Abstract
Catechol-O-methyltransferase (COMT) is an important enzyme participating in inactivation of carcinogenic oestrogen metabolites. In humans there is a single nucleotide polymorphism in COMT gene (COMT va1158met) that has been associated with an increased risk for developing breast cancer. In dogs, there is a single nucleotide polymorphism in COMT gene (G482A), but its relation with mammary carcinogenesis has never been investigated. The aim of this study was to focus on the evaluation of such polymorphism as a risk factor for the development of mammary tumors in bitches and on the analysis of its relationship with some clinicopathologic features (dog's age and weight, number and histologic type of the lesions, lymph node metastasis) of canine mammary neoplasms. A case-control study was conducted analyzing 90 bitches with mammary tumors and 84 bitches without evidence of neoplastic disease. The COMT G482A polymorphism was analyzed by PCR-RFLP. We found a protective effect of the polymorphism in age of onset of mammary tumors, although we could not establish a significant association between COMT genotype and other clinicopathologic parameters nor with mammary tumor risk overall. Animals carrying the variant allele have a threefold likelihood of developing mammary tumors after 9 years of age in comparison with noncarriers. The Kaplan-Meier method revealed significant differences in the waiting time for onset of malignant disease for A allele carrier (12.46 years) and noncarrier (11.13 years) animals. This investigation constitutes the first case-control study designed to assess the relationship between polymorphic genes and mammary tumor risk in dogs. Our results point to the combined effect of COMT genotype with other genetic and/or environmental risk factors as important key factors for mammary tumor etiopathogenesis.
Introduction
Mammary tumor is the most frequent neoplasm in the bitch, representing 25–50% of all neoplasms in female dogs. 20, 24 Steroid hormones play an important role in the pathogenesis of canine mammary tumors, in parallel to what is observed in women. Mammary neoplasms occur almost exclusively in bitches, and most of the rare cases reported in male dogs were associated with testicular neoplasms with concurrent hormonal disturbances. 8, 24 Moreover, the sparing effect of early ovariectomy and the increased risk effect exerted by the administration of long-acting progestins strongly suggest that sex steroid hormones play an important role in mammary carcinogenesis in the dog. 24, 32– 34
In humans, most of the known risk factors for breast cancer (early menarche, late menopause, oral anticonception, and hormone replacement therapy) are related to an increased or prolonged exposure to estrogen. 9, 11, 27 There is a strong and compelling body of information that identifies estrogens as carcinogens, by promoting epithelial cell growth and acting as precursors for mutagenic estrogen metabolites. Estrogens stimulate mammary epithelial proliferation, thereby increasing the chances of genetic errors and enhancing the replication of clones of cells carrying potentially oncogenic mutations. 5 Furthermore, several metabolites derived from oxidative estrogens metabolism constitute potent genotoxic compounds that may act as initiators of genetic mutations. 1, 39
Several studies have demonstrated the existence of variant forms of genes (gene polymorphisms) encoding enzymes involved in estrogen metabolism and biosynthesis. These genetic polymorphisms are responsible for a proportion of interindividual enzymatic variability, thus interfering in the exposure of breast tissue to estrogen. 27, 29, 35
Catechol-O-methyltransferase (COMT) is one such polymorphic gene involved in the inactivation by methylation of catechol estrogens, metabolites that have estrogenic properties and are believed to be carcinogenic. 1, 2 In the active form, catechol estrogens undergo oxidative reactions, resulting in the formation of catechol estrogen quinones. The latter react with DNA to form depurinating adducts and apurinic sites, which can generate mutations playing a critical role in cancer initiation. Moreover, reduction of catechol estrogen quinones to cathecol estrogen semiquinones produces superoxide anion radicals capable of inducing oxidative damage to DNA. 1, 27
In humans, there is a known single nucleotide polymorphism (G→A change) in the COMT gene, resulting in a valine→methionine substitution in codon 158. 19 Toxicologic investigations demonstrated that this gene polymorphism encodes a low-activity form of COMT, 16 and some studies associate it with an increased risk for developing breast cancer. 3, 13, 17 In canine species, there is a recognized single nucleotide polymorphism in the COMT gene resulting in a G→A substitution at the 482nd nucleotide, which has been studied as a possible marker of certain canine behavioral traits. 21 However, to the best of our knowledge, no data describing the involvement of this genetic polymorphism in canine mammary carcinogenesis have been reported yet.
The purposes of this study are to assess the influence of the COMT G482A polymorphism in the development of mammary tumors in dogs and to analyze its relationship with clinicopathologic features of canine mammary neoplasms.
Materials and Methods
A case-control study was conducted analyzing DNA samples from 174 dogs: 90 bitches with mammary tumor (median age 9.0 years) and 84 bitches over 5 years of age (median age 7.1 years) without evidence of neoplastic disease. The dog's breeds represented in both case and control populations were similar. Clinical characteristics, namely age at diagnosis, dog weight, and number of tumors were obtained from medical records.
Peripheral blood samples from all animals were obtained using a standard venipuncture technique and collected in EDTA-containing tubes.
Mammary tumors were obtained during surgery and fixed in 10% buffered formalin. The material was dehydrated, embedded in paraffin wax, cut in 3-μm thick sections, and stained with hematoxylin and eosin. Tumors were classified histologically according to the diagnostic criteria proposed by the World Health Organization classification for mammary tumors of the dog and cat. 25 In bitches with multiple mammary tumors, histologic classification was based upon the lesion associated with the poorest prognosis. 25
Genomic DNA was extracted from white blood cell fraction using a High Pure PCR Template Preparation Kit (Roche, Mannheim, Germany), according to the instructions of the manufacturer. The COMT G482A polymorphism was analyzed through polymerase chain reaction (PCR) followed by restriction fragment length polymorphism (RFLP), based on the protocol previously described by Masuda et al. 21 DNA was amplified in a 50-μl reaction mixture containing 2-μl of genomic DNA, 0.3 μM of each COMT G482A primers (F, 5′-TCCACTTACTCATCCAGCTGG-3′ and R, 5′-TCGAGGCCATCCACTCTCTTG-3′), 0.2 mM of deoxynucleotide triphosphates, 1× Taq buffer, 1.5 mM of MgCl2 and 1 U of Taq polymerase. The amplification was performed in a thermocycler (Biometra, Goettingen, Germany), and PCR steps consisted of initial denaturation at 95°C for 5 minutes, 35 cycles at 95°C for 30 seconds, 53°C for 30 seconds, and 72°C for 1 minute, with a final extension step at 72°C for 5 minutes.
The PCR products were then digested overnight at 37°C with 1 U of BfmI (Fermentas, Burlinton, Canada), a restriction enzyme that recognizes the sequence 5′…C↓T Pu Py AG…3′. The restriction fragments were elecrophoresed in a 1.5% agarose gel with ethidium bromide staining. Analysis of the results was independently performed by 2 of the authors.
Statistical analysis of data was performed using the computer software SPSS for Windows (version 7.0; SPSS Inc., Chicago, IL). Chi-square analysis, with a 5% level of significance, was used to compare categorical variables. The COMT G482A genotypes were associated with age at diagnosis, dog weight (<20 kg vs. ≥20 kg), number of tumors (1–2 vs. >2), histologic type (benign vs. malignant), and lymph node metastasis (presence vs. absence), when data were available. The odds ratio (OR) and its 95% confidence interval (CI) were calculated as a measure of the association between COMT G482A genotypes and the clinicopathologic variables already mentioned. In a subsequent analysis we considered the basic question, “For a newborn bitch, what is the probability that it will experience onset of malignant disease before the age of
Results
The distribution of the COMT G482A polymorphism genotypes (GG, AG, and AA) and the respective allele frequencies in the control group and in the group of animals with mammary neoplasia are displayed in Table 1.
Genotypic and allele frequencies of the polymorphism COMT G482A in control cases and mammary tumor groups.
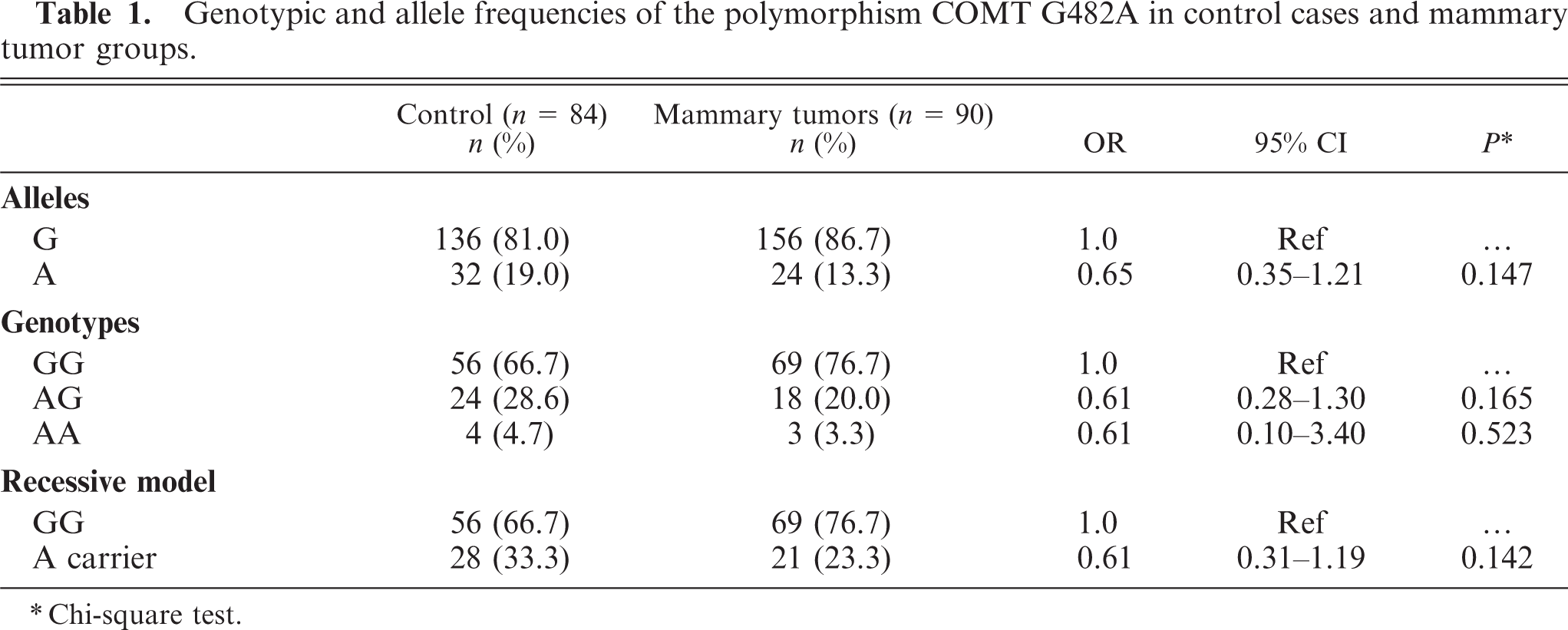
Chi-square test.
In the control group, the frequency of G allele was 81.0%, and the frequency of the allele A was 19.0%, while in the group of animals with mammary tumor, the frequency of the allele G was 86.7%, and the frequency of the A allele was 13.3%. The difference found in the frequency of A carrier and noncarrier genotypes between controls and cases has no statistical significance (
Table 2 represents the results regarding the association of COMT G482A polymorphism with some clinicopathologic parameters among bitches with mammary lesion, namely age, dog weight, number of tumors, histologic type, and lymph node metastasis.
Relationship between COMT G482A polymorphism and clinicopathologic parameters among bitches with mammary tumors.
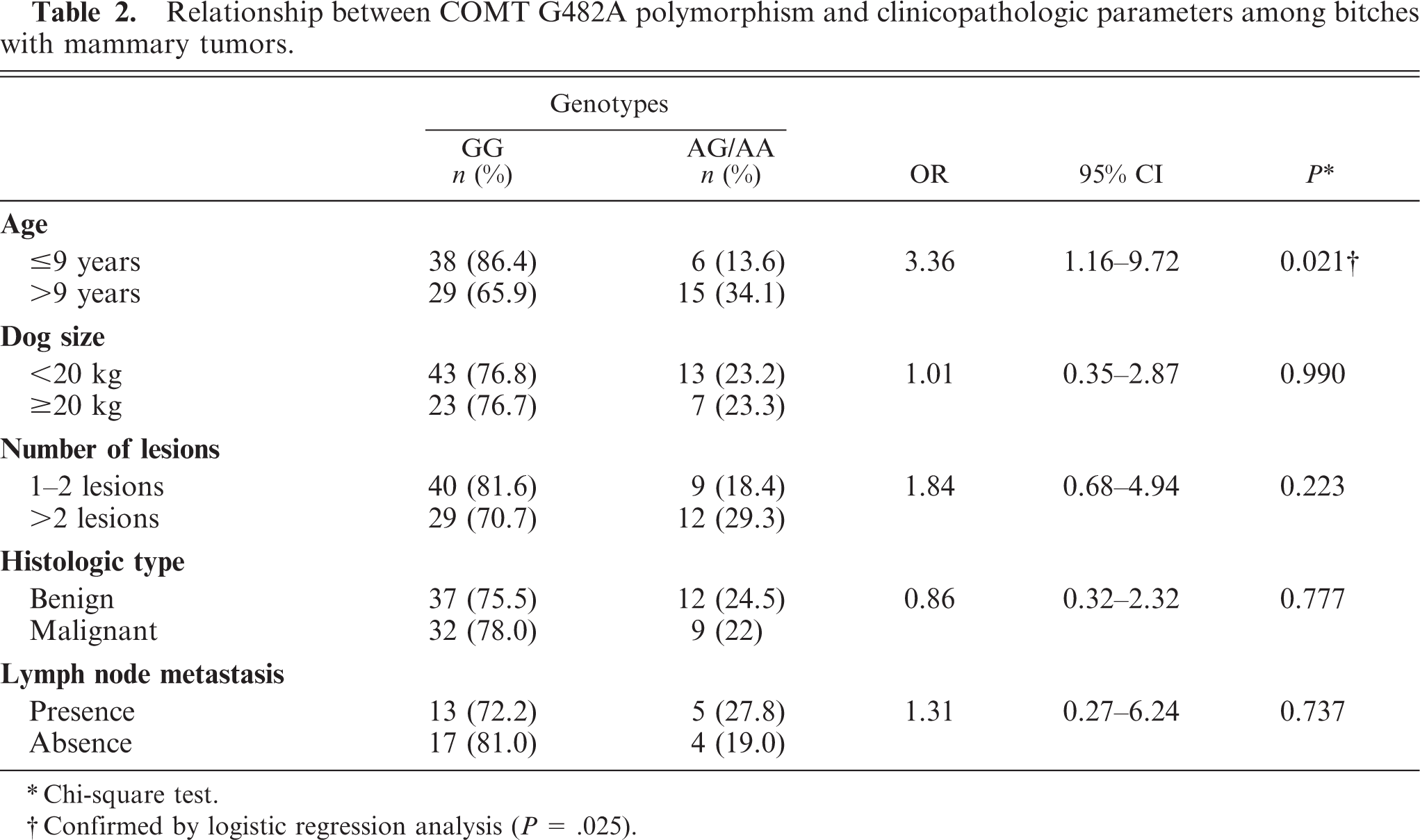
Chi-square test.
Confirmed by logistic regression analysis (
The mean age of cases included in this study was 9.5 years. There was a highly significant statistical difference (
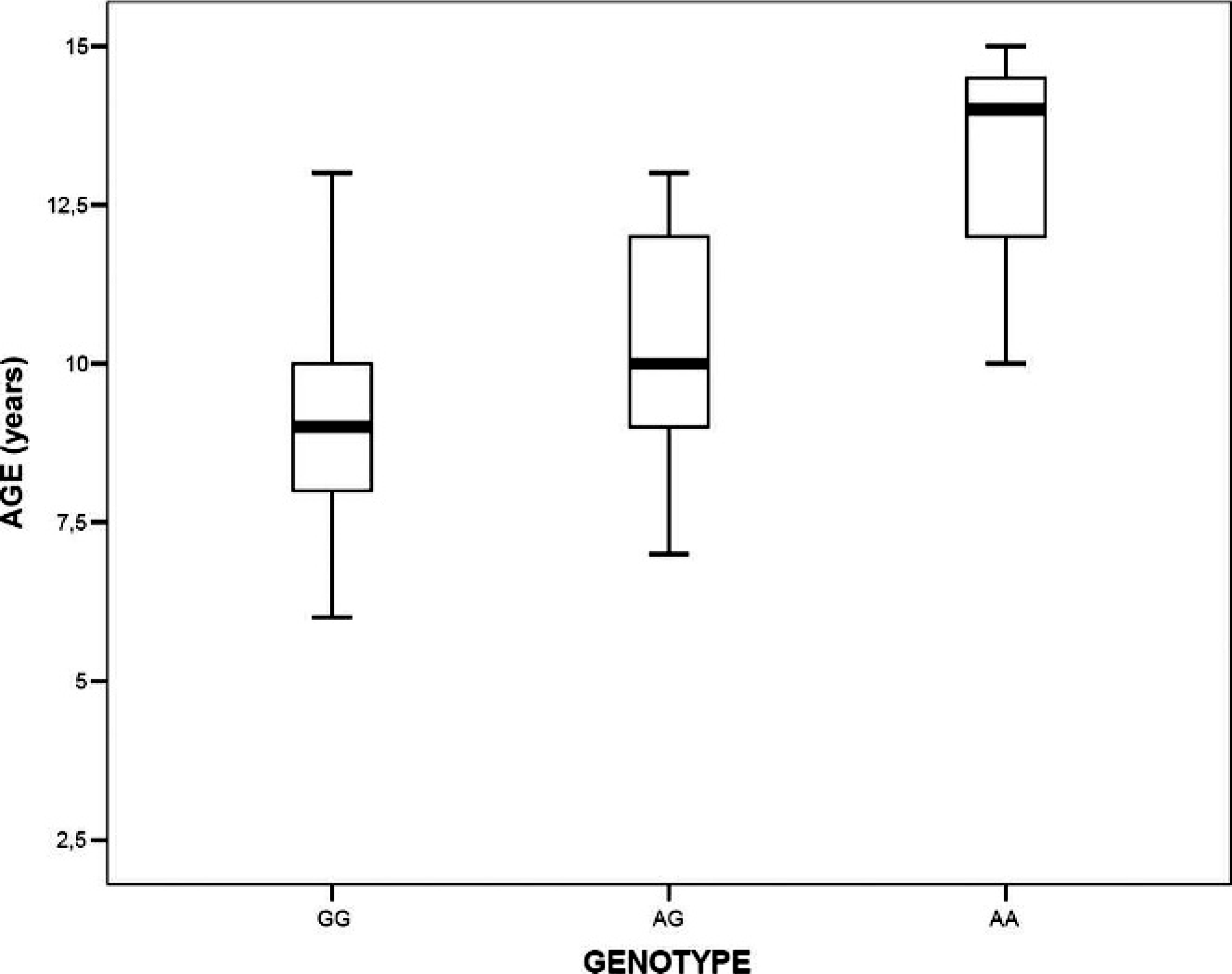
Relationship between COMT genotypes and age of animals with mammary tumor, showing a significant difference in age between dogs carrying genotypes GG, AG, and AA (
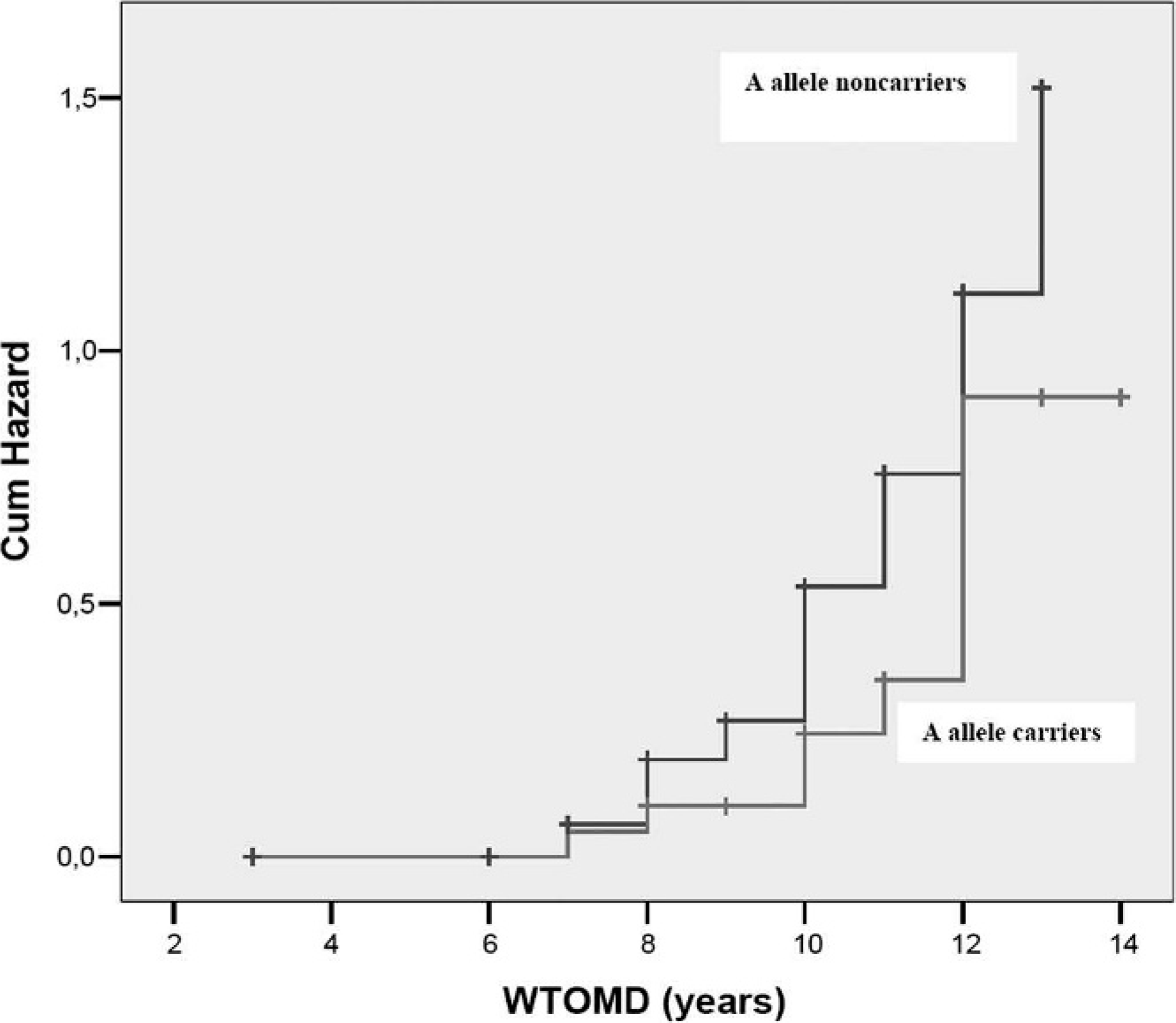
Association of COMT G482A polymorphism and the waiting time to onset of malignant disease, revealing a significant difference in the waiting time for onset of malignant disease for A allele carrier (12.46 years) and noncarrier (11.13 years) animals. Cumulative hazard function plots by the Kaplan-Meier methodology and log rank test (
Grouping animals bearing mammary tumors according to the dog's weight, we found that GG genotype was present in 76.8% of dogs under 20 kg and in 76.7% of those over 20 kg (Table 2). The group of A allele carriers constituted 23.2% and 23.3% of the dogs under and over 20 kg of body weight, respectively. We did not find significant associations between COMT genotype and dog weight (
We verified that genotype GG was present in 81.6% of the animals with 1 or 2 mammary lesions, and in 70.7% of dogs with more than 2 tumors (Table 2). A allele carriers comprised 18.4% of animals with 1 or 2 tumoral masses and 29.3% of those with more than 2 mammary lesions. There was no statistically significant difference between these groups (
Concerning histologic classification of the mammary lesions, we verified that the genotype GG included 37 benign tumors and 32 malignant tumors, while A allele carriers comprised 12 benign and 9 malignant neoplasms (Table 2). No statistically significant differences were observed between these groups (
A surprisingly high percentage of metastatic mammary carcinomas (46.1% of all malignant tumors) was found. The genotype GG was present in 81% of mammary carcinomas free of nodal metastasis and in 76.5% of cases with ganglionar metastasis; in the group of A allele carriers, 23.5% of carcinomas metastized, while 19% did not (Table 2). However, no significant association was found between A allele carriers and presence of lymph node metastasis (
Discussion
There is a strong body of evidence that identifies estrogens as potent carcinogenic compounds, by stimulating cell growth and by acting as precursors for the production of genotoxic metabolites. 1, 5, 39 Furthermore it has been suggested that genetic polymorphisms may influence the susceptibility to breast cancer. 30, 36 COMT is one of several enzymes that participate in inactivation of estrogen genotoxic metabolites. 1, 2 Some investigations in human breast carcinogenesis have recognized an association between a single nucleotide polymorphism in COMT gene (COMT val158met) and an increased risk for developing breast cancer. 3, 13, 17
In dogs there is a known single nucleotide polymorphism in the COMT gene, consisting of a G→A substitution at the 482nd nucleotide, but its relation with mammary carcinogenesis has never been investigated. Therefore, the aim of this study focused on the evaluation of the importance of the polymorphism COMT G482A in the development of mammary tumors in canine species.
The COMT G482A polymorphism was first described in dogs by Masuda et al. 21 who reported a frequency of 90.2% for the G allele and 9.8% for the A allele in a group of 266 animals from 5 dog breeds. We did not find significant differences between the frequency of COMT G482A polymorphism in the control group and in the group of bitches with mammary tumor, revealing that this polymorphism does not seem to play a major role in canine mammary tumor risk overall. Our results are in agreement with data previously reported in some similar human case-control studies. In fact, several epidemiologic investigations have been conducted during recent years in order to evaluate the relationship between COMT genotype and breast cancer risk. However, results from such studies are contradictory, as in spite of the association reported by some authors, 3, 13, 17 others did not find a significant relationship between both parameters. 7, 18, 38 COMT genotype alone does not seem to constitute a determinant factor in canine mammary tumorigenesis, but it may be of importance in conjunction with other relevant genetic background and/or environmental exposures. In fact, some investigators have suggested that the COMT genotype may influence breast cancer risk by affecting levels of exposure to other risk factors. 10, 18, 28 Actually, being a low penetrance gene, COMT action is thought to be dependent on gene–gene or gene–environmental interactions. 4, 15
Despite the fact that we did not find an association between COMT G482A polymorphism and mammary tumor risk overall, a statistically significant relationship was observed when considering the animal's age at the time the mammary tumor was observed. There was a significant difference in age between dogs carrying genotypes GG, AG, and AA (
Our results are in agreement with the hypothesis that estrogens are involved in the development of mammary neoplasms. Recent investigations have reported significantly higher levels of serum steroid hormones in bitches with mammary tumors in comparison with normal dogs. 12, 31 Therefore, we hypothesize that differences in COMT catalytic activity, related to G482A polymorphism, are important determinants of the age of onset of canine mammary tumors in the presence of abnormally high levels of steroid hormones.
An uncommonly, though not unprecedented, high number of metastatic carcinomas (46.1%) emerges from the group of specimens included in this investigation. In a previous study, Misdorp and Hart 26 also reported the involvement of lymph nodes in nearly 59% of dogs with mammary carcinomas treated by block-dissection. Our finding may be explained in part by the fact that most of the animals were periodically re-examined every 3 months, thus improving the chances to detect lymph node metastasis. Besides, in the majority of animals from this investigation, local lymph nodes were surgically removed in a systematically fashion and submitted to histologic examination. Also, because a clinically normal lymph node is not necessarily free of metastasis, and in order to avoid false-negative cases, only specimens including both mammary and local/regional lymph nodes were taken into account in evaluating this parameter. Furthermore, the use of cytokeratin immunostaining allowed a more accurate identification of lymph node metastasis from epithelial tumors. In fact, some of the authors have previously reported that immunohistochemical tests using antibodies to pancytokeratin and cytokeratin 14 allowed the detection of occult micrometastasis in 9.2% of a series of 131 lymph nodes classified as free of metastasis by hematoxylin and eosin staining. 22
Our conclusions point to the combined effect of COMT genotype with other genetic and/or environmental risk factors as important key factors for mammary tumor etiopathogenesis, which is consistent with the multifactorial model that has been proposed for mammary tumor development. 9, 24, 27 However, in spite of the statistical significance of our results, they should be considered as preliminary. Further extensive studies, including a larger number of animals, in which controls and cases should be perfectly matched for age and breed, would be very important in order to confirm our findings and to clarify the influence of the COMT genotype in mammary tumor risk either alone or in combination with other risk factors.
Footnotes
Acknowledgements
We gratefully acknowledge partially funding of this work by the Ministry of Health of Portugal (Comissão de Fomento da Investigação em Cuidados de Saúde: CFICS-226/01), AstraZeneca Foundation, and Calouste Gulbenkian Foundation. The authors would like to thank the Portuguese League Against Cancer (Liga Portuguesa Contra o Cancro – Centro Regional do Norte) for its support.
