Abstract
Prolactin-secreting pituitary adenomas are common spontaneous lesions in aging FVB females. Prolactin-secreting pituitary proliferations play a significant role in mouse mammary tumorigenesis generally producing adenosquamous carcinomas. Since genetically engineered FVB mice are frequently used to study mammary tumor biology, we have examined a cohort of 64 aging wild-type FVB/N females to establish the prevalence and the nature of spontaneous mammary and pituitary tumors. Tissues from mammary and pituitary glands were studied by histopathology and immunohistochemistry. Of the 64 examined mice, 20 had pituitary tumors and 20 had mammary tumors. Mammary and pituitary tumors were associated in 17 mice. All pituitary tumors were prolactin-positive by immunohistochemistry and classified as prolactinomas. Fourteen mammary tumors, including 12 cases with and 2 without concurrent prolactinomas, were adenocarcinomas with different combinations of epithelial growth patterns. Five mice with prolactinomas had mammary tumors characterized by the epithelial-mesenchymal transition (EMT) phenotype. Estrogen receptor alpha (ERα)-positivity was observed for 14 of the 18 mammary tumors tested, including both adenocarcinomas with nuclear immunoreactivity and EMT-phenotype tumors with both nuclear and cytoplasmic immunoreactivity. No immunoreactivity for the progesterone receptor was observed. This study confirms that spontaneous prolactinomas and mammary tumors are both common and significantly associated lesions in FVB mice. Parity and age represented risk factors for the development of these tumors. Compared with previous reports, prolactinoma-associated mammary tumors displayed a broader morphologic spectrum, including cases with the EMT phenotype. The elevated number of prolactinoma-associated and ERα-positive mammary tumors opens intriguing possibilities concerning the role of ERα cytoplasmic localization during EMT tumorigenesis.
Keywords
Introduction
The FVB strain of mice is widely employed for the generation of transgenic mouse models. Advantages in the use of this specific strain include inbred status, high fertility, large size of the pronuclei in fertilized zygotes that facilitates injection of the transgene, and good survival of embryos following injection. 2 A preliminary goal of transgenic mouse model characterization is to assess whether or not the pathologic findings observed represent a direct consequence of the experimental genetic modifications. 5 It is, therefore, crucial to know the baseline pathology of the mouse strain employed, including the spectrum of spontaneous lesions, the extent of interanimal variation, and, hopefully, the causal factors for the variability. 3,17,25
Numerous FVB-based transgenic models have been validated for the study of mammary cancer biology. 4 During the characterization of these models, the contextual examination of appropriate matched control wild-type animals clearly demonstrated that FVB mice spontaneously acquire a series of mammary developmental, inflammatory, and proliferative lesions, potentially leading to misinterpretation of transgene-induced mammary phenotype. 4 The influence of several factors, including specific substrain, age, environment, diet, parity, pups suckling activity, murine mammary tumor virus (MMTV) infection, and concurrent diseases, proved to play a role in the development of this multifaceted scenario of spontaneous mammary lesions. 12,29,36
Previous research clearly established that prolactin stimulation is implicated in mammary tumor initiation and promotion in rodents. 16,20,34,37 Recent surveys demonstrated that the spontaneous prolactin-secreting proliferative lesions of the pituitary gland frequently seen in wild-type aging female FVB mice are commonly correlated with naturally occurring mammary abnormalities. These latter predominately consisted of foci of mammary hyperplasia with squamous metaplasia and mammary carcinomas with adenosquamous differentiation. 23,36 A similar mammary lesion spectrum was also recorded in BALB/c female mice with ectopic prolactin-secreting adenomatous pituitary grafts under the renal capsule. 9,19
However, pathologic data regarding the pituitary gland in age-matched wild-type controls are seldom reported by investigators, leading to a prevailing opinion that not all colonies develop pituitary abnormalities. Because genetically engineered FVB mice are frequently used for the development of transgenic models of breast cancer, the blind use of “historical,” but not age-matched, controls is a dangerous and perhaps misleading practice. Therefore, we carefully studied a cohort of 64 aging wild-type FVB/N females to establish the prevalence and the nature of spontaneous mammary and pituitary tumors, as well as the possible correlation between these lesions.
Material and Methods
Animal breeding and tissue dissection
Cases examined in this retrospective study were selected from the Mutant Mouse Pathology Laboratory Archive of the Center for Comparative Medicine, University of California, Davis (UCD). The reference population included a total of 64 wild-type, 74.3–114.4 (mean 86.2 ± 11.2)-week-old, both nulliparous and multiparous, female, FVB/N mice used as age-matched controls during the characterization of different transgenic mouse models. The animals belonged to a single at-risk cohort from the FVB/N colony at the University of Connecticut. FVB/N mice were obtained from Taconic (Germantown, NY). Animals were housed in plastic cages on newspaper bedding and fed with standard chow and tap water ad libitum. Mice were maintained in HEPA-filtered Thoren units (Thoren Caging Systems, Hazleton, PA). Mice were sacrificed by CO2 asphyxiation followed by cervical dislocation. Mammary glands and pituitaries were removed and fixed in 4% formalin for 24 h, then placed in 70% ETOH and shipped to UCD Mutant Mouse Pathology Laboratory for histopathologic examination.
Animals were bred, housed, and used in accordance with the Policy on Humane Care and Use of Laboratory Animals (Office of Laboratory Animal Welfare, National Institutes of Health).
Case selection, histopathology, and immunohistochemistry
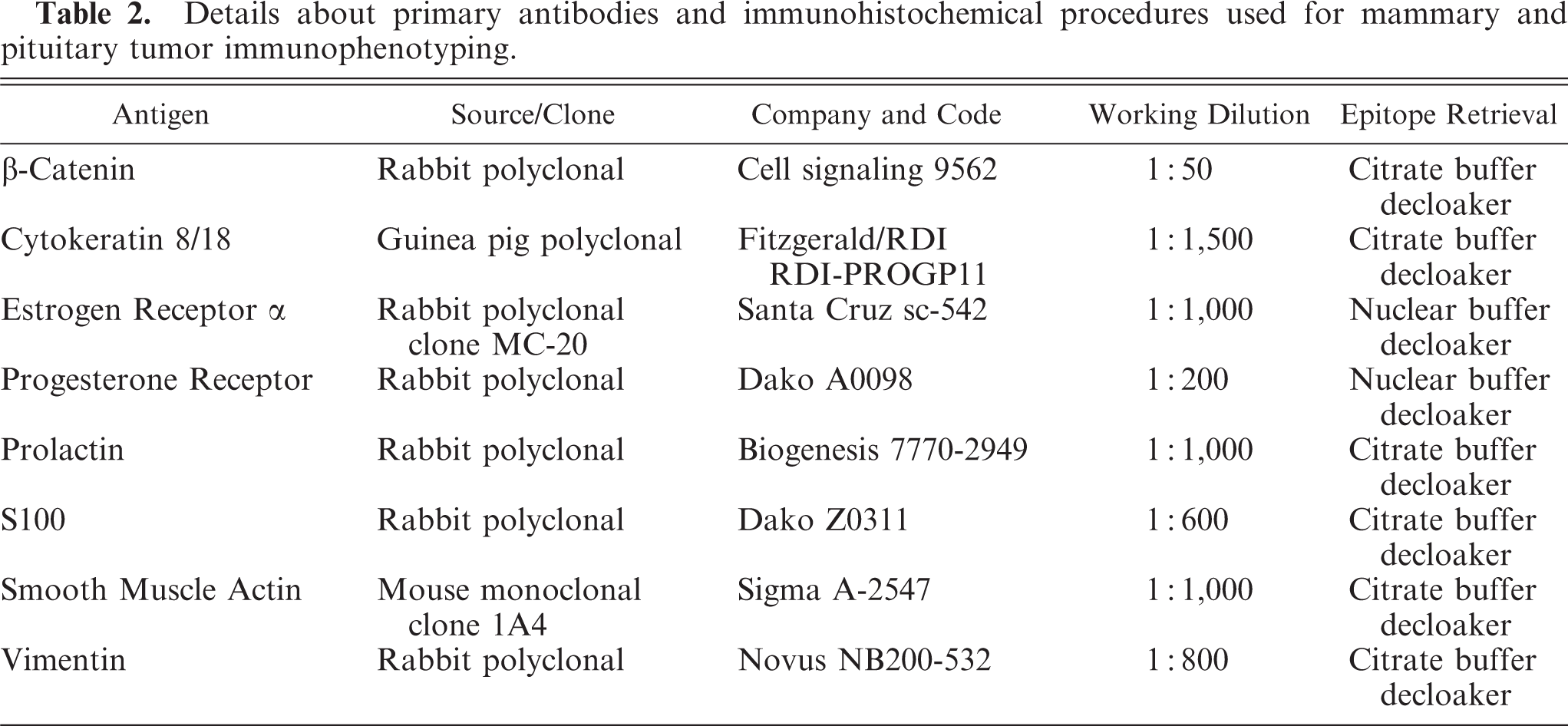
For all the 64 mice considered in this study, detailed mammary and pituitary gland pathologic records were available in the Mutant Mouse Pathology Laboratory Archive (Table 1). Cases were reviewed, and a total of 23 mice out of 64 were selected for further histopathologic and immunohistochemical characterization on the basis of the development of spontaneous mammary, pituitary, or both kinds of tumors (Table 1). Histologic and immunohistochemical methods were performed as previously described. 24 Details concerning primary antibodies and procedures for immunohistochemical stainings are reported in Table 2. Mammary proliferative lesions were classified according to the criteria dictated by the Annapolis Pathology Panel. 4 As reported by subsequent studies, minimal modifications regarding the epithelial-mesenchymal transition (EMT) phenotype definition were introduced in this classification. 11,26 The EMT-phenotype tumor was considered morphologically significant in the presence of epithelial structures displaying loss and disruption of basement membrane and associated with epithelial-to-spindle cell morphology transition and gradual blending or invasion into the surrounding stroma.
Studied cohort of aging female FVB/N mice: comparative evaluation of mammary and pituitary pathologic findings and data concerning parity status and age.
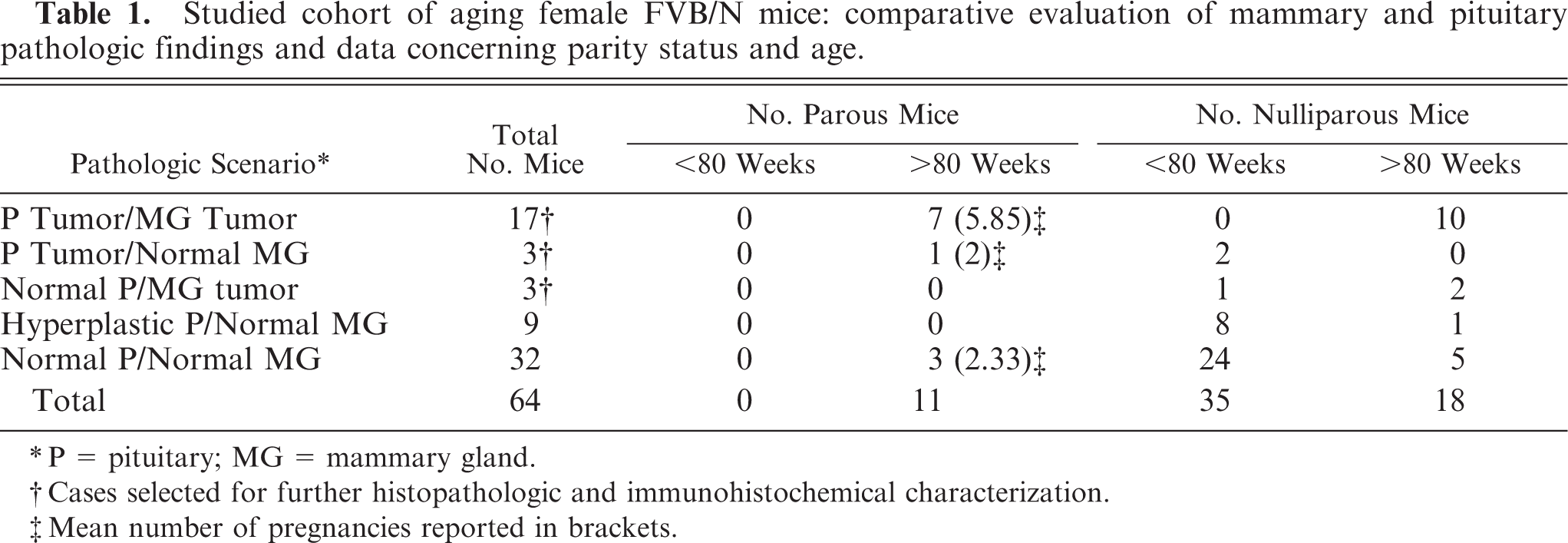
∗P = pituitary; MG = mammary gland.
† Cases selected for further histopathologic and immunohistochemical characterization.
‡Mean number of pregnancies reported in brackets.
Details about primary antibodies and immunohistochemical procedures used for mammary and pituitary tumor immunophenotyping.
Statistical analysis
The prevalence of mammary, pituitary, or both kinds of tumors between different groups of animals was compared by chi-square test. Mean differences in the number of pregnancies were tested by 1-way ANOVA. 33
Results
A comparative overview of the mammary and pituitary gland pathologic findings from the examined cohort is summarized in Table 1. Spontaneous pituitary tumors of the pars distalis were observed in 20 out of the 64 mice (32%). Pituitary hyperplasia of the pars distalis was detected in 9 out of 64 mice (14%). Normal pituitary gland was reported in 35 out of 64 mice (54%). Spontaneous mammary tumors were observed in 20 out of 64 mice (32%). Normal mammary gland was reported in 44 out of 64 mice (68%). This latter group also included cases characterized by minimal nonneoplastic changes such as functional (presence of secretory activity) mammary lobuloalveolar hyperplasia without foci of atypia, segmental ductal ectasia and squamo-inflammatory nodule formation. Out of the 44 mice with normal mammary gland, 32 presented normal pituitary gland, 9 showed pituitary hyperplasia of the pars distalis, and 3 had pituitary tumor of the pars distalis. Out of the 20 mice with mammary tumor, 17 had concurrent pituitary tumor of the pars distalis and 3 presented normal pituitary gland. The prevalence of spontaneous mammary tumors was significantly higher (χ2 = 39.12, P < .0001) in mice with concurrent pituitary tumor (17/20, 85%) than in mice without pituitary tumor (3/44, 7%; Table 1). The prevalence of concomitant mammary and pituitary tumors within the group of parous mice (all aged more than 80 weeks) was significantly affected by the number of pregnancies (Table 1). In fact, among the 11 parous mice included in this study, those with concomitant mammary and pituitary tumors never had fewer than 4 pregnancies, and the mean (±SD) number of pregnancies was significantly higher in these animals than in the others (5.86 ± 0.50 compared with 2.25 ± 0.25; F = 25.68, P < .01). For nulliparous mice, the prevalence of both mammary (χ2 = 26.14, P < .0001) and pituitary (χ2 = 16.86, P < .0001) tumors was significantly affected by age, with only 3 of 35 females displaying neoplastic lesions (2 pituitary adenomas and 1 mammary tumor) before the 80 weeks, and 12 of 18 after that age (10 concurrent mammary and pituitary tumors and 2 mammary tumors; Table 1).
The results of mammary tumor histopathologic examination and classification on the basis of criteria defined by the Annapolis Pathology Panel 4 are summarized in Table 3. Often, more than one histologic pattern was observed in a single mammary tumor. Therefore, the predominant morphologic patterns were considered for classification purposes. Different degrees of invasion into the surrounding fat pad were detectable in most of the mammary tumors. Variable-sized intratumoral areas of necrosis often accompanied by desmoplastic reaction and massive infiltration of inflammatory cells (mostly neutrophils and reactive macrophages) were also frequently observed. In 7 cases, neoplastic emboli lodging in the stromal blood vessels were observed. However, neither regional lymph nodes nor lung were available to assess the presence of metastases.
Aging female FVB mice with spontaneous mammary tumors: data concerning signalment, pituitary pathologic examination, mammary tumor classification, and the relative estrogen receptor (ER) α/progesterone receptor (PR) status.∗
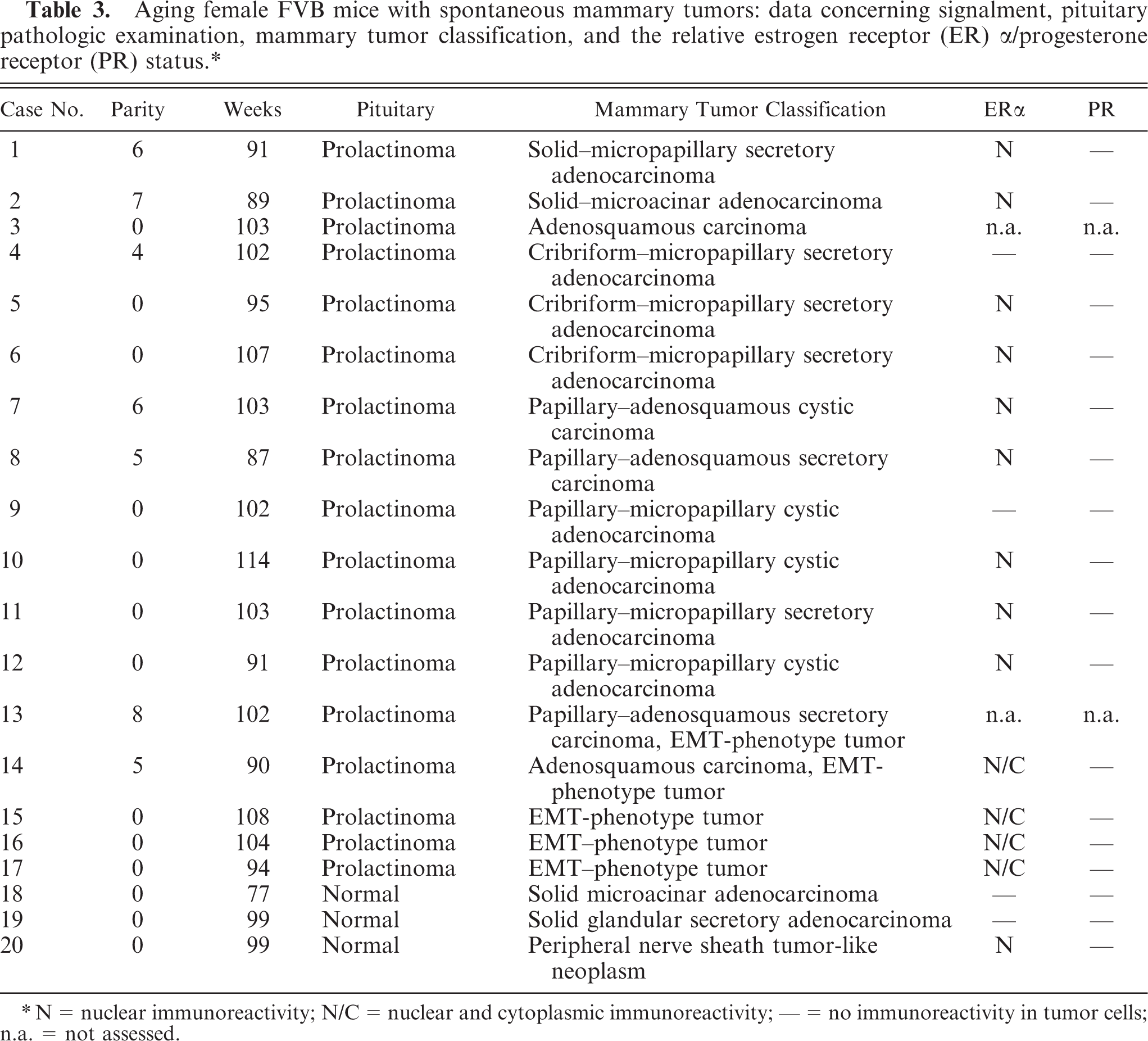
∗N = nuclear immunoreactivity; N/C = nuclear and cytoplasmic immunoreactivity; – = no immunoreactivity in tumor cells; n.a. = not assessed.
Fourteen mammary tumors, including 12 cases with and 2 without pituitary tumors, were adenocarcinomas with different combinations of epithelial growth patterns (Table 3, Nos. 1–12, 18, 19; Fig. 1). Five mice with pituitary tumors had mammary masses primarily consistent with EMT-phenotype tumors (Table 3, Nos. 13–17; Fig. 2). Compared with mammary adenocarcinomas, which tend to be expansile, the EMT-phenotype tumors displayed a greater degree of local aggressiveness, with dissection through adjacent fascia and skeletal muscle. In these latter tumors, the ongoing EMT process was confirmed by different degrees of cytokeratin (CK) 8/18 (Fig. 3a), vimentin (Fig. 3b), and to a lesser extent, smooth muscle actin (SMA) colocalized immunohistochemical expression. A further confirmation of the ongoing EMT process resulted from the marked attenuation of β-catenin immunostaining associated with the gradual epithelial-to-spindle cell morphology transition. A female with normal pituitary had a mammary neoplasm morphologically consistent with a peripheral nerve sheath tumor (PNST) (Table 3, No. 20). This neoplasm displayed a vimentin-positive, SMA-negative, CK 8/18–negative, and S100-negative immunophenotype. Even if neoplastic cell results were invariably negative, S100 staining revealed that tumoral growth was often centered on residual nerve fascicles, individual groups of degenerated axons, or both. By immunohistochemistry, those mammary tumors displaying consistent squamous differentiation (Table 3, Nos. 3, 7, 8, 13, 14) also showed marked cytoplasmic accumulation and nuclear translocation of β-catenin signal.
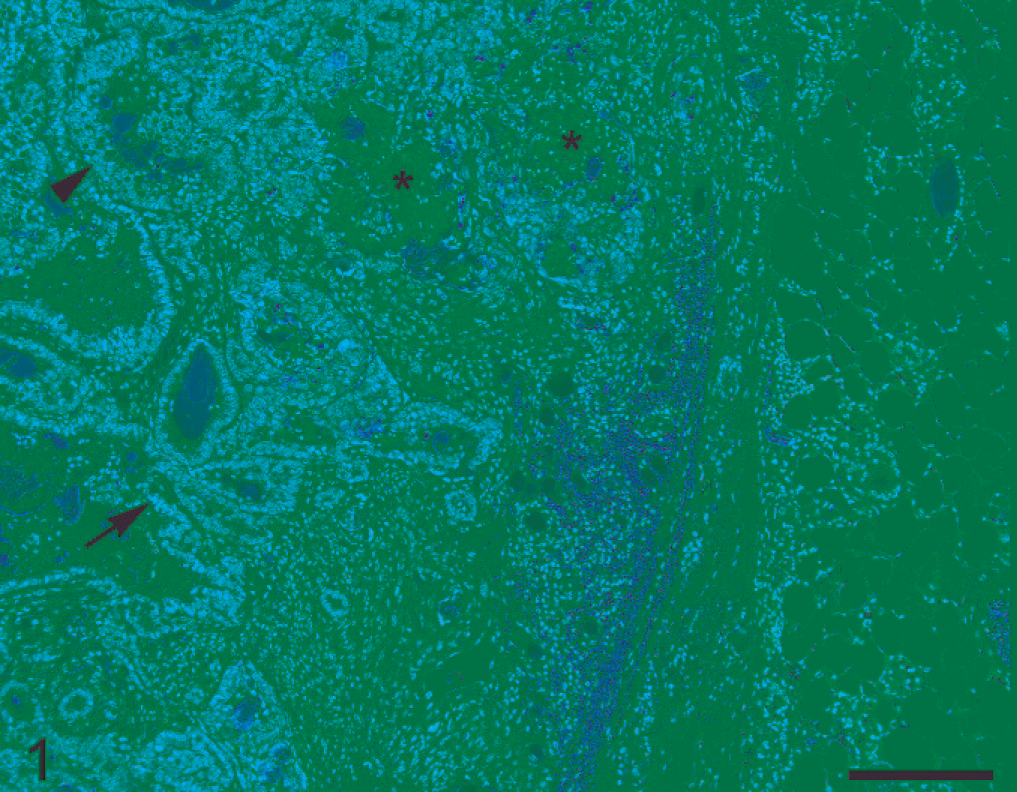
Mammary gland; female FVB mouse No. 8. Mammary adenocarcinomas with different combinations of epithelial growth patterns. Multiple foci of squamous differentiation (asterisks) admixed with papillary glandular units (arrow) showing focal secretory activity (arrowhead). HE. Bar = 200 µm.
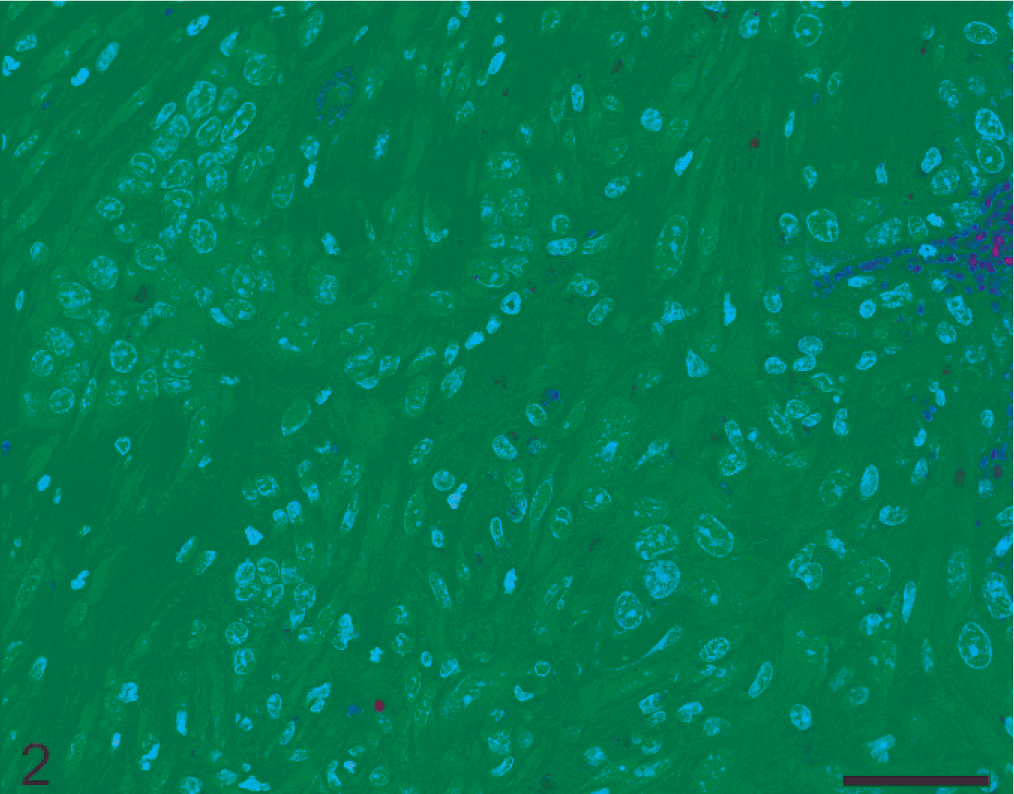
Mammary gland; female FVB mouse No. 13. Mammary tumor characterized by epithelial to mesenchymal transition (EMT) phenotype. Solid epithelial trabeculae displaying a transitional epithelial-mesenchymal cell phenotype with loss of tumor cell cohesiveness and invasion into the surrounding collagenous stroma. HE. Bar = 50 µm.
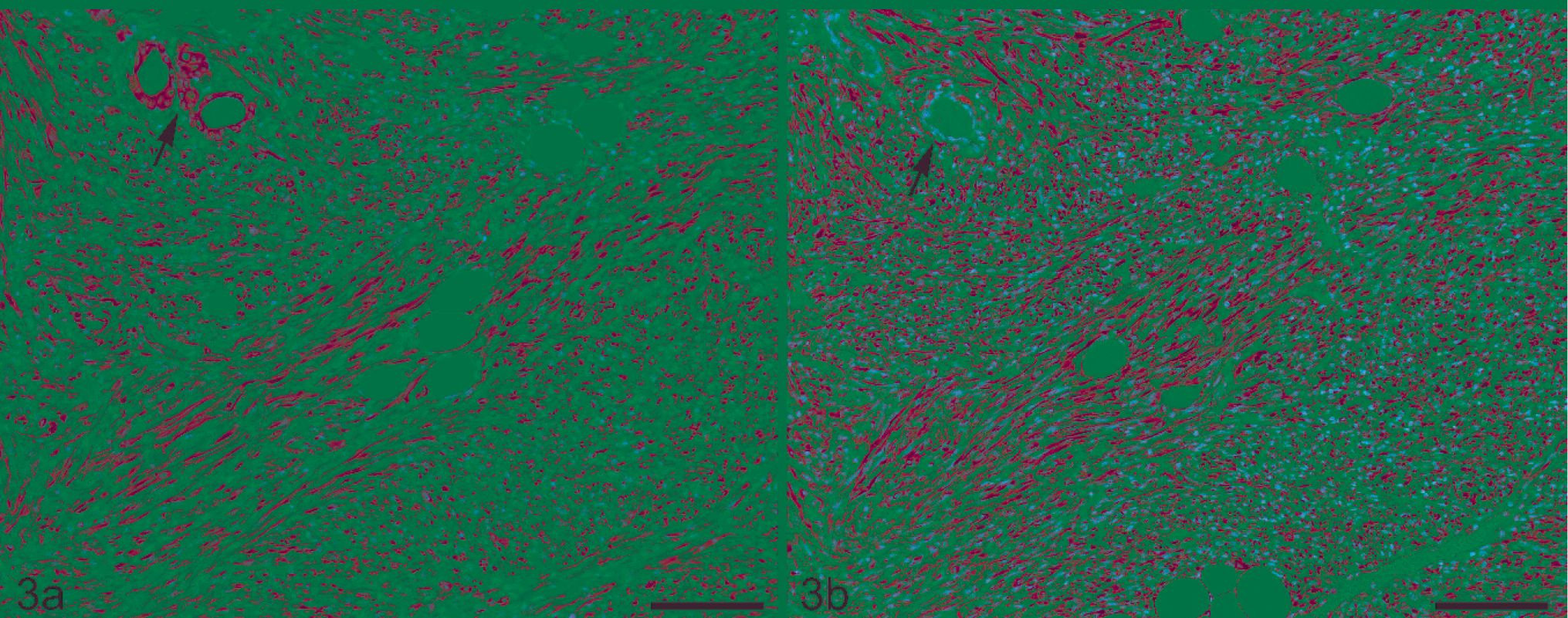
Mammary gland; female FVB mouse No. 14. Fig 3a. Mammary tumor characterized by epithelial to mesenchymal transition (EMT) phenotype. Diffuse reactivity for cytokeratin (CK) 8/18 in both glandular units (arrow) and surrounding bundles of spindle-shaped cells. Immunohistochemistry for CK 8/18, Mayer's hematoxylin counterstain. Bar = 100 µm. Fig. 3b. Same microscopic field depicted in Fig. 3a. The bundles of spindle-shaped cells also express vimentin. Note the vimentin-negative (CK 8/18–positive in Fig. 3a) glandular units (arrow). Immunohistochemistry for vimentin, Mayer's hematoxylin counterstain. Bar = 100 µm.
Immunostaining for estrogen receptor alpha (ERα) and progesterone receptor (PR) was performed only on 18 of the 20 mammary tumors because in 2 cases, no residual neoplastic tissue was available from the paraffin blocks (Table 3, Nos. 3, 13). ERα expression was observed in 14 of the 18 examined tumors (77.8%) (Table 3, Nos. 1, 2, 5–8, 10–12, 14–17, 20). Variable degrees of ERα nuclear expression were recorded for 9 pituitary tumor–associated mammary adenocarcinomas (Table 3, Nos. 1, 2, 5–8, 10–12; Fig. 4), as well as for the PNST-like neoplasm (Table 3, No. 20). For the 4 pituitary neoplasm–associated mammary EMT-phenotype tumors investigated (Table 3, Nos. 14–17), ERα expression in the spindle cell component consisted of marked cytoplasmic (often paranuclear) immunoreactivity, occasionally associated with a less intense nuclear signal (Fig. 5). In EMT-phenotype tumors, marked ERα nuclear expression without any cytoplasmic signal was observed only in those scattered neoplastic structures that still preserved a clear glandular pattern of growth (Fig. 5). A total of 4 mammary adenocarcinomas, 2 with and 2 without concurrent pituitary tumor, were ERα-negative (Table 3, Nos. 4, 9, 18, 19). No immunoreactivity for PR was observed in any of the mammary tumors.
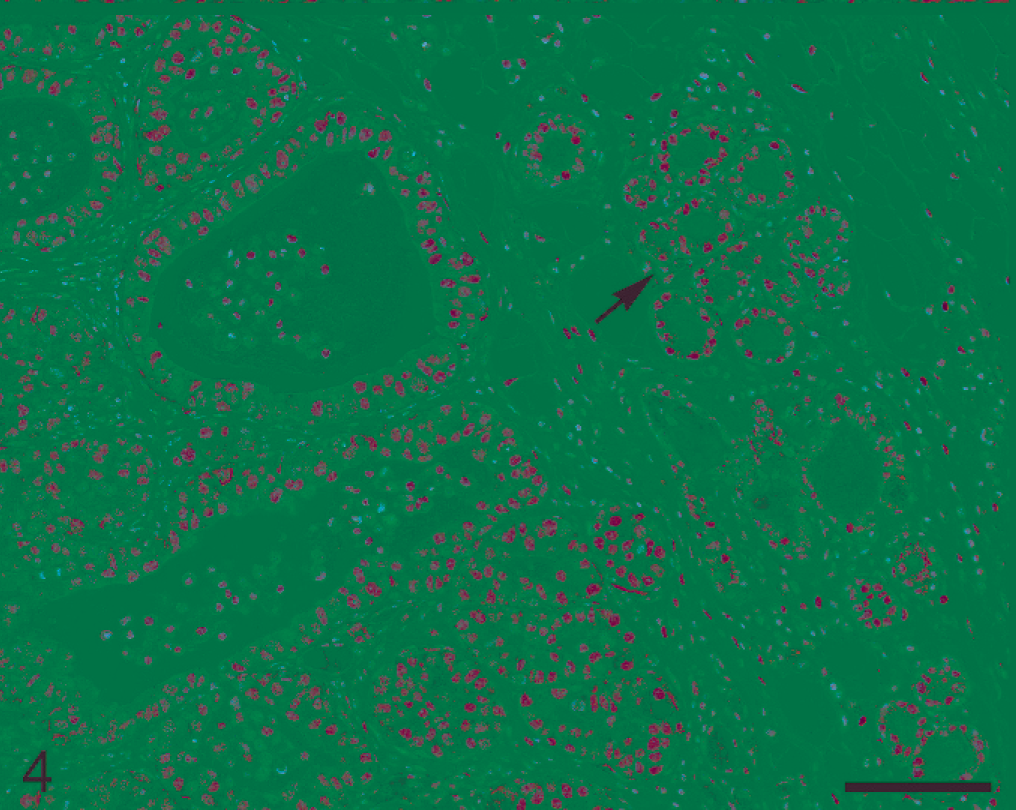
Mammary gland; female FVB mouse No. 7. Mammary adenocarcinoma characterized by both cystic and solid glandular units. Neoplastic epithelial cells show diffuse and marked ERα nuclear expression without any cytoplasmic signal. Nuclear ERα signal is also present in the normal peritumoral mammary lobules (arrow). Immunohistochemistry for ERα, Mayer's hematoxylin counterstain. Bar = 100 µm.
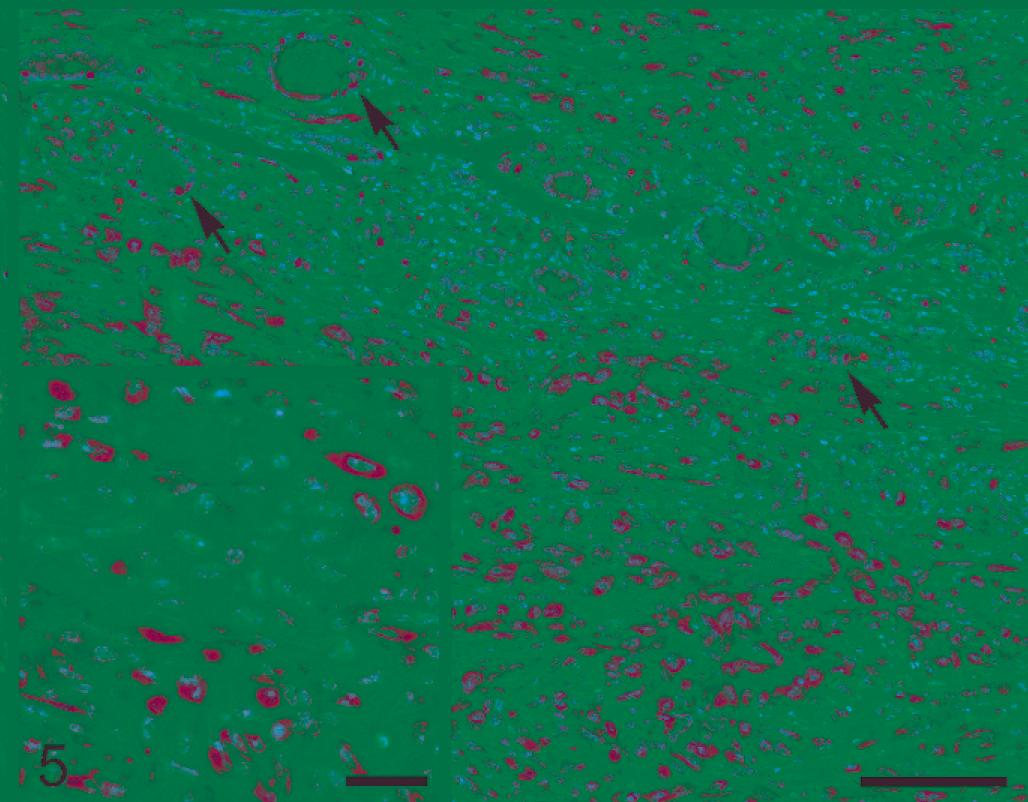
Mammary gland; female FVB mouse No. 14. Mammary tumor characterized by epithelial to mesenchymal transition (EMT) phenotype. Marked cytoplasmic, often paranuclear (inset) immunoreactivity for ERα in the spindle-shaped cell component. Scattered neoplastic cells still display a clear glandular pattern of growth and there is nuclear expression of ERα without any cytoplasmic signal (arrows). Immunohistochemistry for ERα, Mayer's hematoxylin counterstain. Bar = 100 µm. Inset bar = 25 µm.
All 20 pituitary tumors of the pars distalis resulted strongly and diffusely positive for prolactin immunohistochemical staining, leading to their classification as prolactinomas. Histologically, prolactinomas were characterized by multiple, solid, unencapsulated, expansile, micronodular masses effacing most of the pars distalis of the pituitary gland (Fig. 6). Tumor growth was also peculiarly characterized by disruption of the normal pituitary sinusoidal vasculostromal network (Fig. 6). Neoplastic units consisted of densely cellular, irregular sheets and cords of variable-sized, moderately atypical, round to polygonal cells, often containing discrete cytoplasmic accumulations of finely granular eosinophilic material (Fig. 7a). These latter were interpreted as clusters of secretory granules because they proved to be invariably positive for prolactin immunostaining (Fig. 7b).

Pituitary gland; female FVB mouse No. 2. Prolactinoma. The tumor consists of multiple densely cellular micronodular structures with disruption of normal pituitary sinusoidal vasculostromal network. Scattered intratumoral blood-filled lacunae are also evident (arrow). HE. Bar = 200 µm.

Pituitary gland; female FVB mouse. Fig. 7a. Mouse No. 2. Prolactinoma. Higher magnification of Fig. 6. The tumor is arranged in dense irregular sheets and cords of variably sized polygonal cells. Tumor cells display anisocytosis, anisokaryosis, multinucleation and discrete cytoplasmic accumulations of finely granular eosinophilic material (secretory granules) displacing the nucleus at the periphery. HE. Bar = 50 µm. Fig. 7b. Mouse No. 14. Prolactinoma. Same microscopic field depicted in Fig. 7a. Accumulating secretory granules in tumor cells show prominent cytoplasmic immunoreactivity for prolactin. Immunohistochemistry for prolactin, Mayer's hematoxylin counterstain. Bar = 50 µm.
Discussion
Consistent with previous reports, our study provides another large cohort of elderly FVB/N female mice with a high prevalence of prolactinomas associated with concurrent mammary tumors. 25,36 Previous investigations clearly demonstrated that prolactinomas in FVB mice typically represent hormonally active tumors associated with hyperprolactinemia. 29,36 Although data concerning circulating prolactin levels were not available in this study, the large amount of prolactin-positive secretory granules identified in all pituitary adenomas implies ongoing hormonal secretory activity with consequent mammary gland overstimulation. These observations confirm the evidence that, in laboratory mice, abnormal prolactin stimulation is associated with mammary tumorigenesis. 34,37
Data available concerning the concomitant occurrence of spontaneous pituitary and mammary proliferative lesions show great variability among the different strains, substrains, and cohorts of mice examined up to now. 6,7,13,25,29,36 The variability suggests that prolactin alone might not be sufficient to induce a mammary phenotype but that there might be a requirement for many other, as yet not completely characterized, genetic, environmental, metabolic, infectious, and pathophysiologic cooperating factors. 29,32,36 Previous studies pointed out the importance of parity in the generation of spontaneous pituitary and mammary proliferative lesions. In a recent survey, aging wild-type nulliparous FVB/NCr mice frequently developed prolactin-secreting pituitary hyperplasia associated, in most cases, with concurrent squamo-inflammatory nodules formation and functional mammary lobuloalveolar hyperplasia without foci of atypia. Conversely, multiparous FVB/NCr females from the same study displayed an elevated number of prolactinomas and concurrent mammary adenocarcinomas. 36 However, the tumor-bearing cohort was relatively small. Our study, with a relatively large cohort, clearly demonstrated that parity and number of pregnancies represented highly statistically significant risk factors for the concomitant development of pituitary and mammary tumors. Furthermore, in nulliparous females, statistical analyses demonstrated the importance of age for the development of both mammary and pituitary spontaneous tumors, with a very low occurrence in animals younger than 80 weeks. These observations indicate that in FVB/N-based models of breast cancer, the background of spontaneous mammary tumors might be drastically reduced with the use only of nulliparous females and by planning the experiment to conclude before the animals reach 80 weeks of age.
The most unexpected observation of our study was the wider range of mammary tumor phenotypes than previously recorded in the pituitary-associated tumors. Previous investigations indicated that FVB females with prolactinomas and prolactin overstimulation of the mammary gland typically produce mammary adenosquamous carcinomas. 9,19,23 If true in all cohorts, prolactin-related tumors could be readily identified and animals segregated. Importantly, these findings were not confirmed in this study. Indeed, not limited to mammary tumors with adenosquamous differentiation (Table 3, Nos. 3, 7, 8, 13, 14), the pituitary-associated mammary tumor exhibited a broader spectrum, including cases with the EMT phenotype (Table 3, Nos. 13–17) and adenocarcinomas (without relevant adenosquamous differentiation) characterized by complex combinations of epithelial patterns of growth (Table 3, Nos. 1, 2, 4–6, 9–12). This heterogeneous pathologic scenario of spontaneous mammary tumors could add further complexity to the morphologic interpretation of FVB-based transgenic mouse models of mammary carcinogenesis.
A study with a hormonally nonresponsive promoter, neu-related lipocalin (NRL) to drive prolactin (NRL-PRL) expression in mammary epithelia of FVB mice, reported a broad spectrum of mammary tumors, including adenocarcinomas with different epithelial patterns of growth and some “EMT-type” tumors, such as carcinosarcomas and fibroadenocarcinomas. 31 Furthermore, the NRL-PRL transgenic model showed an elevated number of ERα-positive mammary tumors. In a like manner, prolactinoma-bearing mice in our study displayed a high prevalence of ERα-positive mammary tumors (13/15, 87%), including all the EMT-phenotype tumors (Table 3, Nos. 14–17) and the majority of the adenocarcinomas tested (Table 3, Nos. 1, 2, 5–8, 10–12, 14–17). Taken together, these data represent empiric evidence that, in FVB/N mice, prolactin overstimulation could play a role in the development of ERα-positive mammary tumors with multiple morphologic patterns of growth.
ERα-positive mammary tumors are uncommon both in wild-type mice and in genetically engineered mouse models of breast cancer. 35 However, recent investigations underlined that prolactin downstream signaling increases ERα expression levels in breast cancer cells. 14 The molecular mechanisms that link prolactin and estrogens during mammary carcinogenesis are still largely unknown. It has been demonstrated that in breast cancer cells, prolactin and estrogens cooperatively activate the expression of many target genes that are critical for oncogenesis and neoplastic progression, including those influencing cell survival, proliferation, differentiation, angiogenesis, and invasion. 15
ERα-positive mammary tumors observed in our study did not express PR. ERα-positive breast tumors lacking PR expression represent a specific, but rare, clinicopathologic entity that does not respond to endocrine therapy. 18 In the normal mammary gland and in breast cancer, the transcription of the PR gene results from the genomic action of ERα signaling, with the classic ligand-induced ERα nuclear translocation and consequent activation of the estrogen response elements. 8 As a result, loss of PR in the course of malignant transformation and cancer progression reflects a nonfunctional nuclear expression of ERα. 1,16 In our study, nonfunctional ERα expression is therefore hypothesized for those PR-negative mammary adenocarcinomas with a typical ERα nuclear expression. This hypothesis will need to be biologically tested by transplantation of relevant tumors.
The development of EMT-type tumors associated with mammary tumorigenesis in wild-type FVB was another unexpected observation. The EMT-phenotype tumors included in this study had a surprising immunohistochemically detected cytoplasmic expression of ERα. The mouse mammary EMT tumors in all of our other EMT cohorts 11 also had cytoplasmic expression of ERα (P. Damonte and R. D. Cardiff, unpublished), suggesting that cytoplasmic ERα is closely related to the EMT phenotype.
The role of the cytoplasmic expression of ERα in EMT mammary carcinogenesis has not been studied. However, as suggested by recent studies, cytoplasmic redirection and sequestration of ERα in breast cancer cells, might account for the activation of alternative nongenomic biomolecular signals. 30,39 In this sense, a role in metastatic tumor antigen 1 (MTA1)–associated EMT tumor progression has been also proposed for those breast tumors with cytoplasmic subcellular localization of ERα. 22,27 Growing evidence clearly suggest that EMT represents, not only a fundamental process during embryogenesis, but also a mechanism peculiarly adopted by epithelial cells in the course of malignant transformation and tumor progression. This is also true for mammary carcinogenesis, wherein the gradual switch from an epithelial to a mesenchymal phenotype allows the transforming cells to carry out a series of molecular events that account for cell survival, as well as infiltrative and metastatic behavior. 11,21 Although EMT is associated with more aggressive and metastatic behavior in human mammary carcinomas, the same is not true in mice in which, except for local invasiveness, no metastatic behavior has been reported so far. 11
Mammary tumors with consistent squamous differentiation (Table 3, Nos. 3, 7, 8, 13, 14) typically showed marked β-catenin protein cytoplasmic and, to a lesser extent, nuclear accumulation. As already reported in previous studies, the cytoplasmatic accumulation and nuclear translocation of β-catenin protein in mammary tumors with squamous differentiation reflects the activation of the WNT pathway and β-catenin downstream signalling. 28,38 Indeed, it has been demonstrated that nuclear β-catenin stimulates transactivation of genes involved in both tumor progression (cyclin D1 and c-myc ) and squamous differentiation. 10,28
Our study confirmed the high prevalence of spontaneous prolactinomas and concurrent mammary tumors in wild-type aging FVB/N females. In the cohort examined, the age and number of pregnancies represented significant risk factors for the development of these tumors. Compared with previous reports, mammary tumors associated with prolactinomas displayed a broader morphologic spectrum, including cases with the EMT phenotype. These observations reinforce the need for colony-specific, age-matched controls in all experiments with the FVB strain of mice. The peculiar cytoplasmic ERα immunoreactivity observed in prolactinoma-associated EMT-phenotype mammary tumors opens intriguing possibilities about the role of ERα cytoplasmic redirection in EMT cancer progression.
Footnotes
Acknowledgements
This work was supported in part by NIH grant CA55909 (to AA) and the Susan G. Komen Breast Cancer Foundation. We acknowledge Ms Kirsten Saucier and Dr. Elizabeth A. Saria, both from the Center of Molecular Medicine at the University of Connecticut School of Medicine, and Ms. Katie Bell and Mr. Robert Munn from the Center for Comparative Medicine at the University of California, Davis, for technical assistance.
