Abstract
Catechol-O-methyltransferase (COMT) is an important enzyme involved in inactivation of catechol estrogens, which are metabolites with carcinogenic properties. Some investigations in human breast cancer associate a genetic polymorphism in the COMT gene (COMT val158met) with an increased risk and poor clinical progression of the disease. In dogs, there are 2 recognized single nucleotide polymorphisms in the COMT gene (COMTG216A and COMTG482A); however, their influence on the outcome of mammary neoplasms has never been investigated. The purpose of this study is to investigate the influence of COMT in the clinical progression of canine mammary tumors, namely in recurrence, metastasis and survival by testing 2 SNPs (G216A and G482A), and 2 genotypes of the COMT gene. A case series was conducted analyzing genomic DNA samples by polymerase chain reaction-restriction fragment length polymorphism from 80 bitches with mammary tumors. Animals were submitted to an active follow-up study for a period of 24 months after surgery. We observed that bitches carrying both genetic variations simultaneously are more likely to develop recurrence of mammary lesions. Our results demonstrate a possible role for COMT genotypes in the outcome of mammary neoplasms in the dog. Identifying a genetic factor predictive of recurrence may be useful in selecting the most effective surgical approach for canine mammary neoplasms.
Introduction
Mammary neoplasms are the most common tumors in the female dog.15, 18 Several lines of evidence suggest that steroid hormones play a determinant role in the etiopathogenesis of the disease.18 Epidemiologic and experimental studies have identified that increased or prolonged exposure of mammary tissue to estrogens is a risk factor for breast cancer.6, 7, 21 Estrogens mediate carcinogenesis by promoting epithelial cell growth and acting as precursors for mutagenic estrogen metabolites.1, 5, 28 Inactivation of such intermediate metabolites represents an important step in the prevention of breast cancer and suppression of progression.
Inactivation of catechol estrogens, carcinogenic metabolites that have the potential to induce DNA damage and form DNA adducts, is catalyzed by catechol-O-methyltransferase (COMT).1, 2 There is a well-documented single nucleotide polymorphism (SNP) in the human COMT gene (G→A change), resulting in an amino acid substitution (val to met) at position 158. This polymorphism encodes a low-activity form of COMT,11, 14 which appears to result in an increased risk for the development of breast cancer3, 8, 12 and clinical progression of the disease.13, 17
Masuda and colleagues16 investigated the canine COMT gene and successfully amplified 2 SNPs: a guanine-to-adenine substitution at the 216th nucleotide without changes at the amino acid level (G216A) and a guanine-to-adenine substitution at the 482nd nucleotide, resulting in an arginin to glutamine change (G482A).
Since the publication of the Masuda study, we demonstrated an association between COMT G482A SNP and the age of onset of canine mammary tumors.4 However, there have been no other investigations regarding the relationship between COMT genetic polymorphisms and the risk, clinicopathologic features, or progression of canine mammary neoplasms.
The purpose of this study is to investigate the influence of COMT in the clinical progression of canine mammary tumors by testing 2 SNP (G216A and G482A) and genotypes in the COMT gene.
Materials and Methods
A canine mammary tumor case series study, defined as an observational study describing a sequence of case reports that share a common element was conducted analyzing genomic DNA samples from 80 bitches with mammary tumors (mean age 9.6 years). Peripheral blood samples from all animals were collected using a standard venipuncture technique for ethylenediamine tetraacetic acid-containing tubes.
Mammary tumors were obtained by surgery and fixed in 10% buffered formalin. The material was dehydrated, embedded in paraffin wax, cut in 3-μm-thick sections, and stained with hematoxylin and eosin. Tumors were classified histologically according to the diagnostic criteria proposed by the World Health Organization classification for mammary tumors of the dog and cat.19 In bitches with multiple mammary tumors, histologic classification was based upon the lesion associated with the poorest prognosis.19
Genomic DNA was extracted from white blood cell fraction using a High Pure PCR Template Preparation Kit (Roche), according to the instructions of the manufacturer. Both COMT G216A and COMT G482A polymorphisms were analyzed through polymerase chain reaction (PCR) followed by restriction fragment length polymorphism (RFLP), as previously described by Masuda and others.16 Analysis of the results was independently performed by 2 of the authors.
For COMT G216A polymorphism, DNA was amplified in a 50-μl reaction mixture containing 2 μl of genomic DNA, 0.3 μM each of the 2 COMT G216A primers (F, 5′-ATGGGAATGAGGGAGGCAGG-3′ and R, 5′-AGAATGGTGACCTGGAACGG-3′), 0.2 mM of deoxynucleotide triphosphates, 1× Qiagen PCR buffer and 1× Qiagen solution and 1 U of Taq polymerase. The amplification was performed in a thermocycler (Biometra), and PCR steps included an initial denaturation at 95°C for 5 minutes, 35 cycles at 95°C for 30 seconds, 61°C for 30 seconds, and 72°C for 1 minute, with a final extension step at 72°C for 5 minutes. The PCR products were then digested overnight at 37°C with 10 U of the restriction enzyme Eco52I (Fermentas) that recognizes the sequence 5′ … C↓GGCCG … 3′.
Amplification and digestion protocols employed for COMT G482A polymorphism analysis have been previously described.4
Animals were submitted to an active follow-up study for a period of 24 months after surgery. Bitches were clinically evaluated every 3 months by careful physical examination, thoracic radiography, and abdominal ultrasound to investigate the existence of recurrences or distant metastases. Animals that died with suspected distant metastases were submitted to complete necropsy examination, and histologic confirmation of the lesions was obtained.
For statistical analysis of data, 2 SNP (COMT G216A and COMT G482A) and 2 genotypes were considered. Genotype 1 was identified as carriers of at least 1 of the SNP studied while genotype 2 was identified as carriers of both SNP simultaneously. Statistical analysis of data was performed using the computer software SPSS for Windows (version 7.0). SNP and genotypes were associated with the histologic phenotype of the tumor (benign vs. malignant) and with 3 outcome parameters within a 24-month period after surgical excision of the primary neoplastic lesion, including recurrence, defined as subsequent mammary tumor development after mastectomy (yes vs. no); metastasis (presence vs. absence); and survival (alive vs. dead). Chi-square analysis (and Fisher exact test, when appropriated) was used to compare categoric variables. Statistical significance was defined as P < .05. The odds ratio (OR) and its 95% confidence interval (CI) were calculated as a measure of the association between COMT genotypes and the clinicopathologic variables already mentioned. We also estimated the cumulative probability for recurrence, development of metastasis, and survival on the basis of COMT genotype by using the Kaplan-Meier methodology.10
Results
For the population included in this study, the frequency of COMTG216A SNP was 20% for AA, 11.25% for AG, and 68.75% for GG. The frequency of COMTG482A SNP was 2.5% for AA, 22.5% for AG, and 75% for GG. Genotype 1 represented 35%, and genotype 2 represented 21.3% of the population studied.
After careful histologic examination, 43 tumors (53.75%) were classified as benign and 37 tumors (46.25%) as malignant. None of the studied genotypes were significantly associated with the histologic classification of mammary neoplasia.
Figure 1 shows the association of COMT genotypes with the recurrence of mammary tumors within 24 months after surgical excision. Twenty-seven cases were excluded from this part of the study because they were either lost to follow-up (n=21) or died within the follow-up period for causes unrelated to mammary tumor (n = 6). Animals that died unrelated to mammary neoplasia did not show evidence of tumor recurrence.
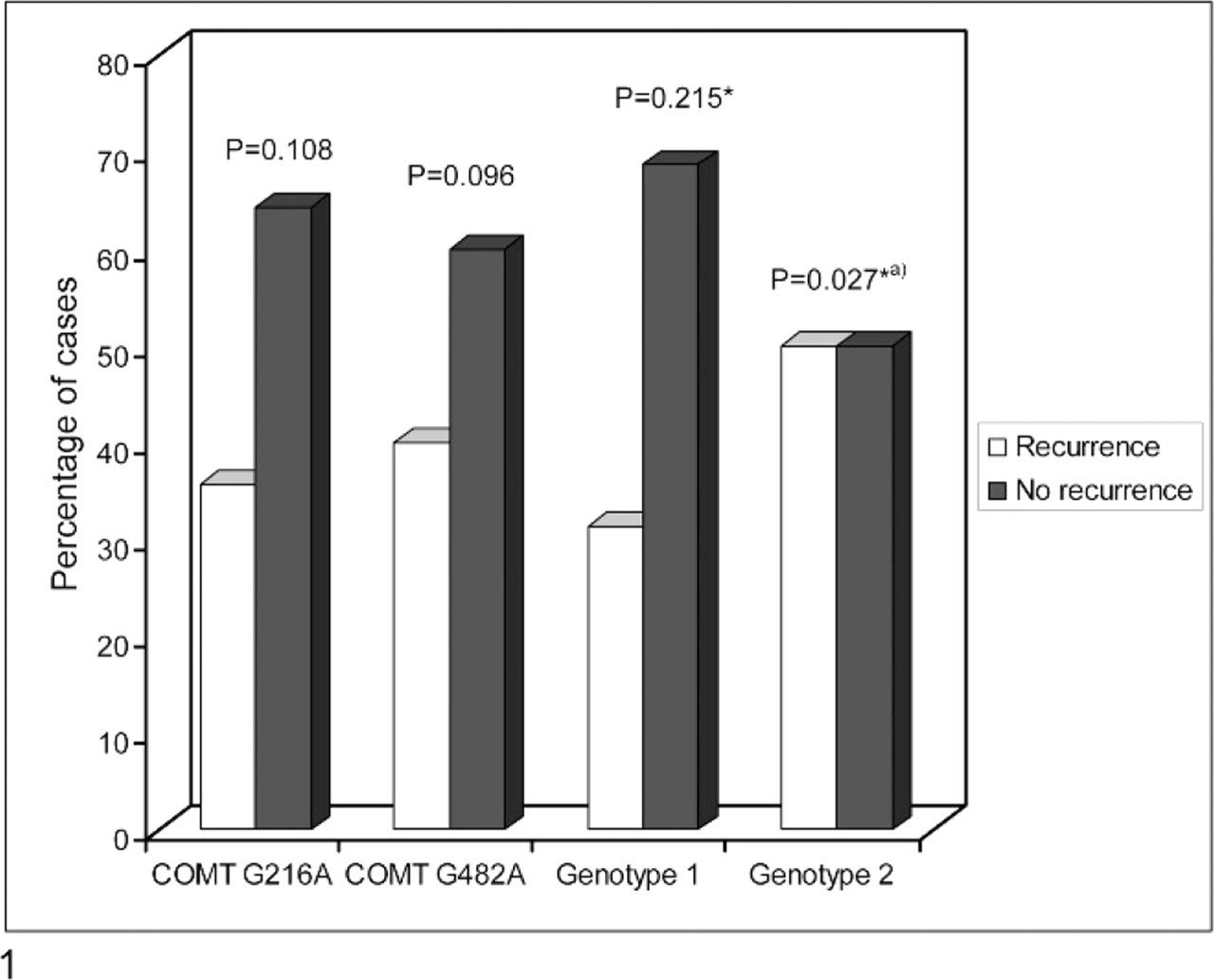
Effect of COMT genotype on recurrences within 24 months after surgical excision of the mammary tumor (∗Chi-square test; a P = .048 [Fisher exact test]).
Recurrence of mammary lesions was observed in 11 of 53 cases. Differences in the recurrence rate observed in animals younger than 9 years (10.5%) or older than 9 years (26.5%) were not statistically significant (P = .307). Animals with genotype 2 represented 36.4% (4/11) of recurrence cases and only 9.5% (4/42) of cases without tumor recurrence, a statistically significant difference (P = .018). The association between genotype 2 and tumor recurrence is shown in Fig. 2. This subgroup of bitches had a fivefold likelihood of recurrence of mammary lesions when compared with the others (P = .027; OR = 5.43; 95% CI = 1.09–26.98). No significant associations were found between COMT G216A SNP, COMT G482A SNP, or COMT genotype 1 and recurrence of mammary neoplasms.
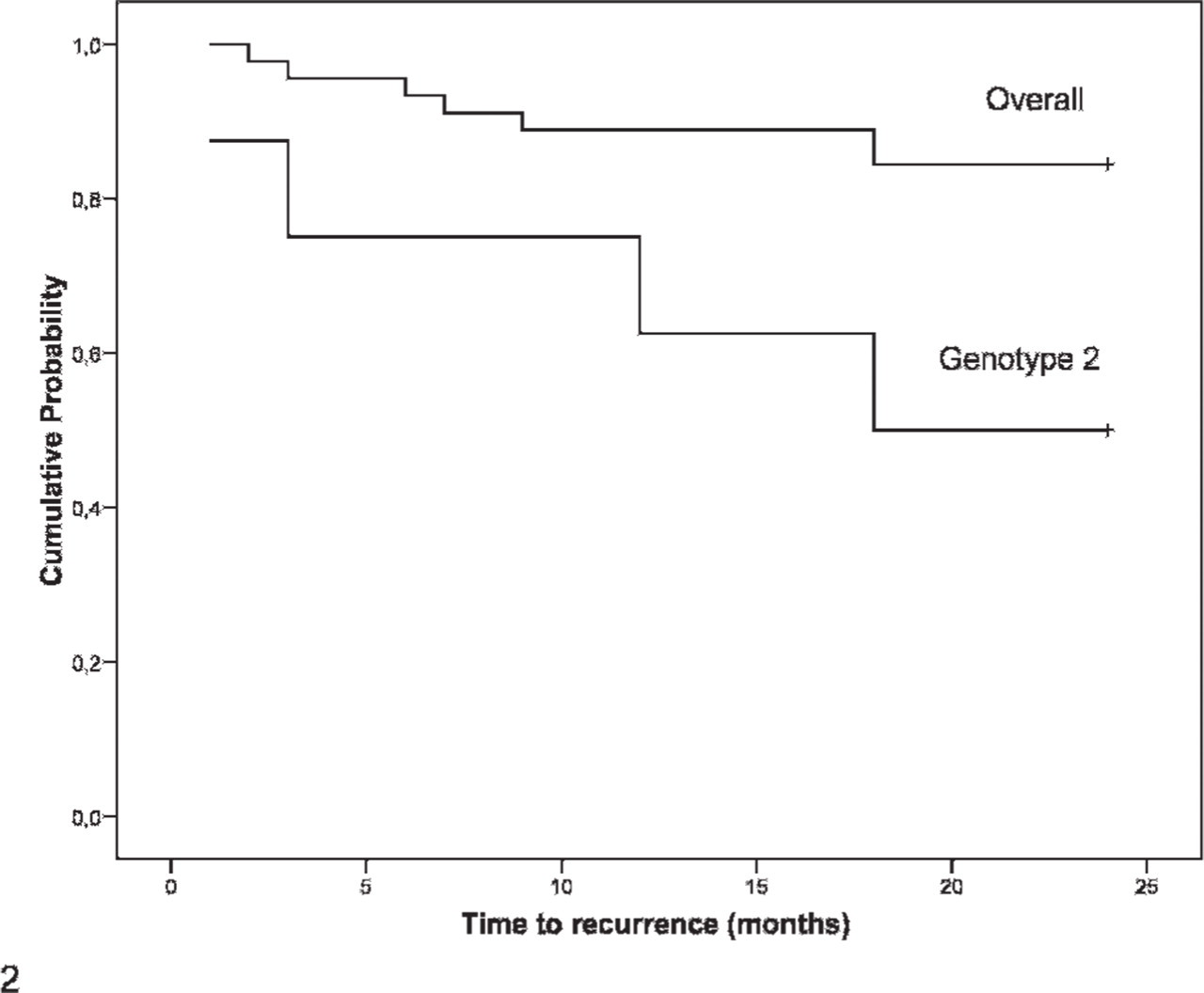
Recurrence of mammary lesions within 24 months after surgical excision (overall vs. genotype 2 [P = .018]).
Within the group of carcinomas (Fig. 3), we found that animals with genotype 2 constituted 36.4% (4/11) of all recurrent carcinomas and only 5.3% (1/19) of nonrecurrent carcinomas, a difference with statistical significance (P = .011). This subset of bitches presented a nearly 11-fold likelihood of recurrence within 24 months of surgical excision of the primary mammary carcinoma (P = .023; OR = 10.86; 95% CI = 1.03–114.58). Figure 4 shows the survival function for recurrence within 24 months of surgical excision of mammary carcinomas, according to COMT genotype.
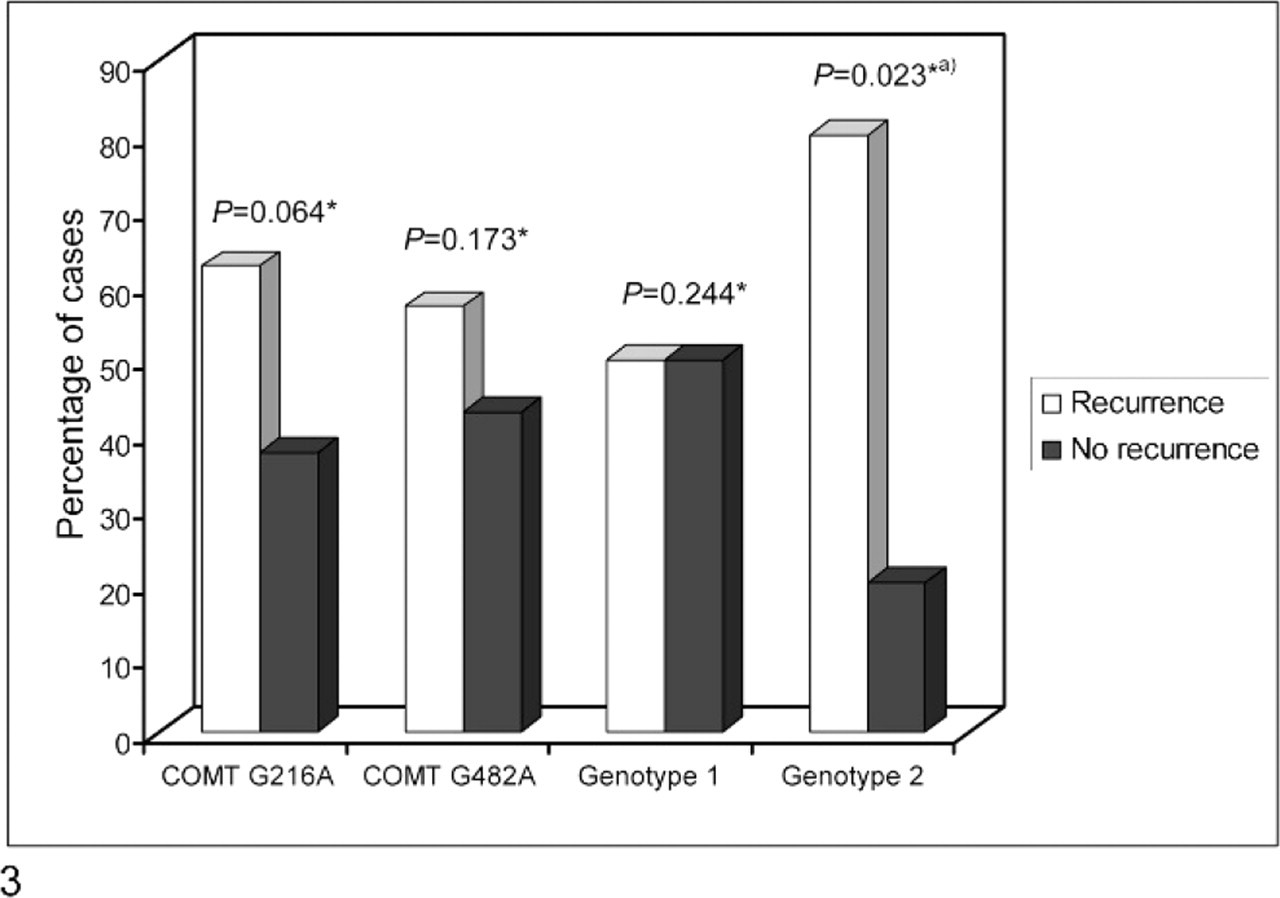
Effect of COMT genotype on recurrences within 24 months after surgical excision of the histologically classified mammary carcinoma (∗Chi-square test; a P = .042 [Fisher exact test]).
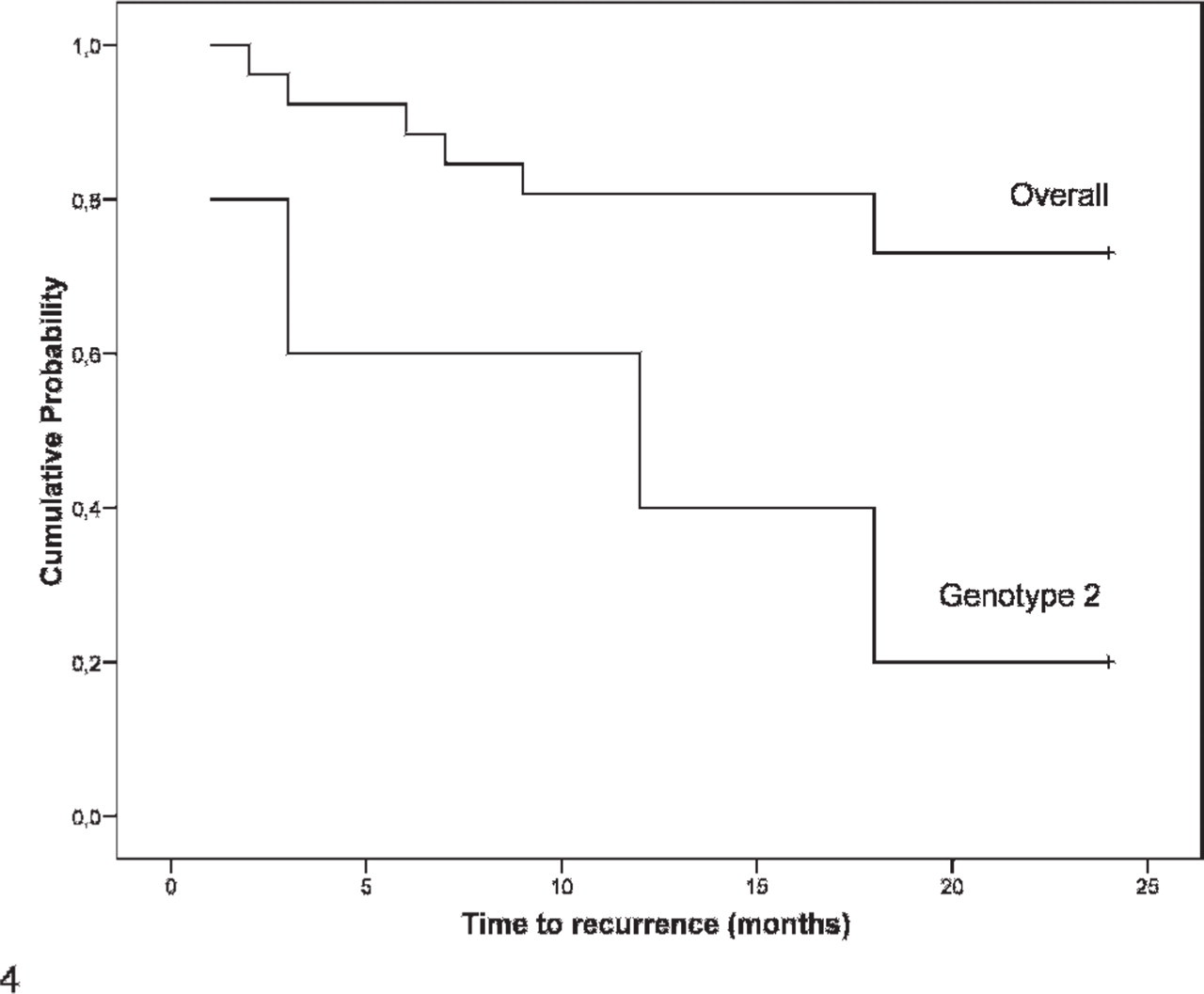
Recurrence of histologically classified mammary carcinomas within 24 months after surgical excision (overall vs. genotype 2 [P = .011]).
We found no effect between the COMT genotypes studied and the development of metastasis or survival of animals with mammary carcinomas within 24 months of surgical excision of the primary mammary tumor.
Discussion
In this study we analyzed 2 COMT SNPs (G216A and G482A) and 2 genotypes and their association with 3 outcome parameters: recurrence, metastasis, and survival within a 24-month period following surgical resection of the primary mammary tumor. Our results strongly suggest that bitches carrying both genetic variations simultaneously are more likely to develop recurrence of mammary lesions. However, we did not find an association between COMT genotypes and metastasis or survival.
Several variant forms of genes (genetic polymorphisms) have been reported to affect the functional activity of enzymes involved in estrogen biosynthesis, metabolism, and bioavailability, particularly COMT. In fact, previous biophysical and kinetic studies demonstrated that the COMT val158met SNP is related to a two- to fourfold decrease in COMT enzyme activity.11, 14 If the inactivation process mediated by COMT is compromised, catechol-estrogens may undergo metabolic redox cycling, generating free radicals and reactive quinones and semiquinones, which cause DNA damage.1, 2 This is biologically consistent with the results of some investigations that have linked COMT SNP with breast cancer risk,3, 8, 12 elevated frequency of lymph node metastasis,17 and reduced disease-free survival.13 It is also conceivable that animals carrying polymorphisms associated with low-activity COMT genotypes may have a poor disease outcome, which was observed in our study for risk of recurrences. COMT kinetic studies in bitches should be done in order to understand the mechanisms of action and functional consequences of COMT polymorphisms in the canine mammary oncogenic process.
Some evidence suggests that COMT is an hormone-sensitive enzyme whose expression is regulated by sex steroid hormones.22, 24 Previous investigations demonstrated that estrogens and progesterone inhibit COMT activity by down-regulating COMT gene transcription in a process mediated by estrogen and progesterone receptors.9, 25, 27 This finding may shed new insight into the regulatory role of sex steroid hormones in the pathophysiology of mammary neoplasms, depending on the animal's hormonal receptor status. It is conceivable that the hormonal receptor status of each animal may be a key determining factor on the influence of COMT genotype in the outcome of mammary neoplasms. This may explain the lack of significant associations between COMT genotype and metastasis or survival observed in the present study. Therefore, in future investigations the subdivision of bitches according to their hormonal receptor status (positive vs. negative) could be a useful strategy to evaluate the prognostic value of COMT genotype in each group of animals.
Most reports on canine mammary tumors focus on the histopathogenic aspects of the lesions, while mechanisms of progression and biological behavior of the disease are not well understood. Therefore, treatment options and information concerning the prognosis of canine mammary tumors are scarce and very limited.18, 26
Surgery remains the treatment of choice for mammary tumors in dogs, and early and complete resection of the lesions with wide margins are advocated.18 However, there is some controversy regarding the selection of regional versus radical mastectomy as the surgical method of choice.20, 23, 26 Our results may constitute a helpful tool in the selection of the most adequate surgical procedure for each individual case. Carriers of both COMT polymorphisms may be recommended for radical rather than regional mastectomy. This surgical approach prevents the need for a second surgical intervention in cases with a likely chance of recurrence which will reduce economic costs and avoid the risk of 2 anesthetic managements and 2 surgical episodes, with their associated wound healing complications. This is an important surgical decision to make, particularly when considering lesions with a high incidence rate in older animals.
Our results demonstrate a possible role for COMT genotypes in the definition of the outcome of mammary neoplasms in the dog, identifying them as a genetic factor predictive of recurrence. Additional studies comprising a larger group of animals are needed to confirm these findings and to identify genetic profiles that may be useful in the prediction of recurrence and, therefore, in the selection of the most effective surgical approach for canine mammary neoplasms.
