, and we suggest that lambda toxin activated the ETX.
Clostridium perfringens, an anaerobic, sporulating, gram-positive bacterium, 15 is classified into 7 toxinotypes (A–G) based on the coding of 1 or more of 6 exotoxins, namely, alpha (CPA), beta (CPB), epsilon (ETX), iota, enterotoxin, and necrotic enteritis B–like. 9 C. perfringens type D isolates encode CPA and ETX. Type D enterotoxemia is one of the most common clostridial diseases of sheep and goats, and has a worldwide distribution.10,13 ETX is considered the main virulence factor of C. perfringens type D, and it is responsible for the clinical signs and lesions of type D disease. 15
The pathogenesis of type D disease is usually associated with an abrupt change in the intestinal milieu, which is believed to be caused by overfeeding of fodder rich in carbohydrates, which, when failing to be digested in the pre-stomachs, passes into the small intestine. This carbohydrate load promotes the proliferation of C. perfringens, accompanied by a large amount of toxin production. 6 ETX is produced in the form of a relatively inactive, ~33-kDa polypeptide, named epsilon prototoxin (pETX). This compound is activated by one of several proteases present in the intestinal environment to the active ~29-kDa ETX by removing 14 C-terminal amino acids.2,3,7 The main protease responsible for activation of pETX is trypsin, although other proteases may also activate pETX. Lambda toxin is a protease produced by some strains of C. perfringens, which has been proven to also activate pETX in vitro. 7 However, to our knowledge, this action has not yet been demonstrated in vivo. Once activated, ETX increases intestinal permeability, which facilitates absorption of ETX across the intestinal mucosa; ETX enters the blood, resulting in the toxin spreading to various target organs, including, but not limited to, the brain, lungs, and heart.2,4 ETX, a member of the aerolysin family of pore-forming toxins, oligomerizes at the cell surface and forms pores in the membrane that promote the loss of intracellular K+ and entry of Na+, Cl−, and Ca++, causing cell necrosis.2,3,8,11
In goats, type D enterotoxemia is characterized by neurologic disease, enteric disease, or a combination of both. Clinical signs include hemorrhagic diarrhea, anorexia, abdominal discomfort, respiratory difficulty, opisthotonos, and/or convulsions. 15 The disease is frequently fatal but occasionally animals may recover. 15
It has traditionally been believed that neonatal animals were not affected by type D enterotoxemia because of the low trypsin activity in the intestinal content given the trypsin-inhibitory action of colostrum and the consequent lack of pETX activation. We present here 2 cases of enterotoxemia in neonatal goats produced by lambda toxin–positive C. perfringens type D. We speculate that lambda toxin was responsible for the activation of pETX.
Two Nigerian dwarf goat kids (kids 1 and 2) from 2 farms in Riverside County, California, were submitted to the San Bernardino laboratory of the California Animal Health and Food Safety Laboratory (CAHFS; University of California–Davis [UCD], Davis, CA) in April 2021 for postmortem evaluation. Both kids were born uneventfully and nursed within 1 h of birth, then became laterally recumbent and weak, had diarrhea, and died when they were 2- and 3-d-old, respectively. An autopsy was performed within 24 h of the death of each animal.
Both kids had mild peritoneal and pleural effusion with fibrin strands adherent to the pleura and serosal surface of the colon, ileum, and mesentery (Figs. 1, 2), and marked semifluid distension of the spiral colon, cecum, and distal ileum. The spiral colon had a dark red serosa and mucosa, and the mesocolon was edematous (Fig. 2). The lungs were congested and edematous. There was hydropericardium (Fig. 3), containing a few strands of fibrin. In addition, kid 2 had a 3-cm long intussusception in the mid-jejunum; proximal to this lesion, the intestine was distended by ~40 mL of pasty yellow content compatible with a mixture of meconium and colostrum; no plant material was observed. No other significant gross abnormalities were observed in either carcass.
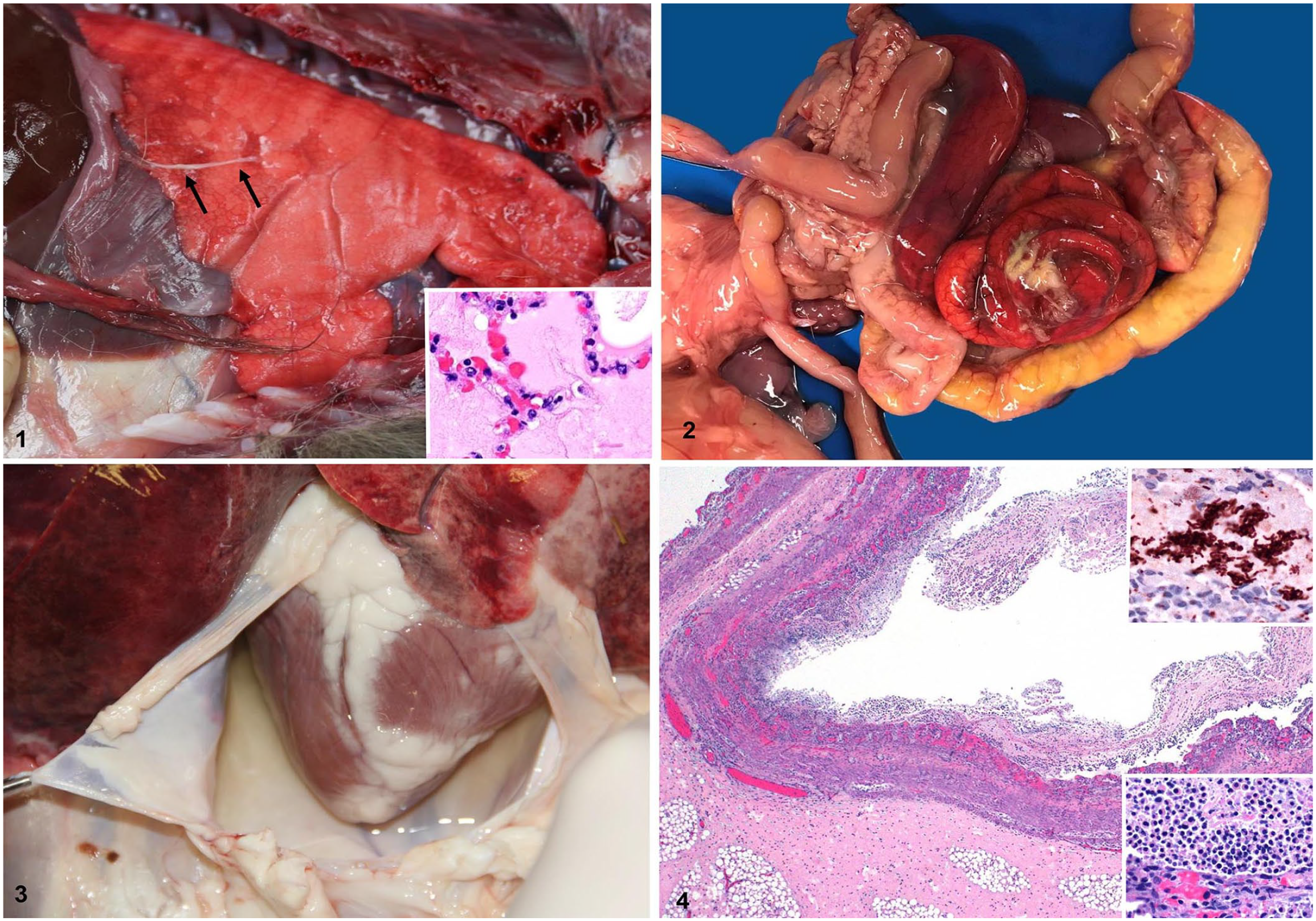
Gross and microscopic changes in 2 neonatal goats with lambda-positive Clostridium perfringens type D enterotoxemia.
Samples of heart, skeletal muscle, kidney, spleen, liver, lung, tongue, reticulum, rumen, omasum, abomasum, duodenum, jejunum, colon, adrenal gland, and the whole brain from both animals were collected and fixed in 10% neutral-buffered formalin for ~24 h. The brain was then sliced at ~0.5-cm intervals and fixed in fresh formalin for another 7 d, after which samples of parietal cortex, corpus striatum, thalamus, brainstem at the level of the anterior colliculus, cerebellum, anterior cerebellar peduncles, pons, and medulla at the level of the obex, were collected. Tissues were processed routinely for the production of 5-μm sections that were stained with H&E. Small and large intestinal sections were also stained with Gram stain, and immunohistochemistry (IHC) for C. perfringens was performed, as described previously. 1
Histologically, the colon of both goat kids had fibrinonecrotizing colitis (Fig. 4) characterized by multifocal-to-coalescing mucosal necrosis, edema, hemorrhage, microvascular thrombosis, and clusters of intralesional gram-positive bacilli, a few of which had sub-terminal spores. Most of these rods stained positively with C. perfringens IHC (Fig. 4). A fibrinonecrotizing pseudomembrane covered the necrotic mucosa (Fig. 4). In addition, villus atrophy was observed in the small intestine. The area of intussusception in the small intestine of kid 2 had diffuse and transmural congestion and hemorrhage. The lungs had multifocal intra-alveolar and interstitial high-protein edema (Fig. 1) and congestion. No significant microscopic abnormalities were observed in any of the other tissues from the 2 kids.
Additional ancillary testing was performed according to CAHFS standard operating procedures. For bacterial anaerobic culture, intestinal contents collected from grossly affected areas of the small and large intestine of each goat kid were inoculated onto pre-reduced anaerobically sterilized Brucella blood agar (Anaerobic Systems), pre-reduced anaerobically sterilized phenylethyl alcohol sheep blood agar (Anaerobic Systems), and egg yolk agar (Anaerobic Systems), and incubated anaerobically at 37°C for 48 h. Intestinal content from each goat kid was also inoculated onto cycloserine–cefoxitin–fructose agar (Veterinary Media Services, UCD) and incubated anaerobically at 37°C for 48 h. All isolates were identified by conventional biochemical techniques and/or MALDI-TOF mass spectrometry. Samples of intestinal contents, lung, and liver from each animal were inoculated onto Columbia 5% sheep blood agar (Hardy) and MacConkey agar plates (Hardy) and incubated aerobically or microaerobically, respectively, at 37°C for 48 h. A PCR assay for Salmonella spp. was performed on small and large intestinal content, mesenteric lymph node, and liver.
No aerobic bacteria were isolated from lung or liver of either kid; the Salmonella spp. PCR assay on colonic content, mesenteric lymph node, and liver was negative. Moderate numbers of C. perfringens were isolated from colonic contents of both kids, and the isolates (5 colonies per animal) were typed by PCR as described previously. 1 Both isolates encoded the genes for CPA and ETX but none of the genes for the other major toxins of C. perfringens, indicating that they were type D. In addition, a PCR assay for the lambda toxin gene was performed on the 2 isolates as described previously, 7 and both were positive.
An ELISA for C. perfringens CPA, CPB, and ETX toxins was performed on small intestinal contents of both kids and on colonic contents from kid 1 using a commercial kit (Bio-X), following the manufacturer’s instructions. The colonic content from kid 1 and the small intestinal content from kid 2 were positive for ETX and negative for CPA and CPB. No parasite eggs were observed on fecal flotation. PCR assays of splenic tissue for border disease virus (Pestivirus ovis) and ovine herpesvirus 2 (Macavirus ovinegamma2) were negative in both animals. A heavy metal screen and selenium concentrations in the liver were within RIs.
The presumptive diagnosis of clostridial enterotoxemia in goats is established based on the clinical history and signs, gross and microscopic postmortem findings, and detection of C. perfringens type D in intestinal content by culture followed by PCR typing. Confirmation of the diagnosis, however, requires the detection of ETX in intestinal content.14,15 In our case, the gross and microscopic lesions, coupled with the isolation of C. perfringens type D from the intestine of both animals, allowed us to establish a presumptive diagnosis of type D disease. Detection of ETX in the intestine of both animals confirmed the diagnosis.
Autopsies were performed close to 24 h after death in both animals. Although this might have altered the relative abundance of intestinal microbiota, the diagnosis was confirmed based on the detection of ETX in intestinal content, and there is no evidence that ETX is produced postmortem in ruminants.
To our knowledge, type D enterotoxemia has not been described previously in neonatal goat kids or lambs. We retrieved no cases of enterotoxemia in neonatal animals in a search of Google, PubMed, CAB Direct, Web of Science, and Scopus, suggesting that this condition has not been reported in goat kids or lambs. The lack of trypsin and other protease activity in the intestine of neonatal animals is usually assumed to prevent pETX activation, which is one of the first steps in the pathogenesis of the disease. Other proteases present in the intestine of ruminants include chymotrypsin, elastase, enteropeptidase, and carboxypeptidases, all of which can activate pETX, but are inhibited by colostrum. 3 However, some strains of C. perfringens type D produce lambda toxin, which has been demonstrated in vitro to activate pETX.5,7 The 2 strains of C. perfringens type D isolated from these 2 goat kids had the lambda toxin gene, suggesting that they were able to produce this toxin. If that were the case, it is very likely that lambda toxin activated pETX. There are no validated methods available to detect lambda toxin in the intestinal content of animals, and we therefore could not test for this toxin in the intestinal content of our cases.
The lambda toxin gene has been described in a comparative genomic analysis of 4 C. perfringens strains, 2 derived from cattle. 12 However, to our knowledge, there is no information in the literature about the prevalence of lambda toxin–positive strains of C. perfringens in goats or neonatal animals in general.
The intussusception and intestinal stasis in kid 2 could have favored the proliferation of C. perfringens and predisposed the kid to enterotoxemia. Moreover, the intussusception could have prevented the normal transit of colostrum from reaching more aboral segments of the small intestine, with the consequence that the trypsin in the area would have remained active. Alternatively, the intussusception could have been a consequence of the enteritis caused by ETX. Type D enterotoxemia has not been reported previously in neonatal kids, to our knowledge, and we suggest that lambda toxin activated the ETX.
Footnotes
Acknowledgements
We thank Dr. Bruce McClane (Medical School, University of Pittsburgh) for his advice on the PCR for the lambda toxin gene PCR, and Mr. Jose Polanco (CAHFS San Bernardino, UCD) for excellent technical assistance.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
