Abstract
Horses are prone to recurrent airway obstruction (RAO), an inflammatory lung disease induced by repeated exposure to environmental mold, dust, and bacterial components. Active disease manifests with mucus hyperproduction, neutrophilic inflammation, bronchoconstriction, and coughing. Chronically affected animals have lung remodeling characterized by smooth muscle hyperplasia, collagen deposition, lymphoid hyperplasia, and impaired aerobic performance. Clara cell secretory protein (CCSP) counters inflammation in the lung, hence we hypothesized that CCSP depletion is a key feature of RAO in horses. Recombinant equine CCSP and specific antiserum were produced, and percutaneous lung biopsies were obtained from 3 healthy horses and from 3 RAO-affected horses before and after induction of RAO. CCSP relative gene expression in tissue, as well as protein concentration in lung lavage fluid, was determined. Immunocytochemical analysis, using both light and immunogold ultrastructural methods, demonstrated reduced CCSP staining in lung tissue of animals with RAO. Immunogold label in Clara cell granules was less in animals with chronic RAO than in normal animals, and absent in animals that had active disease. Median lung lavage CCSP concentration was 132 and 129 ng/ml in healthy horses, and 62 and 24 ng/ml in RAO horses before and after challenge, respectively. CCSP lung gene expression was significantly higher in healthy animals than in animals with chronic RAO. Together, these preliminary findings suggest that reduced production of CCSP and subcellular changes in Clara cells are features of chronic environmentally induced lung inflammation in horses.
Clara cells are nonciliated epithelial cells lining the bronchioles of the distal lung. 23 In most mammals, Clara cells are concentrated at the transitional zone between the conducting and gas-exchanging regions of the lung. There is marked interspecies variation in the proportion of the bronchiolar epithelium composed of Clara cells, and in the size and number of secretory granules. 23 Clara cells comprise 99% of feline, 60% of equine, and 22% of human bronchiolar epithelium, and horses and humans have approximately 28 and 6 granules per cell, respectively, while feline Clara cells entirely lack distinct granules. 22,24,25
The major secretory product of Clara cells is a nonglycosylated low molecular weight (10-kd reduced, 16-kd nonreduced) protein designated Clara cell secretory protein (CCSP), also referred to as CC10, CC16, or uteroglobin. 2 This protein is the prototypic member 1A1 of the secretoglobin (SCGB) family. 2 CCSP is a steroid-inducible homodimer secreted by epithelial cells. In humans, highest expression is in lung and uterus, with lesser amounts detected in prostate. 2 Expression of CCSP coincides with high cytochrome P (CYP) 450 monooxygenase enzyme activity, which renders Clara cells particularly susceptible to injury from metabolism of environmental toxicants. 8,25,28 Among many putative functions of CCSP are regulation of inflammation through inactivation of phospholipases (PL) A2 and C, 18 growth enhancement of pre-implantation blastocysts, 2 binding of lipophilic substances such as polychlorinated biphenyl (PCB), 9 and inhibition of leukocyte chemotaxis. 32 In mice, Clara cells also produce a polymeric mucin, which is normally rapidly secreted and does not accumulate. 7 However, during inflammation, mouse Clara cells may increase mucus production with subsequent cytoplasmic detection; a process resulting in so-called “Clara-goblet cells.” 7 Recently, the value of serum CCSP concentration as an indicator of integrity of the lung-blood barrier was recognized. 3 CCSP was implicated in reduction of airway inflammation in asthma 31 and respiratory syncytial virus infection. 34 CCSP impart resistance to development of pulmonary fibrosis, 13 and is a marker of neoplastic transformation in lung epithelial cells. 15 Although the precise physiologic functions of CCSP remain to be defined, the protein appears to have multiple important roles in health and disease.
Horses are prone to inflammatory and infectious lung disease, but neither physiologic nor pathologic roles of CCSP have been characterized in the horse lung. Recurrent airway obstruction (RAO) or “heaves” is a complex disease of uncertain pathogenesis in horses. 6 The disease is induced and perpetuated by exposure to inhaled antigens, likely a synergistic combination of molds, endotoxin, and fine organic particulate matter common in the barn environment. 27 It is the most commonly diagnosed noninfectious airway disease of mature horses in North America and Europe due to seasonal need for indoor stabling. 17 Disease progression is insidious and may be subclinical with intermittent episodes of inflammation for months to years. Airway inflammation can comprise neutrophils, lymphocytes, mast cells, or eosinophils, but during exacerbations consists predominantly of neutrophils. Lymphoid aggregates form in peribronchial regions, and lung parenchyma gradually remodels as clinical respiratory impairment develops. 11,33 Airway hyperreactivity and bronchoconstriction are features of RAO and may be triggered by the release of mediators such as histamine from mast cells, leukotrienes from neutrophils, or pro-inflammatory cytokines from epithelial or other cells. 16,19,20
Based on knowledge of CCSP in human and mouse respiratory disease, and the unique features of RAO in horses, we hypothesized that CCSP is a crucial component of the equine response to noninfectious lung inflammation. To investigate this hypothesis we characterized the equine CCSP gene, generated recombinant protein and specific antibodies against CCSP, quantitated CCSP gene expression in horse lung, and assessed the protein by immunohistochemistry, immuno-electron microscopy, and lavage fluid enzyme-linked immunosorbent assay (ELISA) in healthy and RAO-affected animals.
Materials and Methods
Animals and procedures
All animal procedures were approved by the University of Guelph Animal Care Committee in accordance with guidelines of the Canadian Council on Animal Care. Animals used were Standardbred horses consisting of 3 mares and 3 geldings between 13 and 18 years of age. Three animals had historic RAO but were free of clinical signs for at least the past 2 months, and 3 had no history of RAO. Horses were maintained in outdoor housing with minimal exposure to dust and mold prior to study. For assessment, animals were acclimatized to indoor hospital surroundings for 24 hours, bedded on wood shavings, fed good quality hay, and provided free access to water. Horses were then subjected to physical exam, pulmonary function testing, broncho-alveolar lavage (BAL) and percutaneous lung biopsy, and classified as healthy controls or as “RAO in remission.” 6 Thereafter, animals were exposed to moldy hay for 3 to 7 days until clinical signs of respiratory impairment were apparent in RAO horses in remission, and the above procedures were repeated. Serum clinical chemistry profiles and complete blood cell (CBC) counts were performed on all horses at baseline.
For pulmonary function testing (PFT), horses were restrained in stocks without sedation. A flexible rubber facemask was fitted over the muzzle and connected to a heated Fleisch No. 4 pneumotachograph (Gould Electronics, Bilthoven, The Netherlands) to measure airflow, with volume derived by the integration of the flow signal (Buxco, Sharon, CT). Transpulmonary pressure (ΔPpl) was measured as the difference between atmospheric and pleural pressures. Pleural pressure was estimated using a latex esophageal balloon (10 cm long, 3 ml of air injected, attached to a 2-m polyethylene catheter, 2.69 mm inner diameter, 3.5 mm outer diameter) inserted to midthorax. The flow and pressure signals were collected and analyzed by respiratory loop analysis (Buxco).
For bronchoscopy and BAL, horses were sedated with xylazine hydrochloride (0.5 mg/kg, given intravenously [IV]), and a 13-mm diameter, 180-cm long sterile bronchoscope (Olympus, Tokyo, Japan) was inserted through the nares and passed to the level of the carina, while a 0.2% lidocaine solution was intermittently sprayed through the biopsy channel of the bronchoscope to alleviate airway discomfort and reduce coughing. The bronchoscope was advanced and wedged in a bronchus, and two 125-ml aliquots of warm sterile saline were instilled and immediately re-aspirated (−10 cm H2O). The volume retrieved was recorded, and the fluid was filtered through sterile gauze and placed on ice. Only samples with 150 to 200 ml fluid recovery, and lack of microscopic red blood cell contamination, were used. BAL fluid analysis included determination of the nucleated cell concentration (Coulter Z2, Mississauga, ON, Canada), and a 200-cell differential count performed on cytocentrifuged preparations of 200-µL aliquots that were stained with Wright's stain.
Percutaneous lung biopsy
Percutaneous lung biopsies were obtained under sedation 24 hours after BAL from the lung opposite to the side of lavage. Local anesthetic (2% lidocaine) was infiltrated into the skin and subcutaneous tissues to the level of the pleura. A small skin incision was made, and 3 Tru-cut biopsy needle samples were collected for fixation in formalin and into RNAlater (Ambion, Streetsville, ON, Canada). A skin suture was placed at the entry site, and horses were monitored for adverse effects over the subsequent 48 hours.
Cloning of equine CCSP
Lung tissue was collected from an adult horse euthanized for an unrelated condition. Total RNA was isolated (RNeasy, Qiagen, Mississauga, ON, Canada) and reverse transcribed to cDNA. Primers for amplification of the coding region were based on the equine uteroglobin sequence (NM_001081858) available in the National Center for Biotechnology Information (NCBI) database and designed with EcoR1 and BamHI sites for subsequent cloning into the pGEX-2T bacterial expression vector (Amersham, Baie d'Urfé, PQ, Canada). CCSP cDNA was amplified with 20 pM of forward primer CC10F-BamHI (5′-T [GGATCC] GGA ATC TGG CCA GAG ATT TGC-3′), 20 pM of reverse primer CC10R-EcoR1 (5′-C [GAATTC] CTA AGC ACA CAG TGG GGC TC-3′), 3 µl template, 1.5 mM MgCl2, 0.25 mM dNTP's, 1× polymerase chain reaction (PCR) buffer and 2.5 U Taq DNA polymerase (Invitrogen, Burlington, ON, Canada) in a total volume of 50 µl. Thermocycling consisted of 6 cycles with 15 seconds at 94°C, 40 seconds at 30°C, and 45 seconds at 72°C, followed by 20 cycles with the annealing temperature adjusted to 45°C, and a final extension of 7 minutes at 72°C. A PCR product of 288 base pair (bp) was excised, and the DNA extracted and purified (Qiagen). The pGEX-2T vectors and cDNA inserts were digested using the appropriate enzymes; then ligated, and propagated into Topo-One-Shot chemically competent Escherichia coli BL21 (Invitrogen), incubated overnight at 37°C and selected on Luria-Bertani (LB) agarose plates containing 100 µg/ml ampicillin. Five clones were grown overnight in 3 ml LB broth with 100 µg/ml ampicillin. Plasmids were screened by EcoR1/BamHI digestion and electrophoresis in 1% agarose gels. Bands of the appropriate size were excised, purified, and sequenced. All sequences were analyzed and aligned with Vector NTI software (Invitrogen).
Production of recombinant equine CCSP
Transformed E. coli BL21 cells were grown for 3 hours in 50 ml 2× yeast/tryptone/ampicillin (YTA) broth at 37°C with gentle shaking. Five milliliters of the culture were transferred to flasks containing 500 ml of fresh 2× YTA medium and incubated overnight at 37°C with gentle shaking. Isopropyl-beta-D-thiogalactopyranoside (IPTG) was added to a final concentration of 2 mM and the culture was incubated at 37°C for 4 hours. Cells were pelleted by centrifugation at 2,080 g for 10 minutes at 4°C, resuspended in one-tenth the original culture volume of 1× PBS plus lysozyme (0.1 mg/ml), and incubated on ice for 30 minutes. The cells were sonicated on ice and treated with 5 ml of 20% TritonX per 100 ml sonicate volume. Cell debris was pelleted by centrifugation at 13,000 g for 10 minutes at 4°C, and the supernatant was filtered (0.45 µM). The remaining filtrate was passed through a glutathione sepharose 4B column (Amersham), washed with 1× PBS, and eluted with 10 mM reduced glutathione (GSH). The eluate was then incubated overnight with 1 ml of 10% thrombin/PBS per 1 ml initial bead volume at 26°C with end-over-end rotation to encourage cleavage of the fusion protein from the glutathione-S-transferase (GST) tag. The bead suspension was centrifuged at 514 g for 5 minutes at room temperature, and the eluate was incubated with 400 µL of benzamidine sepharose beads (Amersham) for 45 minutes at room temperature to capture residual thrombin. Beads were pelleted by centrifugation at 100 g for 10 minutes and the supernatant was collected, concentrated with Amicon Ultra-15 centrifugal filter units (Millipore, Nepean, ON, Canada), and stored at −80°C. To purify the protein, aliquots were resolved by sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) in a 4% stacking, 15% resolving gel under nonreducing conditions. The ∼15 kd band was excised and the protein extracted using a BioRad Model 422 Electro-Eluter according to manufacturer's instructions (BioRad, Mississauga, ON, Canada). The purified protein was collected and stored at −80°C. The final protein concentration and purity were estimated by capillary electrophoresis and densitometry (Bioanalyzer, Agilent, Mississauga, ON, Canada). N-terminal sequencing of the ∼15 kd purified protein was performed through sequential Edman reactions (Nucleic Acid-Protein Service Unit, University of British Columbia, Vancouver, BC, Canada).
Equine CCSP antibody
Antiserum against equine CCSP (eCCSP) was produced by immunizing a rabbit (Pacific Immunology, Ramona, CA) with the synthetic 21-mer peptide EPSKPDADMKAATTQLKTLV-C (NM_001081858, amino acids 26–45). The peptide was synthesized by solid phase technology and coupled to keyhole limpet hemocyanin at the C-terminus. Serum collected after 3 immunizations was affinity purified and titrated before use. To assess antigen recognition of the antibody, aliquots of purified recombinant CCSP were separated by 4% stacking/15% resolving SDS-PAGE under nonreduced conditions and transferred to Immobilon-PSQ polyvinylidene fluoride (PVDF) membranes (Millipore). The blot was blocked overnight with 5% bovine serum albumin (BSA) in 1× Tris-PBS with 0.1% Tween. Primary antibody was diluted 1 : 1,000 in 1× Tris-PBS, 0.05% Tween and 2.5% BSA buffer, and incubated with the blot for 1 hour at room temperature on a shaker. After 3 washes with buffer, goat anti-rabbit IgG coupled to horseradish peroxidase (HRP), diluted 1 : 5,000 in buffer, was added and the blots were incubated at room temperature for 1 hour. After 3 further washes, bound antibody was detected with diaminobenzidine (DAB) chromogen (DakoCytomation, Mississauga, ON, Canada).
Immunohistochemistry
Lung biopsy sections were cut at 5 µm for immunohistochemistry (IHC) and hematoxylin and eosin (HE) staining. IHC sections were deparaffinized, incubated with 0.3% H2O2 in methanol for 20 minutes to quench endogenous peroxidase activity, washed 3 times in 1× PBS buffer and blocked with 10% normal goat serum in buffer for 30 minutes. Then, sections were incubated with the primary antibody at 1 : 1,200 for 30 minutes, washed 3 times in buffer, and incubated for 30 minutes with HRP-labeled secondary antibody (goat anti-rabbit IgG 1 : 150, DakoCytomation). Antibody binding was detected with Nova Red (Vector Laboratories, Burlington, ON, Canada) chromogen, and slides were counterstained with hematoxylin. For negative control blots and sections, pre-immune rabbit serum was used instead of primary antibody, or the primary antibody was pre-incubated with peptide.
Electron microscopy
Clara cells were initially identified by both transmission electron microscopy (TEM) and scanning electron microscopy (SEM). For both SEM and TEM, tissues were fixed in 4% paraformaldehyde and 1% gluteraldehyde. Tissues were postfixed in 1% OsO4, and then dehydrated in an ascending series of acetone. Samples for SEM were then critical point dried and mounted on aluminum stubs with carbon dag and rendered conductive with a thin film of sputtered gold palladium. Specimens were examined and photographed in a scanning electron microscope (JEOL JSM 820, Peabody, MA). Samples for TEM were infiltrated with epon araldite and embedded in BEEM capsules prior to polymerization at 60°C overnight. Areas of interest were selected from 0.5-µm sections stained with toluidine blue and cut using an ultramicrotome. Blocks were trimmed to the area of interest; ultrathin sections were prepared and mounted on grids, then stained in uranyl acetate and lead citrate prior to examination in the transmission electron microscope (JEM 1230, JEOL, Peabody, MA). Images were acquired with a charge-coupled device (CCD) camera (AMT, Danvers, MA).
Immunogold labeling of CCSP was done on lung tissue samples which were fixed in 4% paraformaldehyde/0.1% glutaraldehyde in 0.1 M phosphate buffer for 1 hour at room temperature, rinsed twice in buffer, and infused in a 2.3 M sucrose solution overnight at 4°C. Samples were frozen in liquid nitrogen and immediately immersed in 0.5% uranyl acetate in 100% methanol at −80°C for 3 to 5 days. Samples were slowly warmed to −20°C, immersed in cold (−20°C) 100% methanol for 2 hours, followed by 1 : 1 methanol : Lowicryl HM20 resin for 24 hours. The following day, the tissue was immersed in fresh aliquots of cold (−20°C) 100% Lowicryl HM20 resin twice for 24 hours each. Samples were embedded in BEEM capsules using fresh 100% Lowicryl HM20 and allowed to polymerize under UV light at −20°C for a minimum of 2 days. Ultrathin sections of bronchioles were cut and mounted on Formvar-coated nickel grids, washed twice with 0.15% glycine/0.5% BSA-PBS buffer, and blocked with BSA-PBS buffer for 30 minutes. Primary antibody (eCCSP-1) was diluted 1 : 50 in 5% powdered milk in PBS and incubated with the grids for 1 hour at room temperature, followed by 4 washes with PBS. The grids were then incubated with 10 nm gold-labeled goat anti-rabbit IgG antibody (Amersham), diluted 1 : 20 in BSA-PBS buffer for 1 hour at room temperature followed by a thorough wash in PBS and distilled water. Grids were then counterstained with uranyl acetate and lead citrate. Sections were examined in the TEM and images recorded with a CCD camera. Controls included either the omission of the primary or the secondary antibody.
Quantification of CCSP gene expression
RNA was isolated from lung biopsies (RNeasy, Qiagen) and cDNA was prepared with SuperScript III Reverse Transcriptase (Invitrogen). Primers 5′ CCA TCC TGG CTC TCT GCT G-3′ and 5′ TGC TGT CCT TGG TGT TCT TG-3′ amplified 200 bp of equine CCSP, and primers 5′-GTT TGT GAT GGG CGT GAA CC-3′ and 5′- TTG GCA GCA CCA GTA GAA GC-3′ amplified 257 bp of the equine glyceraldehyde dehydrogenase (GAPDH) gene. Quantitative PCR assays consisted of 0.25 µM of each forward and reverse primer, 2 µl Master Mix containing SybrGreen I (Roche, Mississauga, ON, Canada), 4.5 µl PCR-grade water, and 2.5 µl cDNA. Samples were denatured at 95°C for 10 minutes and cycled 40 times at 95°C for 10 seconds, 60°C for 5 seconds, and 72°C for 12 seconds in a LightCycler (Roche). Melting curve analysis (95°C) was performed to determine specificity of primer binding. To generate standard curves, each gene was amplified from cDNA; the amplicon was gel-extracted (Qiagen), sequenced, diluted from 1 × 10− to 1 × 10−7, and 3 replicates were amplified at each dilution. The average crossing point of each dilution was used to derive standard curves. Triplicates of CCSP and GAPDH were amplified from each unknown sample concurrent with standard curve “calibrators,” and data were analyzed with RelQuant software (Roche).
CCSP ELISA
Wells of polystyrene microtiter plates (Costar, Corning, NY) were incubated with 100 µL of BAL fluid or dilutions of reCCSP (0, 12.5, 25, 50, 100, and 200 ng/ml) in 50 nM carbonate buffer at pH 9.6 overnight at 4°C. Wells were washed with 0.05% Tween-20 in 50 mM PBS, pH 7.4 (T-PBS) and blocked with 5% powdered skim milk in T-PBS for 2 hours. Following 3 washes with T-PBS, 100 µL of eCCSP-AB (diluted 1 : 350 in T-PBS/1% powdered skim milk) was added to each well, and samples were rotated for 1 hour at room temperature on an orbital shaker. After 3 washes in T-PBS, 100 µL of horseradish peroxidase-labeled swine anti-rabbit polyclonal antibody (Dako), diluted 1 : 4,000 in T-PBS/1% skim milk powder was added to each well. Samples were incubated at room temperature for 1 hour, then washed. Finally, 100 µL of tetramethylbenzidine (Pierce, Rockford, IL) was added to each well, plates were incubated for 15 minutes, and the reaction was terminated by addition of 100 µL of 0.5 M sulfuric acid. Absorbance was measured at 450 nm. Incubation of samples with pre-immune rabbit serum in lieu of eCCSP-AB served as a negative control, and readings from blank wells were used to determine background signal. All samples and standards were tested in triplicate. Standard curve analysis was used to extrapolate sample CCSP concentrations.
Statistical analysis
Differences in BAL leukocyte counts, CCSP concentration and gene expression prechallenge and postchallenge, and between different groups of animals at baseline were assessed by analysis of variance in a general linear model (SAS, Cary, NC). Differences were considered significant if P < .05.
Results
Animals
Control horses had no history of RAO, normal physical exam findings, PFT ΔPpl <10 cm H2O, and BAL neutrophil percentage <5%. Horses with RAO had been affected for >2 years prior to study, ΔPpl was >20 cm H2O with >25% BAL neutrophils during disease exacerbation and <10 cm H2O with 5–25% BAL neutrophils prior to challenge. 6 All horses had CBC and serum chemistry results within normal limits. Median BAL neutrophil percentage was 0.7% for control horses, 9.3% (P = .034) for RAO horses in remission, and 78.5% (P = .014) for RAO horses with exacerbated disease.
Recombinant equine CCSP
Immunoblot analysis indicated that the peptide antiserum eCCSP-1 bound to proteins with approximate weights of 29, 12, and 6 kd, which were interpreted to be GST-CCSP fusion proteins, CCSP dimers, and CCSP monomers, respectively (Fig. 1). The molecular weight of CCSP dimers ranges from 10 to 15 kD in primates, rabbits, and rodents, which is compatible with findings here. 10 The recombinant protein was completely cleaved from the GST fusion partner (lanes 1 & 2) and was verified through direct sequential Edman reactions to be CCSP. The protein encoded the entire mature secretory protein of 70 AA.
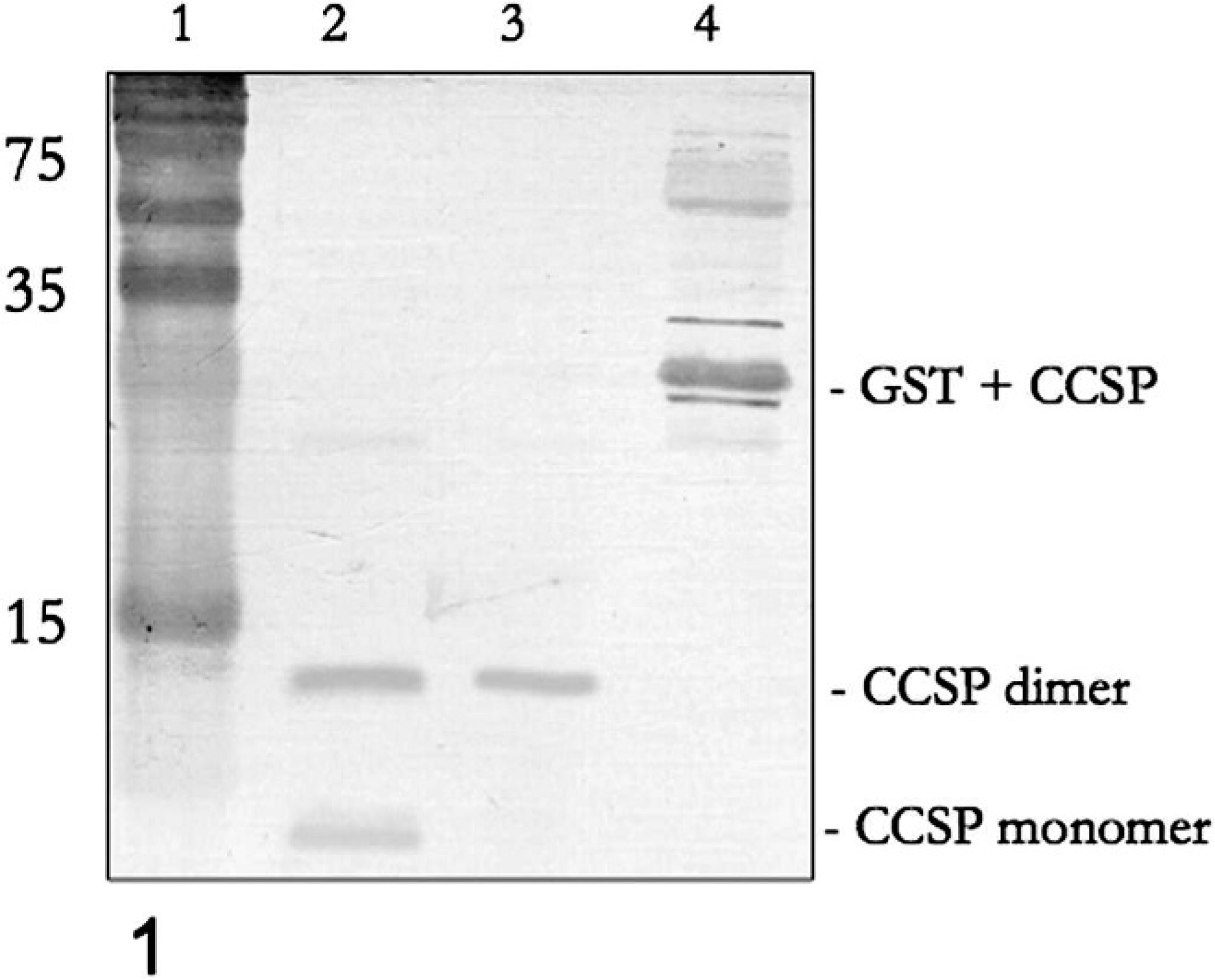
Immunoblot of reCCSP probed with specific antisera. Lane 1, Molecular weight markers (kd). Lane 2, CCSP dimer and monomer (∼12 and 6 kd). Lane 3, CCSP dimer. Lane 4, GST+ CCSP fusion protein (∼29 kd). DAB chromogen.
Immunohistochemistry
The peptide antiserum was reactive with formalin-fixed tissue and bound to nonciliated epithelial cells in lung tissue, but not to ciliated epithelial cells or goblet cells (Fig. 2a). The apical region of the Clara cell cytoplasm typically stained more intensely than the perinuclear or basal region, and staining was frequently granular. In samples from RAO animals, immune staining was markedly reduced relative to samples from control animals, but relatively uniform throughout the cytoplasm (Fig. 2b). Sections from animals after challenge typically had many nonciliated bronchiolar cells completely lacking CCSP immunoreactivity, and scattered individual cells with faint staining (Fig. 2c). Histopathologic changes that distinguished biopsies from healthy horses relative to horses with RAO were alveolar wall thickening, peribronchiolar fibrosis, and presence of peribronchiolar lymphocyte aggregates in the affected animals.
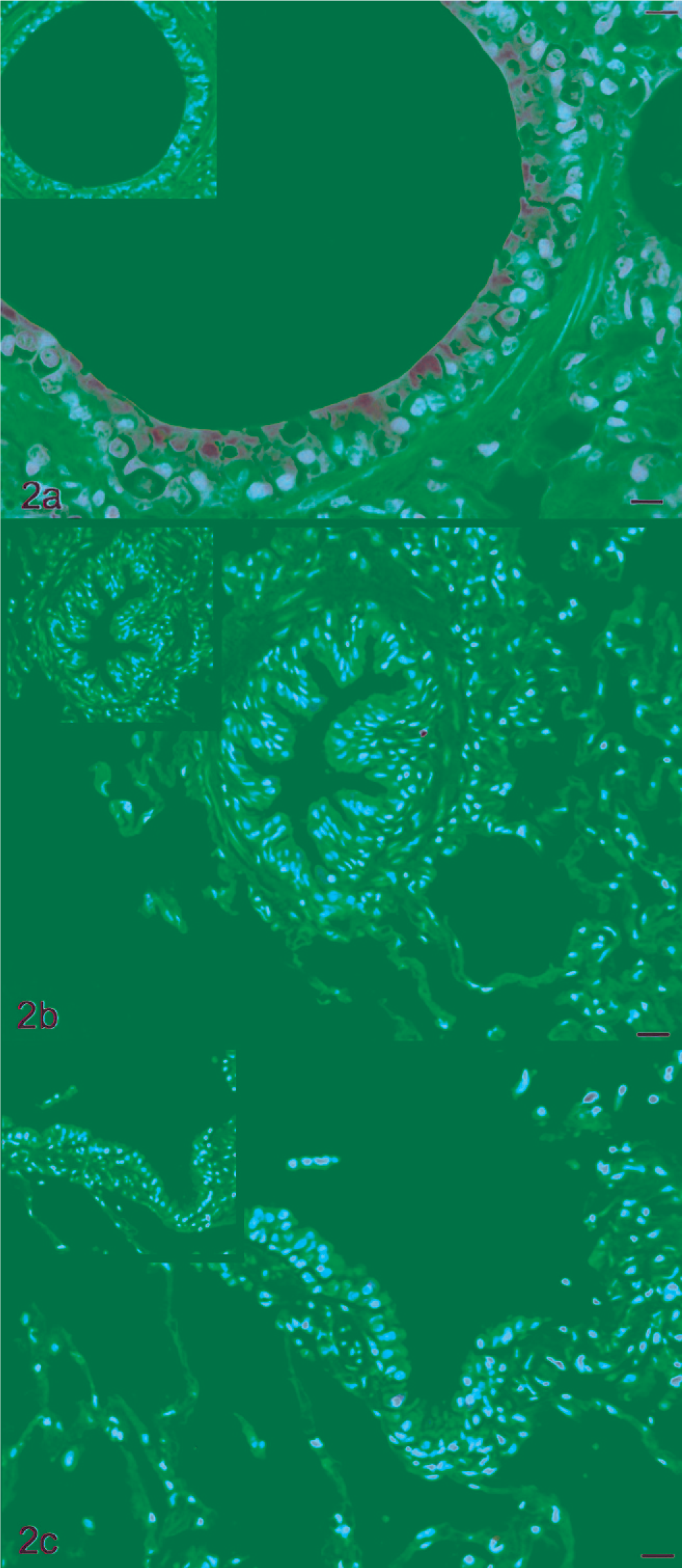
Lung; horse. Immunohistochemical staining of biopsies with antibody to equine CCSP. Hematoxylin counterstain. Fig. 2a. Section from healthy control horse. Bar = 20 µm. Fig. 2b. Section from horse with RAO prior to experimental induction of lung inflammation. Bar = 10 µm. Fig. 2c. Section from horse after experimental induction of lung inflammation. Bar = 10 µm. Inserts are control sections incubated with pre-immune serum instead of primary antibody.
Electron microscopy
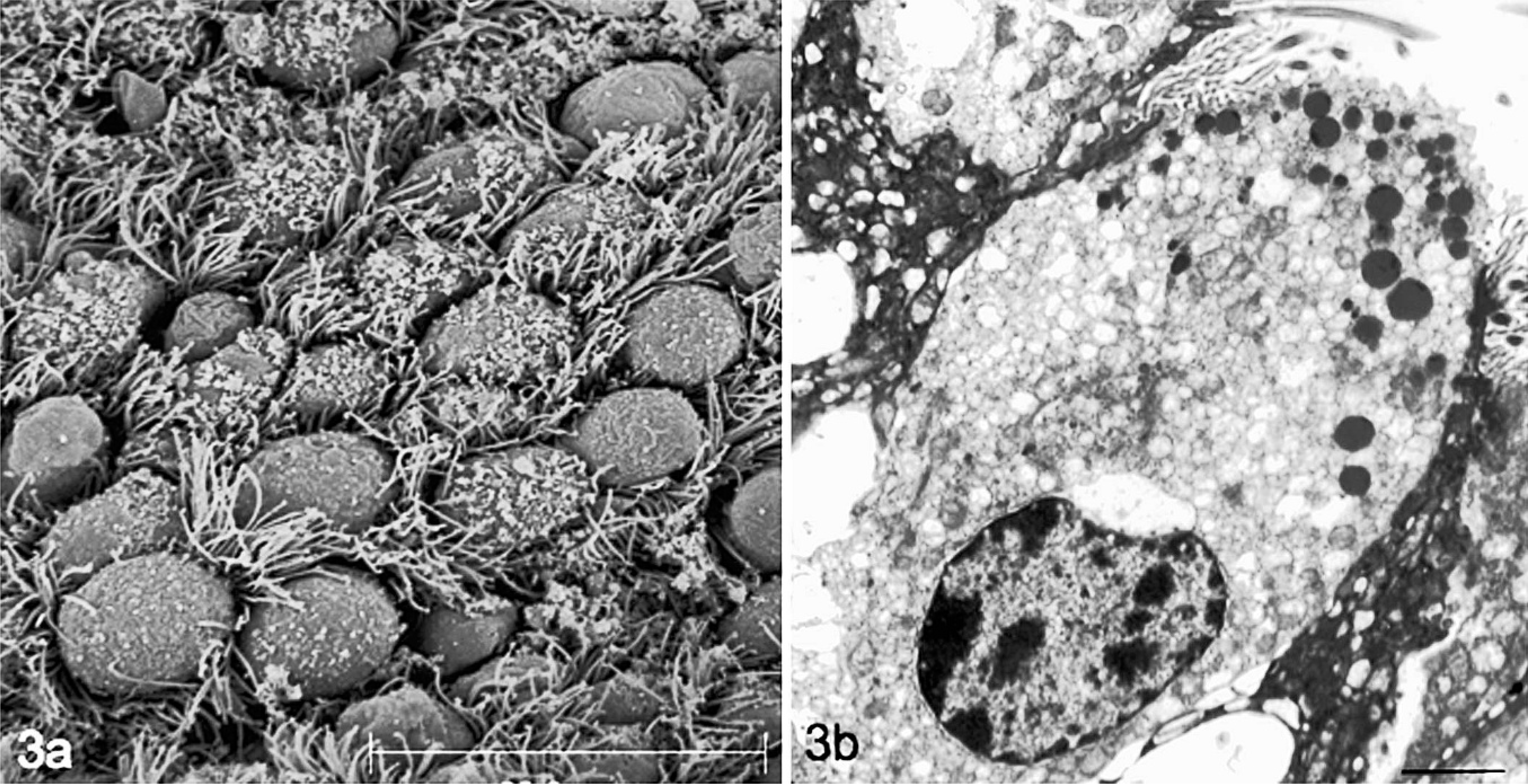
Both SEM and TEM identified Clara cells as nonciliated cells among ciliated epithelial cells in bronchioles from control animals (Fig. 3a). Clara cells had oval nuclei, abundant endoplasmic reticulum, minimal cytoplasmic glycogen, short microvilli, and cytoplasmic granules concentrated on the apical side of the cell (Fig. 3b). Lung biopsies from control animals and animals with RAO in remission and exacerbation were then compared by immuno-electron microscopy (IEM) (Fig. 4). In control horses, Clara cells were relatively abundant (Fig. 4a), and gold-labeled antibody highlighted numerous electron-dense apical granules and spared the remainder of the cytoplasm and nucleus (Fig. 4b). Some gold label could also be appreciated along lengths of endoplasmic reticulum, consistent with the secreted nature of the protein. These ultrastructural features were compatible with previous descriptions of mammalian Clara cells. 25
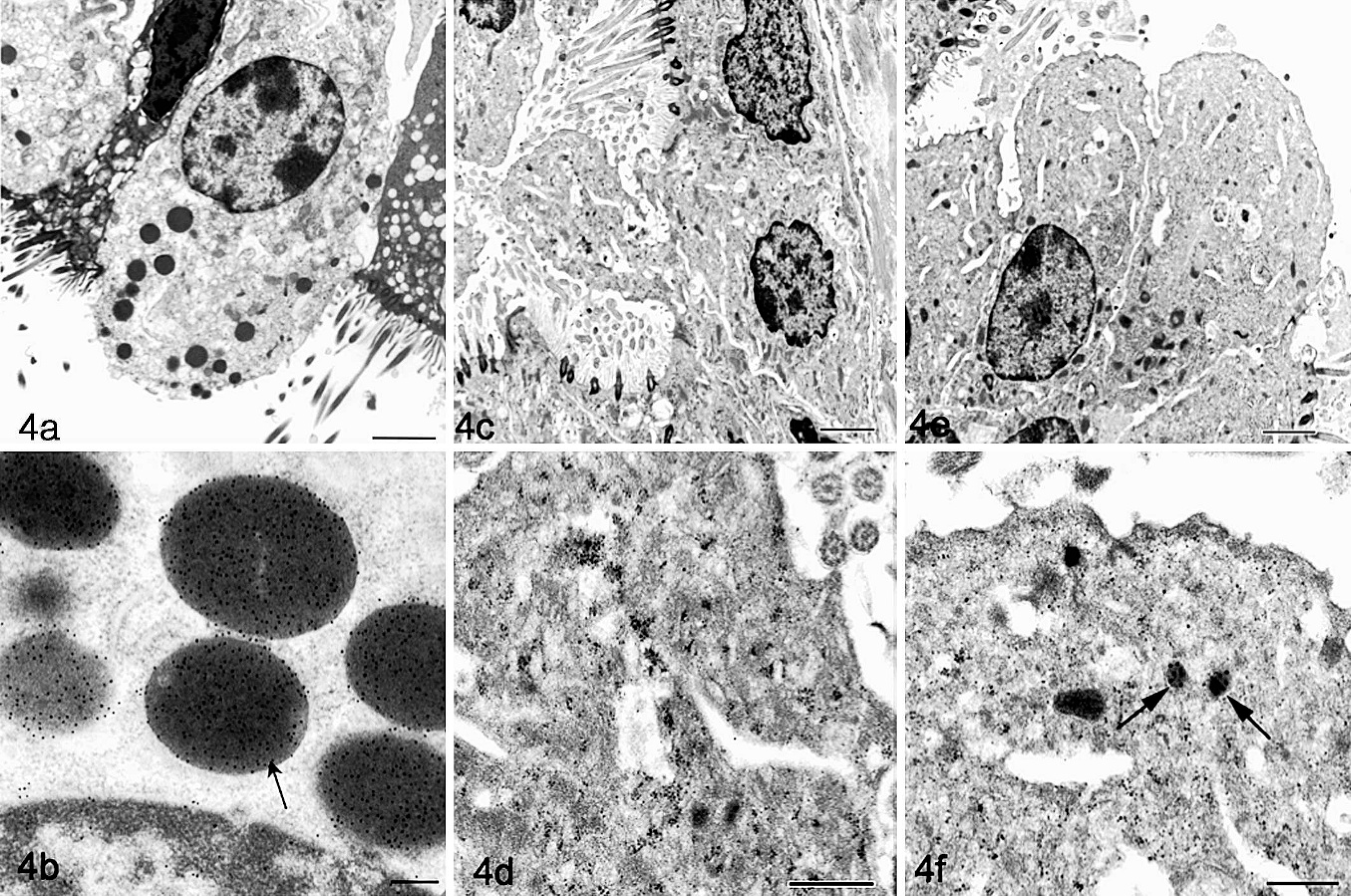
Lung; horse. Immuno-electron microscopic detection of CCSP. Healthy horses (Fig. 4a, b) have Clara cells with electron-dense cytoplasmic granules and abundant endoplasmic reticulum. CCSP-antibody linked to 10 nm gold beads clearly labels the granules (arrow), but not the endoplasmic reticulum, of Clara cells (Fig. 4b). Clara cells from horses with active RAO induced by experimental challenge (Fig. 4c, d) lack detectable cytoplasmic electron-dense granules, and the cytoplasm is almost entirely devoid of gold-label (Fig. 4d). Lung tissue from the same animal with RAO prior to challenge (Fig. 4e, f) lacked distinct cytoplasmic granules, but occasional gold beads were apparent in poorly demarcated cytoplasmic structures (arrow). Bar = 2 µm for Fig. 4a, c, e; bar = 0.2 µm for Fig. 4b, d, f.
Samples from RAO horses after challenge lacked cytoplasmic granules (Fig. 4c), with nearly complete absence of antibody label (Fig. 4d). Ultrastructurally, empty-appearing vacuoles were apparent, but they did not contain immunoreactive CCSP. The same animals with RAO in remission (before challenge) still lacked electron-dense cytoplasmic granules (Fig. 4e), but occasional immunolabel was apparent at higher magnification (Fig. 4f). These accumulations of 3 to 4 gold beads were present in the vicinity of small electron-dense areas and may represent attempts at reformation of granules. IEM findings were consistent among groups of animals, and among multiple sections examined per animal.
Real-time PCR
CCSP gene expression was determined in lung biopsies from control and RAO-affected animals before and after exposure to challenge material. Quantification relative to GAPDH indicated prechallenge and postchallenge mean values of 0.349 and 0.583 in control animals and 0.0485 and 0.1407 in horses with RAO, respectively (Fig. 5a). Although gene expression was lower in horses with RAO, the difference was not statistically different (P = .094). Baseline gene expression in horses with RAO was lower than in healthy controls (P = .256) but increased with challenge (P = .089).
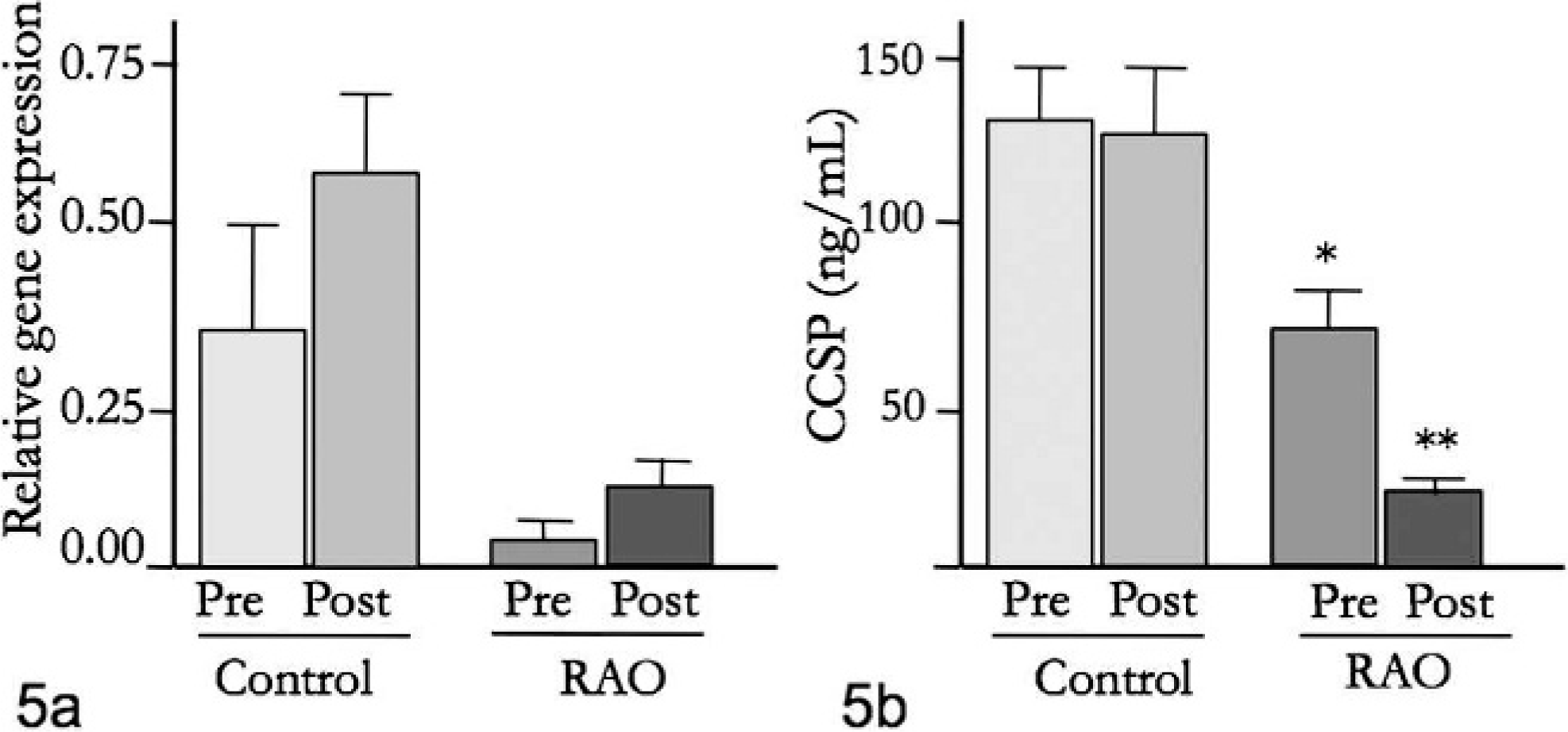
Fig. 5a. Real-time PCR quantification of CCSP relative to GAPDH gene expression in lung tissue from control horses and horses with RAO. Horses with RAO prior to challenge had lower gene expression relative to healthy control animals prior to challenge. Fig. 5b. BAL CCSP concentration determined by ELISA. Baseline BAL CCSP concentration in horses with RAO was lower than in control horses (∗P = .031) and reduced after experimental challenge in horses with RAO (∗∗P = .048).
CCSP ELISA
An antigen-capture ELISA designed for equine CCSP was linear over a range of 12.5 to 200 ng/ml of reCCSP. Antigen was measured directly in BAL fluid from horses. Mean CCSP concentration was 131.1 and 128.3 ng/ml in BAL from healthy horses prior to and after challenge, respectively, and 67.7 and 21.0 ng/ml in BAL from animals with RAO prior to and after challenge, respectively (Fig. 5b). CCSP concentration was significantly higher (P = .031) in control animals than in RAO animals prior to challenge, and changes as a result of challenge were also significant (P = .048).
Discussion
Recurrent airway obstruction in horses is a disease with many similarities to asthma in humans. The pathogenesis is incompletely understood, but respiratory impairment is induced and perpetuated by continual exposure to inhaled antigens, likely a synergistic combination of molds, endotoxin, and fine organic particulate matter. 27 Airway inflammation, hyperreactivity, and bronchoconstriction are the key features of RAO and, similar to allergen-induced lung inflammation in other species, these are triggered by inflammatory mediators and cytokines. 16,19,20 Considering that CCSP appears to have a critical role in response of the respiratory tract to allergens, infectious agents, and toxicants, we hypothesized that loss of CCSP is a key feature of a common allergic lung disease of horses, RAO. To address this hypothesis, we first needed to generate a range of reagents specific for the horse, and then apply these reagents to normal and diseased animal tissues.
We found that CCSP in the horse lung was present in discrete cytoplasmic granules of nonciliated epithelial cells in the distal bronchioles. Since this was not a terminal study, assessment was restricted to tissues accessible by percutaneous biopsy. Such procedure would not be justifiable in clinical patients but provided sufficient sample for all assays described in this study. It was previously reported that in horses Clara cells comprise the majority of epithelial cells at the transition from conducting to gas-exchanging airways, and consistent with this observation, bronchioles were present in all biopsies we obtained. 25 Nonciliated cells comprised between 30 and 60% of epithelial cells in larger bronchioles, but terminal bronchioles were comprised almost exclusively of nonciliated epithelial cells with cytoplasmic granules. The antiserum intensely labeled granules dispersed throughout the cytoplasm and proximal to the apical cell border of nonciliated cells, consistent with a secretory granule. The CCSP anti-serum labeled all granules apparent in the cytoplasm of lung biopsies from either healthy control or RAO-affected horses. Hence, this finding suggests that possibly, in horses, Clara cells neither normally store mucus nor undergo mucus metaplasia during chronic inflammation, in contrast to observations in mice, where a “Clara-goblet cell” becomes morphologically recognizable. 7 In horses with RAO, there was virtually complete loss of cytoplasmic granules and immunolabeling, which was mirrored by BAL CCSP concentration. Immunohistochemical staining indicated that CCSP was present neither in ciliated nor in alveolar epithelium, but there was occasional nonspecific staining of extracellular material in the airways, presumably due to secreted CCSP in airway lining fluid. Faint cytoplasmic staining in samples from clinical remission suggested there might be attempts at regeneration. CCSP inhibits phospholipase A2 (PLA2), 18,28,30 thereby decreasing production of pro-inflammatory mediators such as leukotrienes and prostaglandins. PLA2 inhibition is the main mechanism thought to account for the anti-inflammatory effects of CCSP. 35 It is possible, therefore, that chronic neutrophilic inflammation in RAO induces continual Clara cell production and secretion of CCSP, and that in long-standing lung inflammation, regeneration of stored protein and granule formation are impaired. Furthermore, Clara cells are the primary cells for xenobiotic metabolism in the lung via abundant cytochrome P450 monooxygenase activity. 7,21 This property renders them uniquely susceptible to injury as many of the transformed compounds are converted to toxic intermediates requiring conjugation to reduced glutathione (GSH) by glutathione-S-transferases. 4 Although inhalation of toxic substances is not considered part of the pathogenesis of RAO, chronic exposure to neutrophil-derived reactive oxygen species and proteases may contribute to CCSP depletion. Support for the efficacy of CCSP in countering lung inflammation is derived from in vivo studies of intratracheally administered recombinant human CCSP in conjunction with surfactant therapy in human infants and animal models of pediatric acute lung injury and respiratory distress syndrome. 5,14,26
CCSP gene expression was slightly increased when airway inflammation was induced by challenge, relative to samples collected prior to challenge (Fig. 5a), but a reverse pattern was apparent for CCSP concentration in BAL (Fig. 5b) and tissue (Fig. 4). Such a trend might be consistent with gene induction in Clara cells during active inflammation, but an overall impairment in either the number of Clara cells able to produce CCSP or their relative ability to produce the protein might account for less in BAL fluid. Regulation of CCSP gene transcription is incompletely understood; however, positive and negative regulators have been identified, and changes in their activity might also contribute to reduced CCSP production in disease. 1 Although only a small number of animals were assessed in this study—which must therefore be considered a pilot study with limited statistical power—the findings, nevertheless, strongly suggest that reduced CCSP in lung lavage fluid may be a sensitive indicator of epithelial health in the lung of horses, as in other species. 3,29 While this study was restricted to assessment of CCSP in a noninfectious inflammatory lung condition of horses, it is quite possible that reduction in CCSP also occurs in infectious and other pneumonia and may therefore be a marker of chronic lung inflammation. Clara cells are thought to be lung epithelial progenitor cells, 12 and chronic inflammation may reduce their proliferative and differentiation potential, which would also result in lack of adequate surfactant production. Horses studied here were chronically affected by RAO, but animals during earlier disease stages may have better CCSP and granule regeneration during remission stages. Hence, future studies will be directed at examining samples from a larger number of RAO-affected animals from different disease stages and with different respiratory conditions, and at elucidating the anti-inflammatory properties of CCSP.
Footnotes
Acknowledgements
For technical expertise, we thank L. Huber and T. Bayley at the University of Guelph; and A. Tilups, R. Leung, J. Hwang, H. Rosenberg, and Y. M. Heng at the Hospital for Sick Children. For assistance with the statistical analysis, we thank A. Valliant at the University of Guelph. This work was supported by the Grayson Jockey Club Research Foundation, Equine Guelph, and the Natural Sciences and Engineering Research Council of Canada.
