Abstract
A 6-month-old dairy heifer calf with no premonitory signs was acutely down after the morning feeding and could not rise. On presentation, the heifer was in right lateral recumbency and moribund with opisthotonus and left hind limb paddling. Following euthanasia, gross examination of the brain revealed multifocal loss of gray-white matter distinction and extensive petechiae throughout the brainstem. On histopathological examination, there was striking white matter edema and marked perivascular proteinaceous edema surrounding many arterioles and venules (microangiopathy), mainly in the white matter of the internal capsule, thalamus, midbrain, cerebellum, and cerebellar peduncles. The perivascular neuropil was strongly positive for Alzheimer precursor protein A4. Clostridium perfringens epsilon toxin was detected in the intestinal contents. This is the first report of microangiopathy in postneonatal cattle associated with the detection of epsilon toxin in the intestinal contents.
Clostridium perfringens type D enterotoxemia has been studied mainly in sheep and goats, species in which the condition occurs most frequently. 9 Clinically, the disease is mostly characterized by sudden death or neurological disease in sheep and goats or by diarrhea in goats. Most acute and subacute cases of enterotoxemia in sheep and occasionally in goats are characterized by perivascular proteinaceous edema in the brain (microangiopathy), whereas chronic and occasionally subacute cases in both species have multifocal symmetrical encephalomalacia and white matter edema. 9 When present, brain lesions provide a diagnosis of type D enterotoxemia in sheep and goats. However, when brain lesions are not present, confirmation of the diagnosis in these 2 species requires the detection of epsilon toxin (ETX) in the small and/or large intestinal contents. 9
Despite the numerous studies published on type D enterotoxemia in small ruminants, information on clinical and pathological findings of the disease in cattle is sparse. Disease with clinical signs and histological lesions almost identical to those observed in ovine enterotoxemia was produced experimentally in calves. 4,8 Spontaneous disease with brain lesions like those in sheep was also described in cattle. 1,3 However, a causal relationship between the lesions and C. perfringens type D or ETX was not established in any of these cases, and the cause was undetermined. Recently, microangiopathy similar to that in ovine and caprine enterotoxemia was described in the brain of two 1-day-old calves in which ETX was detected in the large intestinal content. 10 Microangiopathy associated with C. perfringens type D ETX has not been previously reported in older cattle. The present report describes a case of neurological disease in a heifer with ETX detected in the intestine and with brain lesions very similar to those in C. perfringens type D enterotoxemia of sheep.
Case History
A 6-month-old Holstein heifer calf was submitted to the Davis branch of the California Animal Health and Food Safety Laboratory System (CAHFS) for necropsy and diagnostic workup. The calf had been transferred from a mixing pen at the home farm to a feed lot, where it was housed with 25 other calves, 15 days before onset of disease. All calves were on concrete floors with wood shaving and dried manure bedding and received a high-quality alfalfa and oat hay ration with a multimineral preparation ad libitum. The calves were normal during the morning feeding; however, immediately after being fed, this calf was recumbent with labored breathing but no evidence of diarrhea. All other animals in the feedlot were apparently healthy.
Postmortem Examination
Upon arrival to CAHFS, the calf was laterally recumbent with hind limb paddling, obtunded, and opisthotonic. Euthanasia was performed and a necropsy carried out. Samples of pituitary gland, skeletal muscle, tongue, heart, trachea, esophagus, lungs, liver, kidney, spleen, pancreas, lymph nodes, adrenal gland, rumen, reticulum, abomasum, small and large intestines, urinary bladder, and brain were fixed in 10% buffered formalin for 24 hours. Both halves of the brain were sliced at ∼0.5-cm intervals and examined grossly before blocks were obtained from the parietal cortex, corpus striatum, thalamus, midbrain at the level of rostral colliculi, pons, anterior cerebellar peduncles, cerebellum, and medulla at the level of the obex. Tissue samples were processed routinely and embedded in paraffin; 5-μm-thick sections were stained with hematoxylin and eosin (HE). Histological sections were examined to determine the type, location, and severity of lesions. Selected sections of brain were stained with periodic acid–Schiff (PAS) and Holmes silver nitrate/Luxol fast blue (LFB) and processed for immunohistochemistry for the Alzheimer precursor protein A4 (APP). Pooled small and large intestinal content was aseptically collected and cultured anaerobically on blood agar at 37°C for 48 hours. A C. perfringens isolate was typed by polymerase chain reaction (PCR) as described. 5 The same specimen was tested for C. perfringens alpha (CPA), beta (CPB), and ETX toxins by a capture ELISA technique. 9
At necropsy, the calf was in good nutritional condition. Cross-sections of the brain had multifocal loss of gray-white matter distinction in regions of the cerebrum, predominantly in the frontal cortex of the right hemisphere with lesser involvement of the left. Petechiae were widely dispersed throughout the brain stem. In the parietal region of the right cerebral hemisphere was a 4 mm × 7 mm cavitation at the gray-white matter junction. Exposure of the unfixed brain to Wood’s lamp did not produce fluorescence. The lungs were diffusely edematous with a moderate amount of stable froth in the trachea and bronchi. No other significant gross abnormalities were observed.
Significant histological changes were restricted to brain and lungs. Severe multifocal white matter rarefaction due to edema was evident in the cerebrum and cerebellum. Venules and arterioles were prominent and congested with swollen endothelium throughout the brain sections. Frequently, Virchow-Robin spaces were expanded by an eosinophilic, homogeneous, proteinaceous fluid (Fig. 1) or eosinophilic to amphophilic hyaline droplets (Fig. 2). There was also multifocal perivascular hemorrhage. The perivascular edema and the hyaline droplets were PAS positive. The LFB demonstrated marked separation of the cellular processes by edema. Immunohistochemical labeling for APP was strongly positive in the neuroparenchyma around the affected vasculature in both white and gray matter, highlighting the damaged neuronal processes (Fig. 3). The distribution of the perivascular edema, hyaline droplets, and perivascular hemorrhage was mostly restricted to the internal capsule, thalamus, midbrain, cerebellum, and cerebellar peduncles (Fig. 4a, b, c). The gross cavitation in a cerebral gyrus was a sharply demarcated space surrounded by markedly edematous white matter. The changes in the brain were bilaterally symmetrical. The lung had diffuse, mild to moderate edema in the perivascular air spaces and interlobular septa.
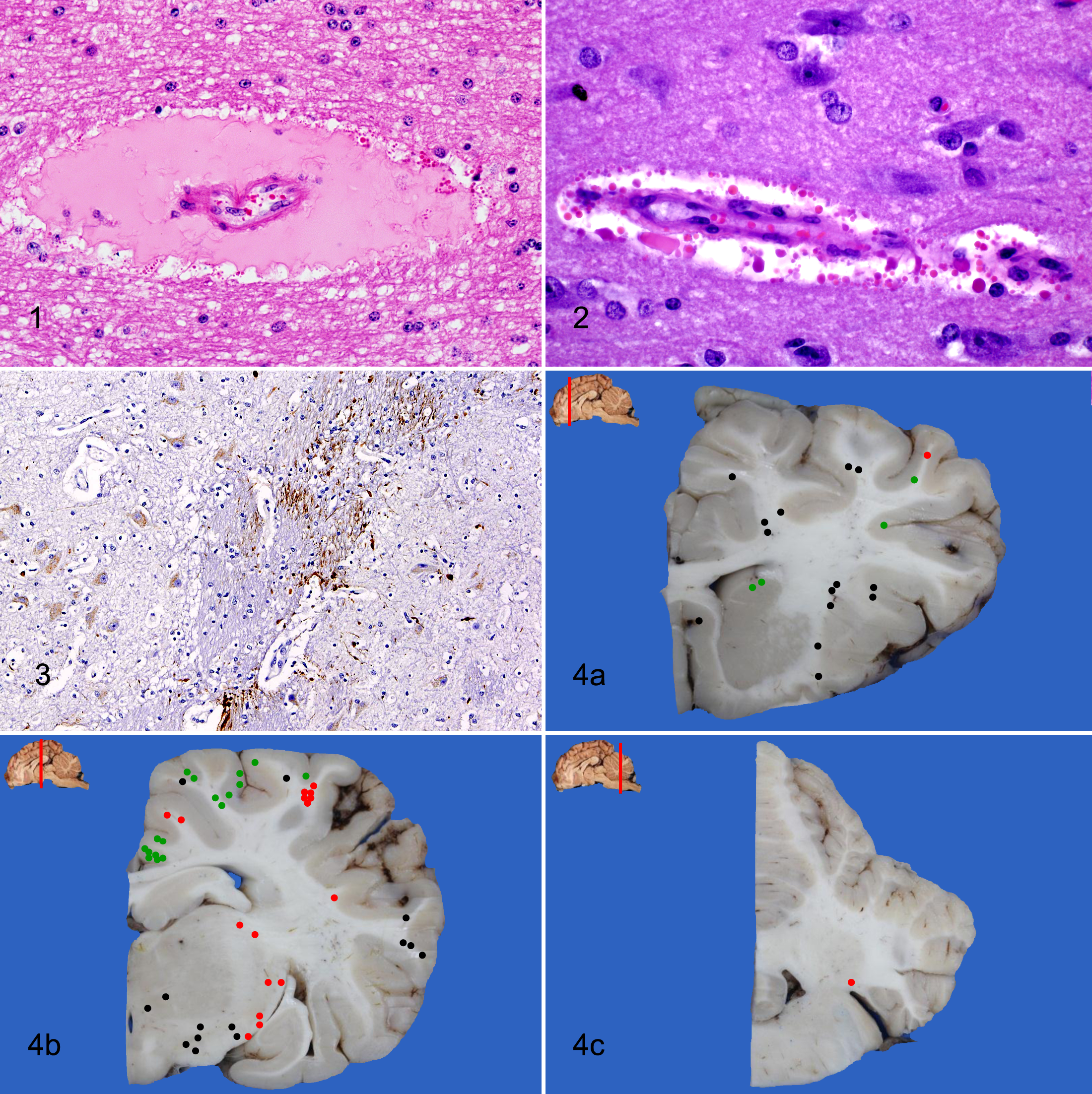
Cerebrum; calf. Arteriole with expanded Virchow-Robin space by a protein-rich eosinophilic homogeneous fluid. HE.
The anaerobic culture of intestinal contents yielded C. perfringens (identified as type A, CPE and beta 2 negative by PCR) and Clostridium sordellii in moderate numbers. CPA and ETX were identified by ELISA in the same specimen. Ancillary laboratory testing ruled out aerobic bacterial infection (lung, liver, meninges), Salmonella spp, bovine viral diarrhea virus, rabies virus, and salt or heavy metal toxicoses.
Discussion
This calf had clinical signs and histological findings in the brain compatible with those of ovine enterotoxemia by C. perfringens type D. The perivascular brain lesions are considered to be pathognomonic for type D enterotoxemia in sheep and goats, 9 and the extensive microangiopathy in the brain was the main reason that ETX testing was pursued in this case. ETX was detected in the intestinal content, which is considered diagnostic for ovine and caprine enterotoxemia. 9 According to Watson and Scholes, 10 demonstration of ETX should not be used in isolation to confirm enterotoxemia in cattle. Although the diagnostic significance of detecting ETX in the intestine of cattle has not been fully determined, its presence has been used to establish a diagnosis of bovine type D enterotoxemia. 10 In the present case, the finding of ETX in the gut was therefore considered highly suggestive, although not confirmatory, of a diagnosis of type D enterotoxemia. Although ETX can also be produced by CPB, this toxinotype also produces beta toxin, which was not detected in the intestine of this animal. Furthermore, CPB has not been isolated in North America, which makes it even more unlikely that the ETX detected in the intestine was produced by this toxinotype of C. perfringens.
C. perfringens type A, but not D, was isolated from the intestine of this calf. It is likely that the type A organism overgrew the type D strain or that the latter was not present in the specific sample that was collected for bacterial culture. This result, however, does not preclude a diagnosis of type D enterotoxemia if ETX is detected in the intestinal content. Furthermore, the distribution of the lesions in the brain had a similar pattern as in sheep and ETX experimental inoculation in cattle. 8 ETX appears to cause similar brain lesions in all ruminants that have been studied, although the reason for this lesion distribution remains unknown. The gross cerebral cavitation in this calf is not a feature of ETX produced-disease and possibly represents congenital porencephaly not associated with C. perfringens type D infection and microangiopathy.
The diagnosis of C. perfringens-induced disease is complicated by the fact that some toxinotypes (eg, type A) are often found in the intestinal tract of healthy animals. However, type D has a much lower prevalence than type A in the intestine of healthy ruminants. 9 It has been assumed that an abrupt feed change to a highly fermentable carbohydrate diet is the main factor that induces bacterial overgrowth and toxin production. Starch overload in the small intestine of sheep and goats has been implicated in providing a most suitable substrate leading to toxin production. 4 However, this has not been proven, and cases of enterotoxemia have been observed in sheep and goats on a constant hay diet. In the present case, the calves received an alfalfa hay and oat hay–based diet with no grain for at least 2 weeks before onset of disease.
Although the pathogenesis of type D enterotoxemia is not fully understood, several studies have demonstrated that ETX induces increased small intestinal permeability followed by absorption of ETX into circulation. 6 This is followed by a direct effect of ETX on brain endothelial cells causing cell damage and necrosis, increased vascular permeability, and cerebral edema as well as binding to myelin, astrocytes, and microglia. 6 The clinical signs of ETX intoxication could be a result of reduced vascular perfusion and hypoxia of the brain parenchyma surrounding affected blood vessels or a direct effect on the neuropil.
The positive perivascular labeling for APP in this study supports a hypoxic mechanism, although the possibility that this distribution reflects the route of delivery of ETX to the neuroparenchyma cannot be excluded. APP is a transmembrane protein whose proposed functions include stabilization of neuronal calcium fluxes, inhibition of the clotting cascade, and cell-cell or cell-matrix adhesion. Normally present at low levels in neurons, its appearance is induced as part of the acute response of the brain to a range of insults including hypoxia-ischemia. 7 Information about the use of APP in cattle is scant, 2 although one of the authors (S.S.) has used it extensively to characterize acute degenerative lesions in bovine brain. Immunohistochemistry for APP could provide a useful tool to detect early brain lesions of type D enterotoxemia, particularly in cases in which intestinal contents for ETX detection are not available and/or microangiopathy is not readily detectable.
Footnotes
Acknowledgements
Special thanks to D. Paulson, P. Yant, R. Cazarez, and J. Shannan for their excellent technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
